Abstract
Human enteropathy-associated T-cell lymphoma (EATL) is classified into 2 distinct subgroups based on their phenotypes (type I and type II). Canine intestinal T-cell lymphoma can be morphologically classified into large and small cell lymphomas (LCL and SCL, respectively). Their association with human EATL or immunohistochemical and biological features has not been well characterized. In this study, the immunohistochemical profiles of 17 cases of LCL and 33 cases of SCL were evaluated with markers used for human EATL classification. Morphologically, LCL was characterized by sheet-like proliferation of large to moderately sized neoplastic lymphocytes, with scant clear cytoplasm and pleomorphic, irregularly shaped nuclei containing distinctive nucleoli and scattered chromatin. In contrast, SCL was characterized by the proliferation of monomorphic small neoplastic lymphocytes, accompanied by infiltration of nonneoplastic plasma cells. Interestingly, 8 cases demonstrated mixed LCL and SCL morphologies. Granular cytoplasmic expression of granzyme B was observed in most LCL and SCL cases. Membranous expression of CD56 was demonstrated in only 2 of 17 LCL and 0 of 33 SCL cases. Coexpression of CD20 by neoplastic T cells was observed in more SCL cases (16/33; 48%) than LCL cases (1/17; 6%). The CD56-positive cells in 2 cases were negative for CD20. Although canine LCL shares common features with human EATL type I, canine SCL cells and human EATL type II differ in their immunophenotype. Canine intestinal T-cell lymphoma had a homogeneous immunophenotype regardless of cell morphology. The findings of this study may indicate large cell transformation of SCL rather than 2 distinct entities.
Canine intestinal lymphoma is of predominantly T-cell origin and is morphologically classified into large cell lymphoma (LCL) and small cell lymphoma (SCL). 56,57 Human enteropathy-associated T-cell lymphoma (EATL) classification has been recently introduced to domestic animals. 8,57 However, classification of canine T/natural killer (NK)–cell lymphoma is insufficient due to a limited number of commercially available antibodies against canine T cells in paraffin sections. 34
EATL is a type of intestinal T-cell lymphoma recognized by the World Health Organization (WHO) classification scheme for humans. EATL is further classified into 2 subtypes, EATL type I (recently renamed as EATL) and type II (recently renamed as monomorphic epitheliotropic intestinal T-cell lymphoma) with distinct clinical and morphological features. 10,13,14,15,20,49,53 EATL type I is characterized by pleomorphic lymphocytes with irregular or angulated nuclei and clear nucleoli. The lesions usually contain sparse inflammatory infiltrates and necrotic regions. 1 Occasionally, the cells are anaplastic. Mitotic figures and necrotic lesions are common. EATL type I is associated with celiac disease in Western populations. 27 Conversely, EATL type II lesions consist of monomorphic, small- to moderate-sized neoplastic lymphocytes with round nuclei and inconspicuous nucleoli, containing minimal inflammatory infiltrates and necrosis. 1 EATL type II is most commonly found in Asian countries. 1 Neoplastic lymphocytes of EATL type I are CD3+CD8+/–CD4–CD56–TCRβ+/– and frequently granzyme B (GRB) positive, while those of EATL type II are CD3+CD8+CD4–CD56+ and TCRβF1 positive or TCRγδ positive. 1,10
In contrast, the immunohistochemical profiles of canine intestinal T-cell lymphoma have remained largely unknown. This study was designed to compare the morphological and immunophenotypic features of canine intestinal T-cell lymphoma and human EATL.
Materials and Methods
Cases
Intestinal tissue specimens were endoscopically collected from dogs presented at Japan Small Animal Medical Center (JSAMC) between 2014 and 2016. All dogs had a chronic history of gastrointestinal signs such as vomiting, diarrhea, and weight loss for more than 3 weeks or evidence of protein-losing enteropathy with low serum albumin levels below 2.7 g/dl. Complete blood count, serum biochemistry, fecal examination, urinalysis, and abdominal ultrasonography were performed to rule out infectious causes, pancreatic insufficiency, and other causes of hypoalbuminemia such as hepatic and renal diseases. Histopathological diagnosis was performed at the Department of Veterinary Pathology, Graduate School of Agricultural and Life Sciences, University of Tokyo. The study population was composed of 50 cases histopathologically and immunohistochemically diagnosed with intestinal T-cell lymphoma (LCL or SCL). Anaplastic large T-cell lymphoma was excluded from the study population. The histopathological criteria are described in the histology section below. Immunohistochemical inclusion criteria included clear membranous expression of CD3 and absence of CD79α. The breeds included in this study were Shiba Inu (n = 17), Miniature Dachshunds (n = 9), mixed breed (n = 4), Chihuahua (n = 3), French Bulldog (n = 3), Toy Poodle (n = 3), Miniature Schnauzer (n = 2), American Cocker Spaniel (n = 1), Beagle (n = 1), English Setter (n = 1), Jack Russell Terrier (n = 1), Labrador Retriever (n = 1), Papillon (n = 1), Pomeranian (n = 1), Pug (n = 1), and Weimaraner (n = 1). There were 29 males (17 neutered) and 21 females (17 neutered) with a mean age of 7.5 years (range, 1.9-13 years). The mean ages of dogs with LCL and SCL were 8.5 and 7.0 years, respectively. There was no age or sex predisposition. Details of the cases are summarized in Supplemental Table S1.
