Abstract
A juvenile raccoon (Procyon lotor) was submitted dead to the Minnesota Veterinary Diagnostic Laboratory for rabies testing without history. The animal had marked hypoplasia of the cerebellum. Histology demonstrated that most folia lacked granule cells and had randomly misplaced Purkinje cells. Immunohistochemistry revealed the presence of parvoviral antigen in a few neurons and cell processes. PCR targeting feline and canine parvovirus yielded a positive signal. Sequencing analyses from a fragment of the nonstructural protein 1 (NS1) gene and a portion of the viral capsid protein 2 (VP2) gene confirmed the presence of DNA of a recent canine parvovirus variant (CPV-2a–like virus) in the cerebellum. Our study provides evidence that (canine) parvovirus may be associated with cerebellar hypoplasia and dysplasia in raccoons, similar to the disease that occurs naturally and has been reproduced experimentally by feline parvoviral infection of pregnant cats, with subsequent intrauterine or neonatal infections of the offspring.
Cerebellar hypoplasia and dysplasia are both well-characterized outcomes of intrauterine or neonatal infections of feline parvovirus in kittens and intrauterine infection by bovine viral diarrhea virus in calves. 4 Parvovirus-associated cerebellar hypoplasia has also been documented on rare occasions in dogs and chickens.16,22 Herein, we report a juvenile raccoon (Procyon lotor) infected with a recent canine parvovirus (CPV) strain (Carnivore protoparvovirus 1) in the brain and manifesting hypoplasia and dysplasia in the cerebellum.
A male juvenile raccoon was shot dead by a private citizen and submitted for rabies testing without further history. The animal was in a fair nutritional state with scant internal adipose tissue stores and weighed 650 g. The animal was estimated to be ~3 mo old based on size and weight.
