Abstract
Multiple myeloma oncogene 1/interferon regulatory factor 4 (MUM1/IRF4) is involved in lymphoid cell differentiation, particularly in the production of plasma cells. We examined the immunoreactivity of mouse monoclonal antibody Mum-1p to MUM1/IRF4 and compared it with expression of CD79a and CD20 in 109 plasmacytomas in 107 dogs. Tissues had been fixed in formalin and embedded in paraffin. One hundred one of 109 (93.5%) tumors were positive for MUM1/IRF4. The staining was nuclear with weak cytoplasmic reaction. Fifty-nine of 105 (56.2%) plasmacytomas were positive for CD79a; only 21 of 108 (19.4%) cases were positive for CD20. MUM1/IRF4 staining was performed on 139 other tumors including B- and T-cell lymphomas, histiocytic proliferations, mast cell tumors, and melanocytic tumors. The only MUM1/IRF4-positive nonplasmacytic tumors were 10 B-cell lymphomas and 1 anaplastic lymphoma. We conclude the following: 1) Antibody Mum-1p is very specific for canine plasmacytomas, 2) antibody Mum-1p is superior in sensitivity and specificity to CD79a and CD20 for the identification of canine plasmacytomas in formalin-fixed, paraffin-embedded tissues, 3) canine lymphomas that express MUM1/IRF4 are few and usually of B-cell origin, 4) other canine leukocytic and melanocytic tumors do not express MUM1/IRF4, and 5) prospective studies are needed to determine whether the expression of MUM1/IRF4, particularly in lymphomas, has prognostic significance.
Extramedullary plasmacytomas (EMPs) are neoplasms of plasma cells at varying stages of differentiation that do not involve bone. 25 EMPs are uncommon in dogs and usually affect the skin, constituting 1.5% of all cutaneous neoplasms. 18 Cutaneous plasmacytomas also have been recognized in cats and horses. 20, 32 Most cutaneous plasmacytomas are solitary; multiple lesions are uncommon. 20 Typical locations of cutaneous plasmacytomas are the pinnae, lips, digits, and chin. 20 Cutaneous plasmacytomas are observed mainly in adult and geriatric dogs. 20 Most canine cutaneous plasmacytomas have benign behavior, although rarely they involve viscera, including bone marrow. 30 Plasmacytomas can affect other organs or tissues including the oral cavity, intestine (particularly the rectum), liver, spleen, kidney, lung, and brain. 25, 42, 43 Canine plasmacytomas have been classified histologically as 5 types: mature, hyaline, cleaved, asynchronous, and polymorphous-blastic. 37 An additional type, monomorphous-blastic, has been included by other authors. 5 Multiple myeloma (MM), a tumor of plasma cells involving at least the bone marrow, is rarer than extramedullary plasmacytoma and has been reported in dogs, cats, cattle, horses, and pigs. 25 Human EMPs are rare, typically solitary tumors found in the upper respiratory tract and less commonly in the gastrointestinal and genitourinary tracts, lungs, lymph nodes, and skin. 28 Human EMPs have an indolent course, with frequent local recurrence; conversion to MM may occur. 28, 56
Plasmacytoma is a tumor of monoclonal immunoglobulin-producing cells, so diagnosis was initially based on immunohistochemical demonstration of cytoplasmic immunoglobulins, particularly clonality for λ or κ light chains of immunoglobulins. 20, 42 Canine plasmacytomas are also positive for CD45 and immunoglobulins G, A, or M and variably express CD18, CD45RA, and CD79a leukocytic antigens. 15, 20, 42 CD79a, a molecule of the B-cell antigen receptor complex that is expressed from the pre–pre-B-cell stage to the plasma cell differentiation stage, 23 has been detected in more than 60% of human EMPs and the majority of MMs. 28 In dogs, CD79a is expressed in up to 80% of plasmacytomas. 20, 46 CD20, a transmembrane phosphoprotein predominantly expressed from the pre-B cell stage to the activated B-cell stage, 23 has been detected immunohistochemically in normal canine B cells and lymphomas 26 ; however, CD20 reactivity in normal or neoplastic plasma cells has not been reported. Many more markers are used in human medicine than in veterinary medicine. Currently, markers used in the characterization of human EMPs and MMs in formalin-fixed, paraffin-embedded tissues include antibodies to Blimp-1, CD20, CD38, CD56, CD 138/syndecan-1, multiple myeloma oncogene 1/interferon regulatory factor 4 (MUM1/IRF4), and antibody VS38c. 7, 19, 28, 44, 47, 51
Interferon regulatory factors (IRFs) are a family of transcription factors involved in regulation of cell growth and immunologic responses. 31 MUM1, also called IRF4, is involved in lymphoid cell differentiation. 16 MUM1/IRF4 is required for immunoglobulin light-chain rearrangement at the pre–B stage of lymphocyte maturation. 33 MUM1/IRF4 is expressed in a subset of germinal center B cells, plasma cells, activated T cells, and a subset of macrophages and dendritic cells. 3, 10, 14, 50, 52 MUM1/IRF4-deficient mice do not form germinal centers, lack plasma cells, and have markedly reduced serum immunoglobulin. 34 MUM1/IRF4 antibodies are used as part of an immunohistochemical panel to characterize human B-cell neoplasms. MUM1 is expressed in diffuse large B-cell lymphoma of postgerminal center (CD10−/bcl-6±/MUM1+/CD138−) and plasmablastic (CD10−/bcl-6−/MUM1+/CD138+) origins. 10, 17, 52 In classic Hodgkin's lymphoma, MUM1/IRF4 is detected in Hodgkin's lymphoma Reed-Sternberg cells. 6, 36, 52 MUM1/IRF4 is expressed in the majority of cutaneous plasmacytomas and multiple myelomas. 36, 52 Human T-cell lymphomas, including adult T-cell leukemia lymphoma and anaplastic large cell lymphoma, are frequently MUM1/IRF4-positive. 52 The only reported nonleukocytic human tumor expressing MUM1 is melanoma. 36, 48 MUM1/IRF4 has been used as a prognostic factor in different studies. MUM1/IRF4 expression correlates with unfavorable prognosis in B-cell chronic lymphocytic leukemia/small lymphocytic lymphoma 8, 24 and probably in diffuse large B-cell lymphoma, 21, 22 although other reports could not find such correlation. 2, 10 In primary cutaneous B-cell lymphomas, MUM1/IRF4 expression is associated with reduced survival times. 49 To our knowledge, the expression of MUM1/IRF4 has not been reported in dogs or other domestic species. The objectives of this study were the following: 1) immunohistochemical detection of MUM1/IRF4 with monoclonal antibody Mum-1p in canine plasmacytomas, 2) comparison of results of immunohistochemical expression of MUM1/IRF4 with that of CD20 and CD79a in canine plasmacytomas, and 3) evaluation of the immunohistochemical staining for MUM1/IRF4 in canine leukocytic and melanocytic tumors.
Methods
Case material came from the archives of the Animal Disease Diagnostic Laboratory-Purdue University and the Department of Pathobiology-University of Illinois. A total of 109 plasmacytomas from 107 dogs were examined (Table 1). These tumors were mostly cutaneous (80) with fewer from the oral cavity (18), rectum (4), spleen (2), lung (1), brain (1), liver (1), and bone marrow (1). Plasmacytomas were histologically classified according to Platz et al. 37 We also evaluated antibody Mum-1p reactivity in 139 other tumors, including lymphoid proliferations (89), histiocytic proliferations (31), mast cell tumors (7), and melanocytic tumors (13).
Site and histologic classification of 109 canine plasmacytomas.∗

∗One dog had tumor in 3 organs.