Histopathology
Endoscopic tissue samples were collected from the stomach, proximal duodenum, distal duodenum, ileum, and colon. Mucosal tissue samples were placed with the villous side upward on filter paper to make a vertical section. A minimum of 6 tissue specimens were collected per region. The samples were fixed in 10% formalin and processed for routine histopathology with paraffin embedding. Sections (4 μm) were deparaffinized, rehydrated through graded alcohols, and stained with hematoxylin and eosin (HE). Tissue specimens used for histopathological evaluation each had at least 3 villi and included the entire depth of the intestinal mucosa. 58 Histopathological diagnosis was performed by 2 veterinary pathologists accredited by the Japanese College of Veterinary Pathologists (K.U. and J.K.C.). Histopathological analysis of the mucosal architecture was based on the histopathological criteria proposed by the WSAVA gastrointestinal standardization group for diagnosing gastrointestinal inflammation in dogs. 12 Degree of inflammatory cell infiltration was determined by observing the plasma cell population among neoplastic lymphocytes. SCL was histopathologically differentiated from chronic enteritis using a diagnostic algorithm of feline intestinal lymphoma. 29 Intestinal lymphomas were diagnosed and classified as LCL or SCL on the basis of their morphological characteristics and nuclear sizes. Lymphoma with nuclei smaller than 2 red blood cells in diameter was defined as SCL, and that with nuclei larger than 2 red blood cells was considered LCL, consistent with previously reported diagnostic criteria. 35,55,57
Immunohistochemistry
Details regarding primary antibodies and antigen retrieval procedures are summarized in Supplemental Table S2. Species specificity of antibodies has been demonstrated by previous studies and by manufacturers. 19,38,48 Immunohistochemistry (IHC) was performed on duodenal and ileal tissue specimens. Following antigen retrieval, endogenous peroxidase was inactivated with 3% hydrogen peroxide in methanol at room temperature for 5 minutes. The sections were blocked with 8% skim milk in Tris-buffered saline (TBS) at 37°C for 30 minutes, then with primary antibodies at 4°C overnight. Following 3 TBS washes, the sections were incubated with Dako EnVision+ system hydrogen peroxidase (HRP)–labeled polymer anti-rabbit/mouse secondary antibodies (Dako Japan, Tokyo, Japan) at 37°C for 40 minutes. The sections were washed 3 times with TBS, followed by visualization using 0.05% 3,3′-diaminobenzidine (DAB) and 0.03% hydrogen peroxidase in Tris-HCl buffer. Normal canine tonsil or intestine was used as a positive control. A negative control was prepared by omitting each primary antibody. Proliferation activity (MIB-1 index) was determined by counting a total Ki-67–positive cells per 1000 neoplastic cells.
Immunofluorescence
To confirm coexpression of CD3 and CD20 in neoplastic lymphocytes, immunofluorescence (IF) staining was performed. After antigen retrieval, the sections were blocked with 8% skim milk in TBS with 1% Tween (TBST). The sections were first incubated with anti-CD20 antibodies at 4°C overnight, followed by anti-CD3 antibodies at 37°C for 60 minutes (Suppl. Table S2). After 3 washes in TBST, the sections were incubated with Alexa Fluor 488–conjugated goat anti-mouse IgG (1:200; Invitrogen, Eugene, OR) and Alexa Fluor 594–conjugated donkey anti-rabbit IgG (1:200; Invitrogen) at room temperature for 1 hour. Expression and colocalization of CD20 and CD3 were analyzed using a laser-scanning confocal microscope (LSM 700; Zeiss, Tokyo, Japan).
Polymerase Chain Reaction for Antigen Receptor Gene Rearrangement
Tissue collection and clonality analysis were performed as previously described. 22 Briefly, tissue samples were endoscopically collected from the duodenum and ileum and stored at –20°C until use. Genomic DNA was extracted from frozen samples using the DNeasy Blood and Tissue Kit (Qiagen, Valencia, CA). Forward primers were labeled with fluorescent dyes at their 5′ ends. All reactions were conducted in duplicate. GeneScan analysis was performed on the ABI 3130xl Genetic Analyzer (Applied Biosystems, Foster City, CA) as a multiplexed run. A clonal T-cell population was defined by the presence of distinct peaks of fluorescence intensity at least 2-fold higher than other background peaks. Monoclonal or biclonal peaks detected either in the duodenal or ileal sample were considered positive results.
Statistical Analysis
Statistical analysis was performed using SPSS software (Version 22.0; SPSS, Inc, an IBM Company, Chicago, IL). Differences between LCL and SCL were considered statistically significant at P < .05.
Results
Ultrasonographic and Endoscopic Findings
Ultrasonography detected jejunal or colic lymphadenomegaly in 21 of 50 cases (42%). Intestinal wall thinning (5/50; 10%) and thickening (6/50; 12%) were detected in a small number of cases. Endoscopically, lacteal dilation was detected in 16 cases (32%). Mild lacteal dilation was characterized by scattered white spots while marked lacteal dilation was characterized by diffuse white nodules in the edematous mucosa (Figs. 1, 2). Endoscopic examination also revealed hemorrhage (11/50; 22%) and ulcer or erosion (6/50; 12%) (Figs. 3, 4).