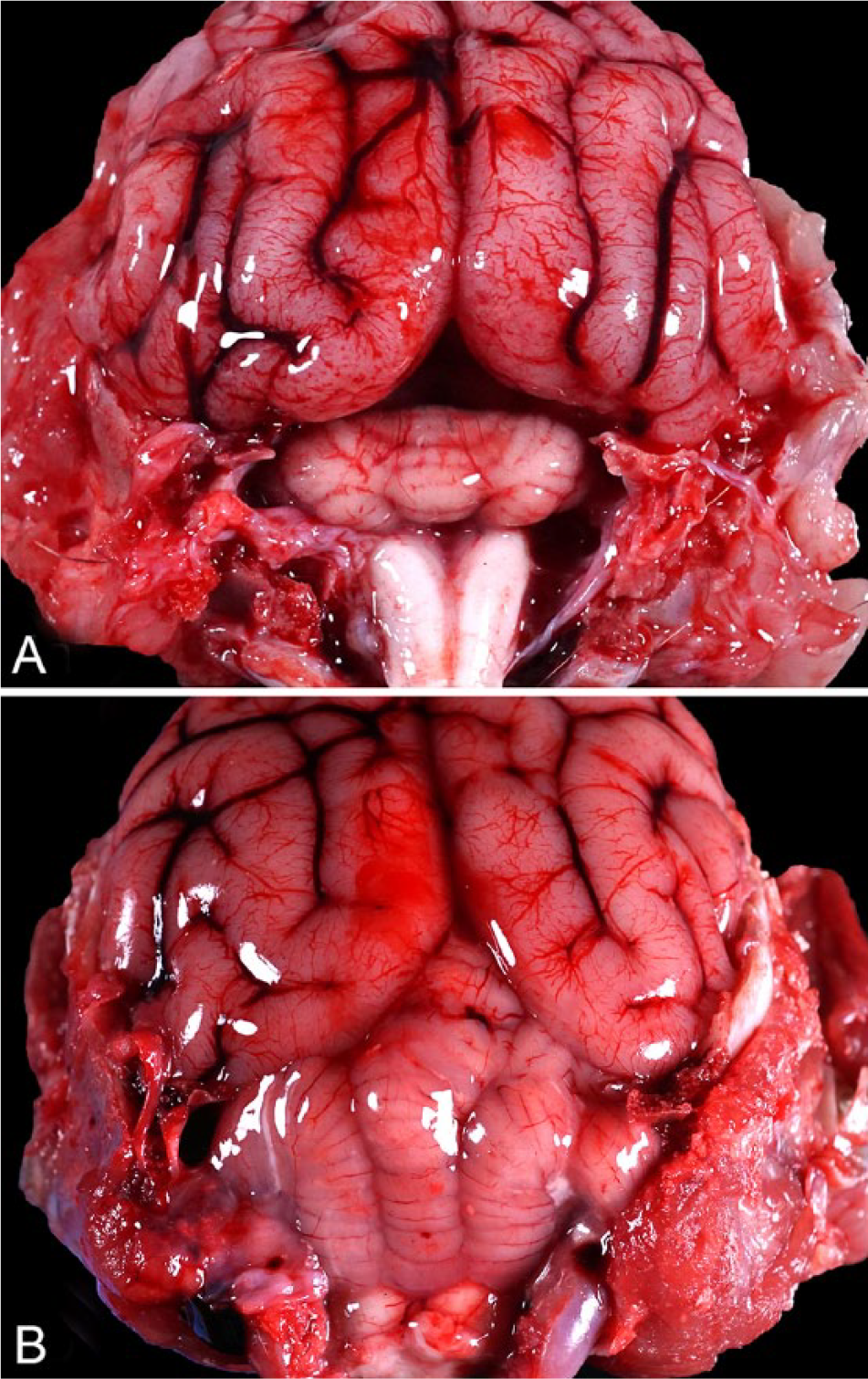
At autopsy, the cerebellum was small and had small poorly defined folia. The space between the cerebellum and the cerebral hemispheres was widened compared to a control raccoon (Fig. 1). The cerebellum was estimated to have approximately one-quarter of the size of an averaged-sized raccoon cerebellum. Gross examination of the remainder of the carcass did not reveal any other significant lesions. Given that the animal was only submitted for the purpose of testing for rabies, only brain samples were collected for further analysis.
Histologically, the brain lesions were limited to the cerebellum (Fig. 2; Supplementary Fig. 1). Gray and white matter layers were thin. Although all folia were affected, subtle differences were present between the ventral and dorsal folia. Granule cells were lacking, except for a few small aggregates in the most ventral folium. Purkinje cells were randomly scattered in the gray matter. In the most dorsal folia, occasional Purkinje cells were swollen because of vacuolation of the perikaryon. Occasional homogeneous eosinophilic ovoid structures, interpreted as necrotic Purkinje cells, were present within the disorganized gray matter of the dorsal folia. Axons in the white matter were poorly myelinated.
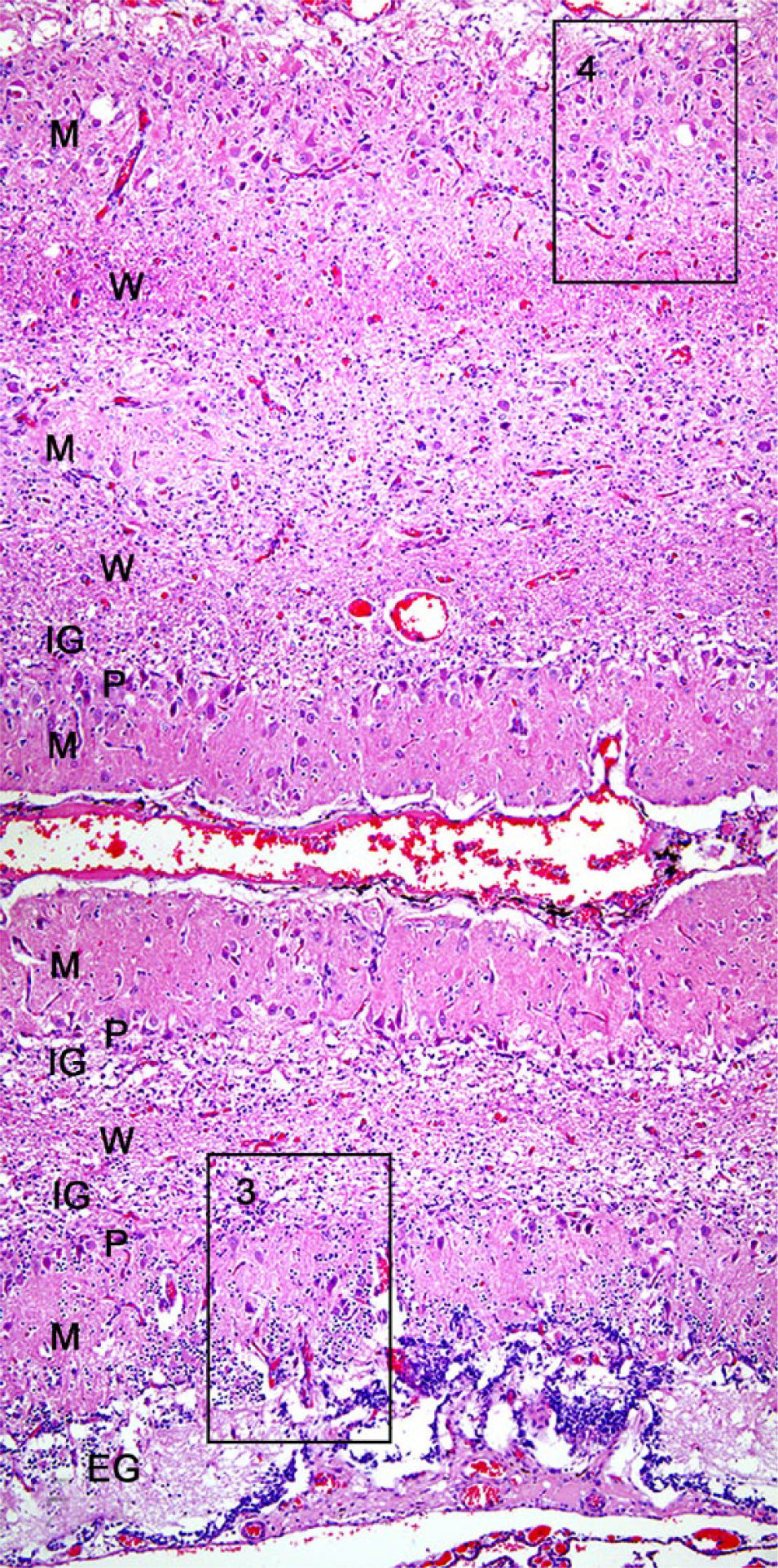
Cerebellar hypoplasia and dysplasia in a juvenile raccoon. Coronal section of the frontal lobe of the cerebellum. The most ventral folium and the 2 overlying folia are depicted. The architecture of the folia is severely altered. The ventral aspect of the ventral folium has a thin external granular (EG) cell layer and a pauci-cellular internal granular (IG) cell layer. Purkinje (P) cells are roughly aligned along the interface of the molecular (M) and IG cell layer, but ectopic P cells are also present within the M layer and are arranged haphazardly throughout the entire width of the gray matter. The overlying folia are characterized by a distinct lack of external and internal granule cells. The white matter (W) layers are thin. H&E. Boxes 3 and 4 are magnified in Figure 3A/B and 3C/D, respectively.