Samples had been fixed in 10% neutral buffered formalin and embedded in paraffin. Four-micrometer-thick tissue sections were stained with HE for routine diagnosis. Immunohistochemistry was done in an automatic stainer following reported protocols. 39– 41 Antibodies and an outline of immunohistochemical procedures are included in Table 2. For plasmacytomas, all cases were examined for expression of MUM1/IRF4, 105 for CD79a, and 108 for CD20. Immunohistochemistry for MUM1 was standardized in a pilot study using formalin-fixed, paraffin-embedded sections of canine reactive lymph node and cutaneous plasmacytoma. 40 For immunophenotyping of lymphomas, CD79a and CD3 were used in all cases and CD20 was used in 57 cases. Lymphomas were considered to be of B-cell origin if tumor cells reacted with CD79a and/or CD20 antibodies and were negative for CD3. T-cell lymphomas reacted with CD3 antibody and were negative for CD79a and CD20. Other markers used in selected cases were CD18, E-cadherin, CD11d, CD45, and PGP 9.5. Antigen retrieval was used as follows: pressure cooker with citrate buffer, pH 6.0 (CD3, CD11d, CD18, CD45, and E-cadherin) and pressure cooker with ethylenediaminetetraacetic acid buffer, pH 9.0 (CD79a, MUM1). Antibodies to CD20 and PGP 9.5 did not need antigen retrieval. The peroxidase-based detection systems used were LSAB2 (CD3, CD20), LSAB+ (CD45, E-cadherin), and ENVISION+ (CD11d, CD18, CD79a, PGP 9.5), all from Dako (Carpinteria, CA). For antibody Mum-1p, we used mouse ImmPRESS polymer-based system (Vector Laboratories, Burlingame, CA). A pilot study using mouse ENVISION+ (Dako), another polymer-based detection system, yielded weak staining when compared with ImmPRESS. In addition, the intensity of the staining was increased (without increase in background staining) by replacing the DAB solution included in the ImmPRESS kit with the DAB solution used for ENVISION+. 40 Slides were counterstained with Mayer's hematoxylin. Immunoreactivity for MUM1/IRF4 was scored in the following manner: 3, >60% positive cells; 2, 30–59% positive cells; 1, 10–29% positive cells; or 0, <10% positive cells.
Antibodies and immunohistochemical methods.
∗HIER = heat-induced epitope retrieval with decloaker (Biocare Medical, Walnut Creek, CA), 20–24 psi, 125°C for 30 seconds, LSAB = labeled streptavidin biotin, EDTA = ethylenediaminetetraacetic acid, MUM1/IRF4 = multiple myeloma oncogene 1/interferon regulatory factor 4.
Results
We examined 109 plasmacytomas from 107 dogs. Five (5/107) dogs had multiple masses; 3 tumors from 1 dog with no cutaneous involvement but multiple affected viscera were evaluated. From the other dogs with multiple plasma cell tumors, only 1 mass per dog was included in this study. Two dogs had 2 cutaneous nodules located very close to each other (neck and lips). Another dog had multiple viscera affected, but only the spleen was examined. The fifth dog had a tumor in the tongue and another in the rectum. Most dogs with plasmacytomas were middle-aged to aged (range, 2.5–16 years; mean, 9.4 years; median, 9 years). The distribution of the lesions is included in Table 1. Eighty tumors were cutaneous, 18 oral, 5 intestinal (rectum), 2 splenic, and 1 each in bone marrow (myeloma), brain, liver, or lung. Cutaneous plasmacytomas were more common in the foot (17), leg (13), and trunk (14), followed by the lips (10), pinna (10), head (7), neck (5), and perineum (4). Nine of 107 dogs (8.4%) had abundant amyloid depositions that were located in the foot (2) and 1 each in the pinna, leg, gingiva, tongue, tonsil, rectum, and perineum. The amount of amyloid varied but, in some cases, contributed more to the volume of the tumor than did the neoplastic cells. A foreign body inflammatory reaction with multinucleated giant cells associated with amyloid deposition was observed in several dogs. On the basis of a published histologic classification, 37 our series had 31 (28.2%) asynchronous, 34 (31.3%) cleaved, 3 (2.8%) hyaline, 21 (19.2%) mature, and 20 (18.4%) polymorphous-blastic plasmacytomas (Figs. 1– 5).
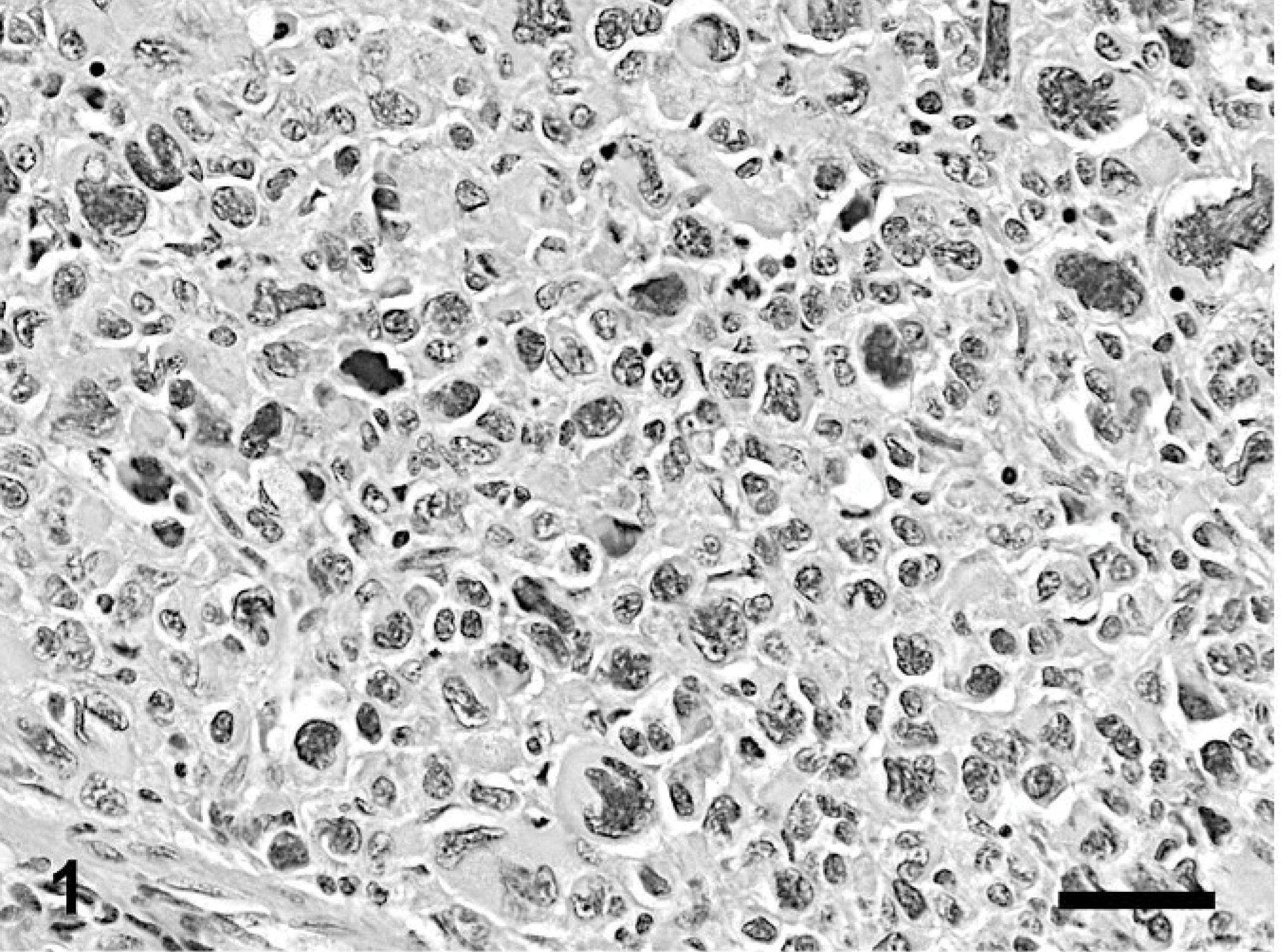
Skin; dog. Plasmacytoma, polymorphous type. HE. Bar = 40 μm.
