Abstract
Signaling lymphocyte activation molecule (SLAM) or CD150 can function as a receptor for the canine distemper virus (CDV) in vitro. The expression of SLAM was studied using immunohistochemistry in order to evaluate the presence and distribution of the receptor in dogs in vivo. Additionally, receptor expression was assessed after experimental infection of dogs with CDV. In 7 control dogs without distemper virus, the receptor was found in various tissues, mostly on cells morphologically identified as lymphocytes and macrophages. In 7 dogs with early distemper lesions characterized by presence of the virus, higher numbers of SLAM-expressing cells were found in multiple tissues recognized as targets of CDV compared with those in control dogs. These findings suggest that SLAM, a putative distemper receptor, is expressed in dogs in vivo. Additionally, virus infection is associated with up-regulation of SLAM, potentially causing an amplification of virus in the host.
Keywords
Canine distemper virus (CDV), an enveloped, nonsegmented negative-strand ribonucleic acid virus, is, like the measles virus (MV), a member of the genus Morbillivirus and often causes a systemic and fatal disease in a variety of terrestrial mammals. 3 Distemper infection in canids affects the gastrointestinal, respiratory, and urinary tracts; skin; and the central nervous system (CNS). 1
Virus entry into the cell occurs via specific receptors. The exact nature of the cellular receptor for CDV is under debate, whereas the CD46 molecule has been identified as a receptor for MV in humans. 4, 7, 8, 11 This receptor is used by attenuated measles strains tested, but only by a few wild-type virus strains. 4 This is in contrast to another MV receptor, the signaling lymphocyte activation molecule (SLAM), which allows virus entry of all MV strains tested, including the wild-type strains. 4 SLAM, which is also known as CD150, is a membrane glycoprotein involved in lymphocyte activation and expressed on various cells of the human immune system. 9, 12– 14, 16
The identity of the receptor used by CDV for binding and entry into cells in dogs is under debate. CD46 and CD9 were discussed as potential receptors for CDV at one time, but both receptors appear not to be involved in CDV infections. 7, 11 In contrast, cells expressing the SLAM receptor become susceptible to virulent and attenuated vaccine strain CDV in vitro. 6 So far, receptor distribution in canine tissues, which is most likely important in distemper pathogenesis, has not been studied. Therefore, we analyzed SLAM expression in dogs, using immunohistochemistry.
Samples of footpad, mesenteric lymph node, stomach, intestine, lung, brain, and urinary bladder were collected from 14 dogs, 7 control dogs (Nos. 1–7), and 7 distemper dogs (Nos. 8–14). Two of the control dogs were not vaccinated against CDV; the vaccination status of the other controls was unknown. These dogs were euthanized or died due to unrelated diseases. Seven other dogs (Nos. 8–14) were animals kept according to the guidelines of the institution's animal care and use committee and were inoculated with the virulent distemper strain A75/17 as described. 2 The dogs were tested by serology negatively for anti-CDV antibodies prior to the inoculation. These animals were euthanized when they showed clinical signs of distemper, including fever, weight loss, diarrhea, and disturbances of the CNS as described. 5 Tissues were collected at necropsy, fixed in formalin, and paraffin-embedded.
Sections were cut and stained with HE for routine histopathologic evaluation. For immunohistochemical assessment, consecutive tissue sections were prepared and stained. An antibody against canine SLAM was prepared using canine SLAM protein expressed in vitro, which was purified and injected into rabbits. In detail, a polymerase chain reaction product of the ectodomain of the DogSLAMtag gene (plasmid kindly provided by Y. Yanagi, Department of Virology, Faculty of Medicine, Kyushu University, Japan) was ligated into a pQE16 vector (Qiagen, Hombrechtikon, Switzerland). The vector was transformed into Escherichia coli M15-competent cells. The sequence was verified by deoxyribonucleic acid (DNA) sequencing analysis. Protein expression was induced by addition of isopropyl β-D-1-thiogalactopyranoside. The cells were harvested and resuspended. Lysozyme was added to the final concentration of 1 mg/ml. The cells were sonicated, centrifuged, and re-dissolved multiple times. The final supernatant was loaded onto a Ni-NTA spin column (Qiagen) and washed. Protein was then eluted and quantified. For generation of antisera directed against the recombinant DogSLAM ectodomain, rabbits were immunized. Control sera of rabbits were collected from the same rabbits before immunization.
A Chinese hamster ovary (CHO) clone stably expressing the molecule was generated as earlier described. 15 The dogSLAMtag gene was cloned into a pcDNA-Neo vector (Invitrogen, Basel, Switzerland). After transfection of this plasmid, CHO cells were incubated in medium containing 1 mg/ml of G418 (Invitrogen). Three weeks later, resistant colonies were sorted by fluorescence-activated cell sorter analysis using anti-HA-FITC antibody (Sigma, Buchs, Switzerland) to select the clone expressing the highest amount of SLAM at the cell surface.
