Abstract
Three cases of feline cerebellar hypoplasia are presented. At the time of examination, the ages of the cats ranged from 2 months to 1 year. Necropsy revealed cerebellar and pons hypoplasia. Polymerase chain reaction for parvoviral deoxyribonucleic acid was positive in cerebellar tissue. Cell-specific immunolabeling was used to characterize the lesions, which were characterized into 2 types. In type 1 lesions, the cortex was nearly agranular, with an extremely thin molecular layer; the Purkinje cells were randomly placed and oriented, and their stunted main dendrite produced a thorn-covered atrophic dendritic tree; the basket cell axons ran randomly and had dysmorphic endings; and myelinated fibers were severely reduced in folia axes. In type 2 lesions, the cortex was hypogranular; the Purkinje cells were linearly organized, but their main dendrite extended too far in the molecular layer before giving up smooth, bent secondary dendrites; many basket cells were located along the cerebellar surface, and their axons ran at right angle to the surface; myelinated fibers were moderately reduced. Defects in climbing fiber synapse translocation and elimination were evident in both types of lesion. This immunohistologic study allowed a comparison between lesions in these spontaneous cerebellar hypoplasia cases with those documented when using silver impregnation studies after perinatal experimental cerebellar damage. Such a comparison is consistent with viral infection that occurs before birth in all 3 cases. Progress in parvovirus biology knowledge suggests that viral NS1 protein cytotoxicity might explain degenerative changes in the Purkinje cells that were present, in addition to the development defect.
A description of feline congenital ataxia can be traced back to 1885. 27 In 1966, evidence for a viral etiology was found. 36 Shortly after, the causative agent was identified as feline panleukopenia virus (FPLV). 35 Cerebellar hypoplasia is the hallmark of this developmental disorder. The developing cerebellum external granular layer proved to be a primary neural target of the viral attack, although changes in Purkinje cells were also regularly reported. 23, 36, 37 As a consequence of the external granule-cell destruction, feline parvovirus–associated cerebellar hypoplasia is generally regarded as a granuloprival disease. 27 Less consistently recognized features are atrophy of the pons, Purkinje cell changes, Purkinje cell heterotopia, and the presence of mineralization in the periventricular area or in the internal capsule. 23, 27, 28, 37, 40, 55
Attempts to reproduce the disease through neonatal FPLV infection of kittens failed to induce the classical clinical signs, although reduced cerebellar size was observed and much information about viral distribution and damage was obtained. 23, 37 In utero FPLV infection in pregnant queens and ferrets 20 days before parturition reproduced signs and lesions observed in naturally occurring cases. 37 Earlier infection (48 days or 38 days before parturition) induced hydranencephaly, as well as cerebellar hypoplasia. 27, 53 In these reports, the lesion description was based mostly on routine staining methods.
Other experimental studies have been helpful in understanding the pathologic processes, for example, experimental destruction of mitotically active cells conducted at various times during cerebellar cortex maturation in laboratory animals, including cats, with X-ray irradiation or the toxic substance methylazoxymethanol. 1, 2, 4– 8, 12 Information can also be drawn from the study of the granuloprival cerebellar hypoplasia of the weaver mutant mouse. 54 The lesion description was based on silver impregnation methods in these latter experimental procedures.
In the present study, we took advantage of 2 methodologic progresses: the advent of polymerase chain reaction (PCR) tests by using paraffin-embedded samples that prove the presence of FPLV in naturally occurring cases, 52 and the mastering of immunohistology that allows cell-specific or cell processes–specific staining. 49 Such cell-specific or cell processes–specific descriptions on PCR-confirmed naturally occurring cases have not been attempted before. This method proved as precise as silver impregnation methods and, hence, made it possible to compare lesions observed in naturally occurring parvovirus-associated feline cerebellar hypoplasia with those after experimental damage to the developing cerebellum in mouse, rat, and cat. 1, 2, 4– 8, 12
Materials and Methods
Case history
Cat Nos. 1 and 2 were litter mates. Ataxia became obvious around 4 weeks of age, when kitten locomotor skill and equilibrium is expected to rapidly improve. 41 Cat No. 1 was euthanatized at 2 months of age. Cat No. 2 had a nonprogressive ataxia but was euthanatized at 1 year of age, after developing feline leukemia virus–associated lymphoma that involved the superficial lymph nodes and kidneys in conjunction with a Haemobartonella-associated anemia. Cat No. 3 was presented while still immature but with a complete adult dentition. This ataxic animal was a newcomer on waste ground and was kept away from food by resident feral cats. Euthanasia was eventually requested. The ataxia in these 3 cats was characterized by a wide base stance, limb-movement hypermetria, and intention tremor of the head.
Histopathology
The brain and the cranial cervical spinal cord were immediately removed after death and fixed in 4% formaldehyde, in 0.1 M phosphate buffer, pH 7.4 for 4 days at 4°C, followed by a 24-hour rinse in 0.1 M phosphate buffer. The brain was cut along the sagittal plane, and the left half was cut transversally along fixed landmarks in 10 parts. The other half was used to prepare parasagittal sections. The different sections were dehydrated and embedded in paraffin. Specimens were cut semiserially at 5 μm; every 20th section was stained with Luxol fast blue and cresyl violet and, some of them, with hematoxylin and eosin.
Immunohistology
The technique used was described elsewhere. 49 Briefly, rehydrated slides immersed in citrate buffer pH 6 were heated close to the boiling point for 15 minutes. After nonspecific site blocking with normal sheep serum (1/20), the primary antibody was applied overnight. Goat anti-rabbit or goat anti-mouse serum was used as a secondary antibody. Binding was visualized by using the peroxidase antiperoxidase method and diaminobenzidine as chromogen. Phosphate buffer saline solution 0.01 M was used for the intermediary rinsing. Type, dilution, source, and relevant specificity of the 7 primary antisera used are reported in Table 1.
Type, dilution, source and specificity of the antibodies used in the present immunohistologic study.
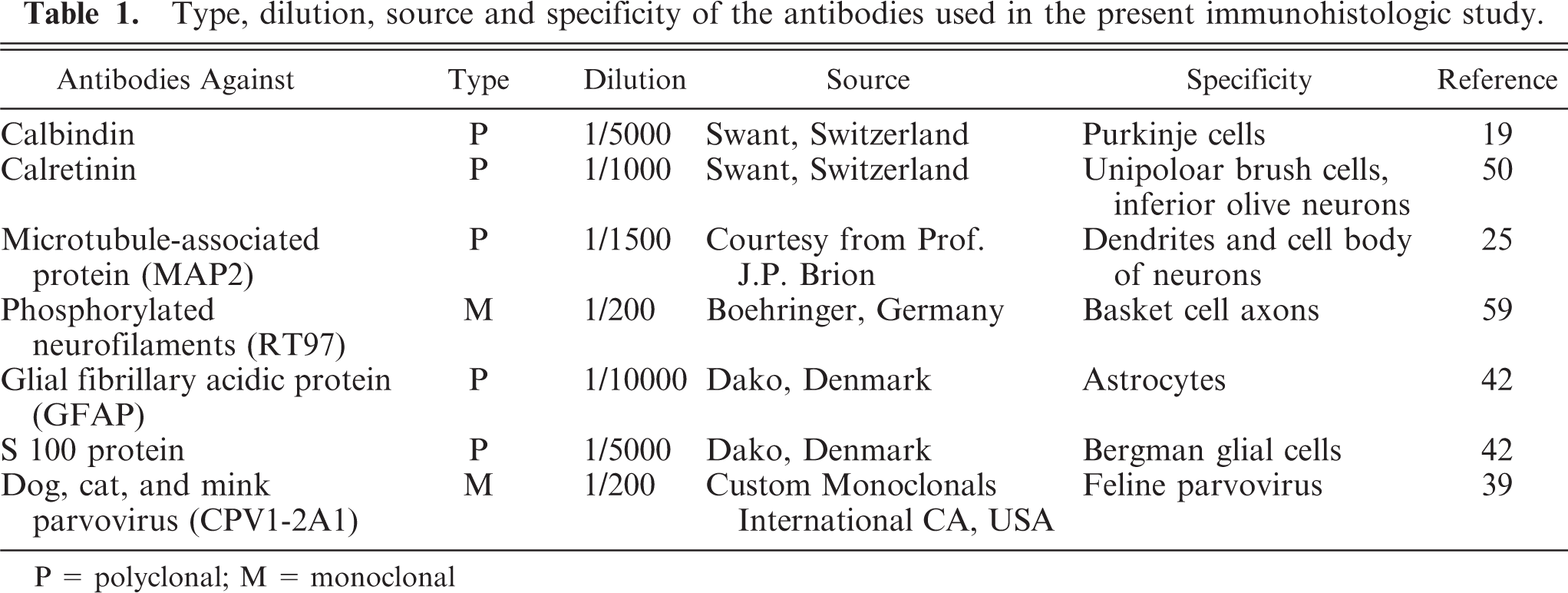
P = polyclonal; M = monoclonal
PCR
The procedure was similar to that already described. 52 Briefly, sections from paraffin-embedded cerebellum from the 3 cats were available for PCR amplification. Samples of the positive (gut from a previously confirmed feline panleukopenia case, a gift from Catherine Botteron, University of Berne) and the negative (cerebellum and gut from healthy cats) controls were similarly formalin fixed and paraffin embedded. Fifteen to 20 sections (15 μm) were deparaffinized and digested with proteinase K (100 μg/ml in lysis buffer) for 24 to 72 hours at 50°C. A primer pair was used to amplify a 214-bp product from the histone 3.3 gene and to assess successful deoxyribonucleic acid (DNA) extraction and sample integrity. 52
DNA extraction was performed with a phenol, chloroform, isoamyl alcohol mix (50, 48, and 2%, respectively) and a chloroform, isoamyl alcohol mix (96 and 4%, respectively) with intermediary centrifugations, followed by isopropanolol precipitation and 75% ethanol washing. The 3 parvovirus primer pairs (Eurogentec, Liege, Belgium) were previously described and amplified a 193-bp product in the VP1 gene, and 215- and 184-bp products of the VP2 gene of the FPLV. 52
PCRs were carried out in a 20-μl reaction mixture that included 4 μl buffer (Promega, Madison, WI), 0.8 μl deoxynucleotide mix (dNTP, 20 mM; Eurogentec), 1 μl forward primer and reverse primer (1 μg/μl), 0.1 μl DNA polymerase (5 μg/μl, Go Taq DNA Polymerase, Promega), and 1 μl DNA extract sample. After a 5-minute denaturation at 95°C, 35 cycles were carried out, including 1-minute denaturation at 95°C, annealing for 1 minute at 55°C, an extension for 1 minute at 72°C, eventually followed by an additional extension for 7 minutes at 72°C. PCR amplification products were analyzed by electrophoresis on a 1.6% agarose gel that contained ethidium bromide (0.075%), and PCR products were visualized under ultraviolet light.
Results
Macroscopic findings
Compared with normal cats, the 3 cats with ataxia had a small cerebellum (Fig. 1). Hemispheres and vermis were similarly affected (Fig. 1a), with severely atrophic folia (Fig. 1b). Cerebral peduncles extended up to the pyramids, without any protrusion at the level of the pons and almost no transverse fibers (Fig. 1c).