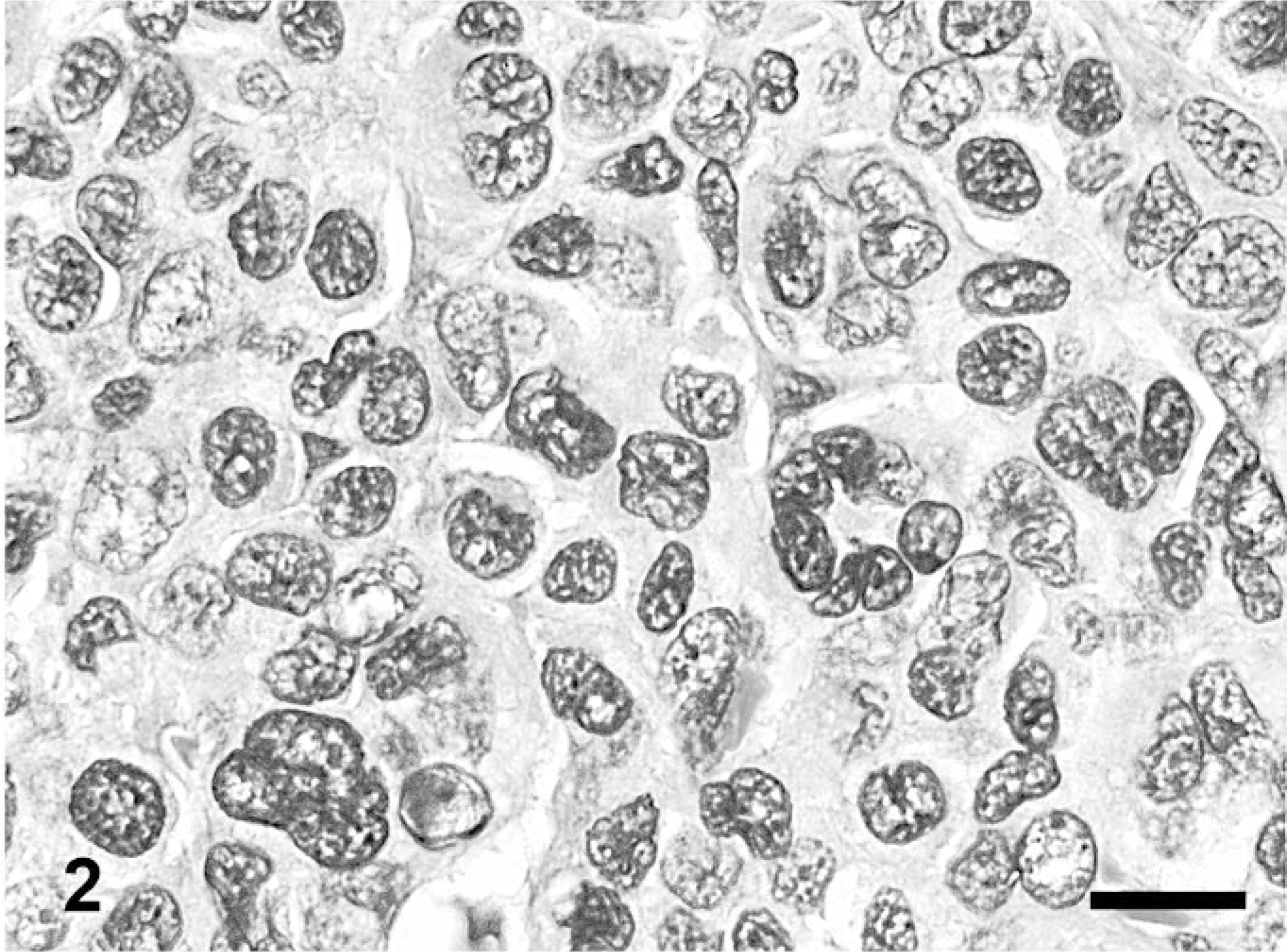
Skin; dog. Plasmacytoma, cleaved type. HE. Bar = 20 μm.
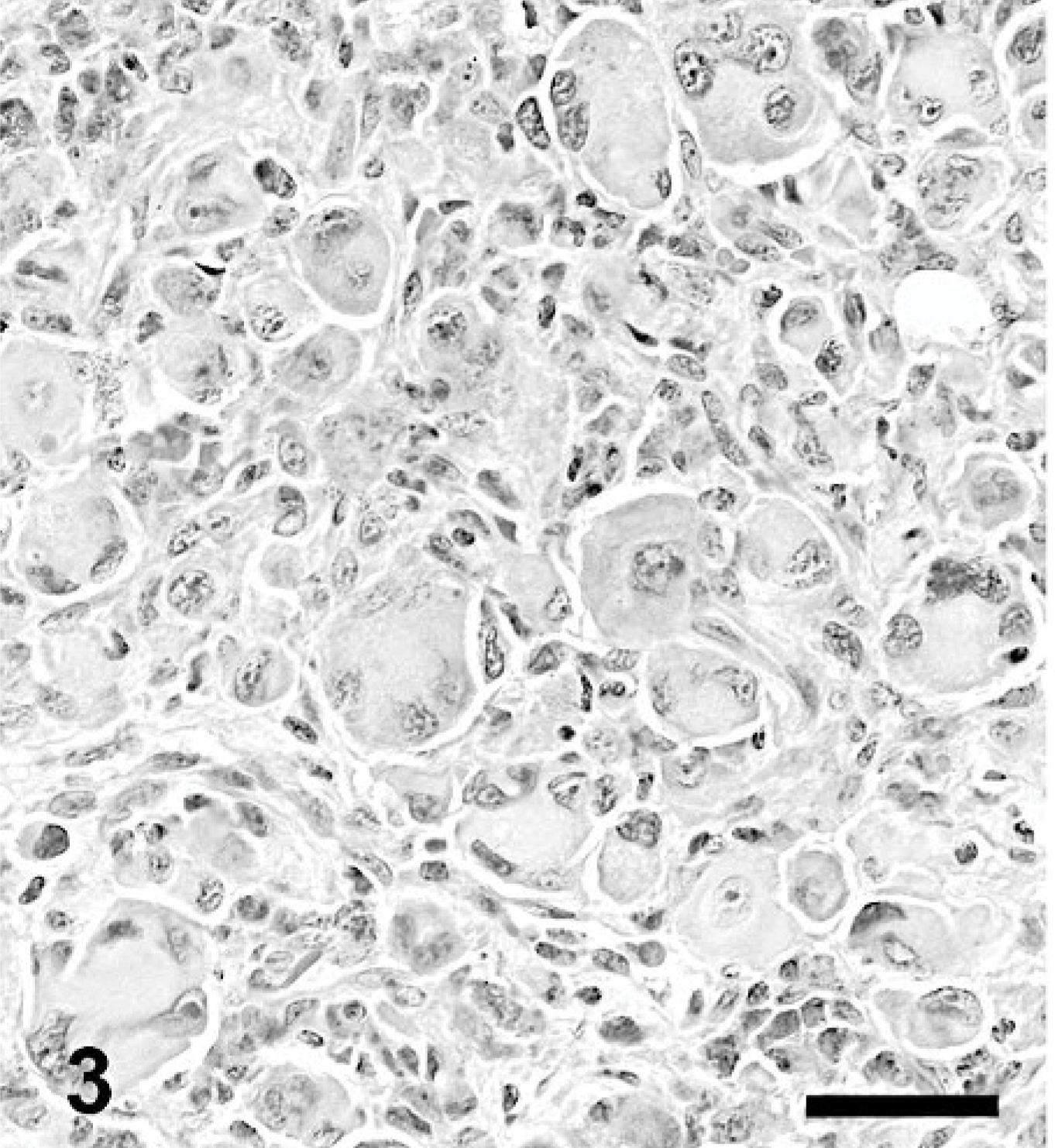
Skin; dog. Plasmacytoma, hyaline type. HE. Bar = 40 μm.