Antibodies were tested using Western blot analysis and immunohistochemistry. For the Western blot analysis, proteins from Vero cells expressing the canine SLAM molecule (VerodogSLAMtag, a kind gift from Veronika von Messling) 19 and nontransfected Vero cells were separated by electrophoresis on an SDS-polyacrylamide gel and blotted onto nitrocellulose transmembranes (Schleider and Schuell, Dassel, Germany). The membrane was incubated with control sera and rabbit anti-DogSLAM antibodies. After incubation with peroxidase-conjugated goat anti-rabbit immunoglobulin G (IgG) (Dako, Zug, Switzerland) for 45 minutes at room temperature, proteins were visualized by enhanced chemiluminescence (Amersham Biosciences, Oetelfingen, Switzerland). Binding of secondary reagents was assessed using an irrelevant antibody (rabbit anti-CDV-F-protein). 10
Transfected CHO cells served as a positive control for the immunohistochemistry. Cell pellets were prepared, fixed in formalin, paraffin-embedded; and sections were cut. Canine distemper virus nucleoprotein was demonstrated using antibody D110. 5 For negative controls, rabbit IgG was used.
Tissue sections were deparaffinized, and endogenous peroxidase was blocked with hydrogen peroxide. Sections were incubated with the primary antibody in a humid chamber following the appropriate pretreatment. This was pronase (tris[hydroxymethyl]aminomethane buffer 10% and 1% proteinase, pH 7.6, 3 minutes at 37°C, (FLUKA, Buchs, Switzerland)) for the anti-SLAM antibody, which was used at a concentration of 1 : 1,000. Distemper nucleocapsid was demonstrated using D110 after heat-induced antigen retrieval (citrate buffer: 0.01M, pH 6.00, microwave 2 × 5 minutes at 750 W) at a concentration of 1 : 5. After application of the secondary biotinylated antibody (LSAB2-Kit, Dako) and streptavidin, the color indicator amino-9-ethylcarbazole was used.
Positively SLAM-labeled cells were counted in the lung, gastrointestinal tract, transitional epithelium, and footpads in 10 standardized microscopic fields of a size of 0.0961 mm. 2 These fields were defined using an ocular morphometric grid and the 40× lens. In order to account for the irregularity of rete ridges of the footpad epithelium, which did not fill a grid at the chosen magnification, 20 fields were counted. The results were statistically analyzed using an unpaired t-test with a Welch correction. Observed differences with a P value of less than .05 were considered significant. As there were only very rare SLAM-positive cells in the brain, these were not counted.
There were no histopathologic changes in the organs from the control dogs (Nos. 1–7). Dogs (Nos. 8–14), which were inoculated with CDV, showed histopathologic changes in all organs examined. Lesions in the brain were mild and consisted of vacuolization of the nervous tissue, mainly in the white matter surrounding the fourth ventricle and of the cerebellar folia, usually without signs of inflammation. Therefore, these lesions were interpreted to represent typical acute distemper changes as described in other studies. 17 The changes within the footpads were mild and consisted of a few inflammatory cells, mainly perivascularly located lymphocytes, as have been described previously. 5 The lymph nodes had moderate lymphoid depletion, usually generalized, and rare syncytial cells. The dogs had a mild interstitial pneumonia with rare intracytoplasmic inclusion bodies within bronchial epithelial cells. The epithelial cells of the urinary tract were characterized by abundant intracytoplasmic inclusion bodies; inflammation was scant. Comparably, many intracytoplasmic and intranuclear inclusion bodies were the main findings in the gastrointestinal tracts of most dogs (Nos. 9–11, 14), whereas inflammation was not prominent. 5
Distemper viral nucleoprotein was absent in any tissue examined from the control dogs (Nos. 1–7). CDV nucleoprotein was identified in inoculated dogs (Nos. 8–14) in all organs studied. Positivity in the brain was restricted to areas with histopathologic changes. Abundant positive cells were found in the footpads (epidermis, sweat glands, endothelial cells), lymph nodes (mainly cells in the parafollicular zone), and urinary tracts (transitional epithelium). Lesser numbers were present in the lung (bronchiolar respiratory epithelium, interstitial lymphocytes) and gastrointestinal tract (epithelial cells, mucosal lymphocytes).
The Western blot analysis demonstrated a strongly positive band with a size in the expected range (according to the predicted amino acids sequence) 15 when protein isolated from SLAM-transfected Vero cells was used, whereas the control cells were negative (Fig. 1). An irrelevant antibody (rabbit anti-CDV-F-protein antibody) had no positive reaction (data not shown). Control CHO cell pellets were negative, whereas CHO cells, which were transfected and expressed canine SLAM, had positive signal in the immunohistochemical staining (Figs. 2, 3).