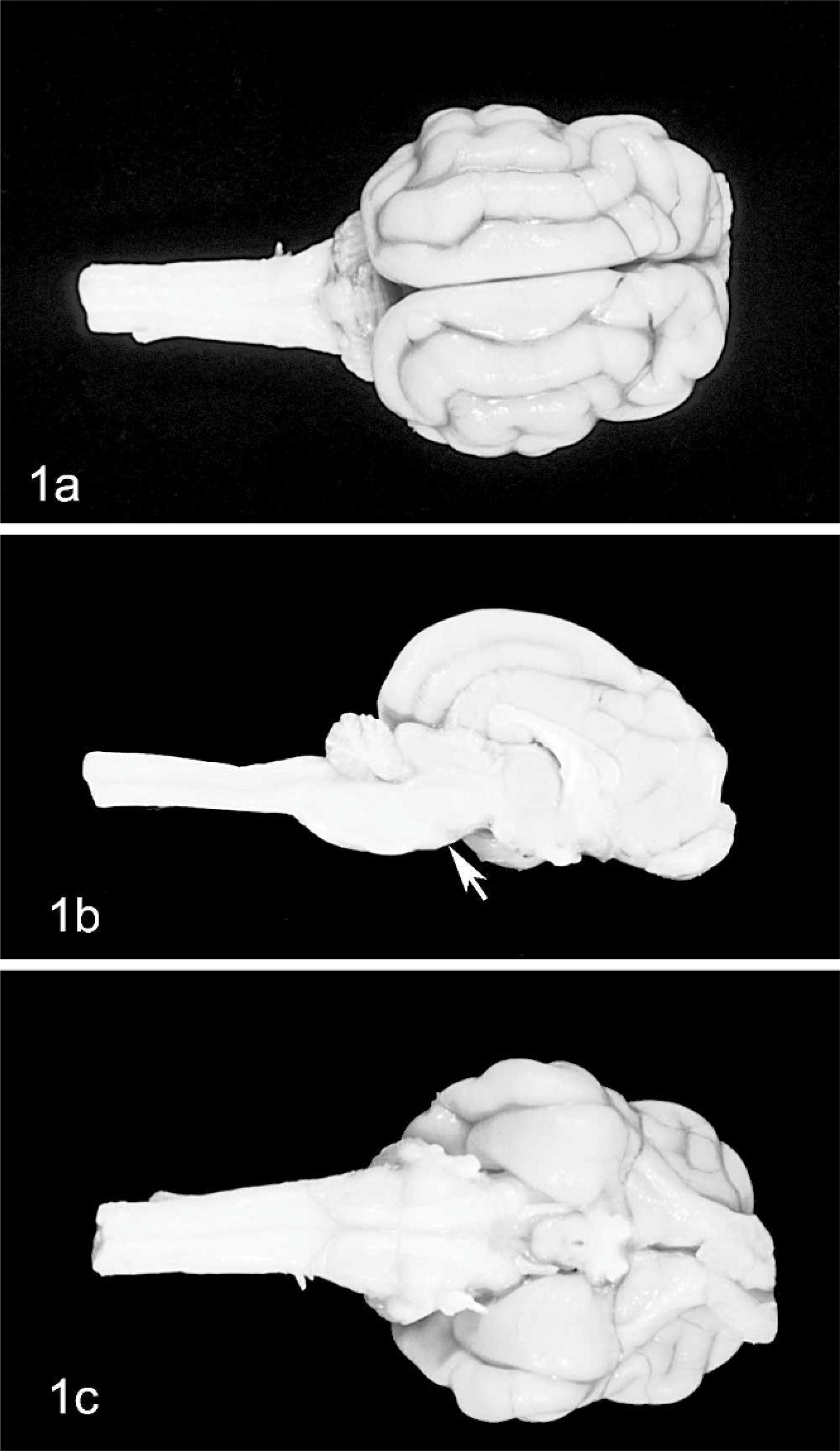
Brain; cat No. 2.
Microscopic analysis
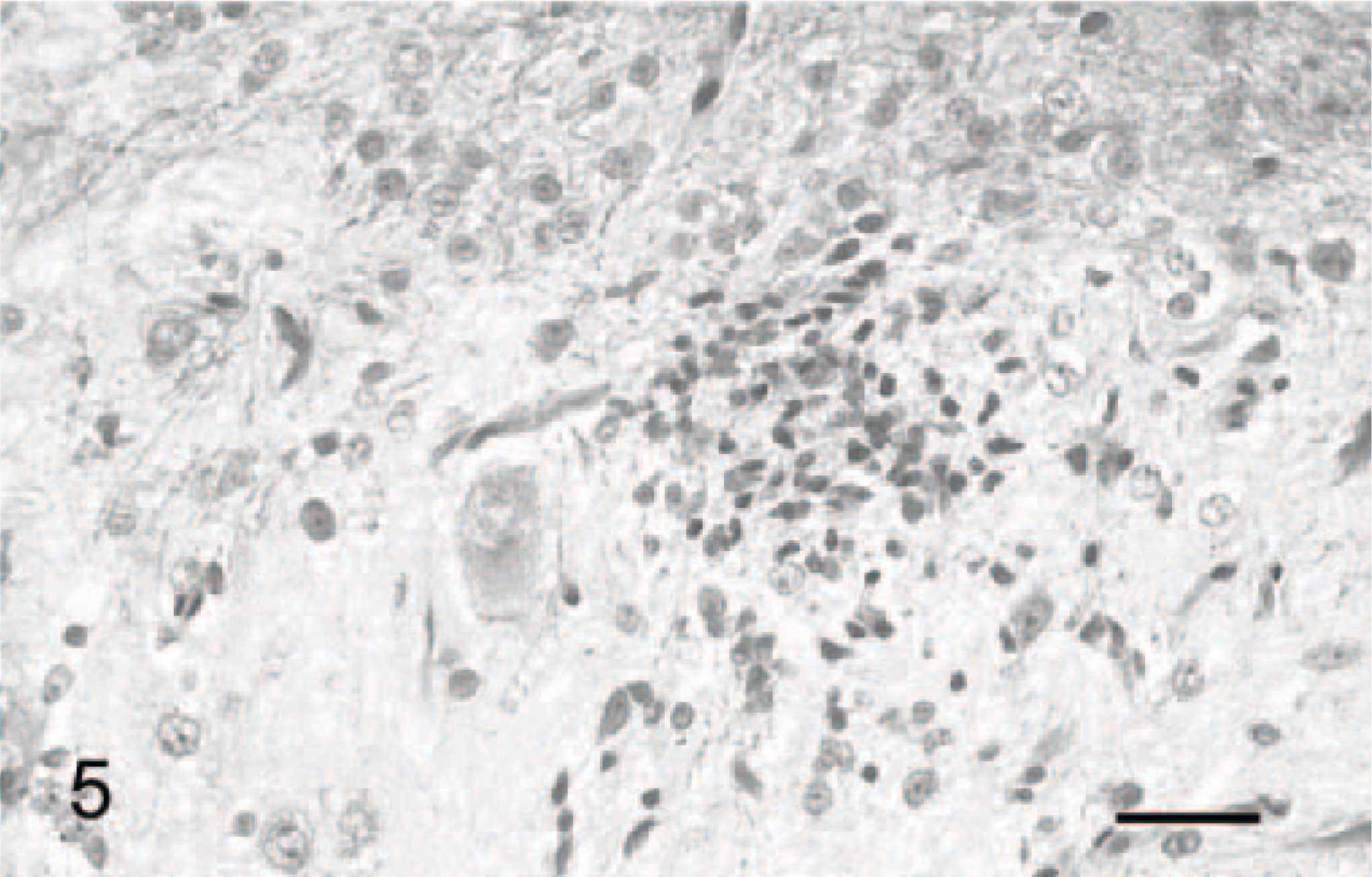
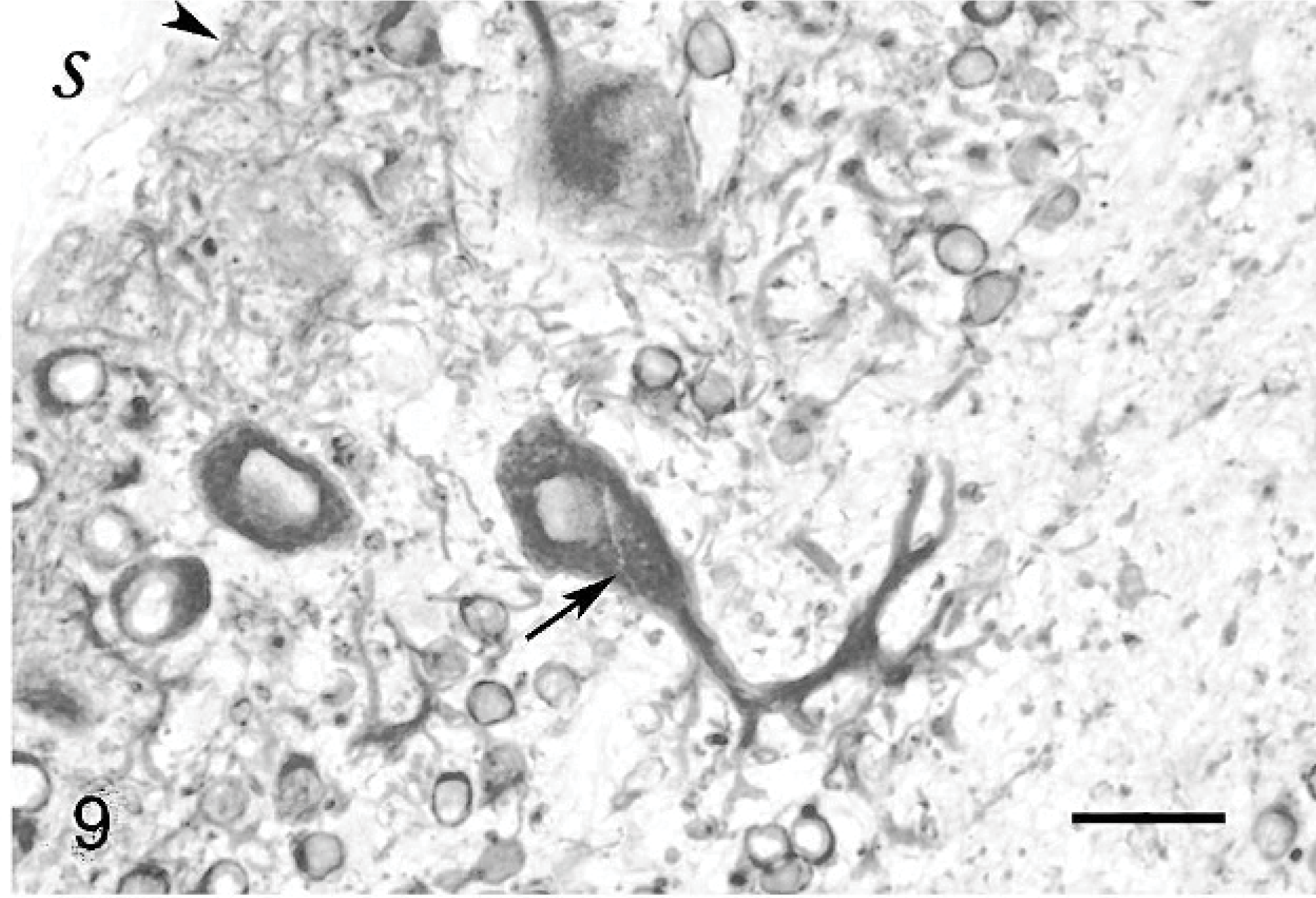
Changes were restricted to the metencephalon. In all cats, the granular layer was reduced, from moderately decreased (Fig. 2) to almost absent (Figs. 3, 4). Purkinje cells were moderately decreased in number and often severely misaligned (Figs. 3, 4). Many of these were vacuolated. In cat Nos. 1 and 3, but not in cat No. 2 (the oldest one of this series), lymphocyte aggregates, up to an half-dozen cells thick, could be seen in the leptomeninges (Fig. 2). Satellitosis of Purkinje cells was relatively often observed, as well as occasional neuronophagia nodules (Fig. 5). No abnormality was observed in the caudal brainstem, which was also devoid of inflammatory cells. Among the precerebellar nuclei, lateral cuneatus, lateral reticular nuclei, lateral vestibular nucleus, and inferior olivary nucleus were unremarkable; however, the pontine nuclei were reduced to a few neurons. Cerebellar white matter was relatively preserved in cat Nos. 1 and 2 (Figs. 2, and 3, respectively) but poorly myelinated in cat No. 3 (Fig. 4). The brachium pontis was abnormally thin.