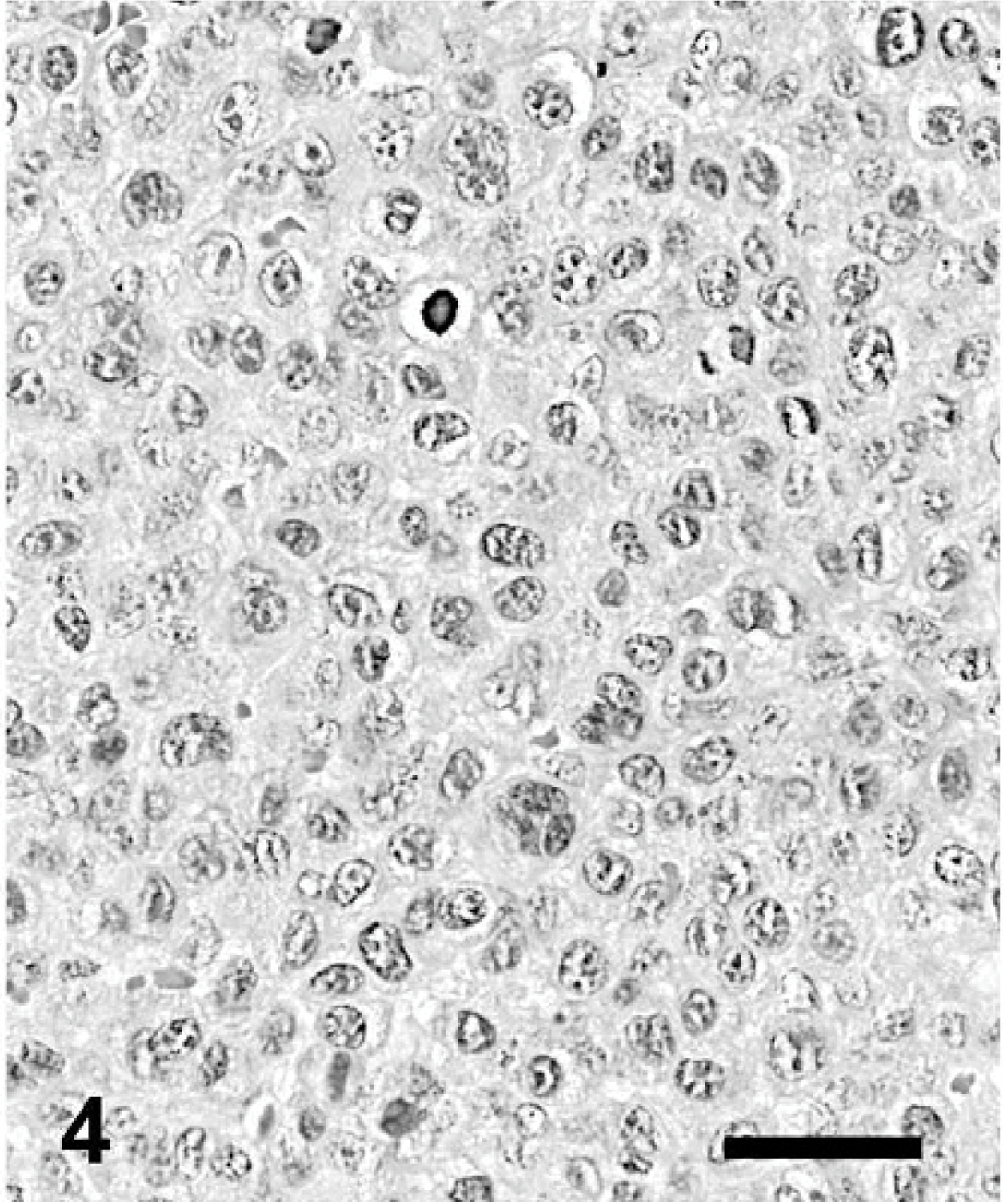
Skin; dog. Plasmacytoma, asynchronous type. HE. Bar = 40 μm.
Skin; dog. Plasmacytoma, mature type. Plasma cells are surrounded by amyloid and foreign body multinucleated giant cells (asterisk). HE. Bar = 40 μm.
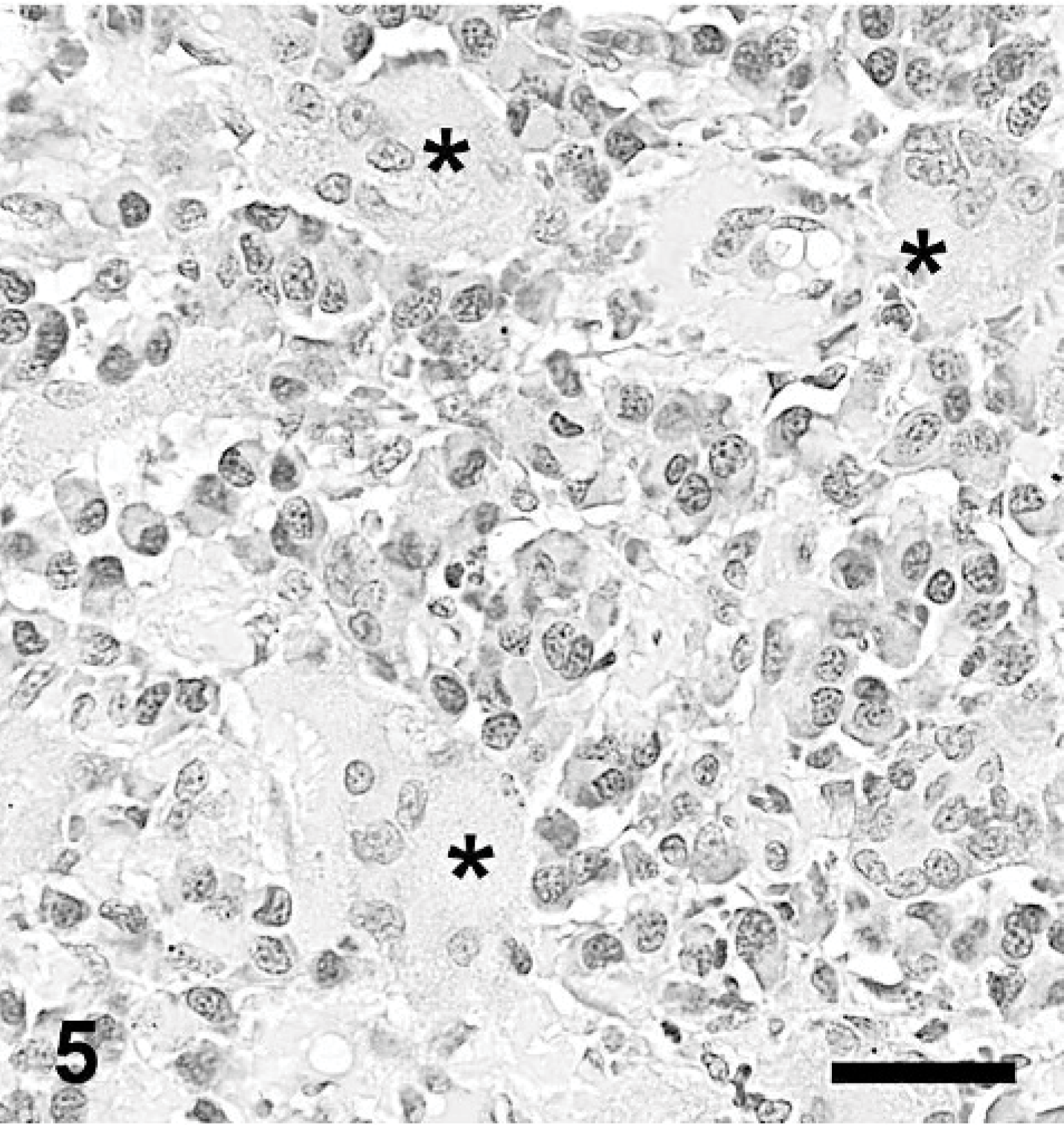
In normal canine lymph nodes, most MUM1/IRF4-positive cells had plasmacytoid cytologic features and were located in medullary cords. MUM1/IRF4-positive lymphoid cells were uncommon in either follicular or interfollicular areas or paracortex. MUM1/IRF4 antigen was detected mostly in the nucleus, with slight and diffuse staining of the cytoplasm, in both normal and neoplastic plasma cells (Figs. 6, 7). Nuclear staining varied in intensity among cells of the same tumor. Binucleated and multinucleated plasmacytoid giant cells also had moderate-to-strong nuclear staining with antibody Mum-1p (Fig. 7). Table 3 shows the results of immunostaining of plasmacytomas with antibodies to MUM1/IRF4, CD20, and CD79a. Of 109 tumors examined by immunohistochemistry, 101 (93.5%) were positive for MUM1/IRF4. Three negative samples belonged to the same dog, making a total of 101/107 (94.39%) cases of plasmacytomas positive for MUM1/IRF4. Sixty-five of 109 plasmacytomas had a positive cell score of 3, 27 had a positive cell score of 2, and 9 had a positive cell score of 1. Mature plasmacytomas had more negative cases (3/21 or 14.28%) than asynchronous (1/31, 3.22%), cleaved (0/34), hyaline (0/3), or polymorphous blastic (2/20, 10%) types. No differences in antibody Mum-1p staining intensity or percentage positive cells among different organs were observed (Figs. 8, 9). Two plasmacytomas that were negative for MUM1/IRF4 were decalcified samples and were also negative for CD20 and CD79a. Fifty-nine of 105 cases (56.2%) were positive for CD79a, and 21/108 (19.4%) plasmacytomas expressed CD20 antigen (Fig. 10). The strongest immunohistochemical correlation among these 3 markers was between MUM1/IRF4 and CD79a; 47/104 (45.1%) plasmacytomas were positive for both markers and negative for CD20. Thirty-two of 104 (30.7%) were positive for MUM1/IRF4 and negative for CD79a and CD20. Only nine plasmacytomas were positive for all 3 markers. Three plasmacytomas negative for MUM1/IRF4 were positive for CD79a and CD20 (Table 3).