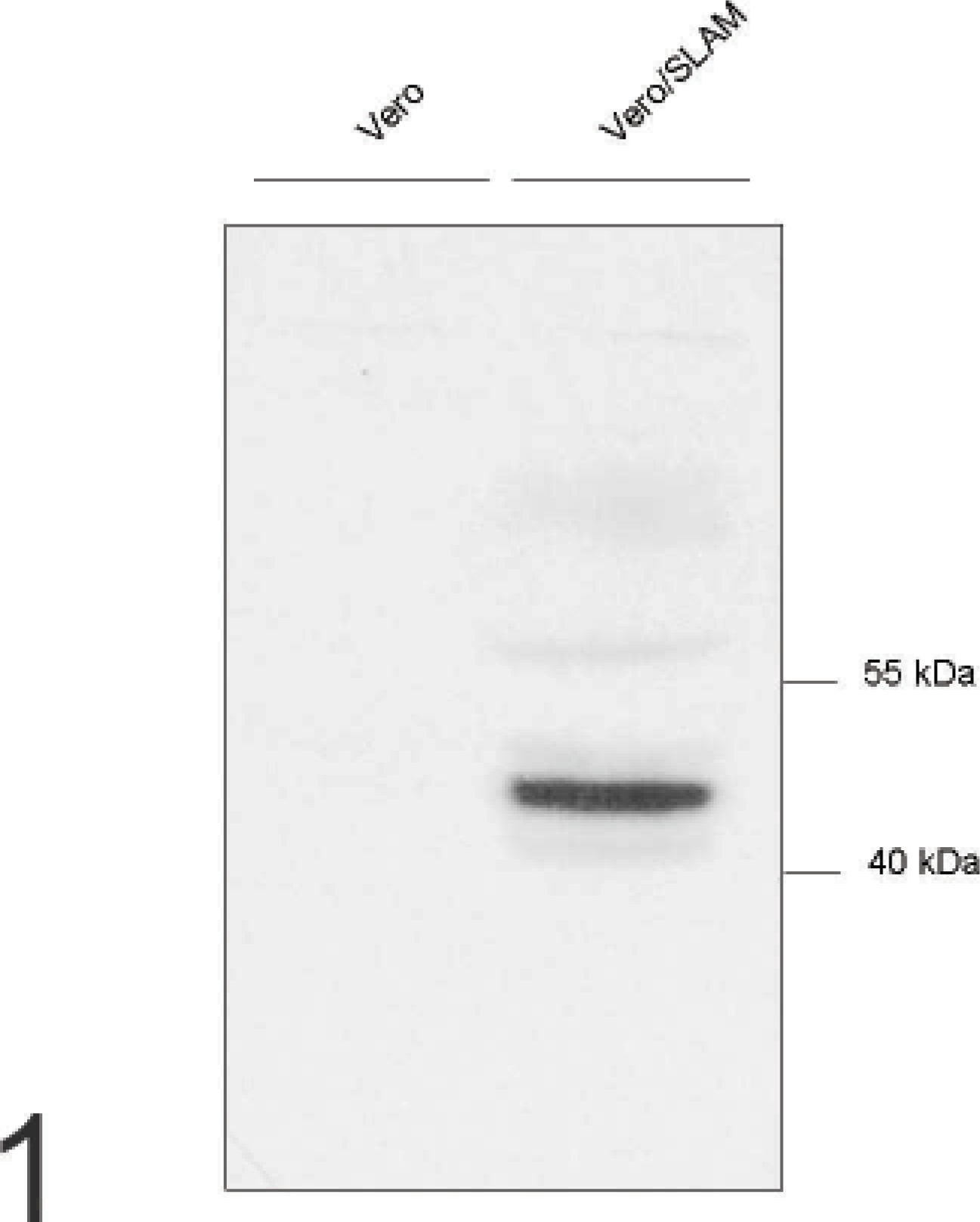
Vero cells, analyzed by Western blot; rabbit anti-canine signaling lymphocyte activation molecule antibody labels 1 strong band in Vero cells transfected with canine signaling lymphocyte activation molecule (right lane) but not in nontransfected control Vero cells (left lane).