Immunohistochemistry (IHC) using markers specific for calcium-binding protein (calbindin), glutamic acid decarboxylase (GAD), glial fibrillary acidic protein (GFAP), and CPV antigen was performed (Supplementary Table 1). Calbindin immunoreactivity is specific for Purkinje cells in the cerebellum. 11 Purkinje cells, like other γ-aminobutyric acid (GABA)-ergic neurons, are also immunoreactive for GAD. 18 These characteristics of Purkinje cells are preserved evolutionarily across various species. GFAP is a marker for astrocytes that is commonly used across a wide range of species. 9 An automated slide stainer (Dako, Carpinteria, CA) or manual benchtop staining were utilized with a peroxidase-labeled polymer conjugate system (Dako) as a secondary antibody (EnVision+ goat anti-mouse: GFAP and calbindin; EnVision+ goat anti-rabbit: GAD) using methods described previously (Supplementary Table 1). 24 The thin layer of gray matter consisted of a complex meshwork of processes of calbindin-positive Purkinje cells, GAD-positive GABA-ergic cells (Purkinje, basket, and stellate cells), and GFAP-positive astrocytes (Fig. 3A–D). Regularly spaced, radiating GFAP-positive processes of Bergmann astroglia were lacking in the molecular layer (Fig. 3B, 3D). Within the ventral folium, some Purkinje cells lined up along a rudimentary granular cell layer–molecular layer interface; other Purkinje cells were scattered randomly. No orderly arrangement of the Purkinje cells was recognizable in the gray matter of the dorsal folia. A few cerebellar neurons and cell processes and a large neuron in the brainstem were parvoviral antigen positive (Fig. 3E, 3F).
