Abstract
A 5-month-old mongrel puppy with a history of respiratory disease presented with progressive neurologic dysfunction. Hematologic results included leukocytosis (neutrophilia with a left shift) and lymphopenia. A mass in the right forebrain, identified by magnetic resonance imaging, was biopsied during decompressive craniectomy. The histologic diagnosis was granulomatous meningoencephalitis with intralesional amoebae. The dog died within 24 hours of surgery. At necropsy, a well-demarcated granuloma was confined to the cerebrum, but granulomatous pneumonia was disseminated through all lobes of the lung. Concurrent infections included canine distemper, canine adenoviral bronchiolitis, and oral candidiasis. Canine distemper virus probably caused immunosuppression and increased susceptibility to secondary infections.
History, Clinical Findings, Diagnostic Imaging, and Surgical Pathology
A 5-month-old, intact female, mongrel dog, which was born in Texas and moved to Indiana, had a history of chronic respiratory disease with more recent neurologic dysfunction. Neurologic signs had progressed to an obtunded mental status, nonambulatory tetraparesis with loss of conscious proprioception in the hind legs, ventral strabismus, absent menace response, and myoclonus in the face and limbs.
Hematologic results included leukocytosis (neutrophilia with a left shift) and lymphopenia. Upon magnetic resonance imaging, a rim-enhanced, heterogeneous mass with perilesional edema was evident in postcontrast images in the right forebrain (frontal/parietal lobes).
The differential diagnosis for the cerebral mass included brain abscess associated with bacterial invasion or an expanding middle ear infection, fungal granuloma, and embryonal neoplasms. 8 The puppy was scheduled for decompressive craniectomy to debulk the mass and obtain a diagnostic biopsy specimen. At surgery, abscess was ruled out based on the gross appearance and texture of the mass, which harbored a mucinous content rather than purulent exudate. Histologically, the cerebral parenchyma was heavily infiltrated by macrophages with fewer neutrophils and lymphocytes and generally without organization into discrete granulomas. Additionally, there were numerous (1–10 per 400× field), spherical, unicellular organisms that ranged from 10 to 25 μm in diameter. The organisms were consistent with amoebae, with a nucleus, a large intensely stained endosome, and abundant vacuolated cytoplasm (Fig. 1). The histologic diagnosis for the biopsy specimen was granulomatous meningoencephalitis with intralesional amoebae; the differential diagnosis was narrowed to cerebral amoebiasis attributable to Acanthamoeba spp, Balamuthia mandrillaris, or Naegleria fowleri. The dog died approximately 24 hours postoperatively.
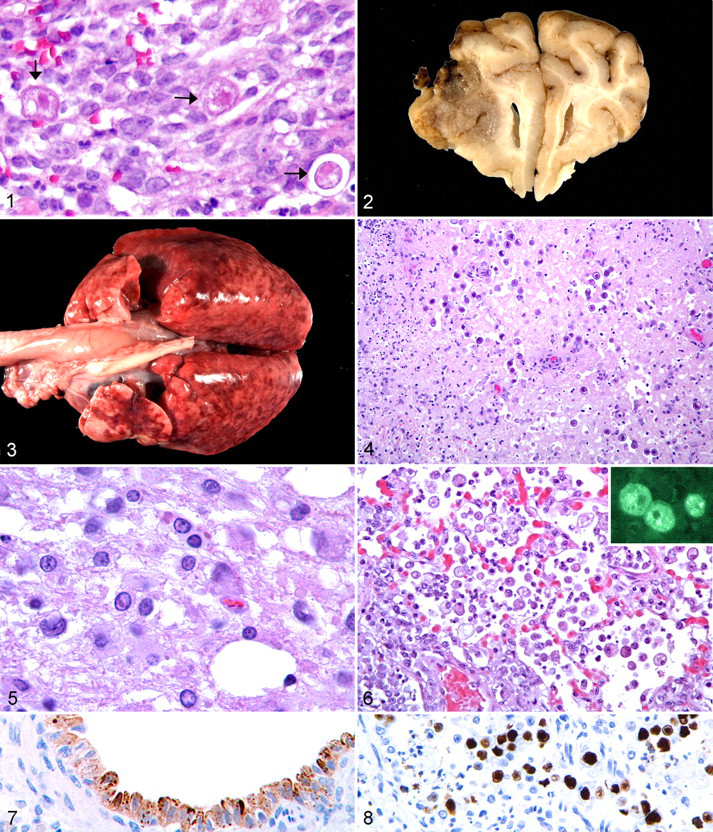
Necropsy Findings
At necropsy, the cerebrum contained a dark brown to tan, 1.3-cm (diameter), roughly spherical mass that extended from the dorsolateral aspect of the right frontal lobe into the thalamus (Fig. 2). The lungs were poorly collapsed and mottled pink to dark red; all lobes had numerous pinpoint to 3-mm (diameter) coalescing red to gray-tan nodules (Fig. 3). Other pertinent gross findings included thymic atrophy and pale-tan to dark brown mottling of the liver.