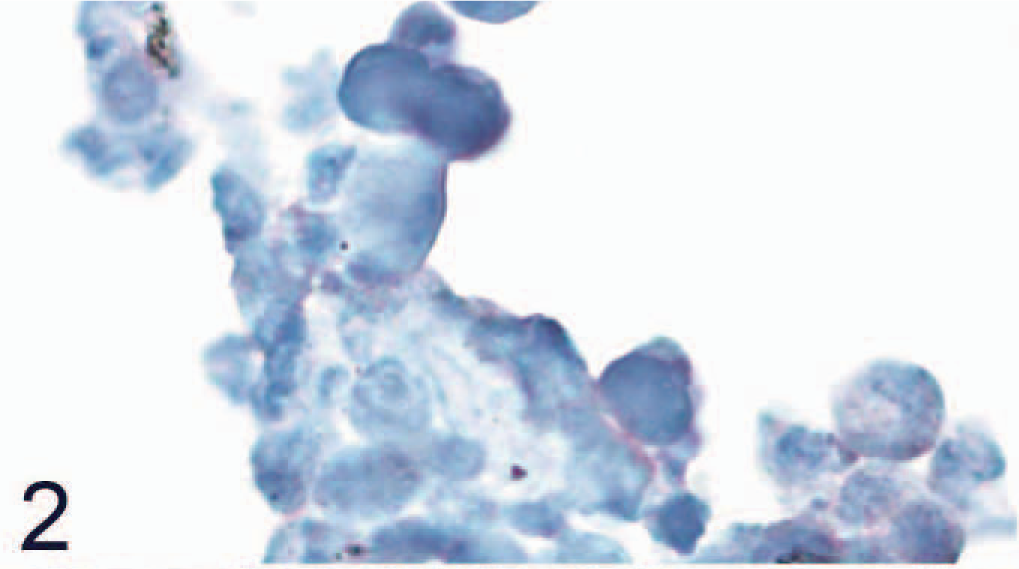
Nontransfected control Chinese hamster ovary cells that do not express canine signaling lymphocyte activation molecule (SLAM) are negative for SLAM by avidin-biotin-peroxidase complex immunohistochemistry. Rabbit anti-canine SLAM antibody and AEC, counterstain with hematoxylin.
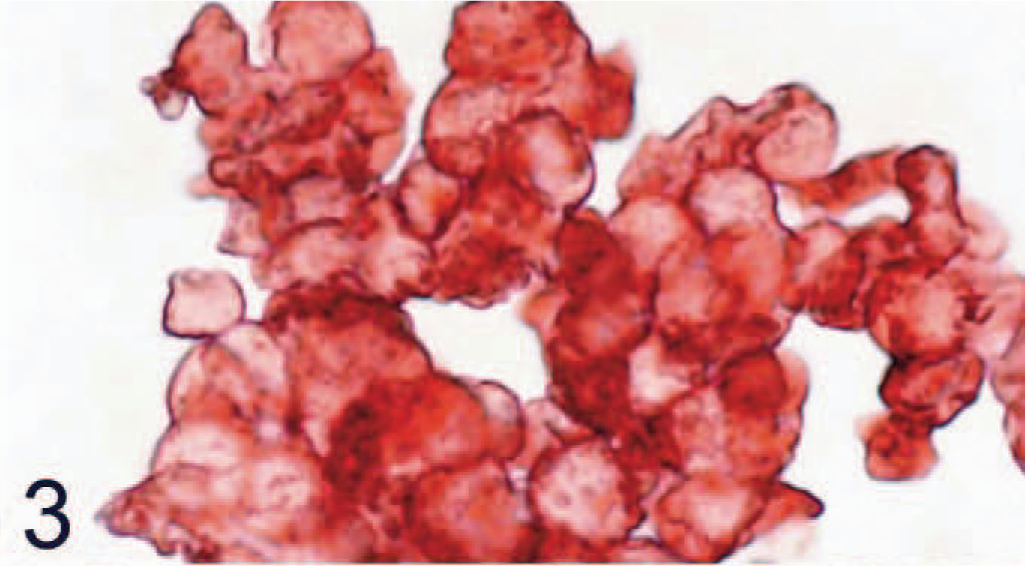
Chinese hamster ovary cells transfected with canine signaling lymphocyte activation molecule (SLAM) are all positive for SLAM by avidin-biotin-peroxidase complex immunohistochemistry. Rabbit anti-canine SLAM antibody and AEC, counterstain with hematoxylin.
SLAM labeling was also found in multiple organs, including footpad lymph nodes (Figs. 4, 5), lung (Figs. 6, 7), transitional cell epithelium of the urinary tract, and in the stomachs and intestines of control dogs (Nos. 1–7) and CDV-infected dogs (Nos. 8–14). The amount of positive cells within these organs is detailed in Fig. 8. The number of SLAM-positive cells was higher in CDV-infected dogs compared with control dogs; this was statistically significant in all organs but the stomach, which exhibited only a numeric difference. Morphologically, SLAM-positive cells resembled lymphocytes and macrophages. In addition, SLAM positivity was occasionally observed in endothelial cells and in transitional epithelial cells. Footpad epidermis was not positive for SLAM in control or CDV-infected dogs.