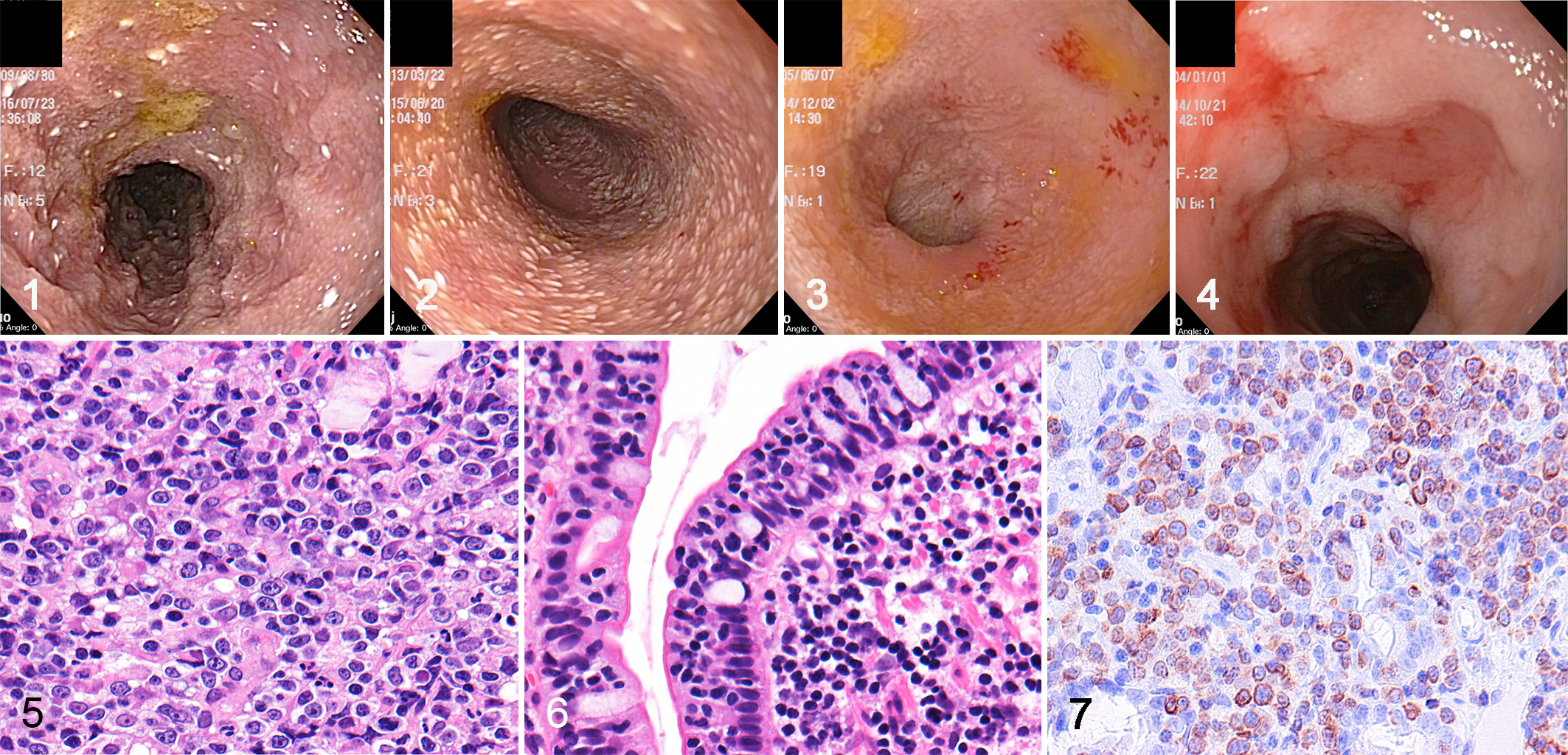
Small cell lymphoma (SCL), duodenum, case 19. Mild lacteal dilation is characterized by scattered white spots in the edematous mucous membrane.
Histopathology
Of the 50 cases of intestinal T-cell lymphoma, 17 (34%) were LCL and 33 were SCL (66%). Both the duodenum and ileum were affected in most of the cases, including 14/17 (82%) of those with LCL and 23/33 (69%) of those with SCL. Neoplastic lesions were also found in the stomach and/or colon in 2 of 17 LCL (11%) and 5 of 33 SCL (15%) cases. Apart from neoplastic lesions, hyperplastic polyps were found in 2 of 17 SCL cases (6%).
LCL was characterized by diffuse proliferation of neoplastic lymphocytes. Cells were moderate to large size and pleomorphic, with round to ovoid nuclei containing scattered chromatin and distinct nucleoli (Fig. 5). The neoplastic cells had slightly eosinophilic pale granules in the cytoplasm in a small number of cases (3/17; 18%). Epitheliotropic lymphocytes infiltrated the villous and/or crypt epithelium in 16 of 17 LCL cases (94%) while epitheliotropism was absent in 1 LCL. Conversely, SCL was characterized by small, round, monomorphic cells with round nuclei containing dense chromatin (Fig. 6). Epitheliotropic lymphocytes were present in all SCL cases, forming intraepithelial nests (≥5 clustered lymphocytes) in 10 of 33 (30%) cases and intraepithelial plaques (≥5 adjacent epithelial cells overrun by clustered lymphocytes) in 23 of 33 (70%) SCL cases. Mixed large and small cell phenotypes were observed in both LCL (2/17; 12%) and SCL (6/33;18%) cases. In those cases, diagnosis was made based on the predominant cell population. Marked villous atrophy was observed in all LCL and 19 of 33 (58%) SCL cases. The remaining 14 SCLs (42%) had mild or moderate villous atrophy. Ulcerated lesions were more common in LCL (8/17; 47%) compared with SCL (1/33; 3%). Increased number of nonneoplastic lymphocytes and plasma cells was present in 5 LCL (5/17; 29%) and all SCL cases. Eosinophil infiltration was infrequent in LCL (1/17; 6%) and absent in SCL. Marked changes in enterocyte appearance, including cuboidal morphology and cytoplasmic vacuoles, were frequent in LCL (6/17; 35%) compared with SCL (1/33; 3%). Multifocal crypt distention or crypt necrosis was common in both LCL (9/17; 53%) and SCL (18/33; 55%). Moderate to marked lacteal dilation was frequent in both LCL (7/17; 41%) and SCL (20/33; 61%).
