Abstract
This study evaluates the immunoreactivity of 12 sex cord–stromal tumors of nonhuman primates (11 granulosa cell tumors and 1 luteoma). The markers selected are used in the characterization of gonadal tumors in dogs and other species, including cytokeratins AE1/AE3, GATA-4, inhibin-α, neuron-specific enolase, protein gene product 9.5, and vimentin. A normal nonhuman primate ovary was used as a control and to optimize immunolabeling. Staining was graded as follows: 0 (nonstaining), 1+ (< 10% positive cells), 2+ (10%–50% positive cells), and 3+ (> 50% positive cells). Calretinin, GATA-4, neuron-specific enolase, and vimentin were the most consistently expressed markers (12 of 12). Cytokeratins AE1/AE3 were also consistently expressed (11 of 12). Inhibin-α and protein gene product 9.5 were expressed in 8 and 10 sex cord–stromal tumors, respectively. Results indicate that immunoreactivity of nonhuman primate sex cord–stromal tumors is similar to that observed in other species and that calretinin, GATA-4, and neuron-specific enolase are the most consistently expressed markers in nonhuman primate sex cord–stromal tumors.
The most commonly reported ovarian tumors in nonhuman primates are granulosa cell tumors and teratomas, and the majority are reported from baboons and macaques. 1,2,12,13 Ovarian sex cord–stromal tumor (SCST) is the general term applied to ovarian tumors comprising neoplastic populations of granulosa cells, theca cells, their luteinized derivatives, and fibroblasts. 7 The embryologic derivation of these tissues is uncertain, and they may arise from the mesonephric or celomic epithelium. 7 Histologically, ovarian SCSTs resemble the normal cellular elements from which the neoplastic cells arose and may or may not be hormonally active. 7 Reports of the immunoreactivity of ovarian SCSTs are limited to humans, dogs, and 1 rhesus macaque report. 3,5,6,10,12,16 Inhibin-α and calretinin are markers consistently expressed in human SCSTs; cytokeratins (CKs), vimentin, S100, and desmin react variably in human SCSTs. 4,6 Inhibin-α alone is utilized to confirm SCSTs in dogs. 16 Macaque neoplastic granulosa cells were variably reactive for CK, vimentin, and inhibin. 12 This report evaluates the immunoreactivity of 12 ovarian SCSTs of nonhuman primates with markers commonly used in the characterization of human SCSTs.
Twelve nonhuman primate ovarian tumors, including 11 granulosa cell tumors and 1 luteoma, were diagnosed on the basis of the current World Health Organization classification for tumors of the genital system of domestic animals. 7 All examined ovaries were from zoos in the United States and submitted as formalin-fixed specimens to the Northwest ZooPath for histologic examination. For each ovary examined, 5-μm-thick sections were obtained and stained with hematoxylin and eosin and by immunohistochemistry (IHC) for calretinin (rabbit polyclonal antibody 18-0211; Zymed, San Francisco, CA), CKs (mouse monoclonal antibody, clone AE1/AE3; M3515, Dako Corporation, Carpinteria, CA), GATA-4 (goat polyclonal antibody sc-1237; Santa Cruz Biotechnology, Santa Cruz, CA), inhibin-α (mouse monoclonal antibody MCA951; AbD Serotec, Raleigh, NC), neuron-specific enolase (NSE; mouse monoclonal antibody, M0873; Dako), protein gene product (PGP) 9.5 (rabbit polyclonal Z5116; Dako), and vimentin (rabbit monoclonal antibody, clone RM-9120; Neomarkers, Lab Vision Corporation, Fremont, CA) according to procedures previously published. 14
Table 1 summarizes the relevant clinical history of the 12 ovarian SCSTs included in this study. The ovaries were from multiple species, including 7 squirrel monkeys (Saimiri spp.), 2 spider monkeys (Ateles spp.), a red-handed tamarin (Saguinus midas), a cottontop tamarin (Saguinus oedipus), a Sumatran orangutan (Pongo abelii), and a chimpanzee (Pan troglodytes). The ages of the nonhuman primates ranged from 4 to 55 years. The luteoma was from a squirrel monkey, and the control ovary was from the red-handed tamarin. Histologically, the granulosa cell tumors were similar and comprised well-demarcated islands, sheets or anastomosing cords of columnar to spindloid or polygonal cells with an oval nucleus, variation in nuclear diameter and chromatin content, indistinct nucleoli, and ample pale lacy eosinophilic cytoplasm and faint cell borders (Fig. 1). Islands of cells were separated by a fine to abundant fibrovascular stroma. Morphologically, these 11 granulosa cell tumors had features typically observed in other nonhuman primate studies. 13 The normal architecture of the ovary containing the luteoma was partially replaced by multiple lobules and packets of polygonal cells with well-demarcated cell borders, vacuolated cytoplasm, and a nucleus with coarse chromatin and variably conspicuous nucleoli. Luteinized cells were supported by a small amount of fibrovascular stroma.