Lymph node; dog No. 3. Canine distemper virus–negative. Only a few cells are positive for signaling lymphocyte activation molecule (SLAM) by avidin-biotin-peroxidase complex immunohistochemistry. Rabbit anti-canine SLAM antibody and AEC, counterstain with hematoxylin.
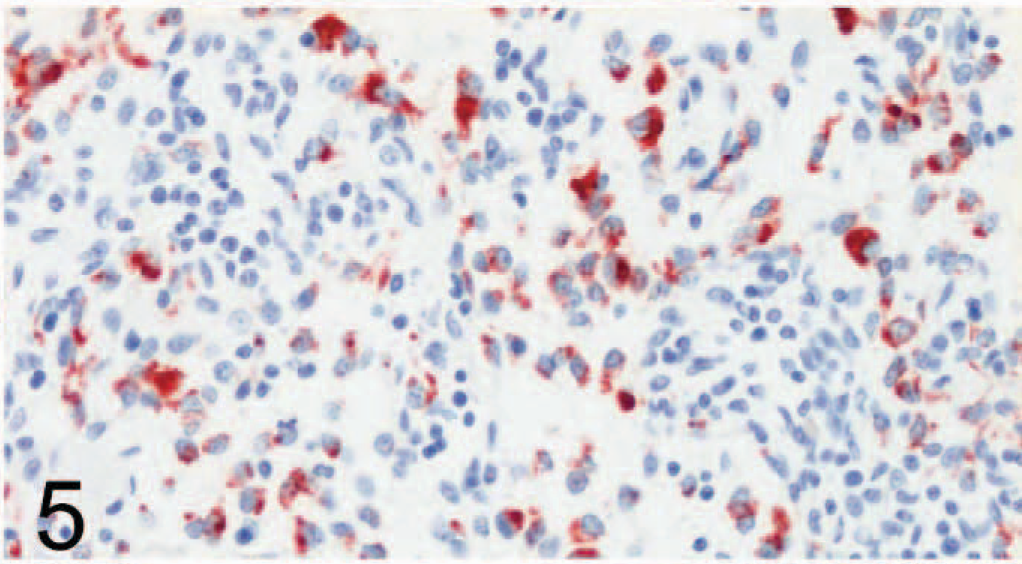
Lymph node; dog No. 9. Canine distemper virus–positive. Many cells are positive for signaling lymphocyte activation molecule (SLAM) by avidin-biotin-peroxidase complex immunohistochemistry. Rabbit anti-canine SLAM antibody and AEC, counterstain with hematoxylin.
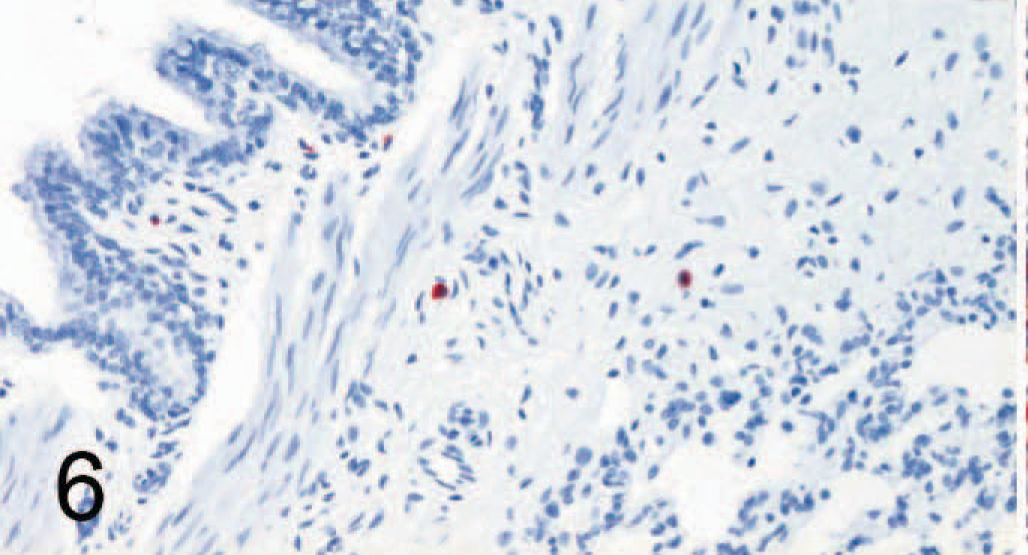
Lung; dog No. 3, Canine distemper virus–negative. Only rare cells are positive for signaling lymphocyte activation molecule (SLAM) by avidin-biotin-peroxidase complex immunohistochemistry. Rabbit anti-canine SLAM antibody and AEC, counterstain with hematoxylin.