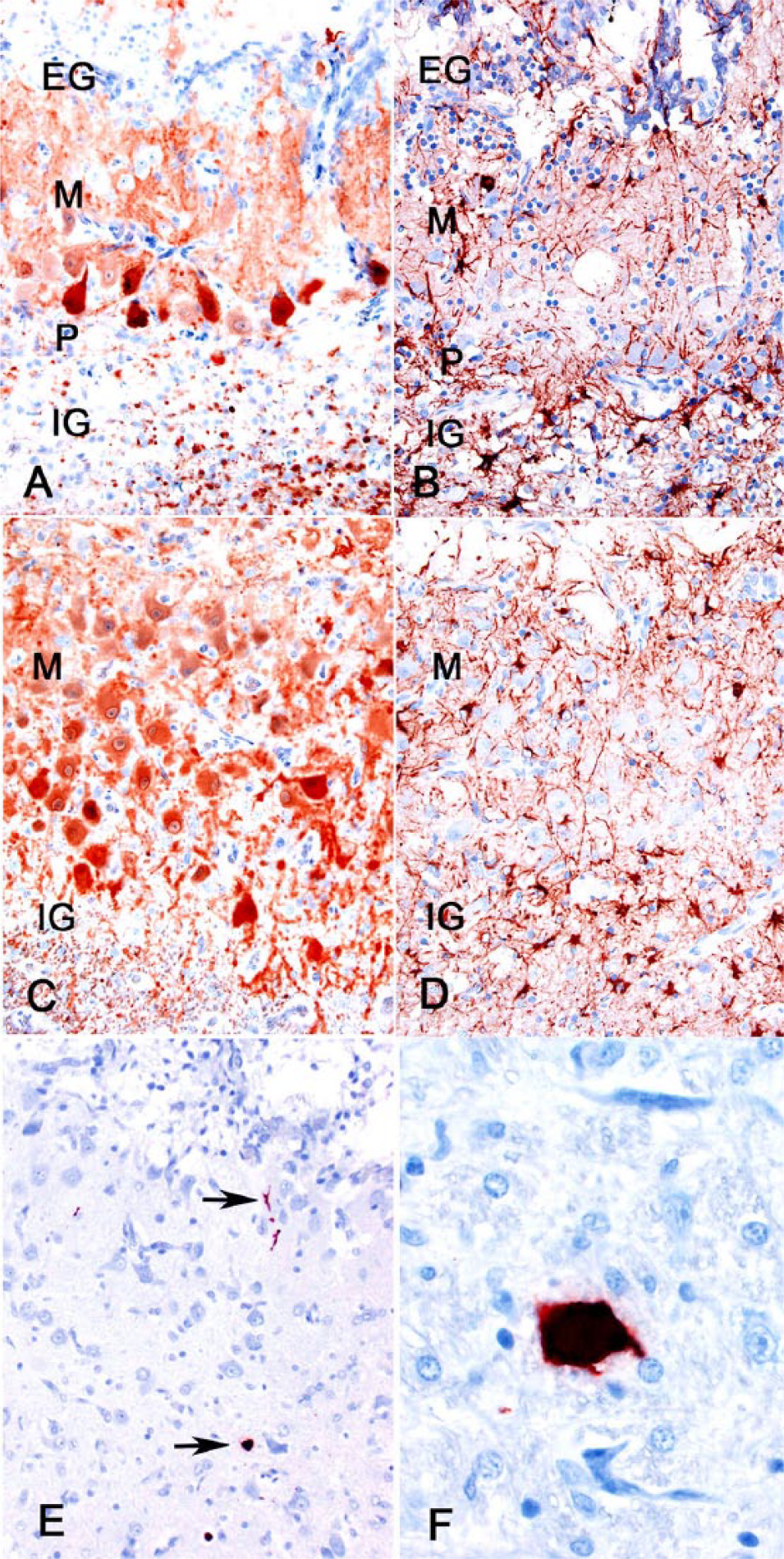
Cerebellar hypoplasia and dysplasia in a juvenile raccoon.
To examine further for the presence of parvovirus, DNA was extracted from unfixed cerebellar tissue (E.Z.N.A. tissue DNA kit, catalog D3396-01; Omega Bio-tek, Norcross, GA), as per the manufacturer’s instructions, and quantified (> 10 ng/µL; NanoDrop 2000/2000c spectrophotometer; Thermo Scientific, Waltham, MA). The isolated DNA was used as template for PCR reactions (Q5 high fidelity DNA polymerase; New England BioLabs, Ipswich, MA), under the conditions recommended by the manufacturer (30 cycles), and the following set of primers, specific for NS1 and VP2 parvoviral DNA: 5′-ATAAAAGACAAACCATAGACCGTTACTGAC-3′ and 5′-ACTCGCTTGCACGTCTTT-3′ (for NS1 gene, ~2,000 base amplicon), and 5′-CAGCCTGCTGTCAGAAATGA-3′ and 5′-CTTAACATATTCTAAGGGCAAACCAACCAA-3′ (for VP2 gene, ~2,000 base amplicon).