Cerebellar cortex; cat No. 3. Glial nodule in the Purkinje cell layer, suggesting destruction of some Purkinje cells. Luxol fast blue-cresyl violet. Bar = 35 μm.
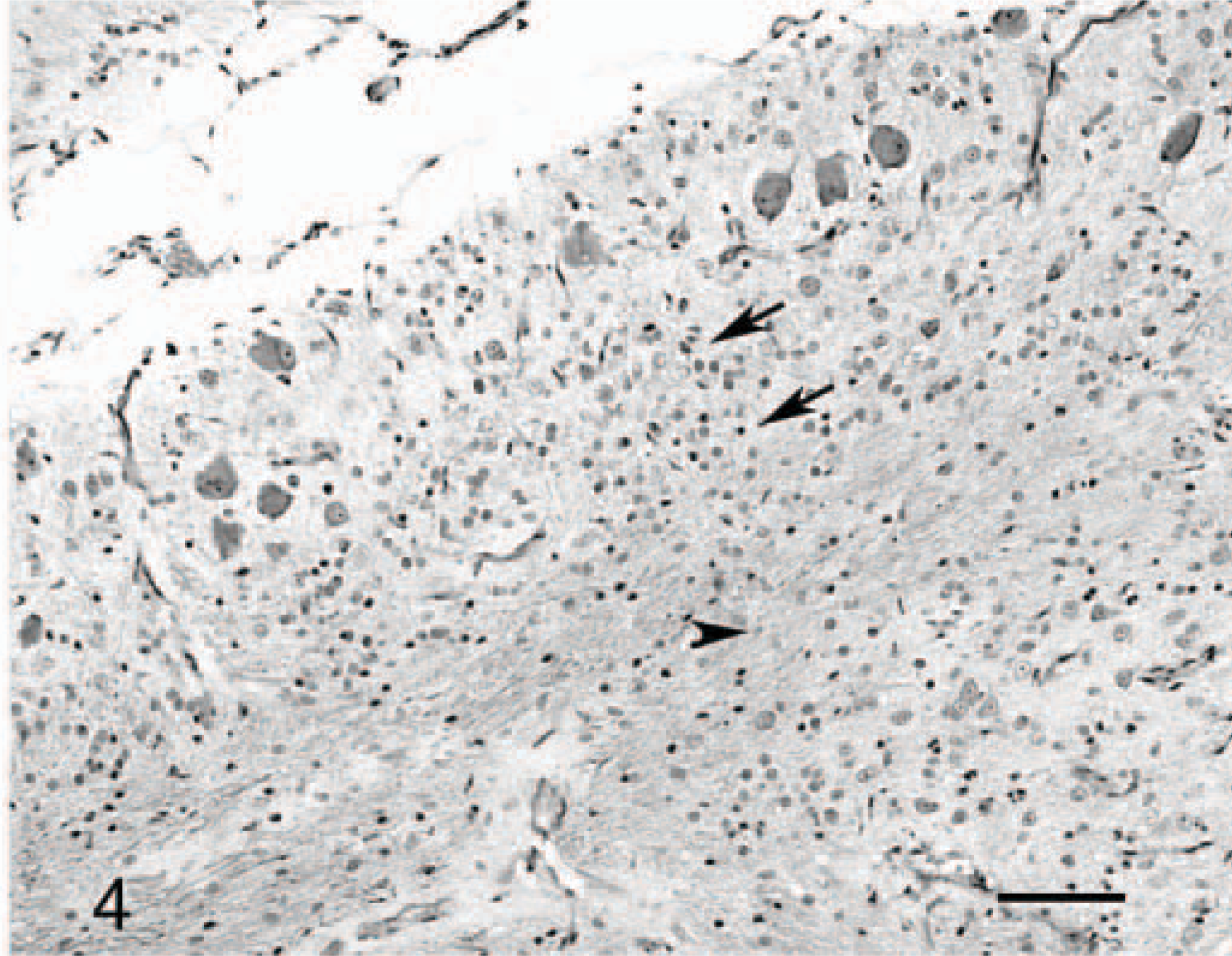
Cerebellar cortex; cat No. 3. Absence of distinct granular layer (arrow) and poorly myelinated white axes (arrow heads). Luxol fast blue-cresyl violet. Bar = 90 μm.
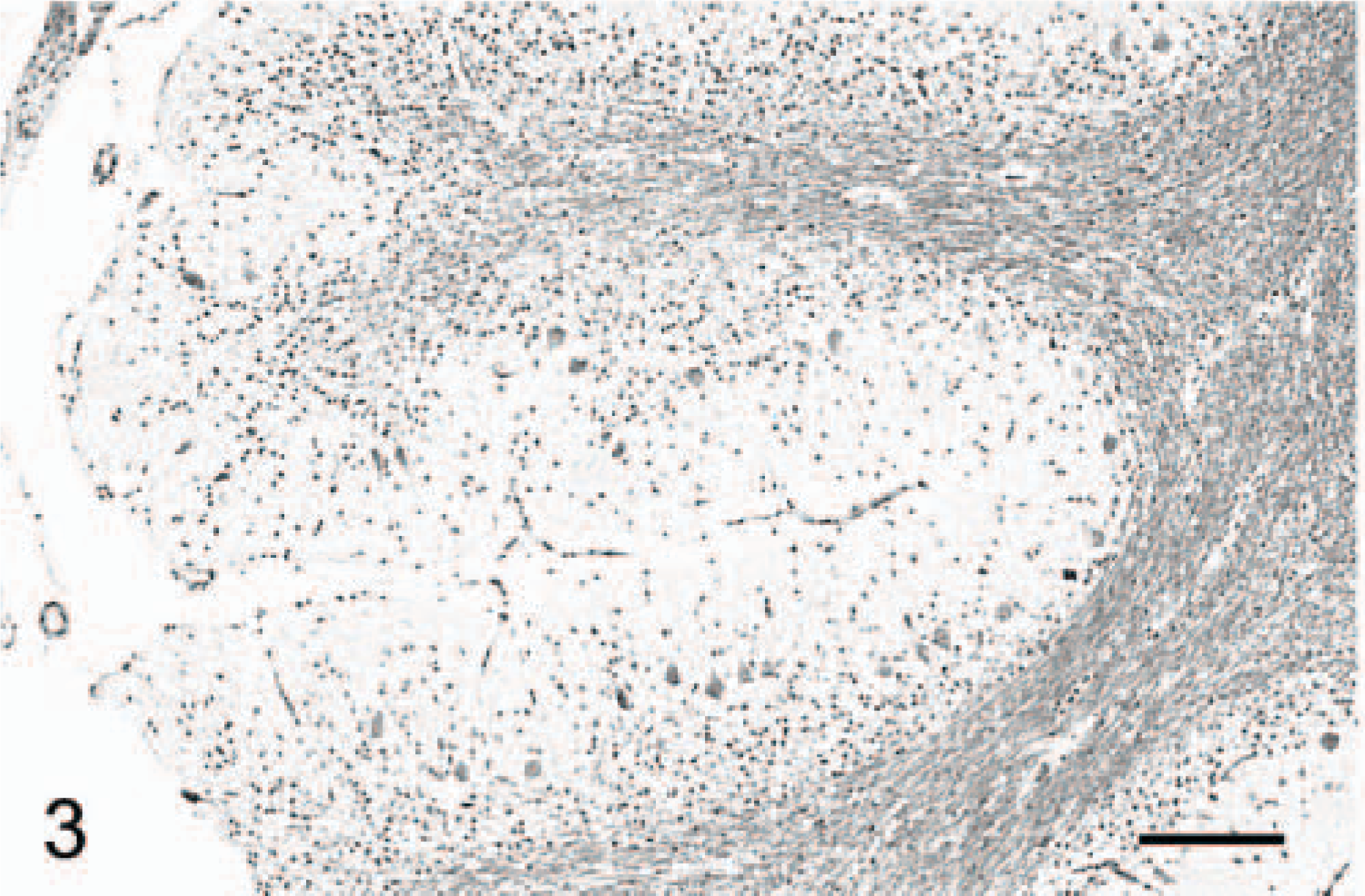
Cerebellar cortex; cat No. 2. In this 1-year-old cat, no inflammatory cell could be found. Luxol fast blue-cresyl violet. Bar = 190 μm.
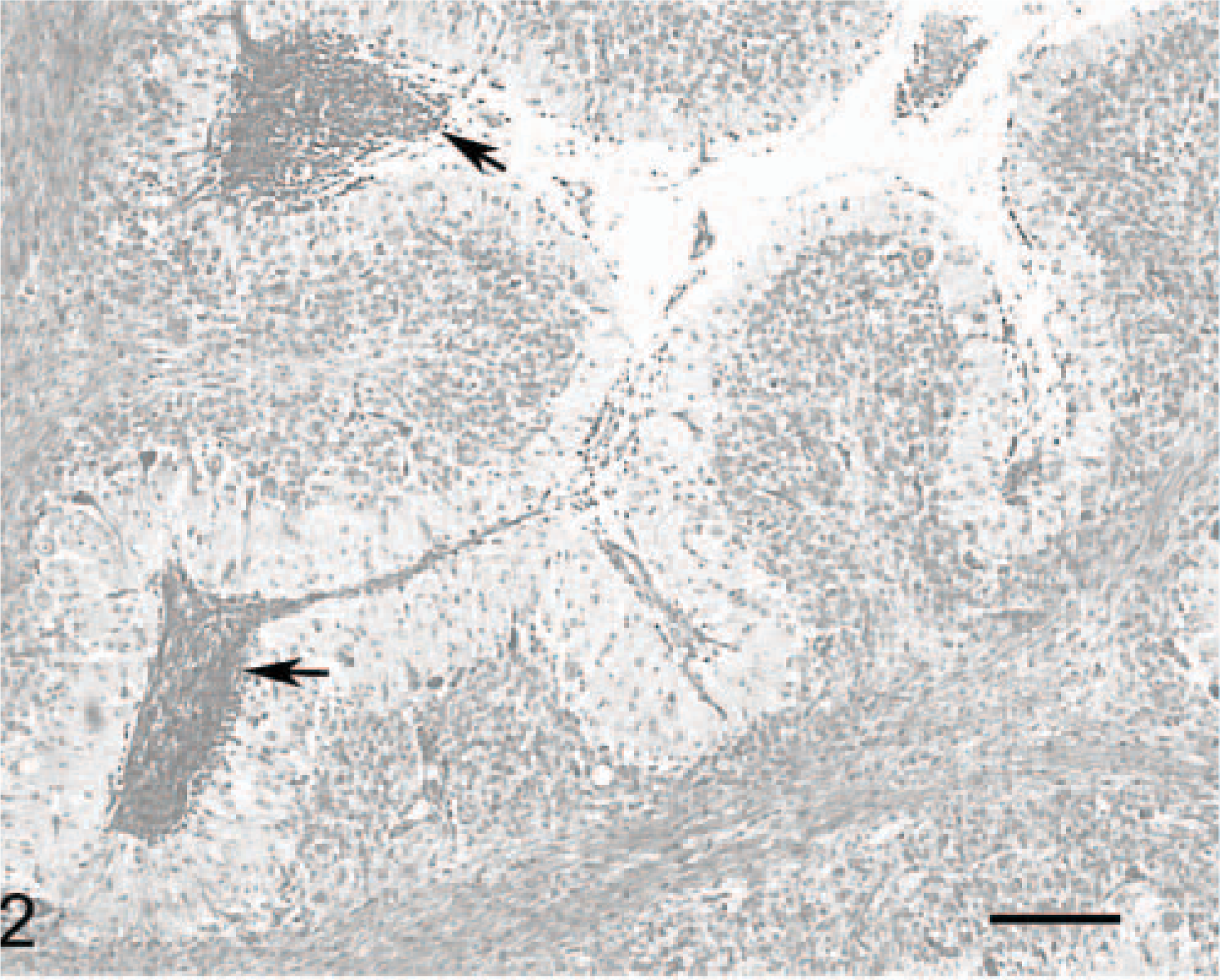
Cerebellar cortex; cat No. 1. The overall anatomy is preserved along the atrophic folia. Note the inflammatory infiltrates in the meninges of this 2-month-old kitten (arrows). Inflammatory cell infiltration was not retrieved in the 1-year-old cat (No. 2) of the series. Luxol fast blue-cresyl violet. Bar = 200 μm.