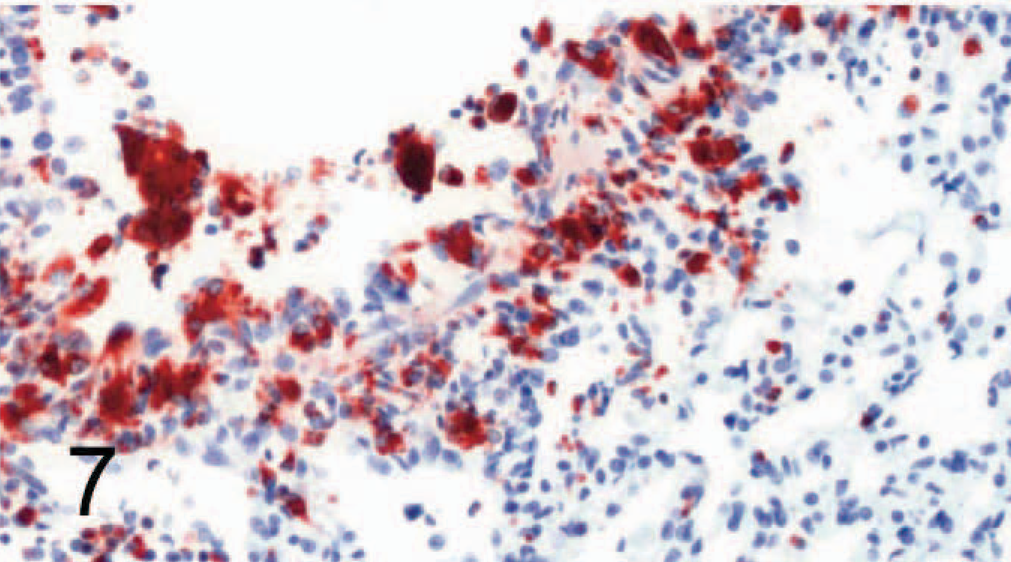
Lung; dog No. 8. Canine distemper virus–positive. Many cells, mainly around bronchioli, are positive for signaling lymphocyte activation molecule (SLAM) by avidin-biotin-peroxidase complex immunohistochemistry. Rabbit anti-canine SLAM antibody and AEC, counterstain with hematoxylin.
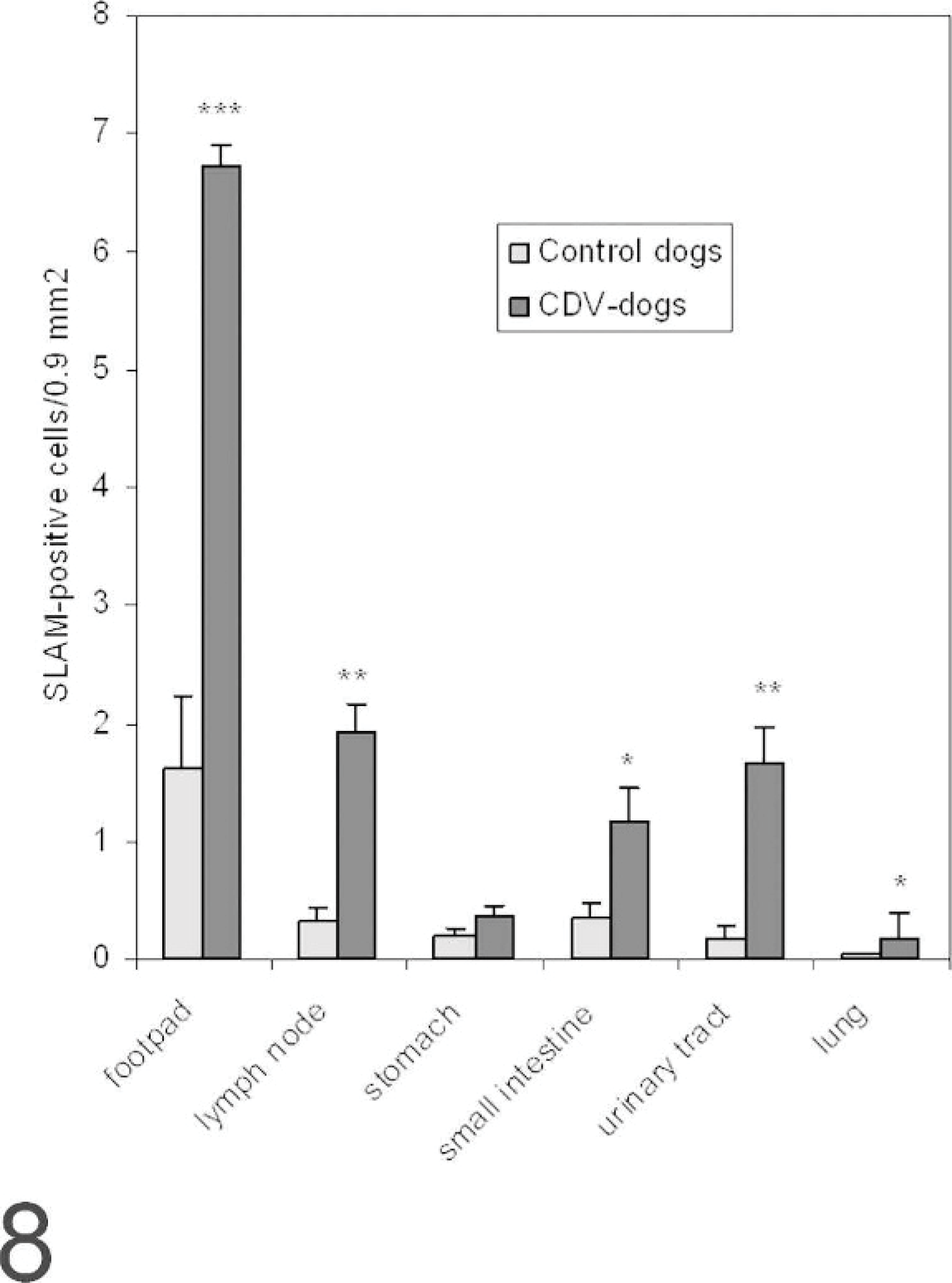
Distribution of signaling lymphocyte activation molecule (SLAM)–positive cells in various organs known as canine distemper virus (CDV) target organs. The number of cells positive for SLAM by immunohistochemistry is higher in all organs tested in CDV-positive dogs; this difference was statistically significant in the footpad, lymph node, small intestine, urinary tract, and lung. (mean ± SEM, ∗∗∗ = P < .001, ∗∗ = P < .01, ∗ = P < .05).