Relevant Clinical Data in Nonhuman Primates With Ovarian Sex Cord–Stromal Tumorsa
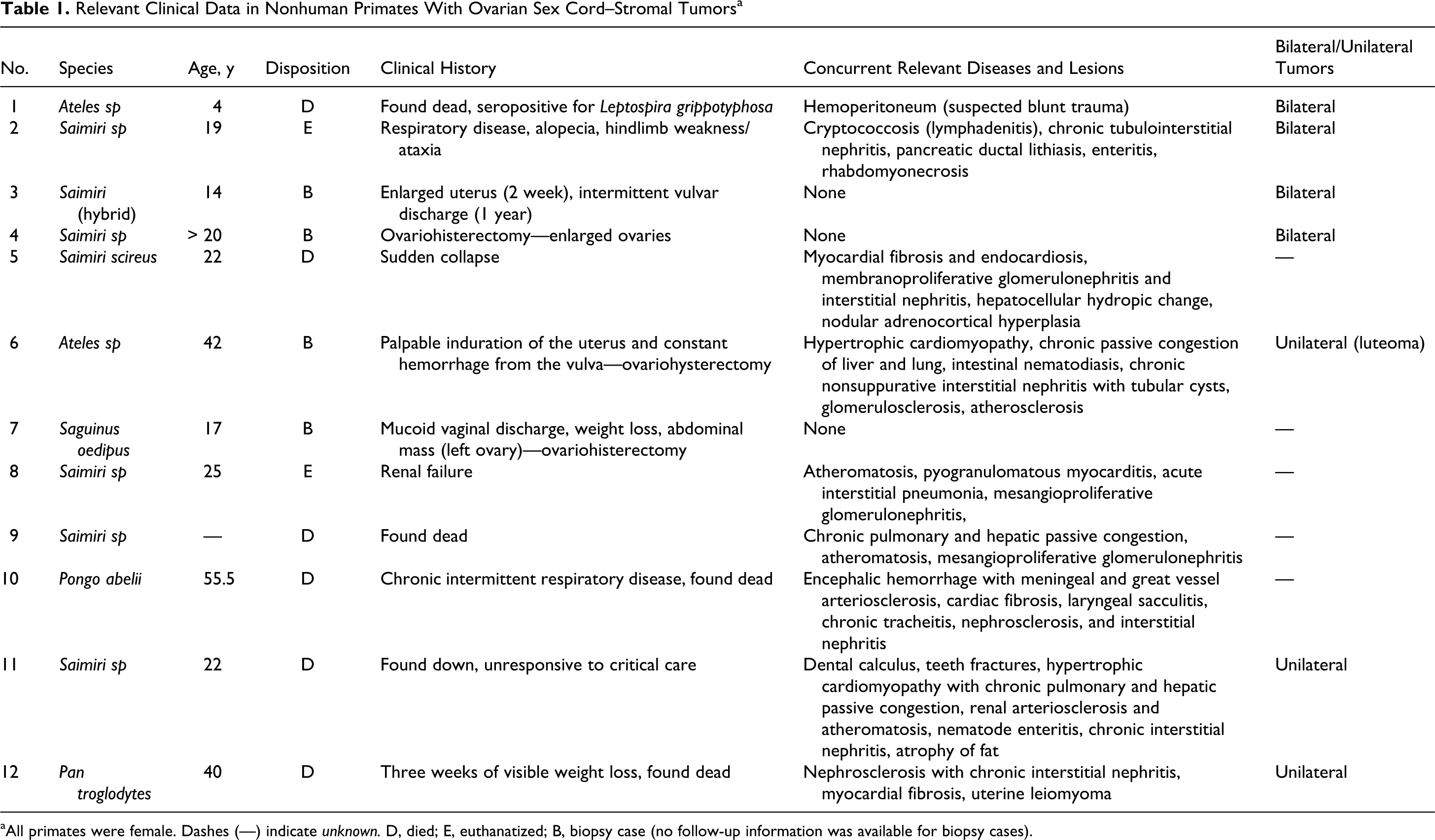
aAll primates were female. Dashes (—) indicate unknown. D, died; E, euthanatized; B, biopsy case (no follow-up information was available for biopsy cases).