Purkinje cells
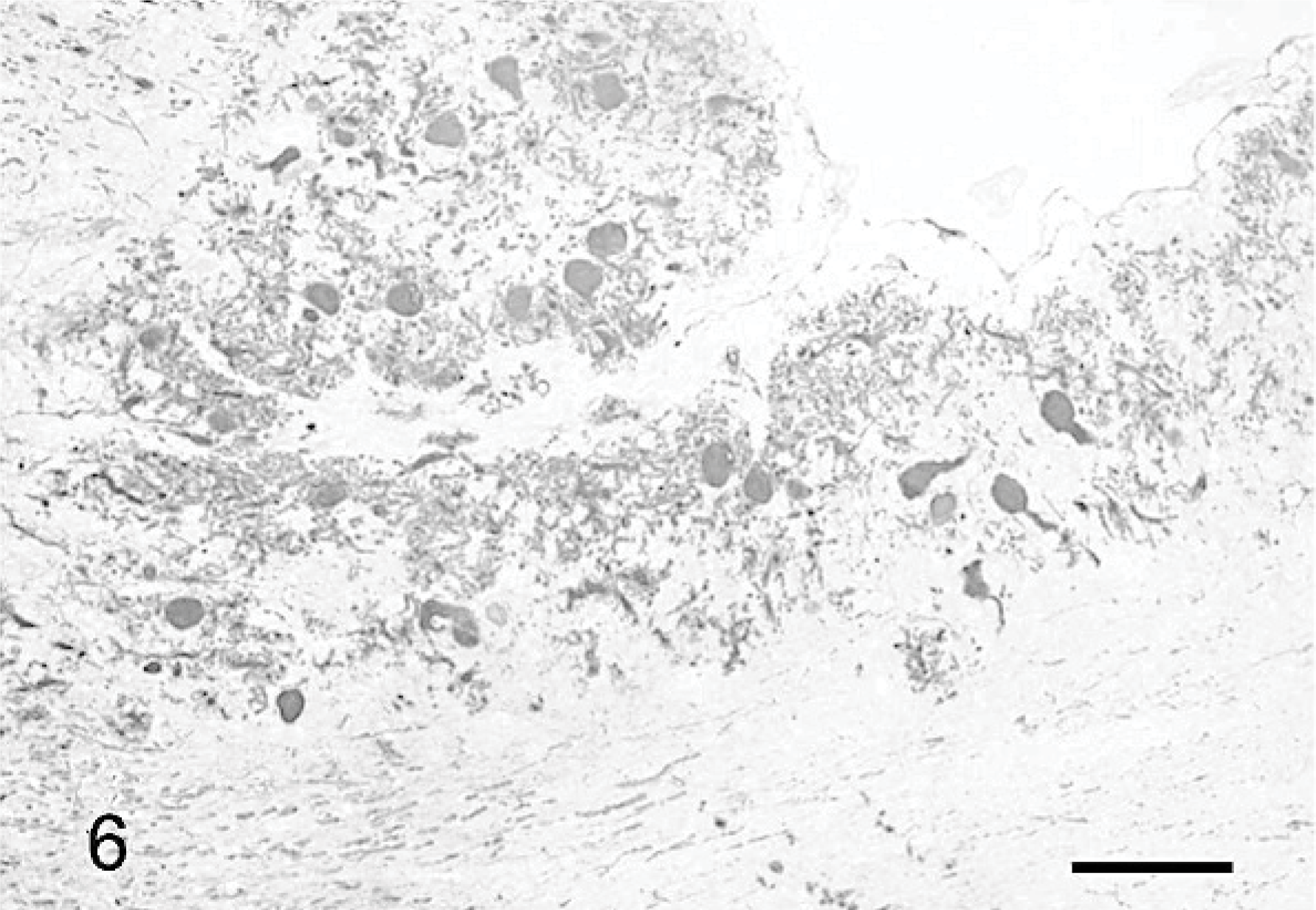
Purkinje cells and their processes were demonstrated by using anticalbidin and anti-MAP2 Ab. In normal cats, Purkinje cells are linearly organized between the superficial molecular layer and the deeper granule-cell layer, and they extend their dendritic tree toward the pial surface. Two different organizations of residual cells from the markedly atrophic cerebellar cortices could be distinguished. In some areas of the cerebellar cortex of cats No. 1 and 2 and most of the cerebellar cortex of cat No. 3, the Purkinje cells were scattered randomly, without definite orientation (Fig. 6). Conversely, in most parts of cat No. 1 and the cerebellar cortex of cat No. 2, and in a few areas of the cortex of cat No. 3, the Purkinje cells had their normal linear arrangement, with their dendrites extending to the thin molecular layer (Fig. 7). However, even in these regions, Purkinje cells were abnormally shaped.
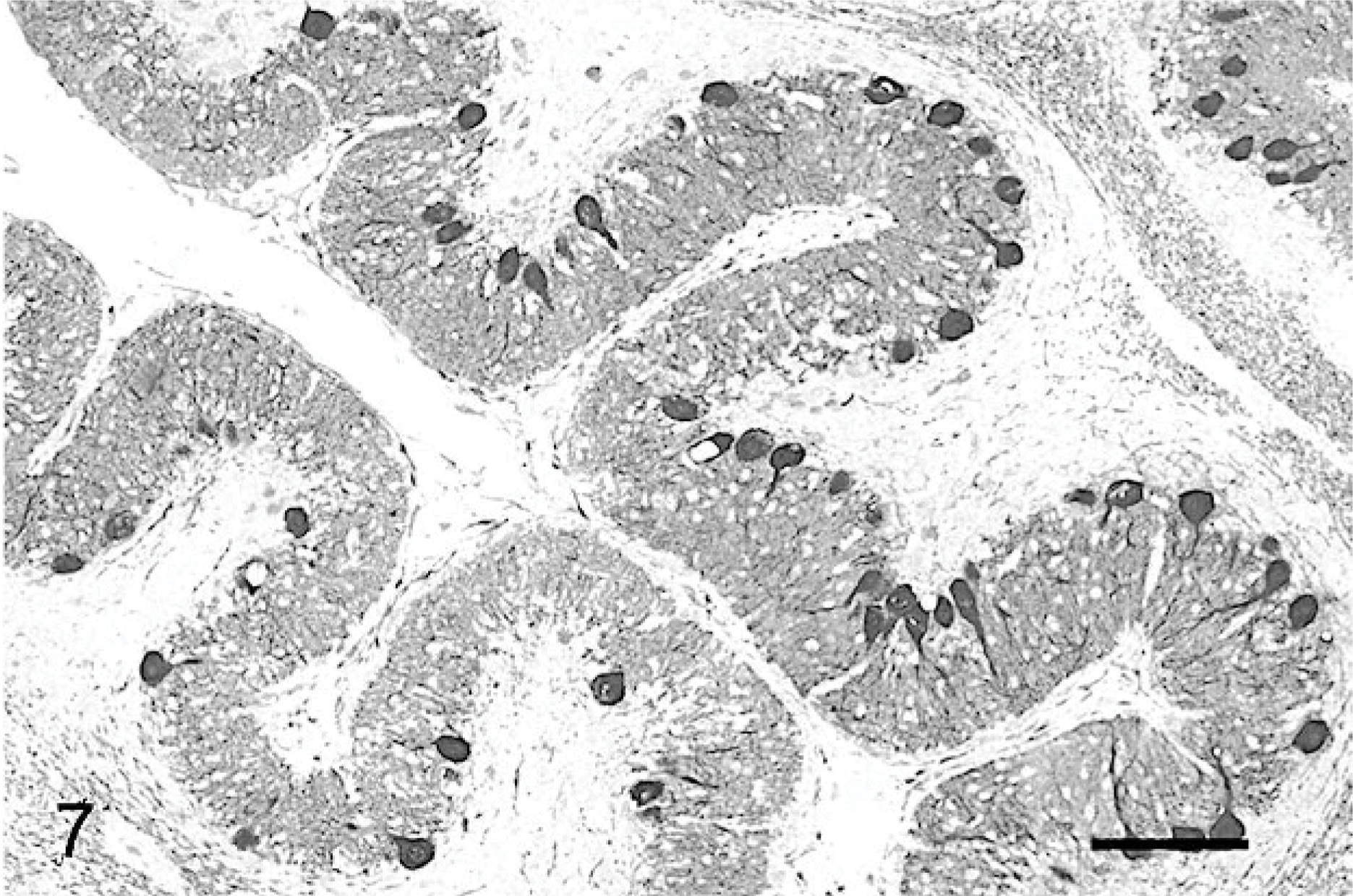
Cerebellar cortex; cat No. 1. Normal orientation of the Purkinje cells, with dendritic trees projecting into a well-differentiated molecular layer. Anticalbindin, PAP method, diaminobenzidine visualization, no counter staining. Bar = 200 μm.
Cerebellar cortex; cat No. 3. Purkinje cell heterotopia and absence of recognizable molecular layer (compare with Fig. 7 ). Anticalbindin, PAP method, diaminobenzidine visualization, no counter staining. Bar = 200 μm.
Purkinje cells that did not have normal orientation (Figs. 8, 9) were characterized by a short main dendrite, irregularly branched, with thorn-covered secondary dendrites (Fig. 8). The dendritic branching sometimes occurred in close contact with the white matter of the folial axes (Fig. 9). Purkinje cells that were physiologically oriented generally had a long, straight main dendrite and secondary dendrites that came out near the cerebellar surface, giving the dendritic tree the characteristic appearance of a weeping willow (Fig. 10). Irregular branching was also present. Purkinje cells with 2 main dendrites were also rather numerous (Figs. 10, 11). Sometimes, a single bent main dendrite ran along the cerebellar surface (Fig. 12).