Immunohistochemical staining with MUM1/IRF4, CD79a and CD20 in canine plasmacytomas.

∗MUM1/IRF4 = multiple myeloma oncogene 1/interferon regulatory factor 4.
†Five of the 109 plasmacytomas were not evaluated for all 3 markers.
‡Two of the triple-negative tumors had been decalcified.
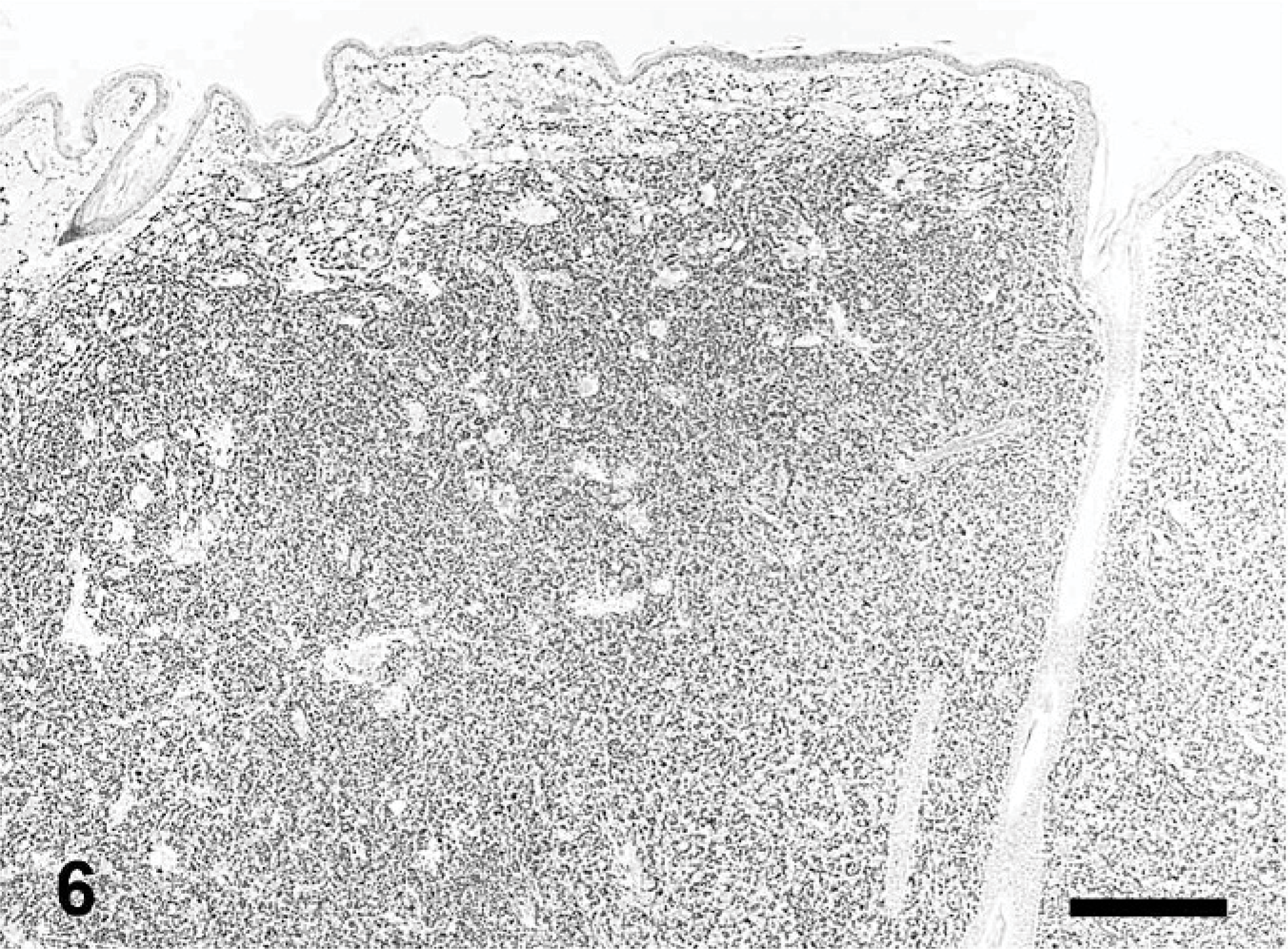
Skin; dog. Plasmacytoma. The dermis is diffusely infiltrated by neoplastic cells. Strong staining with Mum-1p antibody. Immunoperoxidase for multiple myeloma oncogene 1/interferon regulatory factor 4, Mayer's hematoxylin counterstain, HE. Bar = 400 μm.