Histopathologic Findings
Histologically, in the cerebral granuloma, the gray and white matter was effaced by coalescing foci of necrotizing granulomatous inflammation with 5–10 amoebae per high-power field. The amoebae had a prominent endosome with abundant vacuolated cytoplasm (trophozoites) or eosinophilic amorphous cytoplasm with a dense basophilic refractile wall (cysts) (Fig. 4). In adjacent, less affected neuropil, astrocytes were prominent; neuronal somata were swollen or shrunken and hypereosinophilic. Few swollen axons (spheroids) were associated with the affected gray matter. Some vessels were surrounded by macrophages. Focally in the white matter, especially in the internal capsule, but also in the corona radiata, there was spongiosis with dilated myelin sheaths; eosinophilic intranuclear inclusion bodies were observed in some astrocytes in these areas. The meninges, mainly the pia mater, were expanded by increased numbers of lymphocytes, plasma cells, macrophages, and amoebae (mostly cysts). A few meningeal vessels were expanded by poorly organized thrombi. In addition to the internal capsule and corona radiata, spongiosis was noted in white matter of the cerebellum and pons, with dilated myelin sheaths, decreased staining with Luxol fast blue, astrogliosis, and microgliosis. Eosinophilic intranuclear inclusions were observed in astrocytes in these areas (Fig. 5).
In the lung, numerous amoebic trophozoites and cysts were associated with granulomatous pneumonia (Fig. 6). Some bronchioles were filled with cellular debris, sloughed epithelial cells, and a few neutrophils and macrophages. Amphophilic to eosinophilic intranuclear inclusions (8–12 μm in diameter) were observed especially in the sloughed bronchiolar epithelial cells. In areas of the lung that were less severely affected by granulomatous pneumonia, there was interstitial pneumonia; bronchiolar epithelial cells and macrophages in these areas contained 5–6 μm, eosinophilic, mostly cytoplasmic inclusions. Other pertinent histologic findings included lingual hyperkeratosis and candidiasis, tracheal squamous metaplasia, multifocal hepatic necrosis, and splenic and nodal lymphoid depletion and histiocytosis.
Immunohistochemistry, Immunofluorescence, and Molecular Diagnostics
Based on the postmortem histologic findings, granulomatous meningoencephalitis and pneumonia were attributed to amoebiasis. Infection with canine distemper virus was considered the cause for demyelinating encephalitis and interstitial pneumonia; canine distemper viral antigen was identified immunohistochemically using Mouse:CDV-NP (1:200) from Veterinary Medical Research and Development in affected cerebellar and pulmonary parenchyma (Fig. 7). Necrotizing bronchiolitis was attributed to infection with canine adenovirus; adenoviral antigen using Mouse 20/11 and 2/6 (1:4000) from Chemicon was detected immunohistochemically in affected airways (Fig. 8).
To identify the amoebae, immunofluorescence and real-time polymerase chain reaction (PCR) tests were conducted on lung tissue by the National Center for Zoonotic, Vector-Borne and Enteric Diseases at the Centers for Disease Control and Prevention (Atlanta, GA). Immunofluorescence testing was positive with polyclonal anti-Acanthamoeba castellanii antibodies (Fig. 6 inset), and negative with anti-Naegleria fowleri and anti-Balamuthia mandrillaris antibodies. Similarly, with real-time PCR tests, the lung was positive for Acanthamoeba spp and negative for Balamuthia mandrillaris and Naegleria fowleri.
Discussion
Acanthamoeba sp. are free-living protists, commonly found in fresh water, salt water, soil, dust, and sewage. 1,6 The life cycle consists of a free-living cyst and a vegetative trophozoite that feeds on bacteria in aquatic environments and acts as an opportunistic pathogen. Canine infections are caused by Acanthamoeba castellanii, Acanthamoeba culbertsoni, and Acanthamoeba sp. Genotype T1. 6,9 Further classification beyond the genus was not pursued in this case.
Acanthamoeba species typically infect the lung and brain. 1,5 Systemic acanthamoebiasis is opportunistic and typically a result of inhalation of cysts from the air or exposure to the organisms while swimming in contaminated water. Spread to the brain can occur via the olfactory nerves or hematogenously from a primary lung infection. Acanthamoeba epizootics have been described in greyhounds, and single cases have been observed in German Shepherd Dogs, Akitas, Labrador Retrievers, and mixed-breed dogs. 1,6
Acanthamoeba spp are ubiquitous, but acanthamoebiasis often occurs in the southeastern United States. Thus, a critical part of the history in this case is the dog’s origin in Texas. In addition to encephalitis and pneumonia, Acanthamoeba can cause keratitis in healthy humans, especially those who use soft contact lenses. Humans with organ transplants, acquired immunodeficiency syndrome (AIDS), diabetes mellitus, or other immunosuppressive conditions are susceptible to disseminated acanthamoebiasis. 6,7
Young dogs, typically those with an underlying disease, are predisposed to acanthamoebiasis. Although acanthamoebiasis was the major infection in this puppy, the initial pathogen was probably canine distemper virus, which resulted in immunosuppression and increased susceptibility to canine adenovirus-2 and Acanthamoeba infection; however, congenital immunosuppression may have resulted in increased susceptibility to disease. 2 –4 Canine distemper virus, a morbillivirus, commonly results in respiratory and gastrointestinal disease, immunosuppression, and demyelinating leukoencephalomyelitis. 2
Footnotes
Acknowledgements
We thank Dee DuSold and the Animal Disease Diagnostic Laboratory histopathology section for histologic and immunohistochemical preparations and Rama Sriram and Rebecca Bandea of the National Center for Zoonotic, Vector-Borne and Enteric Diseases for technical help with the immunofluorescence and real-time PCR tests.
The authors declared that they had no conflicts of interests with respect to their authorship or the publication of this article.
The authors declared that they received no financial support for their research and/or authorship of this article.