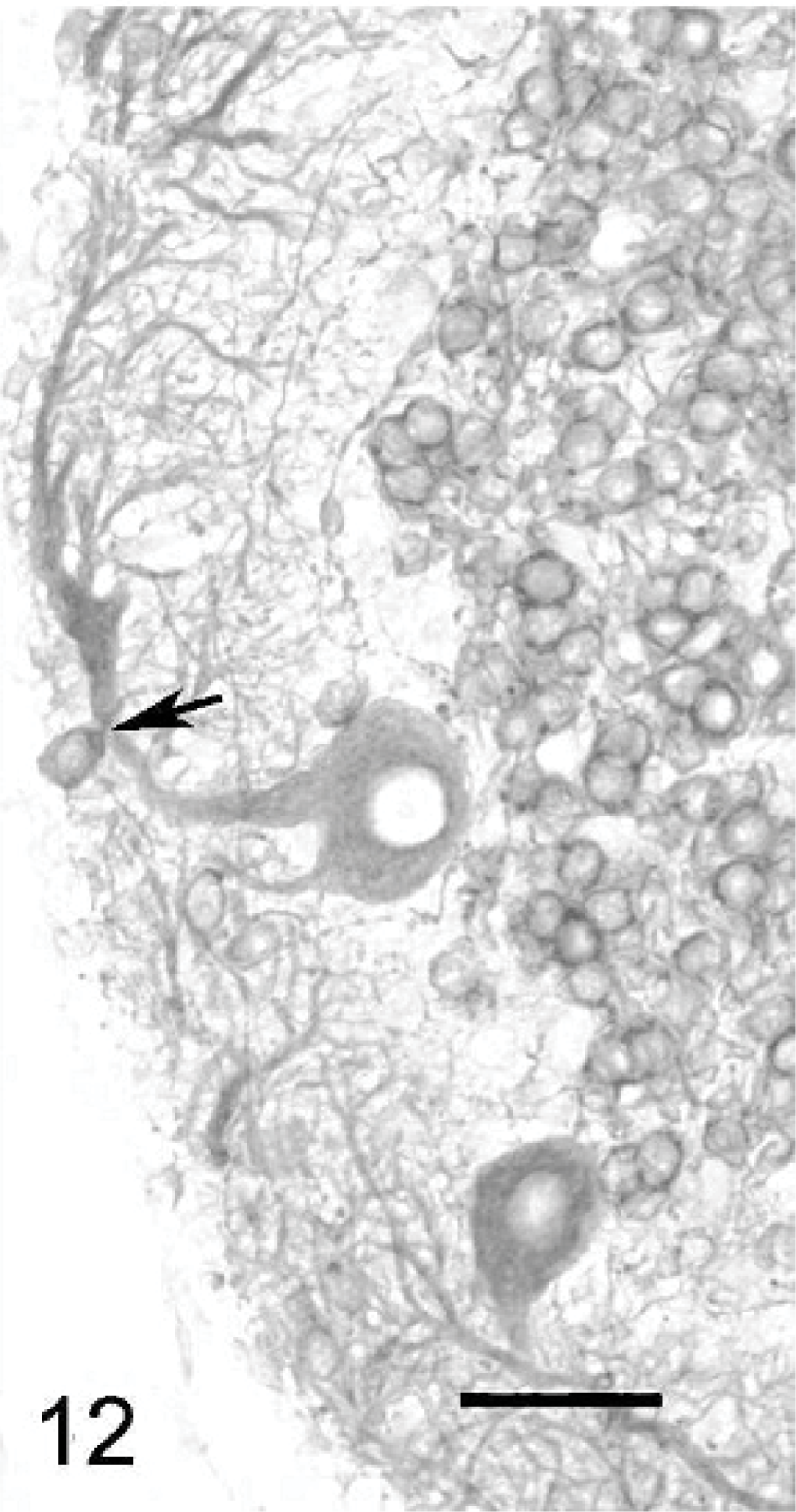
Cerebellar cortex; cat No. 1. Purkinje cell with a bent dendrite coursing close to the cerebellar surface. Arrow: displaced basket cell body. Anti-MAP2, PAP method, diaminobenzidine visualization, no counter staining. Bar = 45 μm.
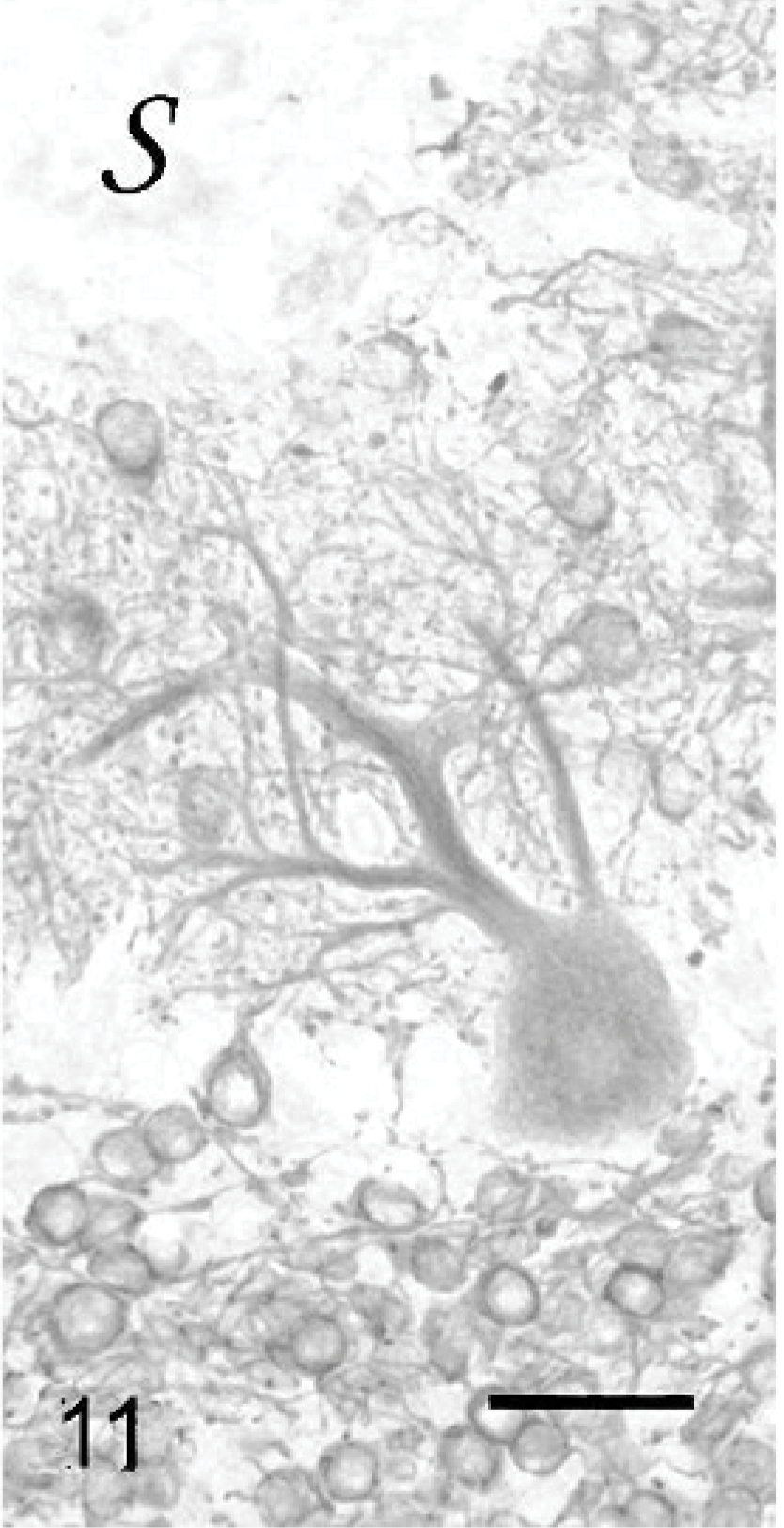
Cerebellar cortex; cat No. 1. Purkinje cell with multiple main dendrites, beside dendritic tree abnormalities. s: cerebellar surface. Anti-MAP2, PAP method, diaminobenzidine visualization, no counter staining. Bar = 50 μm.
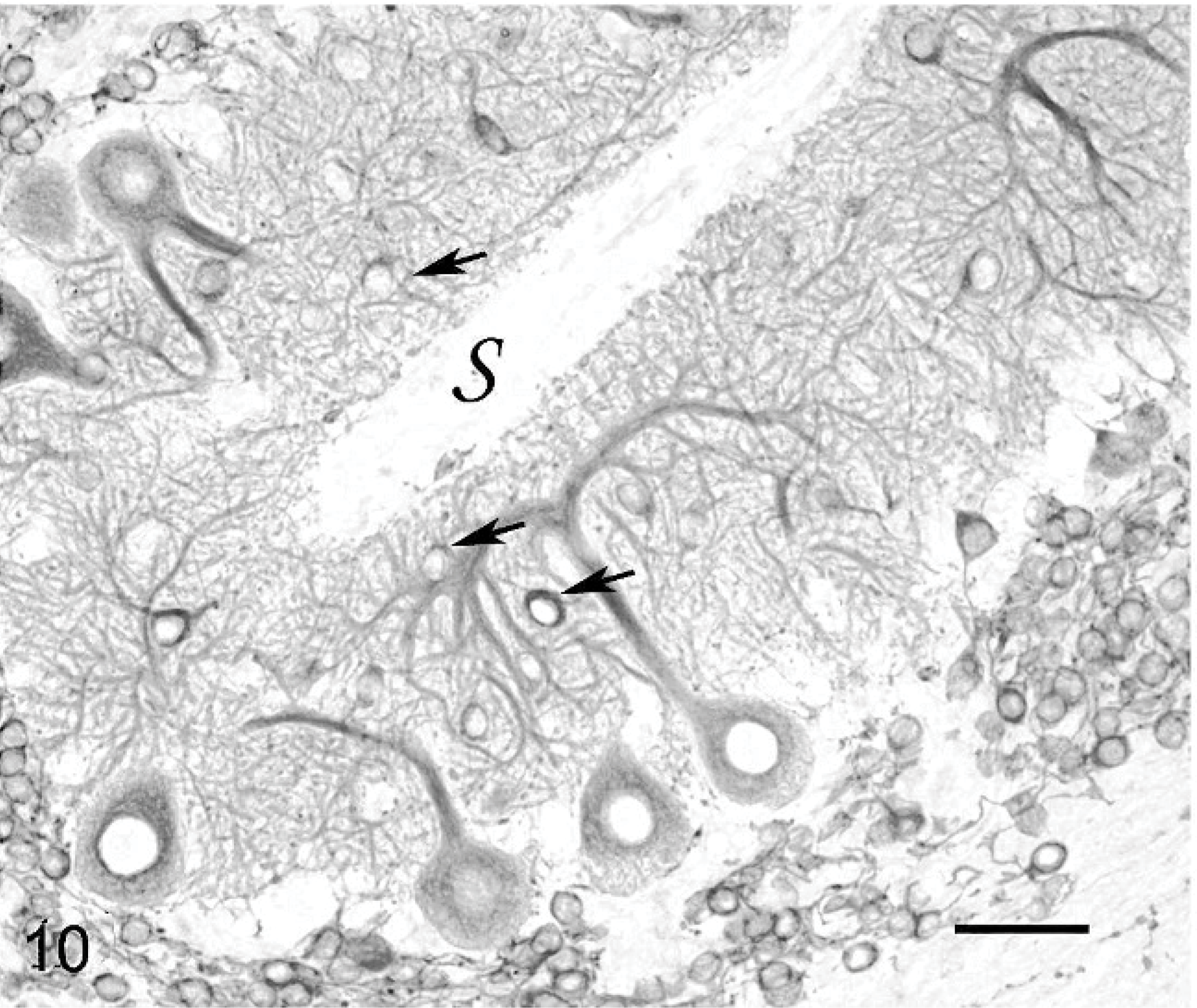
Cerebellar cortex; cat No. 1. Weeping willow Purkinje cell. Arrows show displaced basket cell bodies. s: cerebellar surface. Anti-MAP2, PAP method, diaminobenzidine visualization, no counter staining. Bar = 40 μm.
Cerebellar cortex; cat No. 1. Disoriented Purkinje cell (arrow). Absence of internal granular layer (arrow heads). s: cerebellar surface. Anti-MAP2, PAP method, diaminobenzidine visualization, no counter staining. Bar = 62 μm.