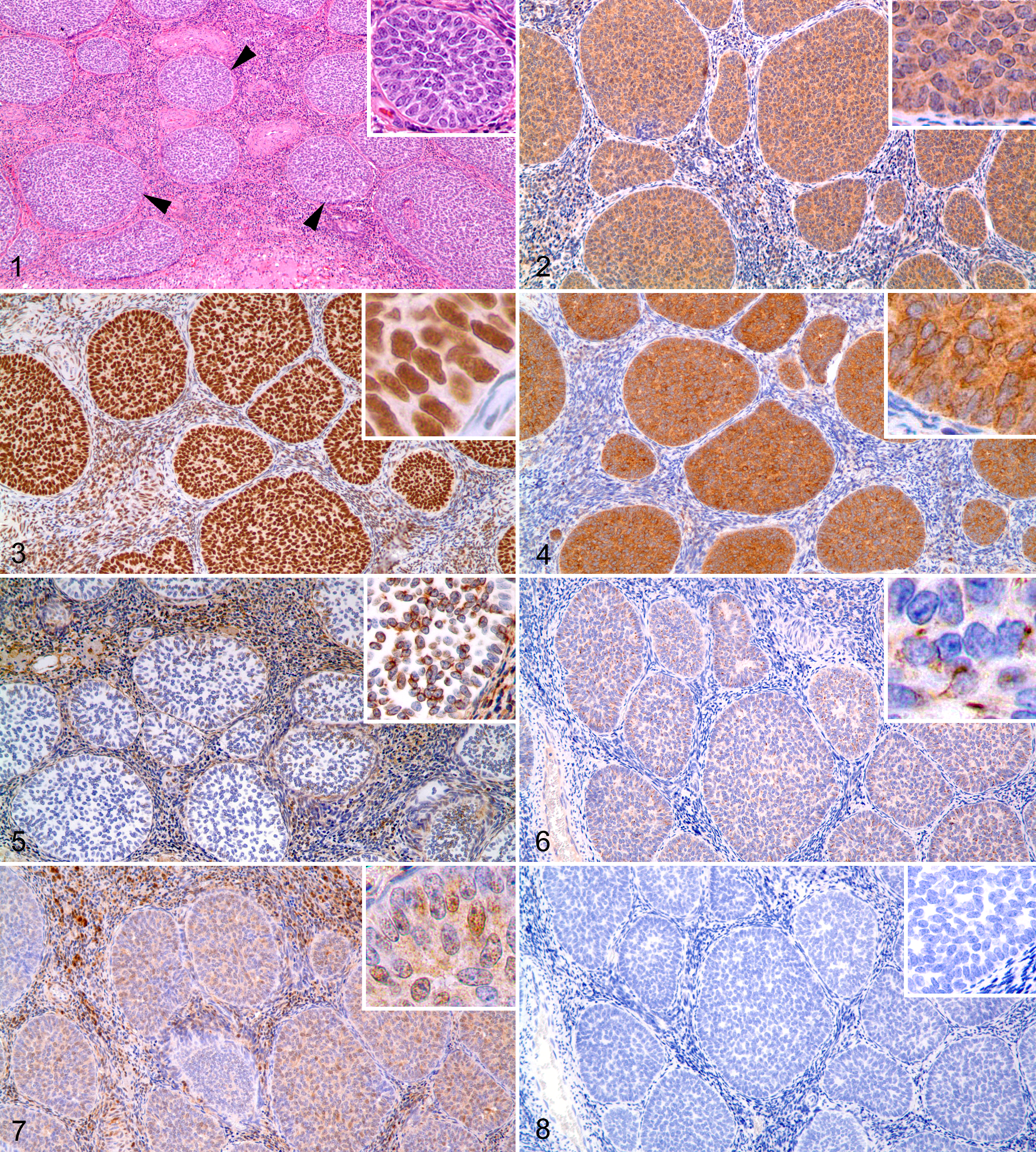
In the normal ovary, sex cord–stromal cells labeled positively with calretinin (diffuse cytoplasmic), GATA-4 (diffuse nuclear, weak cytoplasmic), NSE (diffuse cytoplasmic), PGP 9.5 (diffuse nuclear and cytoplasmic), and vimentin (weak cytoplasmic). CKs and inhibin-α did not label sex cord–stromal cells of the control ovary. Two authors (AD, JRV) independently scored each specimen for all immunohistochemical markers, and scores were averaged for the final grade. Immunolabeling was graded as follows: 0 (nonstaining), 1+ (< 10% positive cells), 2+ (10%–50% positive cells), and 3+ (> 50% positive cells).