IHC and IF
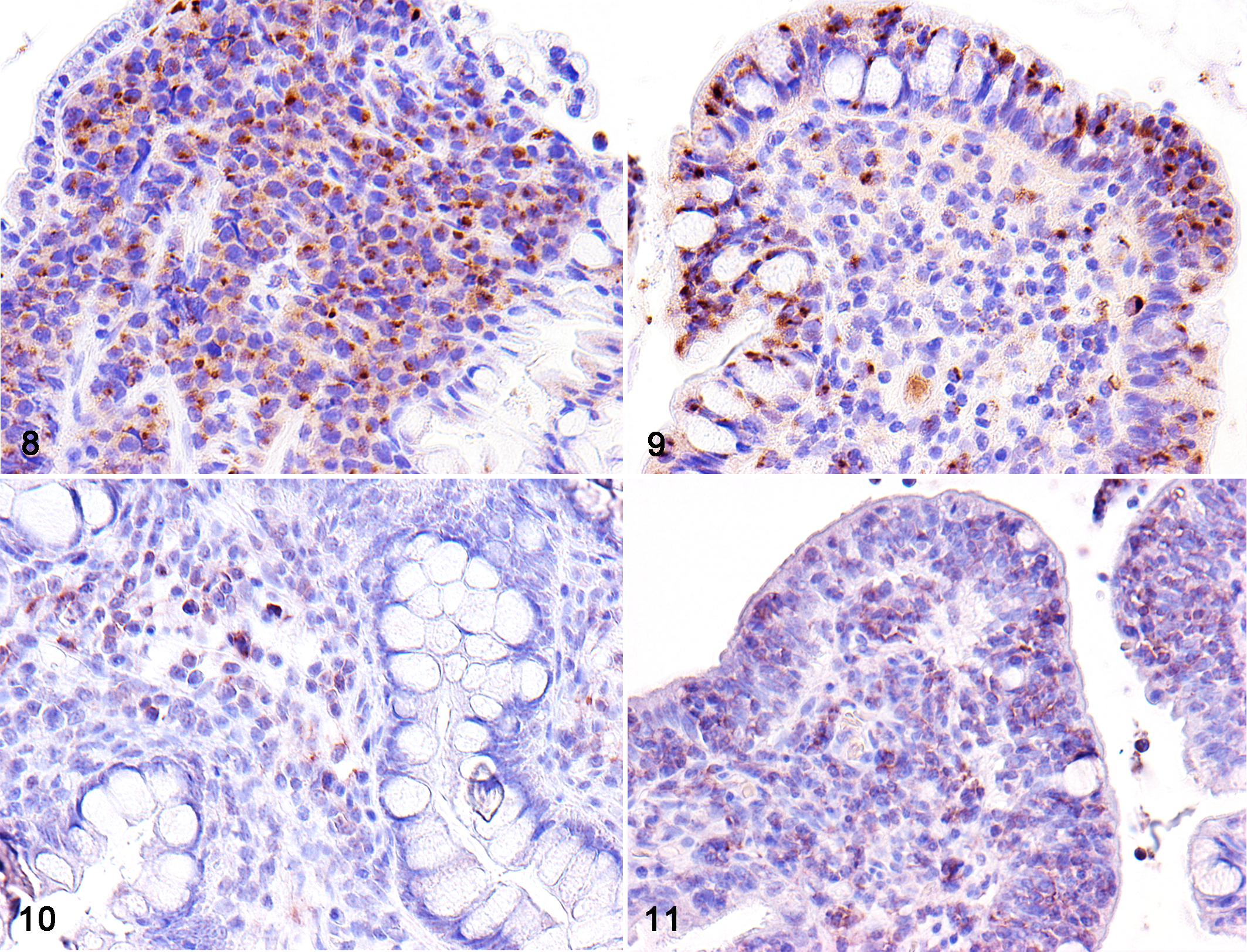
IHC results are summarized in Table 1. All neoplastic lymphocytes displayed a T-cell phenotype (CD3+, CD79α–; Fig. 7). Neoplastic cells had granular cytoplasmic expression of GRB in the majority of LCL (14/17; 82%) and SCL (27/33; 82%) cases (Figs. 8, 9). Membranous CD56 expression was observed in a small number of LCLs (2/17; 12%) while it was completely absent in SCL. CD56 expression in the neoplastic lymphocytes was weaker compared to positively labeled NK cells in the normal intestine.
Immunohistochemistry and Immunofluorescence Findings in Canine Intestinal T-cell Lymphoma.
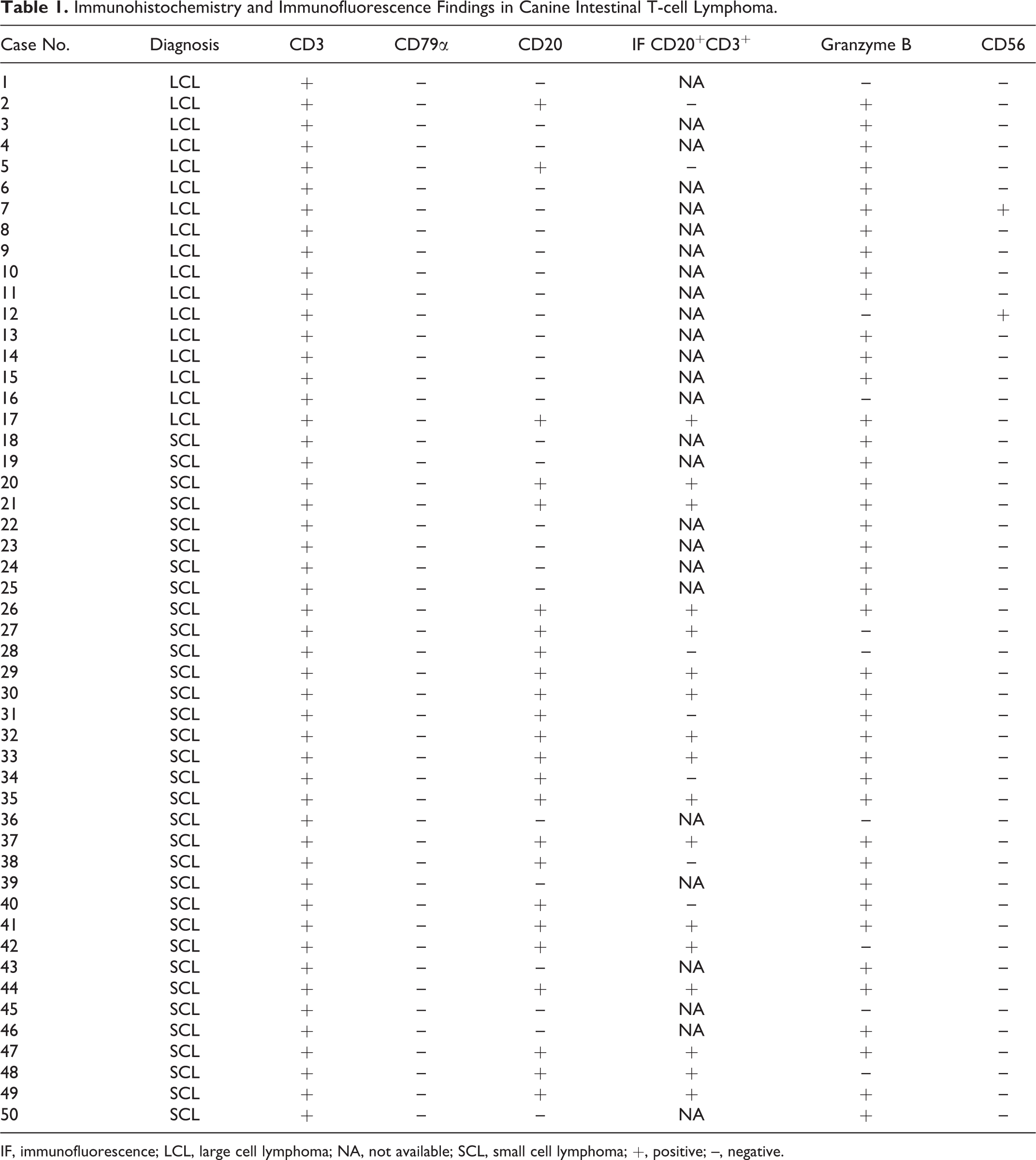
IF, immunofluorescence; LCL, large cell lymphoma; NA, not available; SCL, small cell lymphoma; +, positive; –, negative.