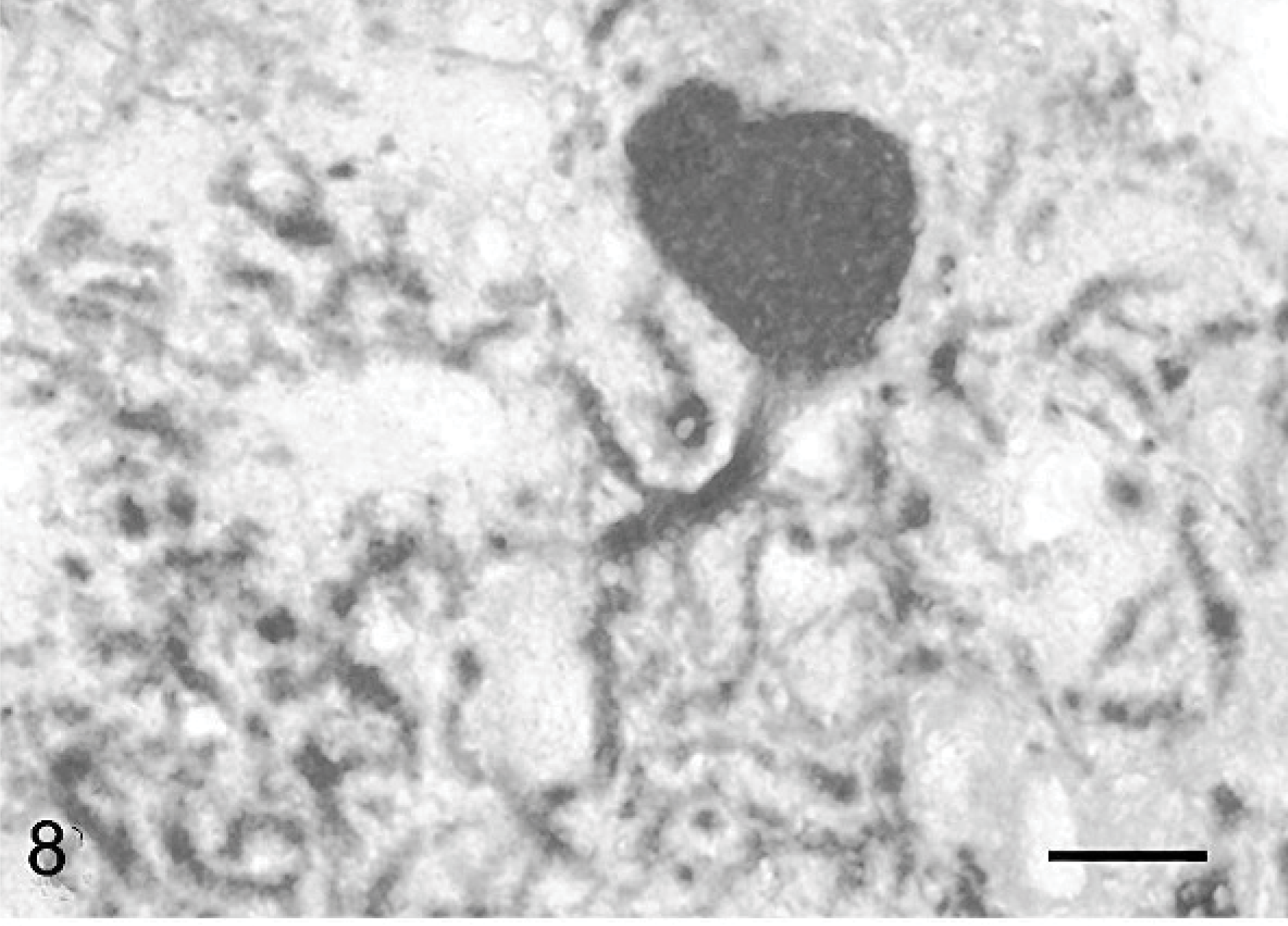
Cerebellar cortex; cat No. 3. Purkinje cell with a short main dendrite and hyperspiny dendrites oriented toward the white axis. Anticalbindin, PAP method, diaminobenzidine visualization, no counter staining. Bar = 15 μm.
Granule cells
In normal cats, granule cells form a thick layer of densely packed cells beneath the Purkinje cell layer. Granule cells, like other neurons, were demonstrated through the use of anti-MAP2 Ab. Granule cells were sometimes entirely missing, especially where Purkinje cells were randomly scattered (Fig. 9). In areas where Purkinje cells were linearly orientated, granule cells generally formed a thin layer beneath the Purkinje cell perikaryons (Figs. 10– 12). No displaced granule cells were seen in the molecular layer, even in the youngest cat. Rarely, at the tip of some folia of all cats, a few granule cells were the only neurons remaining in the cortex.
Basket and stellate cells
Basket and stellate cells are found in the molecular layer. Basket cells are seated relatively deeper in this layer and send axons that run parallel to the pial surface before ending in a rich set of terminals entwisting the Purkinje cell perikaryons. Basket cells were rarely seen. They were located in a superficial position, at the level where the main dendrites were branching, i.e., at mid level of the molecular layer (Fig. 10) or even immediately below the surface (Fig. 12). The basket cell axons appeared extremely abnormal when contrast enhanced with antiphosphorylated neurofilaments mAb (Fig. 13). In areas where the Purkinje cells exhibited a long, straight dendrite, basket cell axon divisions ran at a right angle to the cerebellar surface as a consequence of the abnormal superficial position of their cell body (Fig. 13). Where the molecular layer was extremely thin, the basket cell axon sometimes ran along the cerebellar surface. In both cases, normal baskets were not observed around the Purkinje cell bodies. They were often thin and poorly developed, entwisting the Purkinje cell main dendrite and ending around the Purkinje cell perikaryon in an impoverished plexus (Fig. 13). In the most degenerated folia, no basket cell axons were visible at all.
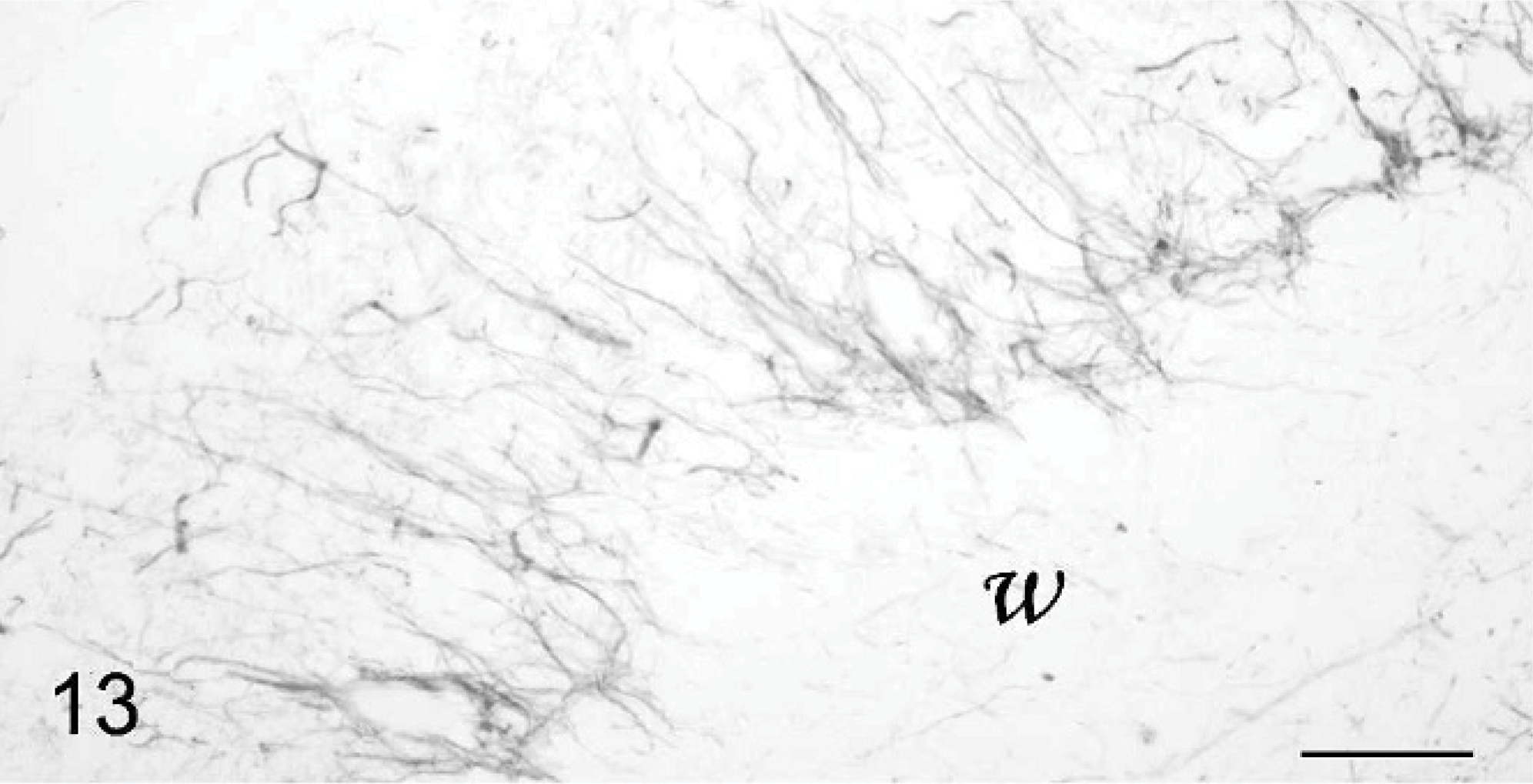
Cerebellar cortex; cat No. 2. Abnormal basket cell axons, oriented perpendicularly to the surface and ending in impoverished plexi. w: white matter. Antiphosphorylated neurofilaments, PAP method, diaminobenzidine visualization, no counter staining. Bar = 50 μm.
Unipolar brush cells
The unipolar brush cells of normal cats are labeled by anticalretinin Ab. 30 In the 3 cats, the unipolar brush cells were normally located in the nodulus and flocculus, and had a normal size and shape.
Climbing fibers
Climbing fibers also are stained by anticalretinin Ab. 30 A high number of positive fibers were seen around the Purkinje cell bodies and along their main dendrite (Figs. 14– 16). They were located in all parts of the cerebellum, strictly in association with Purkinje cells. In contrast to normal climbing fibers, 18, 31 two of them could be present along the same main dendrite (Figs. 14, 15). Climbing fibers were either slender and straight or coarse and meandering. Many thick dots, similar to varicose synapses, were observed on both types at the level of the Purkinje cell bodies (Figs. 15, 16) and main dendrites (Figs. 14, 16).

Cerebellar cortex; cat No. 3. Climbing fiber along a Purkinje cell perikaryon and along its disoriented dendritic tree. w: white matter. Anticalretinin, PAP method, diaminobenzidine visualization, no counter staining. Bar = 50 μm.
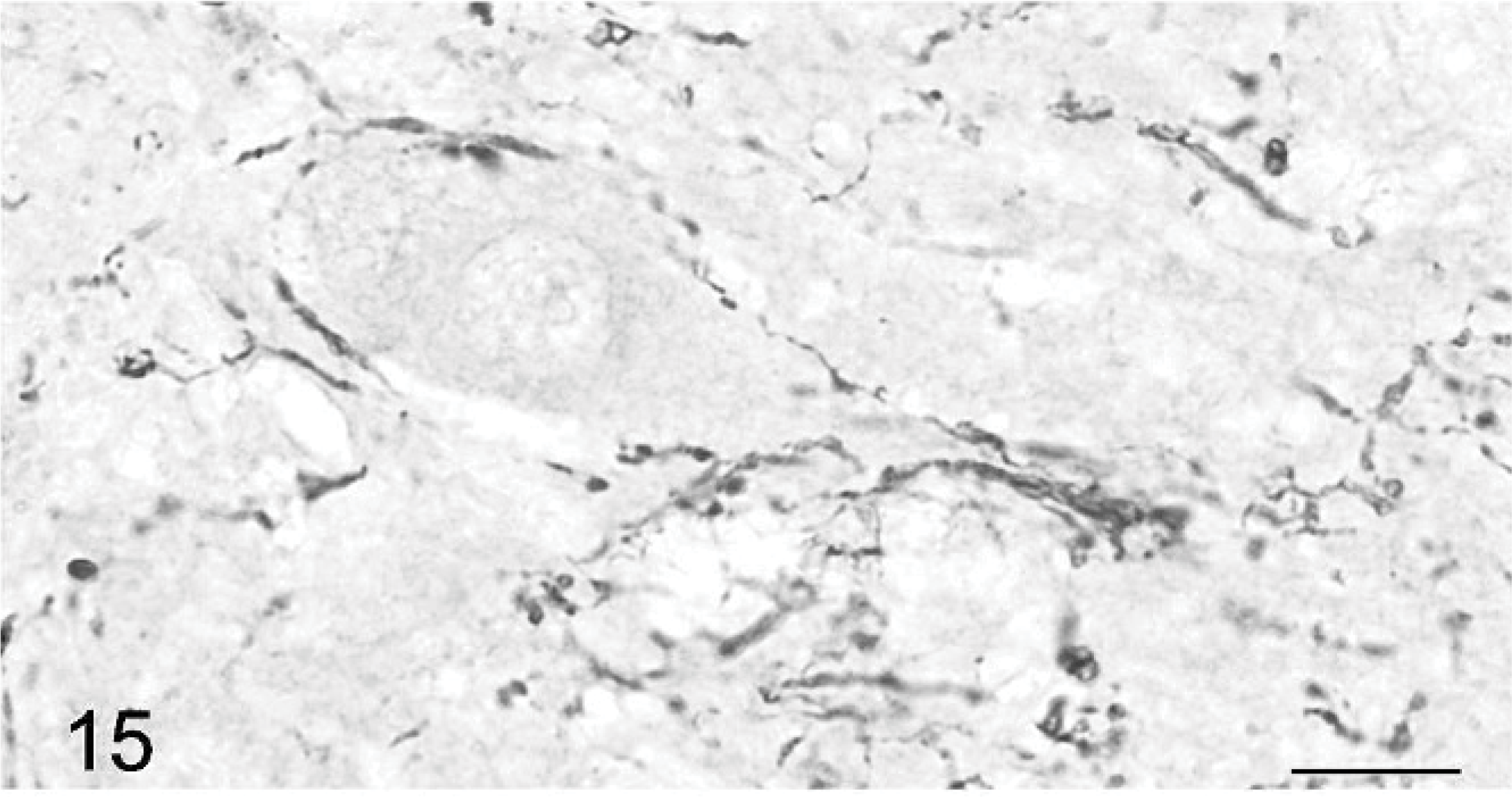
Cerebellar cortex; cat No. 1. Climbing fiber showing varicosities around a Purkinje cell perikaryon and along its dendritic tree. Anticalretinin, PAP method, diaminobenzidine visualization, no counter staining. Bar = 15 μm.