Results of immunohistochemical studies are in Table 2. Calretinin (diffuse cytoplasmic), GATA-4 (diffuse nuclear, weak cytoplasmic), and NSE (diffuse cytoplasmic) were strongly expressed in the cytoplasm of all 12 SCSTs (Figs. 2–4). Vimentin (weak cytoplasmic) was also detected in all 12 SCSTs (Fig. 5). CKs (diffuse to focal cytoplasmic) were positively labeled in 11 SCSTs with variable intensity (grades 0–3) (Fig. 6). PGP 9.5 was expressed in 10 of 12 SCSTs with variable intensity (grades 0–3) (Fig. 7). Eight of 12 SCSTs were positive for inhibin-α with variable intensity (grades 0–3) (Fig. 8). Specifically, the sex cord–stromal cells of the only luteoma in this series expressed calretinin, CKs, GATA-4, NSE, PGP 9.5, and vimentin but did not express inhibin-α.
Five of the 7 IHC markers tested in this study (calretinin, CKs, GATA-4, NSE, and vimentin) consistently labeled nonhuman primate ovarian SCSTs. In humans, calretinin is detected in benign mesothelial cells and mesothelioma and is routinely used in an IHC panel, in conjunction with inhibin-α, to aid in the diagnosis of SCSTs. 3 –5 GATA-4 is a transcription factor that regulates gene expression and proliferation that has been shown to protect human granulosa cell tumors from apoptosis. 9,10 GATA-4 is commonly expressed in the heart, intestinal epithelium, and gonads of humans and some domestic animals. 8,14 NSE is a common neuroendocrine marker. Many carcinomas and sarcomas are typically positive for NSE; therefore, this marker lacks specificity. 11 SCSTs, especially granulosa cell tumors, can be positive for pancytokeratin, as are epithelial-derived tumors. 4 Vimentin is an intermediate filament that is characteristic of but not limited to mesenchymal tumors and therefore has limited specificity. Inhibin-α is considered a sensitive and somewhat specific marker for human and canine SCSTs. 6,15 Inhibin-α is involved in follicular growth and differentiation, and granulosa cells decrease their expression of inhibin-α as follicles differentiate. 16 The lack of immunoreactivity in 4 of 12 nonhuman primate ovarian SCSTs may indicate that sex cord–stromal cells of the unreacting tumors may originate from more differentiated follicles. An alternative explanation could be that inhibin-α is not expressed in some of the nonhuman primate species examined or that the antibody used did not cross-react with some nonhuman primate species. To evaluate these hypothesis, we examined inhibin-α expression in normal ovaries of 3 species (P. abelii, S. oedipus, and Pan troglodites); normal ovaries from other species were not available. Results indicate that at least in S. oedipus, the lack of inhibin-α expression in SCSTs is also observed in normal ovarian follicles. Normal ovaries from the other 2 species examined expressed this protein as their corresponding SCSTs.