Large cell lymphoma (LCL), ileum, case 6. Immunohistochemistry (IHC) reveals granular cytoplasmic expression of granzyme B (GRB) in large neoplastic lymphocytes.
Of the 2 CD56-positive LCL cases, case No. 12, which had both smaller and larger neoplastic cells, displayed distinct differences in staining intensity according to cell morphology. Specifically, smaller cells in the duodenum had stronger CD56 expression than that of larger cells in the ileum (Figs. 10, 11).
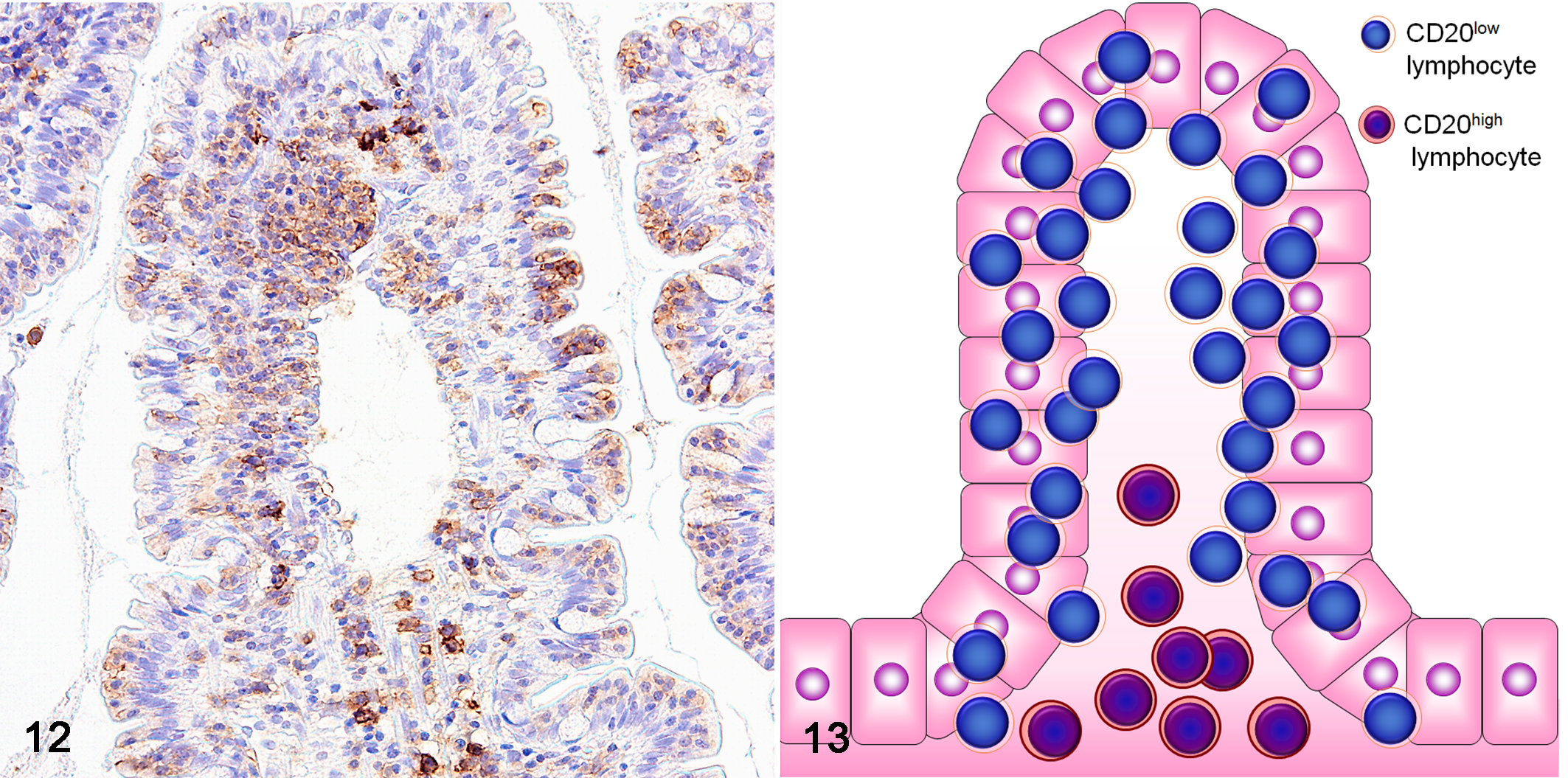
Neoplastic T-cells coexpressed CD20 in 3 of 17 LCLs (18%) and 21 of 33 SCLs (64%) (Fig. 12). Compared with the background B cells in the lamina propria, which showed clear CD20 expression (CD20high), the neoplastic T cells had weak CD20 expression (CD20low) (Fig. 13). LCL expressed a significantly higher MIB-1 index (55.1%) than SCL (29.8%) (P < .01).