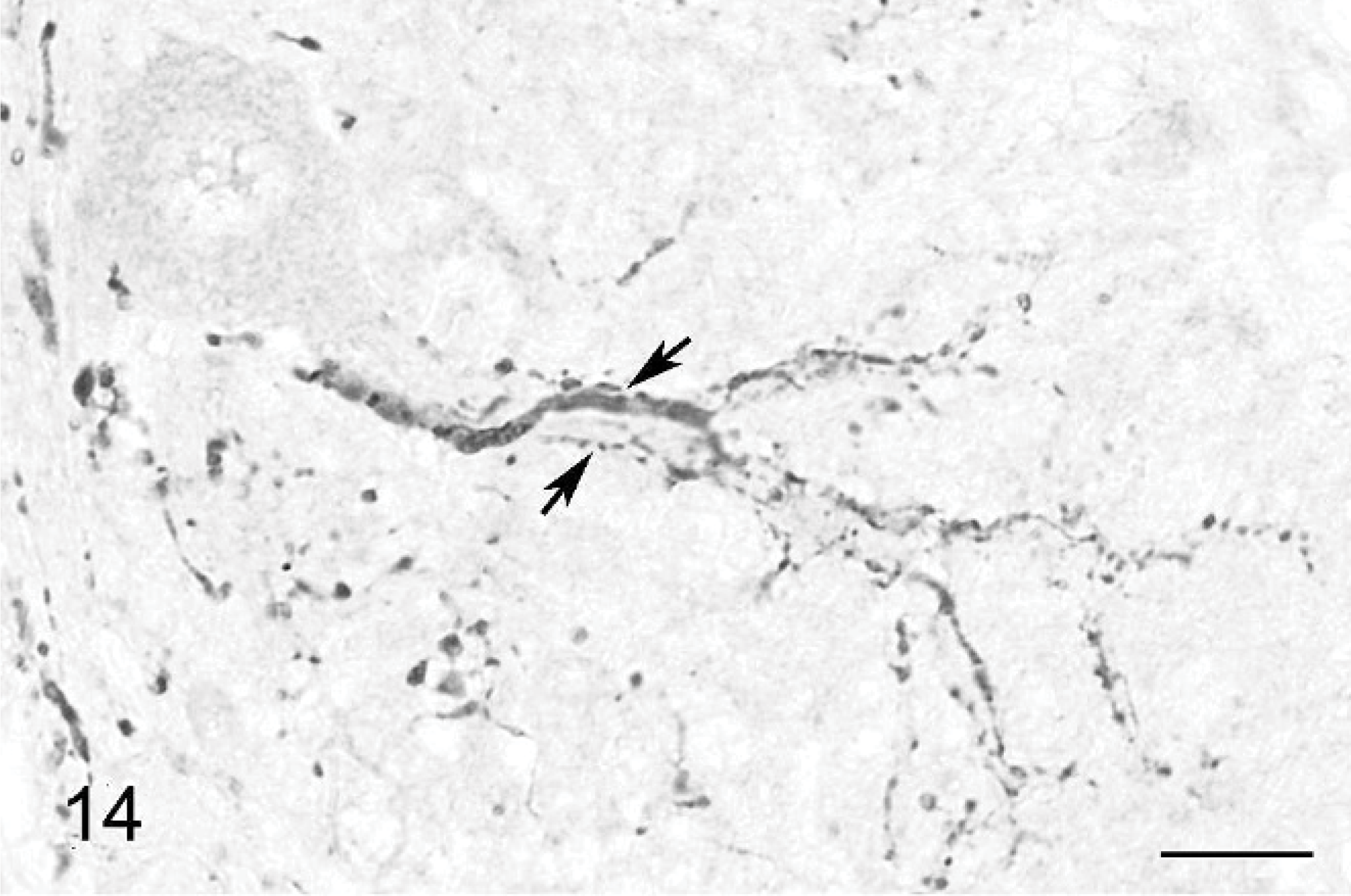
Cerebellar cortex; cat No. 1. Climbing fibers with different diameter running along a Purkinje cell dendritic tree (arrows). Anticalretinin, PAP method, diaminobenzidine visualization, no counter staining. Bar = 15 μm.
Gliosis
Gliosis was moderate in cat Nos. 1 and 2 and impressive in cat No. 3. In the latter, anti-GFAP Ab revealed numerous glial fibers and astrocytes, mostly in the white matter but also in the residual cortex (Fig. 17). Fibers of the Bergmann glia were not distinguishable whereas abnormal astrocytes were present even close to the cerebellum surface (Fig. 18). By using anti-S100 Ab, we could not visualize the layer of Bergmann glia cell bodies that is visible at the level of the Purkinje cell layer in normal cat. 49 Bergmann glial cells were actually randomly scattered, making any distinction between the granular and the molecular layer impossible when using the anti-S100 Ab. In the 3 cats, the inferior olive was normal, but the pontine nuclei were reduced to a very small number of cells and the brachium pontis was extremely thin.
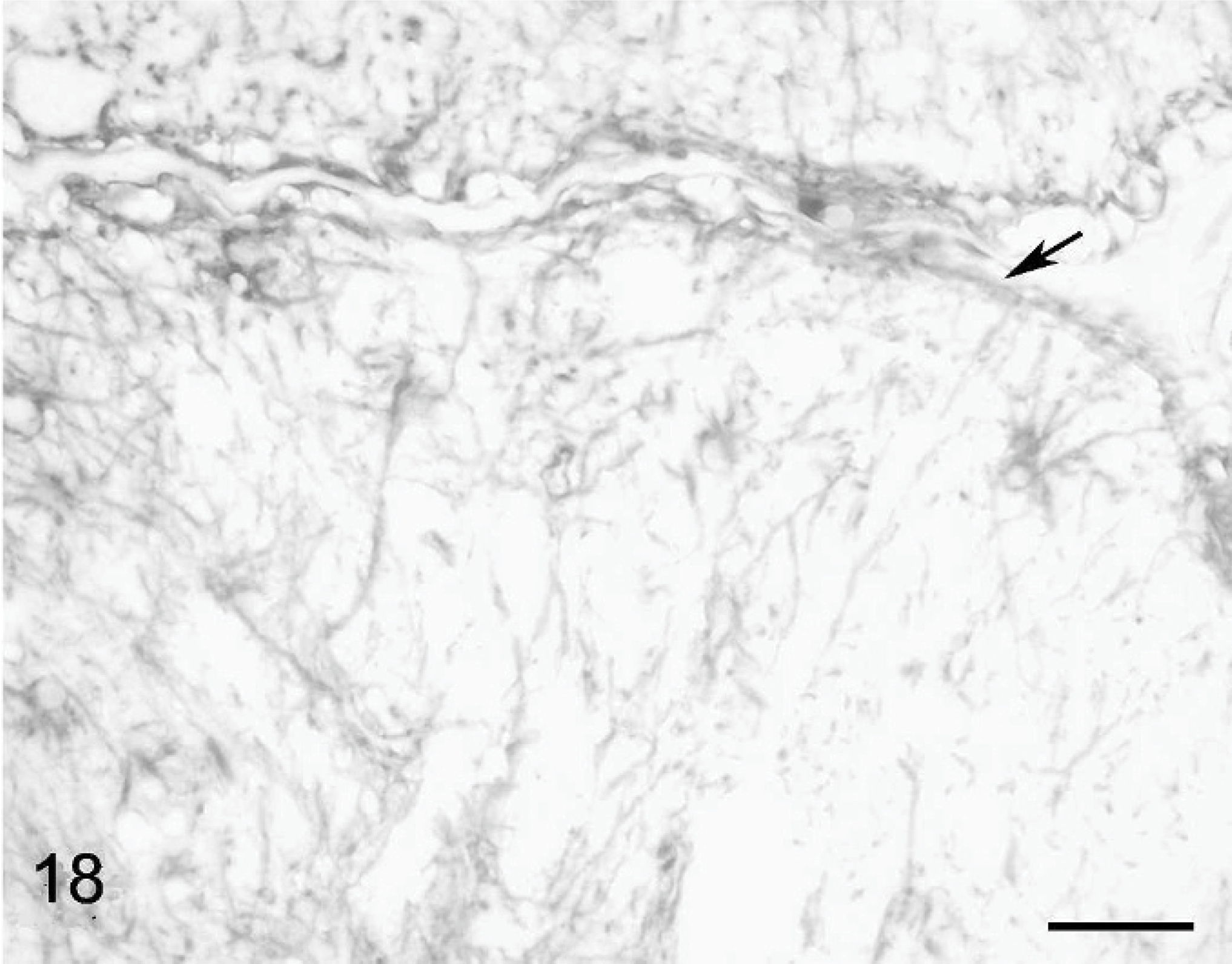
Cerebellar cortex; cat No. 3. Gliosis present up to the cerebellar surface (arrow). Anti-GFAP, PAP method, diaminobenzidine visualization, no counter staining. Bar = 25 μm.

Cerebellar cortex; cat No. 3. Intense gliosis; the Bergman glia scaffold is not recognizable. Anti-GFAP, PAP method, diaminobenzidine visualization, no counter staining. Bar = 80 μm.
In brief, 2 kinds of cerebellar cortex patterns could be discriminated. In type I, there were almost no granule cells left, the Purkinje cells were randomly distributed, with a disoriented, stunted, thorn-covered dendritic tree; the gliosis was prominent; and the folial axes were demyelinated. In type II lesions, granule cells were more numerous and Purkinje cells were linearly arranged, with an overlong but correctly oriented main dendrite, giving bent, smooth secondary dendrites, whereas gliosis and folial axes demyelination were more discreet.
PCR analysis
The 214-bp product from the histone 3.3 gene was amplified from all cerebellar samples and from positive and negative controls. Amplification of DNA extracted from the gut of the confirmed feline panleucopenia case and from the cerebella of cat Nos. 1 to 3 yielded PCR products of the expected size when using each of the 3 primers pairs for parvovirus (Fig. 19). Amplification of DNA extracted from the normal cat cerebellum and from the normal cat gut yielded no products with the use of any of the 3 parvoviral pairs of primers.