Averages of Immunohistochemical Grades for Each Examined Nonhuman Primate Ovarian Sex Cord–Stromal Tumora
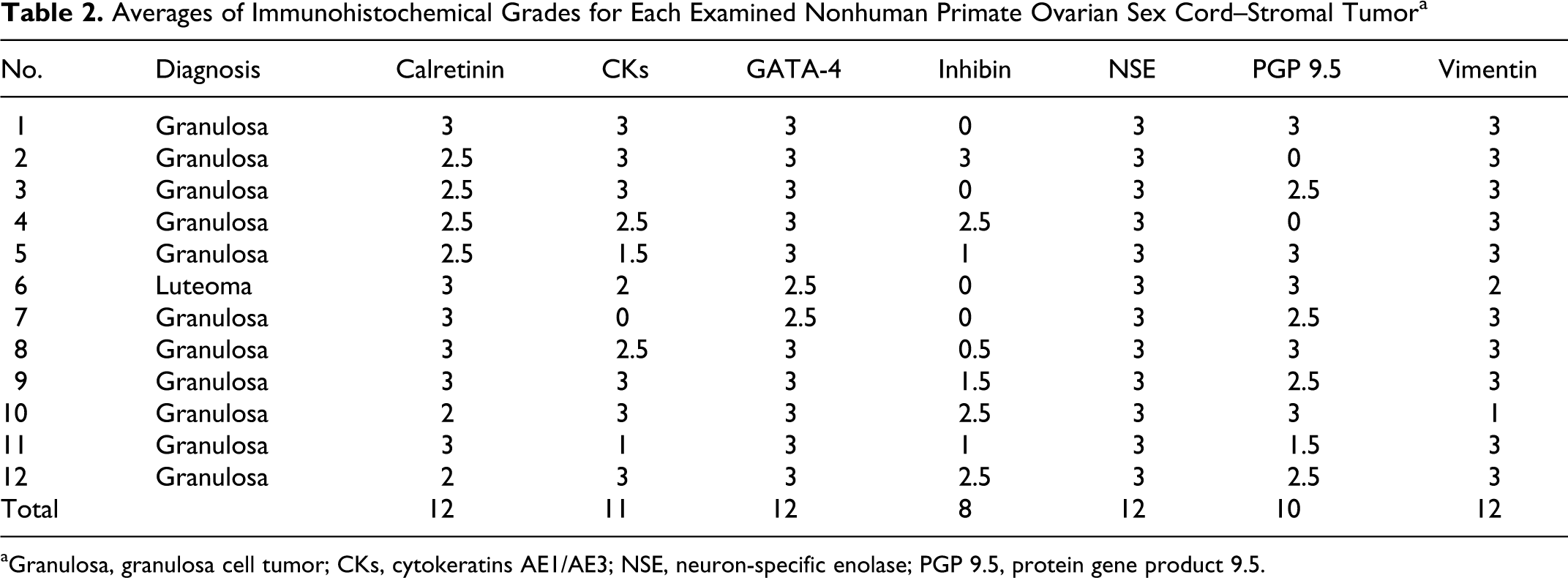
aGranulosa, granulosa cell tumor; CKs, cytokeratins AE1/AE3; NSE, neuron-specific enolase; PGP 9.5, protein gene product 9.5.
In summary, this is the first report to evaluate the immunoreactivity of 12 nonhuman primate ovarian SCST. Calretinin, GATA-4, and NSE were the most consistently expressed markers of nonhuman primate SCSTs with the strongest immunoreactivity. CKs and vimentin were also consistently expressed in SCSTs; however, the specificity of these 2 markers is low. Inhibin-α and PGP 9.5 were variably expressed suggesting less diagnostic utility.
The numbers of specimens included in this study were restricted due to limited access to nonhuman primate ovarian tissues. Further studies are needed to compare additional ovarian tumors, including primary and metastatic tumors, as well as more SCSTs to determine the utility of immunohistochemistry in the diagnosis of primary and metastatic nonhuman primate ovarian tumors.
Footnotes
Acknowledgements
We thank Dee DuSold at the Purdue University Animal Disease Diagnostic Laboratory for performing histologic and immunohistochemical procedures, and clinicians for submission of tumors.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