Small cell lymphoma (SCL), ileum, case 42. Weakly CD20-positive lymphocytes are observed in the epithelial layer, while a small number of strongly CD20-positive lymphocytes are present in the lamina propria. Immunohistochemistry for CD20.
Those cases with CD20-positive T cells in IHC were examined by double-labeled IF for CD3 and CD20. CD20 coexpression was defined as expression of both CD3 and CD20 in more than half of the neoplastic cell population. CD20 and CD3 double-positive neoplastic cells were more frequently found in SCL (16/33; 48%) than in LCL (1/17; 5.8%) (P < .05) (Fig. 14). None of the CD56-positive T cells showed CD20 expression in IHC and thus were not further evaluated by IF. In those cases with GRB-positive neoplastic cells that were tested by IF, CD20-positive T cells were positive for GRB in 1 of 1 LCL and in 13 of 16 SCLs (81%).
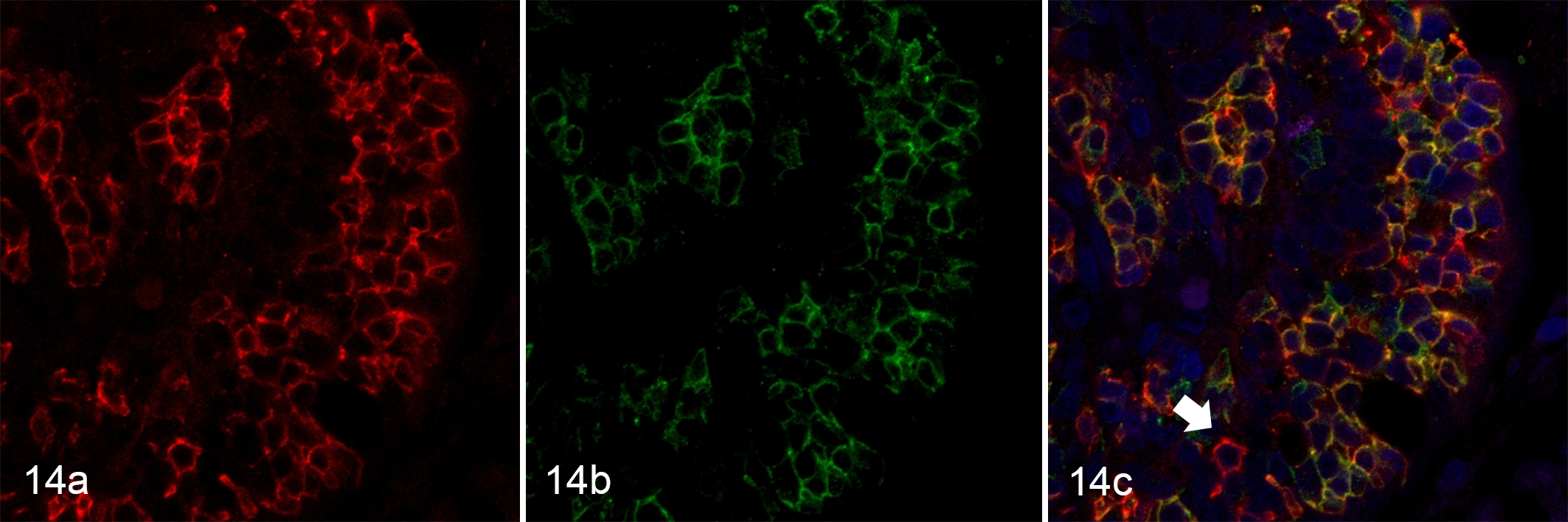
Small cell lymphoma (SCL), duodenum (villus), case 42. (a) Neoplastic cells with membranous CD20 labeling are present within the epithelium of the villus. (b) Neoplastic cells located in the epithelium also express CD3. (c) Neoplastic cells in the epithelial layer coexpress CD20 (red, Alexa Fluor 594) and CD3 (green, Alexa Fluor 488). Colocalization is indicated by yellow staining. There are a small number of B cells positive only for CD20 (red staining, arrow). Nuclei were counterstained with DAPI (blue).
In summary, the immunophenotypes of neoplastic cells in LCL were as follows: CD3+GRB+ (12/17; 70%), CD3+GRB– (2/17; 12%), CD3+GRB+CD20+ (1/17; 6%), CD3+GRB+CD56+ (1/17; 6%), and CD3+CD56+ (1/17; 6%), and those in SCL were as follows: CD3+GRB+ (14/33; 42%), CD3+GRB+CD20+ (13/33; 39%), CD3+GRB–CD20+ (3/33; 9%), and CD3+GRB– (3/33; 9%). Of those cases with mixed large and small cells, both types of cells shared the same immunophenotype except in the 3 SCLs (case Nos. 30, 47, 48) that showed decreased or absent expression of CD20 in large cells compared with small cells.
PCR for Antigen Receptor Gene Rearrangement
Clonality analysis was conducted for all LCL and 31 SCL cases. A clonal TCR gene rearrangement was detected in 14 of 17 LCL (82%) and 20 of 31 SCL (65%) cases. Clonal rearrangement of both the TCR gene and IgH gene was detected in 2 of 17 LCL (12%) and 6 of 31 SCL (19%) cases. A clonal IgH gene rearrangement was detected in 1 of 31 SCL (3%) cases. In 1 of 17 LCL (6%) and 4 of 31 SCL (13%) cases, neither TCR nor IgH clonal gene rearrangements were detected.