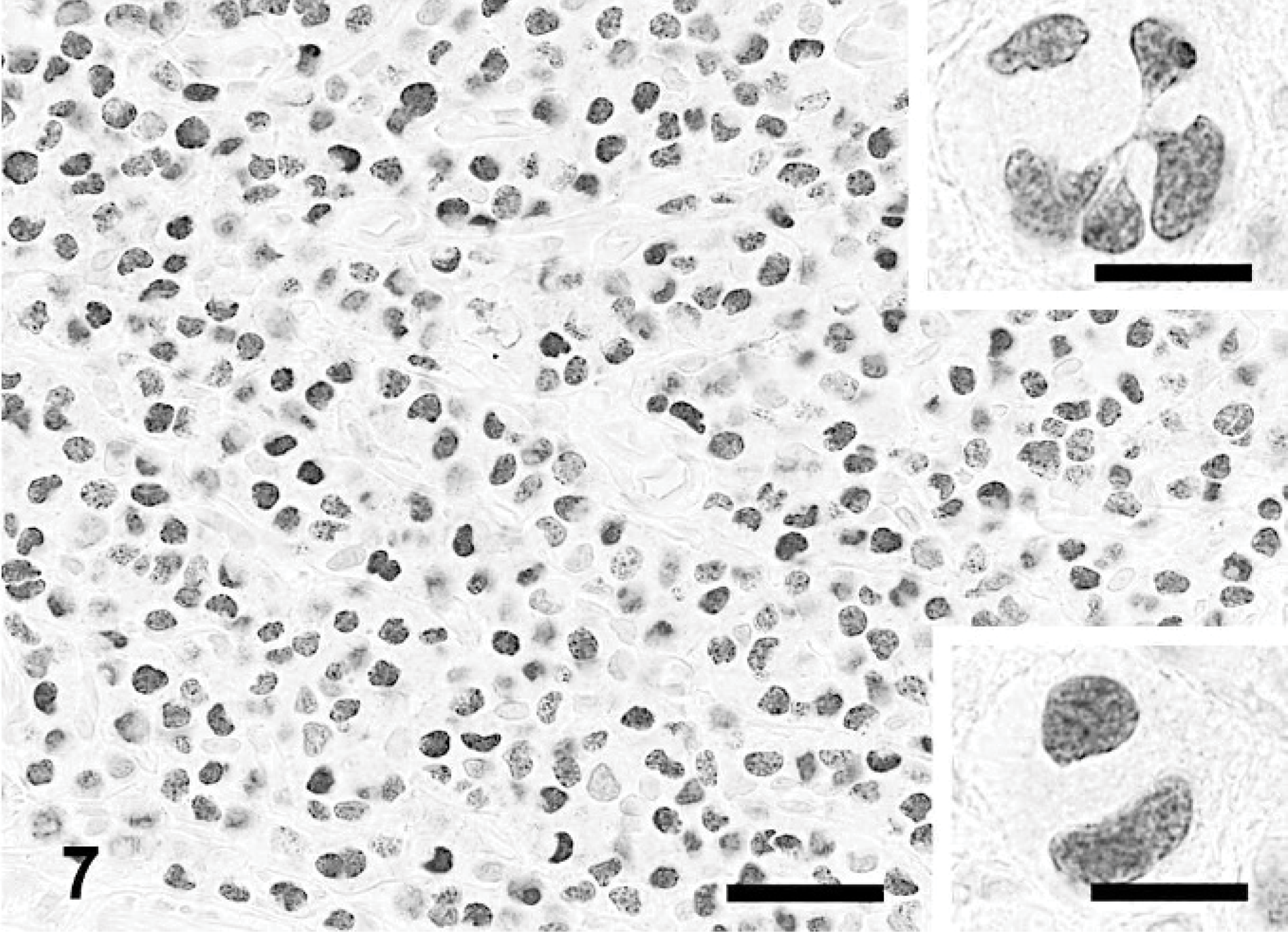
Skin; dog. Plasmacytoma. Detail of staining with antibody Mum-1p. Strong nuclear and weak cytoplasmic staining in most cells. Bar = 40 μm. Upper inset: Staining of multinucleated giant cell. Lower inset: Staining of binucleated giant cell. Immunoperoxidase for multiple myeloma oncogene 1/interferon regulatory factor 4, Mayer's hematoxylin counterstain. Bar = 20 μm.
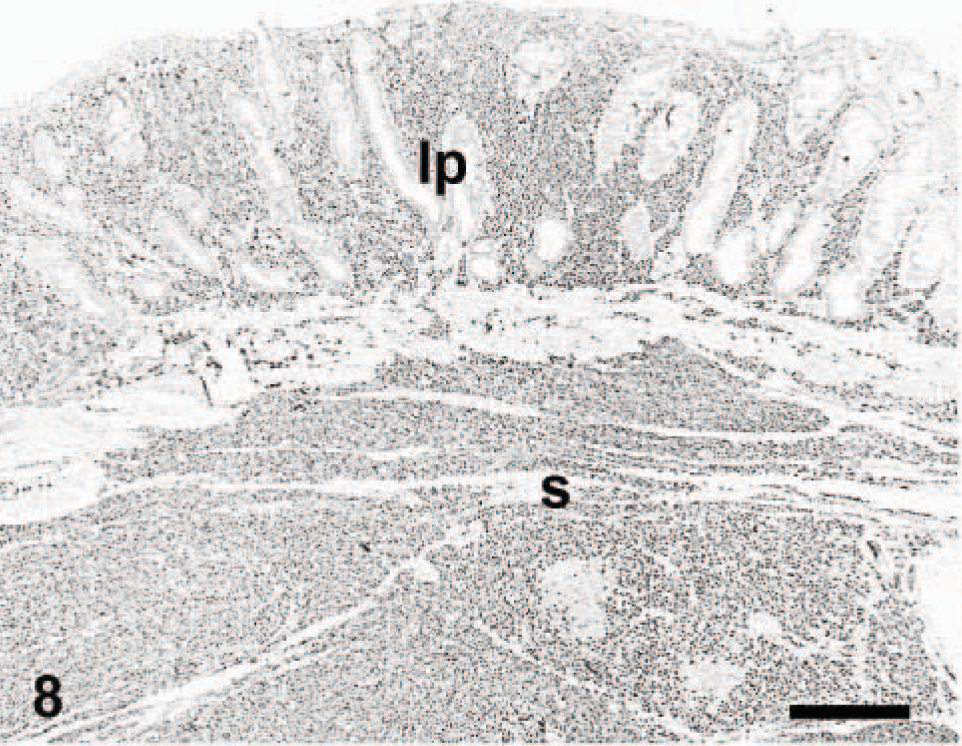
Rectum; dog. Plasmacytoma. Neoplastic cells in the lamina propria (lp) and submucosa (s) are positive. Immunoperoxidase for multiple myeloma oncogene 1/interferon regulatory factor 4, Mayer's hematoxylin counterstain. Bar = 400 μm.
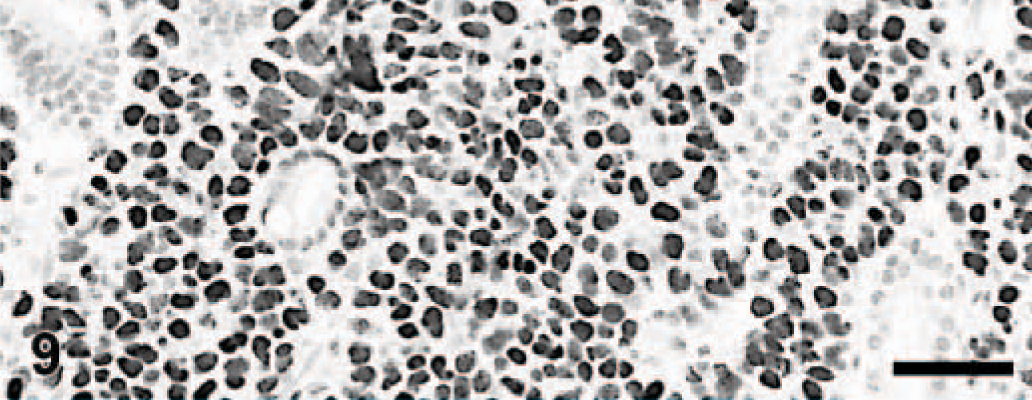
Rectum; dog. Plasmacytoma. Numerous variably stained cells are interspersed among mucosal glands. Immunoperoxidase for multiple myeloma oncogene 1/interferon regulatory factor 4, Mayer's hematoxylin counterstain. Bar = 80 μm.