In brains of control dogs, only rare individual cells within blood vessel walls of the brain stained positively for SLAM; the parenchyma did not show positivity. In the brains of CDV-infected dogs, the areas characterized by histopathologic changes in the white matter surrounding the fourth ventricle, within the medulla oblongata and the cerebellar folia, which also contained nucleoprotein, were positive for SLAM (Figs. 9, 10).
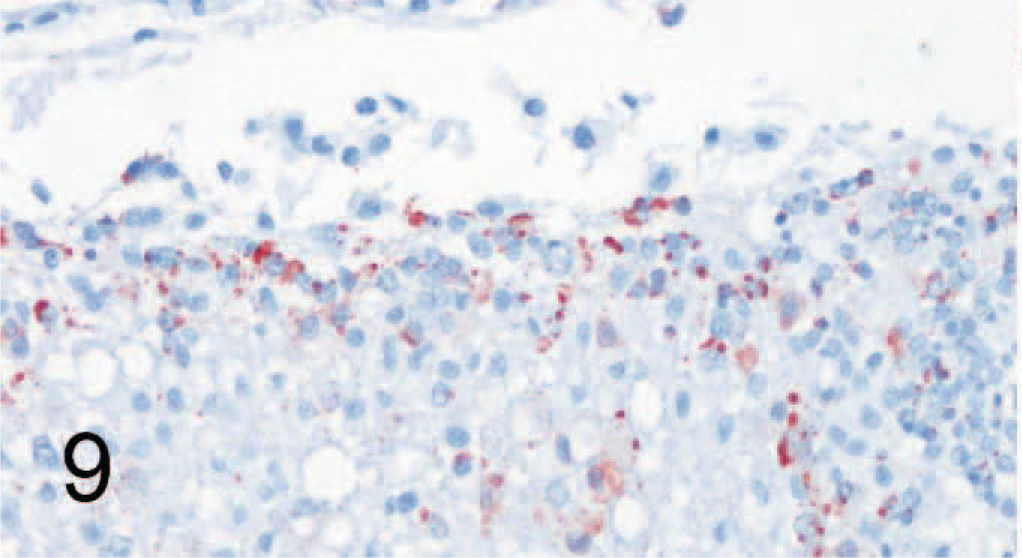
Brain (serial section of tissue depicted in Fig. 10); dog No. 9. Canine distemper virus (CDV)–positive. A moderate amount of cells in the neuropil are positive for CDV nucleoprotein by avidin-biotin-peroxidase complex immunohistochemistry. Antibody D110 and AEC, counterstain with hematoxylin.