The PCR amplifications showed positive results for both viral genes, confirming the presence of parvoviral DNA, whereas controls showed no DNA amplification. Amplification products were purified with 0.45 × volume AMPure XP beads (Beckman Coulter, Brea, CA), and the nucleotide sequences, determined using Sanger sequencing, included nucleotides 350–1,190 (NS1 gene) and 2,995–3,715 (VP2 gene). Sequences determined were aligned to a CPV reference sequence (CPV type 2; GenBank M38425.1) and to references of feline parvovirus (FPV; M38246.1) and CPV type 2a (GenBank MN451663.1) using Geneious Prime v.2019.0.4. 14 The analysis showed 4 polymorphic sites in the NS1 gene that were synonymous, and where the sequences were more closely matched with CPV than FPV. The VP2 capsid gene sequence had the characteristics of a recent CPV (CPV-2a–derived). 23 Sequences of VP2 capsid protein showed substitutions of amino acids at residues 80, 87, 93, 101, and 103 (Supplementary Table 2). These substitutions distinguished the virus from both FPV and the original CPV-2 viral strain. The sequence of the parvovirus from this raccoon is consistent with sequences found in contemporary dogs and in raccoons in a previous study of natural host samples. 1 An additional substitution of unknown significance was seen of VP2 capsid gene at residue 232.
Enteric infections with carnivore protoparvoviruses are common in raccoons.2,3,13,17 Both feline and canine parvovirus are readily capable of infecting raccoons naturally.2,5 Onward transmission of introduced CPVs occurs among raccoons, and raccoon-adapted variants of the virus have been detected.1,2 To our knowledge, cerebellar hypoplasia and dysplasia have not been described in raccoons. 10 The brain findings warrant a diagnosis of cerebellar hypoplasia because of markedly reduced cerebellar size, and cerebellar dysplasia because of the markedly disturbed microarchitecture of the gray matter in the histologic sections and as substantiated by the immunohistochemical calbindin-, GAD-, and GFAP-specific findings. IHC, PCR, and sequencing provide evidence that this lesion may be associated with CPV. Cerebellar hypoplasia and dysplasia are well-known sequelae of intrauterine or neonatal infection with feline parvovirus in cats, but are relatively rare because most kittens are protected by maternal immunity, which prevents infection of kittens during gestation or shortly after birth.6,7,12 Cerebellar hypoplasia is very rare in dogs, and a link between this lesion and intrauterine or neonatal parvoviral infection is less well established, although parvoviral DNA has been detected in formalin-fixed, paraffin-embedded cerebellum samples of 2 Bluetick Coonhound littermates. 22
Intrauterine infection of the feline fetus with feline parvovirus and of the hamster fetus with a rat parvovirus results in the death of the mitotically active granule cell precursors of the external germinal cell layer and of Purkinje cells of the cerebellum.8,15,19 Orderly migration of cells of these precursor cells to the granular cell layer in the developing brain is essential for appropriate stimuli for other cells to locate to the proper locations in the brain. 19 The infection and resultant death of the cells of the external germinal layer precludes this stratification process. 18 In kittens with naturally occurring cerebellar hypoplasia and dysplasia, viral antigen is usually absent in the brains, but viral antigen was detectable in granule cells and Purkinje cells in kittens with cerebellar hypoplasia up to 3 wk after experimental inoculation of neonatal kittens with feline parvovirus.7,8,15,20,21
Supplemental Material
Supplemental_material – Supplemental material for Cerebellar hypoplasia and dysplasia in a juvenile raccoon with parvoviral infection
Supplemental material, Supplemental_material for Cerebellar hypoplasia and dysplasia in a juvenile raccoon with parvoviral infection by Arno Wünschmann, Robert Lopez-Astacio, Anibal G. Armien and Colin R. Parrish in Journal of Veterinary Diagnostic Investigation
Footnotes
Acknowledgements
We thank Olivia Graham for identifying the case during the rabies sample collection.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