Discussion
Canine intestinal T-cell lymphomas can be classified into LCL and SCL based on their cell morphologies. 57 Recently, human EATL classification has been introduced for the classification of intestinal T-cell lymphoma in domestic animals. 8,57 However, the lineage and underlying pathological mechanisms of canine intestinal T-cell lymphoma have not been well established. The morphology of neoplastic lymphocytes in canine intestinal lymphoma was quite comparable to human EALT; however, several differences were observed. Monomorphic proliferation of medium-sized lymphocytes, which is characteristic in human EATL type II, was rarely seen in the present canine study. 1 Inflammatory infiltrates are less severe in human EATL type II compared to EATL type I. 1 In contrast, there was an increase in plasmacytic infiltrates in canine SCL compared to LCL. An earlier study reported eosinophil infiltration in LCL cases. 45 There was 1 LCL with increased eosinophil infiltration, but its clinical significance was not further determined. Marked villous atrophy and enterocyte degeneration were more commonly seen in LCL than in SCL. The degrees of lacteal dilation or crypt distention, however, were not necessarily associated with the morphological features of canine intestinal lymphoma. LCL cases had an increased number of Ki-67–positive intraepithelial lymphocytes compared to SCL. This finding may indicate that, unlike the human EATL subtypes, the canine subtypes may differ in behavior, with LCL having more aggressive behavior than SCL.
GRB, a serine protease, is frequently present in cytoplasmic granules in cytotoxic lymphocytes. 57 Human EATL types I and II are both positive for GRB. 1,54 Similarly, canine LCL and SCL were predominantly positive for GRB, which indicates a cytotoxic immunophenotype in most canine intestinal T-cell lymphomas regardless of cell morphology.
EATL type II neoplastic lymphocytes often have a CD56+ phenotype. 9,14 CD56 is a neural cell adhesion molecule expressed on the surface of NK cells, neurons, and skeletal muscle cells. 31,33,50 CD56 is also expressed in a subset of peripheral cytotoxic T lymphocytes that function without major histocompatibility complex (MHC) restriction. 32 EATL type II neoplastic cells might arise from CD8+ cytotoxic T cells, which may upregulate GRB and CD56 during tumor progression. 54 In this study, 2 LCL cases (case Nos. 7 and 12) were positive for CD3 and CD56. Differentiation of CD56+ T-cell lymphoma from NK/T-cell lymphoma could be challenging. Epstein-Barr encoding region (EBER) is useful for differentiating NK/T-cell lymphoma from CD56-positive EATL type II in humans. 1 However, the utility of EBER has not been further evaluated in the current study. It is interesting to note that case No. 12, which had both larger and smaller neoplastic cells, showed a distinctive staining pattern of CD56 expression, in which the smaller cells in the duodenum had stronger CD56 expression than the larger cells in the ileum. Complete absence of CD56 in neoplastic cells in SCL cases may indicate a fundamental difference between canine SCL and human EATL type II.
CD20, a transmembrane protein, has a role in transmembrane Ca2+ movement 5 and is often used as a marker of B-cell lymphoma. CD3+CD20+ T-cell lymphoma is an infrequent condition that has been described in human EATL cases. 9,54 The clinical implications of CD20-expressing T-cell lymphoma remain unclear. 52 CD20-expressing cutaneous and intestinal T-cell lymphomas have been described in dogs. 4,44 In the present study, IF revealed consistent coexpression of CD20 and CD3 in 5.8% of LCL and 48% of SCL cases. Inconsistency in CD20 expression between IHC and IF could be associated with difference in affinity of the antibody or by difference in cell population within the areas sampled for IHC and IF. CD20 expression is considered rare in human EATL, although 1 study described aberrant coexpression in 24% of EATL type II. 54 Human studies have suggested an increase in circulating CD20low T cells with age and neoplastic transformation of these T cells. 47,51 Alternatively, a high frequency of CD20 coexpressing canine SCL may partly be explained by nonneoplastic proliferation of activated T cells, accompanied by TCR gene rearrangements. The increased CD20-expressing T cells in monkey lymph nodes following stimulation with simian immunodeficiency virus suggested that CD20 is associated with T-cell activation. 39 Alternatively, CD20 positivity in T cells could be a nonspecific reaction. 23 Immunophenotypically, a larger population of CD20low-positive T cells expressed CD8 in human cases. 26 Unfortunately, CD8 expression in canine intestinal T-cell lymphoma could not be investigated in this study due to lack of a commercially available antibody to CD8.
A negative result for a clonal TCR gene rearrangement was detected in 6 cases. However, those results do not rule out T-cell lymphoma. Several studies described lower sensitivity of clonality analysis in intestinal T-cell lymphoma compared with lymphoid tumors in other sites. 18 This could be explained by the absence of neoplastic lymphocytes in the biopsy samples due to patchy distribution of intestinal T-cell lymphoma, polyclonal background, insufficient coverage of the primers, primer site mutation, or chromosomal aberrations. 24,28 Similarly, presence of clonality is not necessarily indicative of proliferation of neoplastic cells. Although TCR gene rearrangement was detected in 94% and 84% of LCL and SCL cases, respectively, a clonal TCR/IgH gene rearrangement has been reported in nonneoplastic disorders in both domestic animals and humans. 2,3,6,7,16,21,25
The WHO classification describes both subtypes of EATL (type I and II) as tumors of intraepithelial T cells, with predominantly cytotoxic T-cell features of the αβ phenotype. 11,27 However, a recent study has demonstrated that neoplastic lymphocytes in EATL type II are more commonly of the γδ phenotype. 9 Although not commonly available, identification of the αβ or γδ phenotype would allow further classification of the neoplastic lymphocytes in canine intestinal T-cell lymphoma.
There was an inconsistency between canine intestinal T-cell lymphoma and human EATL. Neoplastic cells of canine intestinal T-cell lymphoma predominantly had a GRB+CD56– immunophenotype regardless of the cell morphology. Moreover, the mixed small and large cell cases were not uncommon. CD20-expressing T cells in SCL could potentially indicate a distinct subpopulation of T cells or activated T cells. In humans, the 5-year survival rate for EATL is approximately 10%, with EATL type II carrying a less favorable outcome. 43 In contrast, canine SCL showed a better prognosis compared with LCL in a recent study. 38,41 SCL has been recognized as a slowly progressive intestinal T-cell lymphoma that may arise from inflammation. 17,37 The prolonged clinical course in canine SCL might create a favorable situation for transformation into more malignant LCL over time in a subset of dogs.