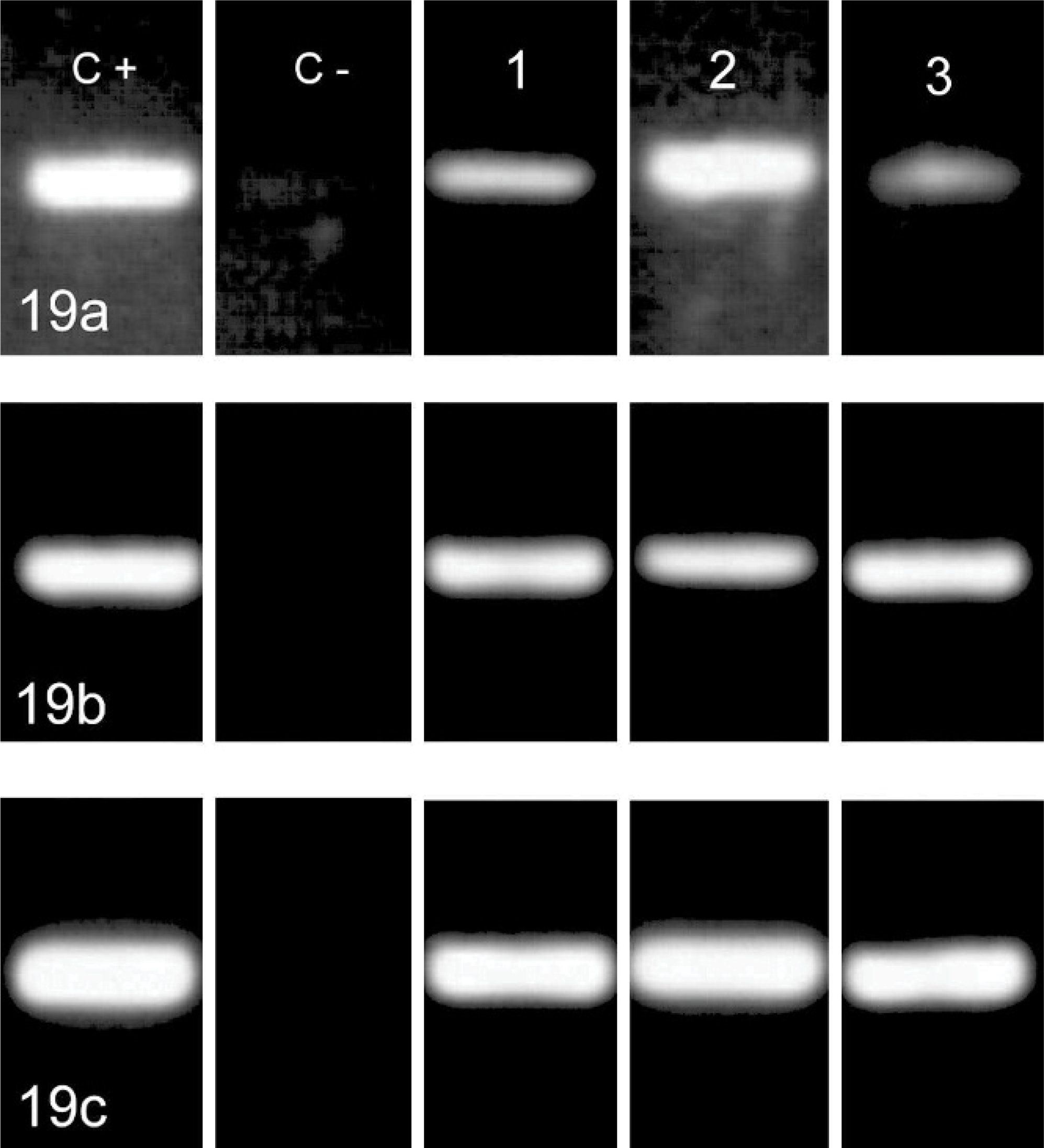
PCR amplification for parvoviral DNA by using 3 primer pairs yielding a 193-bp product in the VP1 gene (a), and 215-bp (b) and 184-bp (c) products of the VP2 gene. C+, C−: positive and negative controls (FPLV infected cat intestine and normal cat cerebellum respectively). 1, 2, 3: clinical case Nos. 1, 2, and 3.
Immunostaining for parvovirus
By using the antiparvovirus mAb, positive enterocytes were demonstrated in the parvovirus-infected cat gut, but no positivity was found in the samples of cat Nos. 1–3 or the negative controls (result not shown).
Discussion
The use of cell-specific or cell processes–specific immunostaining allowed description of the morphology of cerebellar cortex cells in 3 naturally occurring, PCR-confirmed cases of FPLV-associated cerebellar hypoplasia, with an accuracy comparable with that of silver impregnation methods. It confirmed lesions previously reported in cats, namely the reduction of granule cell numbers, Purkinje cell heterotopia, and a high number of vacuolated Purkinje cells. 23, 27, 28, 36, 37 In addition, it showed changes in Purkinje cell dendrites, basket cell axons, and climbing fibers, which were undetectable by routine staining methods. Identifying these changes allows comparison of lesions in spontaneous cases and those described when using silver impregnation methods after experimental damage to the developing cerebellum in laboratory animals.
To complete their DNA replication and induce host-cell lysis, parvoviruses need to invade cells that enter the S phase of the mitotic cycle because they rely on host-cell proteins expressed only during this phase. 14, 22, 51, 56 Granule precursor cells located in the cerebellar external granular layer are the major target for parvovirus replication during the perinatal period. 3, 6, 18, 48 Acute cell lysis with significant virus particle production takes 2 days and occurs in pregranule cells recently arisen from the external granular layer. 48 This explains the reduction of granule cell numbers observed in the present cases and in previous reports. 23, 27, 28, 37
Selective external granular layer destruction can be achieved by X-ray–irradiation, 1, 2, 4– 8 methylazoxymethanol acetate administration, 12, 61 and viral infection. 23, 37, 48 The ensuing cerebellar cortical abnormalities are unrelated to the method used; they are dependant only on the time of infliction during the cerebellar development. The pivotal development time seems to be the time of spreading of the Purkinje cell somas from a multilayered structure to a monolayer. 3, 18
An agranular cerebellum, similar to our type I lesion pattern was always observed when complete destruction of the external granular layer occurred before the formation of this monolayer. 1, 2, 4, 5, 60 Conversely, a hypogranular cortex, similar to our type II lesions, was obtained when destruction of the external granular layer occurred after the monolayer formation. 1, 2, 5, 7, 12 In the latter case, the already settled granule cells survived and the Purkinje cell lining up was preserved, although the Purkinje cell main and secondary dendrites exhibited abnormal features. 1, 2, 5, 7, 12 The development stage corresponding to the Purkinje cell monolayer formation occurs before birth in the feline species. 18, 57 This explains why X-ray irradiation or experimental FPLV infections given 1 day before delivery or from the first postnatal day induced neither near complete granule cell loss nor Purkinje cell heterotopia. 6, 23, 36, 37 Conversely, intrauterine FPLV infection 20 days before delivery induced agranular cortex similar to our type I lesions in experimental cats. 37
The abnormal features of neurons in the hypogranular cortex described in the present report are identical to those observed in experimentally induced hypogranular cortex, i.e., from postnatal day 6 to postnatal day 15 in rats 1, 2 or between the first postnatal day and the 3rd postnatal week in kittens. 7 During this period, the Purkinje cells develop a single apical dendrite oriented toward the pial surface. 9, 18, 34 In rats, as in cats, the length of the Purkinje cell primary dendrite was related to the time of irradiation, the longest dendrites being observed with the earliest exposures. 2, 5, 7
Consequently, the presence of type I lesions in the 3 cats of the present study is consistent with viral infection that occurs before birth, but the concomitant presence in the 3 cats of type II lesions, with impressive Purkinje cell main dendrite length suggests an infection that occurred close to the day of birth. The prominent proportion of type I lesion in cat No. 3 and of type II lesion in cat Nos. 1 and 2 suggests that the viral infection occurred closer to the day of birth in cats Nos. 1 and 2 than in cat No. 3.
Basket cell features observed when using antiphosphorylated neurofilament antibodies in our cats (especially in cat Nos. 1 and 2), namely displaced cell bodies, main axons oriented perpendicular to the cerebellum surface, and atrophic plexus, were not previously described in naturally occurring feline hypogranular cerebellar hypoplasia. These changes are a direct consequence of abnormal branching of the Purkinje cell main dendrite. Basket cell somas in X-irradiated and normal animals settle in the molecular layer at the level where the main dendrite of the Purkinje cell bifurcates into secondary branches. 1, 2, 4, 5, 8 If this bifurcation occurs too close to the pial surface, as in hypogranular cerebella, then the basket cell perikaryons become misaligned; their main axon sends divisions that run along the elongated Purkinje cell main dendrites, resulting in an impoverished set of terminals around the Purkinje cell perikaryons. 1, 8
Developmental alterations and defects in climbing fiber synapse translocation and elimination, evidenced through anticalretinin antibodies in the present cats, are documented in the feline species for the first time. Translocation of climbing fiber terminals from Purkinje cell perikaryon toward its dendritic tree, as well as synapse elimination that leads to the one to one relationship between inferior olive neurons and Purkinje cells are well described developmental events. 18, 20, 43 Our original observation supports a stop in maturation of climbing fibers in the 3 naturally infected cats. Alteration of these processes were observed in various rodent and ferret models of hypogranular cerebellum. 13, 16, 61
Neurons in the pontine nuclei that rely on their granule cell targets to develop were almost completely absent in all cats in this study, as also reported in agranular mutant mice. 46 In contrast, other precerebellar nuclei, like the lateral vestibular, lateral reticular, and lateral cuneate nuclei were preserved. These neurons extend their process before cerebellar cortex differentiation, with neurons in deep cerebellar nuclei as the primary target. 33 Later on, as spinocerebellar fibres invade the cerebellar cortex, Purkinje cells take the role of organizer in the agranular cerebellum of mice. 10 Consistently, no abnormality was found in the spino- and vestibulocerebellar pathways of our cats. Similarly, the survival of the inferior olivary nuclei in the 3 cats supports that abnormal Purkinje cells were a convenient target for the climbing fibers as reported in agranular mutant mice. 15
A high number of vacuolated Purkinje cells was observed in the 2 youngest cats of the present study, as in the previous report about postnatal experimental FPLV infection in kittens. 23, 27, 36, 37 Even if the Purkinje cell density appeared relatively preserved in these atrophic cerebella, the total surface of the cerebellum was reduced to the point that the number of Purkinje cells could be assumed to be far below normal. This low Purkinje cell number cannot result from granule cell death, because the number of Purkinje cells is unaffected, even if all granule cells are destroyed by X-ray or are genetically absent. 4, 54 They, however, could be related to viral transcription that occurred after the acute viral replication. Purkinje cells express the transferrin receptor that is used by canine and feline parvovirus for cell entry. 21, 32, 47, 58 Consequently, they are permissive to parvovirus invasion, although they should not allow viral replication, being postmitotic cells. Nevertheless, transcription of viral proteins occurs, as shown by the presence of viral antigen, both in the surviving granule cells and the Purkinje cells up to 3 weeks after experimental infection. 23, 27 We did not observe such positive viral immunohistology in our cats, either because of their older age or because of methodologic differences. However, persistence of viral genome segments in the cerebellum was shown through PCR, a PCR diagnosis being viewed as the most reliable technique among all the possible diagnostic tests for parvovirus. 26, 38
Moreover, Purkinje cells express low levels of E2F-1 transcription factor, 11 the main activator of the parvovirus P4 promoter. Provided its P4 promoter is activated, the nonstructural parvoviral protein NS1 is produced at a low level during the G0 and G1 phases of the cell cycle. 24, 29 NS1 is known to be highly cytotoxic and able to induce cytoskeleton alterations and cell changes. 17, 44, 45 Consequently, the current knowledge about parvovirus biology raises an hypothesis about the long-recognized Purkinje cell alteration and disappearance.
Footnotes
Acknowledgement
We thank Dr. C. Rusbridge for editing the English language of the manuscript.