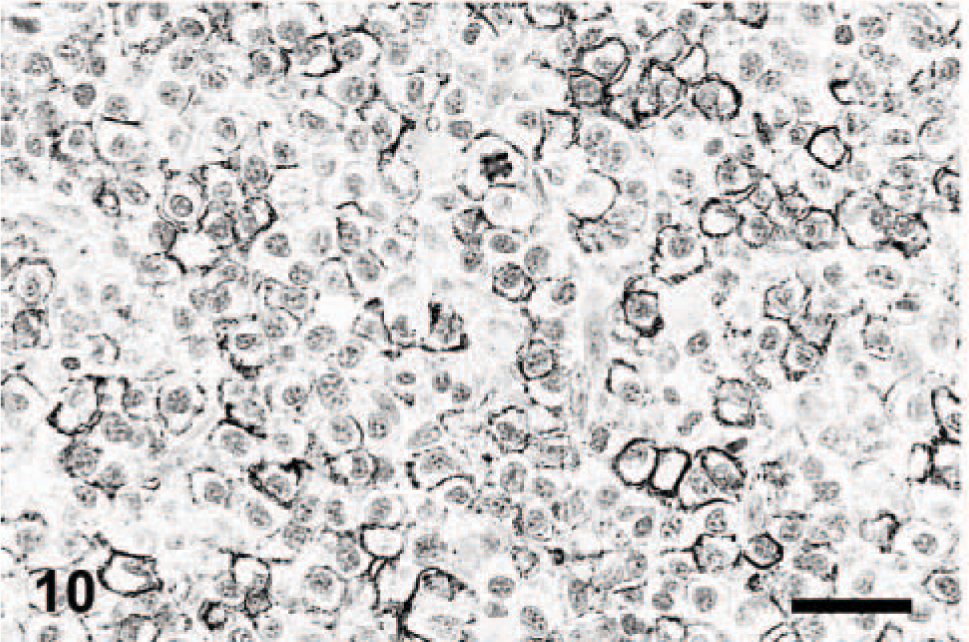
Skin; dog. Plasmacytoma. Distinct cytoplasmic membrane staining in numerous cells. Immunoperoxidase for CD20, Mayer's hematoxylin counterstain. Bar = 40 μm.
A total of 139 nonplasmacytoma canine round cell proliferations and melanocytic tumors were examined to validate the immunohistochemical detection of MUM1/IRF4 in plasmacytomas. Tumor classification was based on histologic features and immunophenotyping. These tumors included anaplastic lymphoma (2), atypical follicular hyperplasia (2), B-cell lymphoma (47), hemophagocytic histiocytic sarcoma (2), histiocytic sarcoma (7), cutaneous histiocytoma (17), cutaneous histiocytosis (3), mast cell tumor (7), melanocytic tumor (13), reactive histiocytosis (2), T-cell epitheliotropic lymphoma (17), T-cell lymphoma (19), and thymoma (1). The only MUM1/IRF4-positive tumors were B-cell lymphomas (10 of 47 B-cell lymphomas, 21.2%; 10 of 85 total lymphomas, 11.7%) and 1 of 2 anaplastic lymphomas (Figs. 11– 13). When lymphomas were classified according to the current World Health Organization classification, 53 MUM1/IRF4-positive lymphomas were diffuse large cell B lymphoma (DLCBL) of centroblastic type (4), DLCBL of immunoblastic type (2), follicular lymphoma (2), and 2 lymphomas with plasmablastic differentiation. In the follicular lymphomas, cytologically identifiable centrocytes (and not centroblasts) were the only positive cells in 1 dog. None of the marginal zone lymphomas reacted with the antibody Mum-1p. The number of positive cells in lymphomas varied, but the staining was usually intense and unequivocal. An anaplastic lymphoma that expressed both CD3 and CD79a antigens had strong reaction in >90% of cells for MUM1/IRF4.
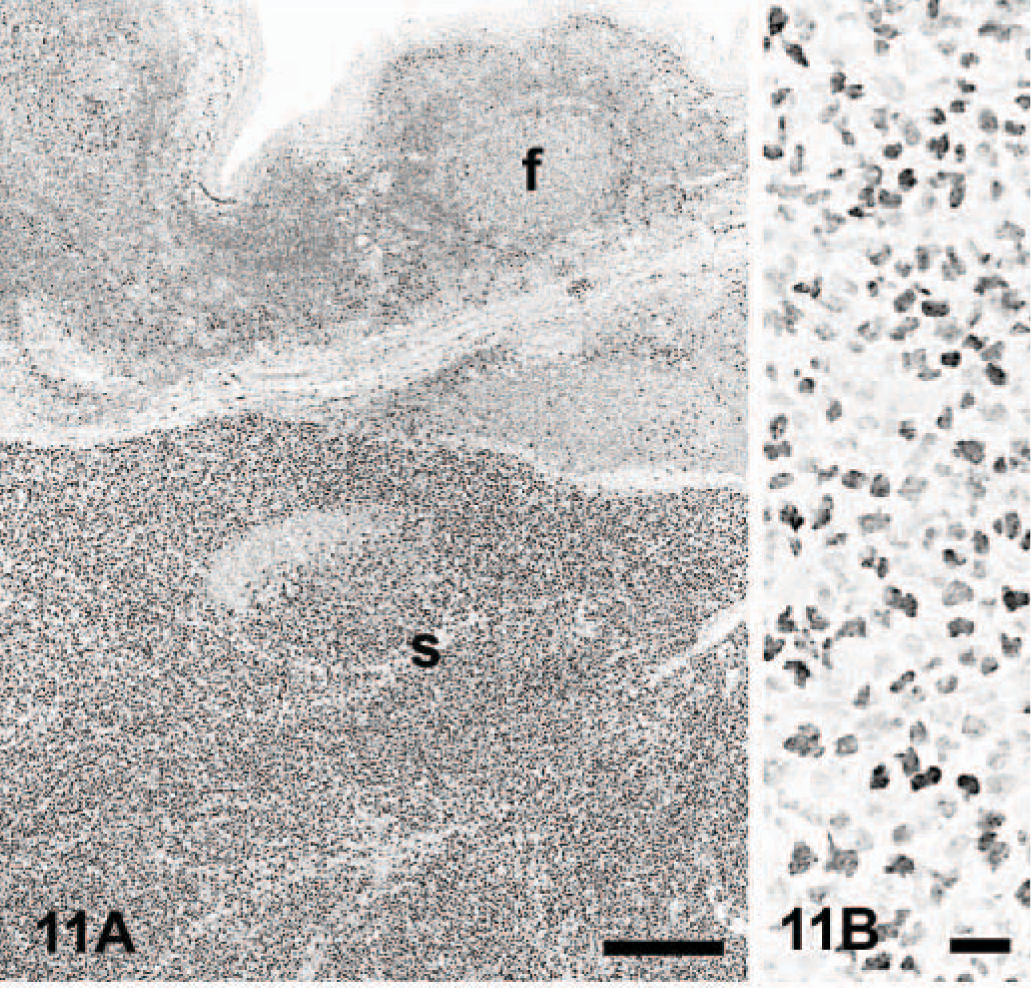
Tonsil; dog.

Lymph node; dog. Follicular lymphoma. Many neoplastic lymphocytes react with antibody Mum-1p. A thin rim of nonneoplastic lymphocytes in the periphery of the follicles has rare positive cells (arrows). Immunoperoxidase for multiple myeloma oncogene 1/interferon regulatory factor 4, Mayer's hematoxylin counterstain. Bar = 160 μm.