This may suggest loss of transient CD20 expression in a subset of SCL according to transformation into LCL (Fig. 15). Alternatively, the slowly progressive clinical course of SCL may indicate a benign entity that is independent of later LCL development. Indolent T-cell lymphoproliferative disorder (TCLPD) of the gastrointestinal tract has been recently included in the revised WHO classification of human lymphoid neoplasms. 46,53 This is a rare clonal condition, usually composed of CD8+ T cells. 36,53 It has an indolent clinical course, and malignant transformation remains unclear. 46 Morphological and clinical similarities have been suggested between feline EATL type II and TCLPD. 59 Although canine SCL is morphologically slightly different from TCLPD, these 2 conditions clinically share several common features.
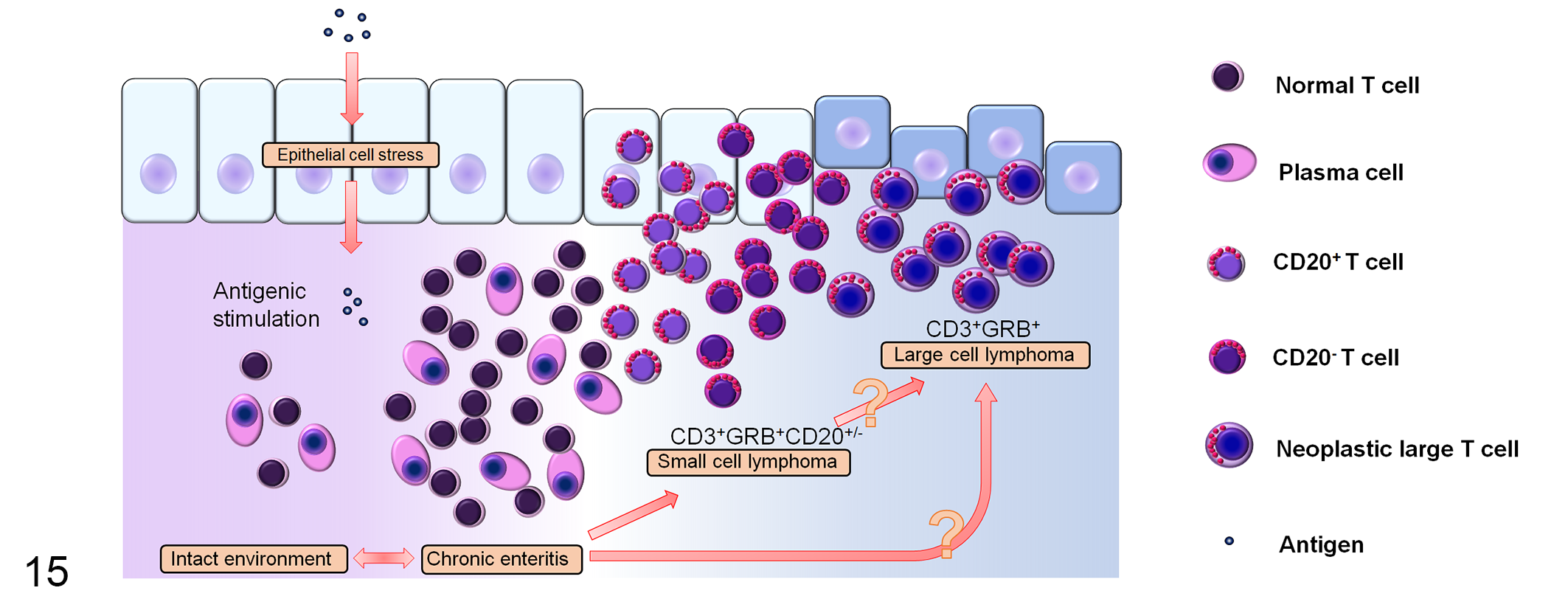
Schematic diagram depicting the proposed development of canine intestinal T-cell lymphoma. Inflammation and/or other pathological processes may promote progression of intestinal T-cell lymphoma. Neoplastic cells predominantly have granzyme B+CD56– immunophenotype regardless of cell morphology. With progression of small cell lymphoma (SCL), there is an increase in the number of CD20 coexpressing T cells, which may represent a distinct subpopulation of T cells or activated T cells. A subset of SCL transforms into the more aggressive large cell lymphoma and loses CD20 expression.
EATL types I and II are distinct entities with different ethnic distributions and immunohistochemical features. In addition, recent human studies have identified variations in predominant JAK/STAT mutations between types I and II. 30,40,42 Further investigation of genetic aberrations and immunophenotypes (CD8/4 and TCR αβ/γδ) of SCL and LCL will be valuable for classification of canine intestinal T-cell lymphoma (Table 2).
Comparison of Typical Findings in Human EATL and Canine Intestinal T-Cell Lymphoma.

Abbreviations: EATL, Enteropathy-associated T-cell lymphoma; IEL, intraepithelial lymphocyte.
a CD56 expression was noted in an extremely small number of large cell lymphoma cases.
In conclusion, canine intestinal T-cell lymphomas had diverse morphologies, ranging from small monomorphic intraepithelial lymphocytes to large atypical cells with extensive Ki-67 expression. Lack of CD56 expression in SCL cells suggests differences between canine intestinal SCL and human EATL type II. Moreover, LCL and SCL shared a relatively homogeneous immunophenotype, and mixed large and small cell cases were not uncommon. These findings suggest large cell transformation of SCL in canine intestinal T-cell lymphoma. Although identification of the TCR-αβ or TCR-γδ phenotype and additional immunophenotypical characterization will be necessary for complete classification, our study provides valuable information on the cellular origins of canine intestinal T-cell lymphoma.
Supplemental Material
Supplemental Material, DS1_VET_10.1177_0300985818800015 - Immunohistochemical Profiling of Canine Intestinal T-Cell Lymphomas
Supplemental Material, DS1_VET_10.1177_0300985818800015 for Immunohistochemical Profiling of Canine Intestinal T-Cell Lymphomas by Isao Matsumoto, Ko Nakashima, Yuko Goto-Koshino, James Kenn Chambers, Hajime Tsujimoto, Hiroyuki Nakayama, and Kazuyuki Uchida in Veterinary Pathology
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