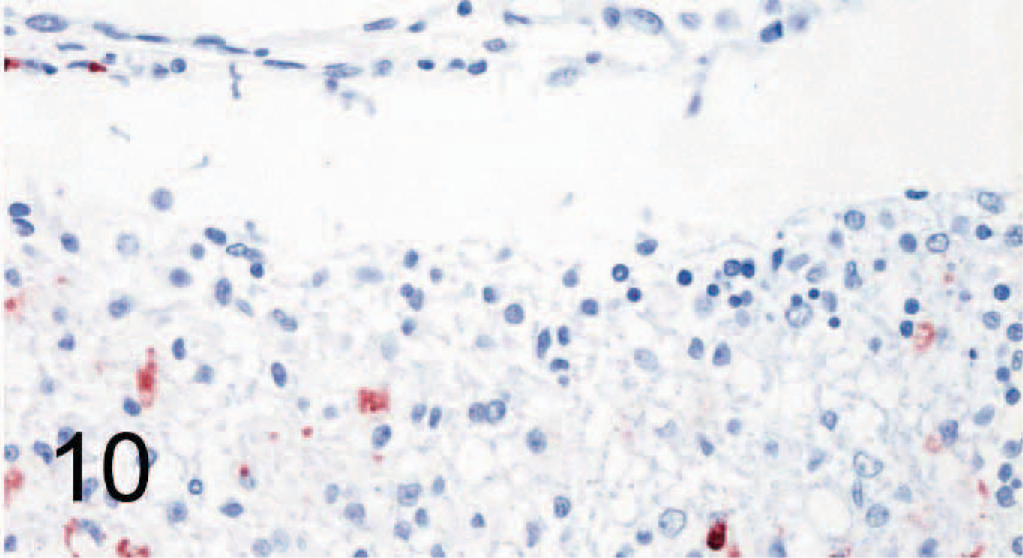
Brain (serial section of tissue depicted in Fig. 9); dog No. 9. Canine distemper virus–positive. Cells in the same location as depicted in Fig. 9, but in lower numbers, are positive for canine signaling lymphocyte activation molecule (SLAM) by avidin-biotin-peroxidase complex immunohistochemistry. Rabbit anti-canine SLAM antibody and AEC, counterstain with hematoxylin.
So far, SLAM has been identified as a potential receptor for CDV in dogs in vitro only. 6, 12 Until now, the role of SLAM as a CDV receptor was described in ferrets. 18 In this study, SLAM was demonstrated in various canine tissues in vivo in noninfected control and CDV-infected dogs using immunohistochemistry. This finding suggests that this molecule may serve as a receptor for CDV in natural distemper infection in vivo. SLAM was present on cells morphologically resembling lymphocytes and macrophages from control dogs in various tissues. These cells are well-known targets of CDV, and the virus could potentially use SLAM as a receptor in these cells during the early infection. The presence of SLAM in CDV-negative control dogs indicates that this molecule could represent one early receptor encountered in CDV infection. Additionally, SLAM expression was found on transitional urothelial cells of control dogs, which again are commonly infected in distemper virus infection.
In brains of noninfected control dogs, only very few cells within blood vessel walls stained positively for SLAM. After CDV infection, abundant SLAM expression could be demonstrated within the lesions. This finding suggests that in dogs, as has been observed in humans with subacute sclerosing panencephalitis, Morbillivirus infection of the brain results in increased SLAM-positivity in lesions.
Up-regulation of SLAM expression was observed in early CDV-infection of experimentally infected dogs in many tissues known to be target tissues of CDV. Receptor up-regulation might render more cells susceptible towards a CDV-infection, thus leading to a potentially higher viral load and increase in lesions. MV was reported to either down- or upregulate SLAM expression on cells. 9, 13 It is unclear at the moment whether the up-regulation of SLAM in dogs in contrast to the observed down-regulation after MV infection is due to differences of the virus (CDV versus MV) or the host (dog versus human) or reflects differences between the in vivo and in vitro situation.
Interestingly, SLAM could not be identified by the method used in the epidermis of footpad keratinocytes. These represent well-known targets for CDV and were also strongly positive for CDV nucleocapsid in the distemper dogs used in this study. This finding suggests the existence of another molecule that allows viral entry into these cells. Therefore, the presence of a second, hitherto unidentified, receptor for CDV in the dog can be postulated.
In summary, SLAM or CD150 is expressed in normal dogs and therefore can serve as a receptor for CDV infection in vivo. In dogs with acute CDV infection, an up-regulation of the receptor was observed. This indicates the possibility of existence of a cycle that could result in amplification of the virus in the host.
Footnotes
Acknowledgements
This work was in part supported by grants from the Swiss National Science Foundation (31-58657.99 and 32-61961.00). We thank Y. Yanagi and V. von Messling for the kind donation of plasmid and cell line, respectively.