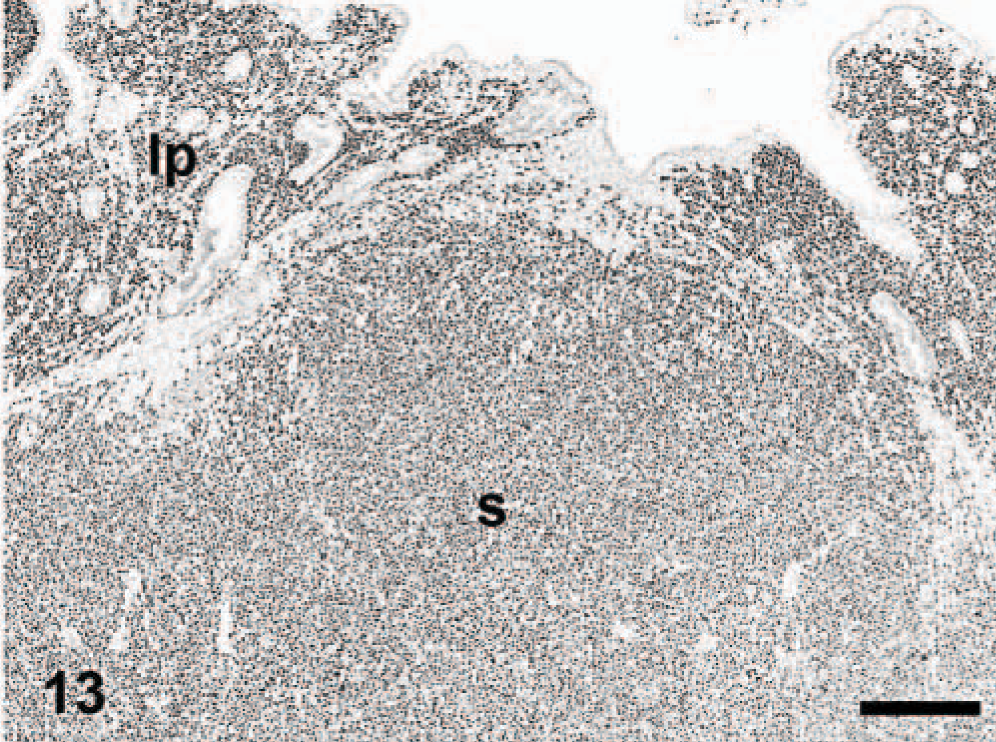
Intestine; dog. Anaplastic large cell lymphoma. Diffuse staining in the lamina propria (lp) and submucosa (s). Immunoperoxidase for multiple myeloma oncogene 1/interferon regulatory factor 4, Mayer's hematoxylin counterstain. Bar = 400 μm.
Discussion
The most common histologic forms of plasmacytomas in our series were cleaved and asynchronous. These 2 forms are reportedly the most common canine plasmacytoma types. 37 Histologic grading of canine plasmacytomas does not seem to correlate with biologic behavior, which usually is benign. 1, 37 In our series, only 2 cases involved multiple organs, and neither of these had cutaneous lesions. Although follow-up attempts were not made, we did not find in the databases used in this study evidence of recurrence of the tumors. Amyloid is observed in about 10% of canine plasmacytomas 20 ; a similar percentage of cases with amyloid deposition was present in our series. The mean age of presentation of plasmacytomas was 9.4 years, similar to the 9.2 years reported in another large study 18 in which the head was the site most frequently involved; however, in the current study, the most common location was the leg. Most human EMPs are in the upper respiratory tract, oral cavity, or gastrointestinal tract. 9, 27 Human cutaneous plasmacytomas are rare and usually solitary; visceral metastases or progression to multiple myeloma is observed in one third of cases. 56, 57
Immunohistochemical detection of immunoglobulin heavy and light chains has been the standard test for plasma cell differentiation. 1, 4, 5, 13, 29, 35, 38, 42, 45 However, nonspecific staining may result from adsorption of immunoglobulins to the cell surface, internalization by phagocytes, or binding of immunoglobulins to a variety of cell types via Fc receptors. 46 Although reactivity of CD79a in canine plasmacytoma and B-cell lymphomas is well established, there are large variations in the percentage of cases and number of cells stained with this marker, with up to 80% of plasmacytomas expressing CD79a. 20, 42, 46
To our knowledge, this is the first report of MUM1/IRF4 immunohistochemical detection in canine plasmacytomas and other canine round cell tumors. In addition, we compared reactivity of MUM1/IRF4 with that for other B-cell markers, CD79a and CD20, in a large series of canine plasmacytomas. More than 94% of canine plasmacytomas were positive for MUM1/IRF4. Two of the negative cases were digital lesions that had been decalcified; these cases were also negative for CD79a and CD20, which suggests the possibility that decalcification hindered immunohistochemical detection of these antigens. This was a retrospective study, and no attempts were made to determine the significance of expression of MUM1/IRF4 in canine plasmacytomas and lymphomas. In humans, MUM1/IRF4 expression correlates with unfavorable outcome or decreased survival in B-cell chronic lymphocytic leukemia/small lymphocytic lymphoma, diffuse large B-cell lymphoma, and primary cutaneous B-cell lymphomas 8, 21, 22, 24, 29 ; other investigators could not find such correlation. 2, 10 Although MUM1/IRF4 is typically expressed in postgerminal center lymphomas, 10 2 of the 10 B-cell lymphomas that were positive for this marker were high-grade follicular lymphoma.
In the current study, the number of CD79a-positive plasmacytomas was markedly lower than that for MUM1/IRF4. This was not unexpected considering the reported expression of CD79a during lymphoid maturation. 23 CD79a is expressed from the pre–pre-B-cell stage to the plasma cell stage, but it is not expressed in all plasma cells and their tumors. 19 However, other authors reported 13 of 16 canine plasmacytomas positive for CD79a. 46 This discrepancy could reflect differences in scoring criteria.
Of the 3 B-cell markers used in this study, CD20 was the least likely to be expressed in canine plasmacytomas. CD20 is expressed from the pre–B-cell stage to the activated B-cell stage 23 and is not detected in human EMPs or multiple myelomas, although a few normal plasma cells can show reactivity. 9, 27, 28, 55 Typically, human plasmacytomas have a MUM1/IRF4+, CD79a+, CD20− immunophenotype, with expression of other markers such as CD138 and VS38c. 7, 19, 28, 44, 51, 55 Our results correlate fairly well with those of the human literature, particularly for MUM1/IRF4 and CD20. We did not find references of CD20 reactivity in canine plasmacytomas.
Specificity of the antibody Mum-1p for plasma cells and their tumors was evaluated by examining its reactivity in other leukocytic and melanocytic tumors because some T- and B-cell lymphomas, in addition to melanomas, have been reported to express MUM1/IRF4 in humans. 6, 36, 52 Only 10 B-cell lymphomas and 1 anaplastic lymphoma from a total of 139 leukocytic and melanocytic neoplasms expressed MUM1/IRF4. In our series, diffuse large cell B-cell lymphoma was the most common nonplasmacytic tumor to express MUM1/IRF4. None of the marginal zone lymphomas in our series (6 nodal, 1 splenic) were positive for MUM1/IRF4. We did not observe plasmacytic differentiation in these tumors, but some human extranodal marginal zone lymphomas have this differentiation and MUM1/IRF4 is the most commonly expressed plasma cell marker. 11, 12 A result that deserves further examination is the strong detection of MUM1/IRF4 in centrocytes in a single case of advanced or late-stage (III) follicular lymphoma (FL). Currently FL is identified on oversight staining by a tissue architecture with tightly facetted and coalescing follicles that have lost most or all of the mantle cell cuffs and have a uniform population of cells across the follicular diameter. 54 In contrast, nodes with even atypical follicular hyperplasia retain an antigen-related polarity most evident in those centers immediately beneath the node capsule. This polarity is characterized by segregation of cells with centroblastic nuclei into the deep or dark pole area and those with centrocytic nuclei into the superficial or light-pole area. Since most follicular center cells stain positively with B-cell markers in both benign and neoplastic follicles, the availability of a stain that marked only the centrocytes of neoplastic follicles would be a valuable addition to animal diagnostic hematopathology. However, this finding needs to be confirmed using a larger series of FL. In humans, up to 95% of melanomas express MUM1/IRF4. 36, 48 We examined canine oral melanomas and cutaneous melanocytomas, and none showed reactivity for antibody Mum-1p.
Although other nonleukocytic tumors were not examined in our series, these results indicate the following: 1) Antibody Mum-1p is very specific for canine plasmacytomas, 2) antibody Mum-1p is superior in sensitivity and specificity to CD79a and CD20 for the detection of canine plasmacytomas in formalin-fixed, paraffin-embedded tissues, 3) the sensitivity of antibody Mum-1p to detect canine plasmacytomas was 93.5%; when combined with CD79a, it increased to 95.5%, 4) few canine lymphomas, usually of B-cell origin, express MUM1/IRF4, 5) canine melanomas and other leukocytic tumors do not express MUM1/IRF4, and 6) prospective studies are needed to determine whether the expression of MUM1/IRF4, particularly in lymphomas, has prognostic significance.
Footnotes
Acknowledgement
We appreciate the technical expertise of Dee DuSold in performing immunohistochemistry.
