Abstract
Clostridium difficile is an enteric pathogen affecting a variety of mammals, but it has only recently been diagnosed as a cause of neonatal typhlocolitis in pigs. The most important virulence factors of C. difficile are 2 large exotoxins, toxin A (TcdA) and toxin B (TcdB). TcdA is a potent enterotoxin with effects on host tissues that are dependent upon receptor-mediated endocytosis of the intact toxin. TcdB is an effective cytotoxin, but it apparently does not bind receptors on intact mucosal epithelium. TcdB is much less toxic in vivo unless there is underlying damage to the mucosa, and it is not essential for the virulence of C. difficile. One hypothesis to explain the resistance of most species as neonates (e.g., humans and hamsters) is that they may lack significant numbers of TcdA receptors. The susceptibility of neonatal pigs suggests cells of the gastrointestinal mucosa express sufficient numbers of toxin receptors for lesion development. Immunohistochemical (IHC) assays documented specific binding of TcdA, but not TcdB, to the epithelium of the small and large intestine. The carbohydrate Galα1–3/β1–4GlcNAc-R has been described as an important receptor for TcdA. However, IHC indicated a distribution on cell surfaces much different from that of TcdA binding, suggesting a specific interaction of toxin with an alternative receptor.
Clostridium difficile–associated disease (CDAD) is a major health problem among human hospital patients and a growing concern for the husbandry of many other mammals. 13, 25 The etiologic agent is a Gram-positive bacillus that is an obligate anaerobe and forms environmentally hardy spores. Recently it has been identified as an etiologic agent of diarrhea in neonatal pigs and represents a significant concern to the pork industry. 24, 31
CDAD is associated with the elaboration of 2 large exotoxins, toxin A (TcdA) and toxin B (TcdB). The toxins have similar structures with 3 distinct domains. 29 The carboxy-terminus includes a series of tandem repeats and, in the case of TcdA, is associated with receptor binding. This portion of the TcdB molecule is very different from that of TcdA, and no receptor molecules have been identified for TcdB. The amino-terminus, however, is very well conserved between TcdA and TcdB; it is the enzymatic portion and is responsible for mono-glucosylation of a specific class of host-cell signal-transduction molecules, the Rho-subtype GTPases. 4, 14 The mid portion of the 2 toxins includes hydrophobic regions hypothesized to be associated with membrane insertion and/or translocation. 15, 29
Receptor-mediated endocytosis is an essential feature of toxin activity. A number of receptors for TcdA have been proposed or positively identified, including Galα1-3β1-4GlcNAc-R (α-galactosyl or the α-Gal epitope), Galβ1-4{Fuα1-3}GlcNAc (Lewis X), Galβ1-4GlcNAcβ1 (Lewis Y), Galβ1-14GlcNAcβ1-3Galβ1-4(Glc) (Lewis I), and sucrose isomaltase. 16, 17, 28 Mutations in the receptor-binding portion of the molecule, or the presence of antibodies specific for the receptor-binding portion, are sufficient to protect susceptible cells. 2, 7, 29 Lysosomal acidification is essential for conformational changes in both toxins that preclude enzymatic activity and/or translocation. 6, 12, 22 Following endosomal acidification of TcdB, a functional subunit of the toxin is present in the cytosol of host cells. 19
Much of the attention with regard to TcdA pathogenesis has been devoted to α-galactosyl, but the other receptors are fully functional and are important to toxin activity. This is clearly illustrated by the conspicuous absence of α-galactosyl from tissues of humans and all Old World primates, even though these species are fully susceptible to CDAD. 1, 13, 27 Pigs, however, have a greater abundance of α-galactosyl than do most other species. 8, 26 This carbohydrate is one of the most troublesome antigens leading to graft rejection in xenotransplantation of porcine tissues into humans and Old World primates. 8 Because pigs are known to have a great deal of this antigen on cell surfaces, we hypothesized that this might be the most significant receptor of TcdA in porcine tissues. If α-galactosyl is expressed in significant concentration at an early age, it might also explain why pigs are susceptible as neonates, unlike most other species. We present here evidence that TcdA binds specifically and abundantly to the brush border of the intestinal mucosa of neonatal pigs. However, this binding does not correlate with the distribution of α-galactosyl.
Materials and Methods
Animals
Five, clinically normal, 12- to 24-hour-old pigs were selected on the basis of the absence of TcdA and TcdB, as determined by a commercial ELISA (Techlab, Blacksburg, VA, USA) and the absence of C. difficile, as determined by bacteriologic culture. All pigs were immediately euthanized by CO2 asphyxiation and served as a source of normal tissues. Five, clinically normal, adult, female, Syrian hamsters were euthanized by CO2 asphyxiation. The absence of C. difficile infection was confirmed as before. Sixteen hamster pups, approximately 12 hours old, were also euthanized by CO2 asphyxiation.
Tissue processing
All animals were dissected immediately after euthanasia, and samples of stomach, duodenum, jejunum, cecum, ascending colon, and descending colon were fixed in 10% neutral buffered formalin. Samples of liver were also collected from the pigs and the adult hamsters. A midline incision was made on neonatal hamsters, and they were fixed whole in formalin. Within 48 hours of placement in formalin, tissue samples were dehydrated by passage through serial baths of graded alcohol and xylene. The dehydrated samples were embedded in paraffin, and 5-μm-thick sections were stained by hematoxylin and eosin (HE). Unstained serial sections were retained for immunohistochemistry (IHC).
Bacterial culture
Fecal samples from all animal subjects were examined by bacteriologic culture on taurocholate cycloserine cefoxitin fructose agar and C. difficile selective agar (brain heart infusion, C. difficile antimicrobic supplement CC, sodium taurocholate, and bovine blood) as previously described. 9, 32 After 48–72 hours, suspect colonies were picked and transferred to brain heart infusion agar with yeast extract (0.5%), cysteine hydrochloride (0.05%) (BHI-CYE), and bovine blood (2%). Identification of C. difficile was based on colony morphology, odor, weak alpha hemolysis, and production of L-proline aminopeptidase (REMEL, Lenexa, KS, USA). For toxin production, C. difficile was cultivated for 5–7 days in dialysis tubing (molecular-weight cutoff, 15,000 d) suspended in a solution of brain heart infusion (3.75%), yeast extract (0.5%), and BHI-CYE broth (0.01%).
Bacterial strains
TcdA and TcdB were isolated from C. difficile strains VPI 10463 (ATCC, Manassas, VA, USA) and JGS890, from a field case of CDAD in a neonatal pig. Strain JGS692, a nontoxigenic isolate from a pig, was the source of non-TcdA and non-TcdB proteins used in negative controls for IHC.
Cell culture
Activity of C. difficile toxins was assessed as toxicity for Chinese hamster ovary (CHO) cells. CHO cells were maintained in Iscove's Modified Dulbecco's Medium (IMDM, Cambrex, Walkersville, MD, USA) with fetal bovine serum (10%) and 100-μg gentamicin per milliliter. For toxicity assays, CHO cells were trypsinized, washed, and suspended at a concentration of 2 × 105 cells per milliliter; 100 μl of suspension was dispensed into each well of a 96-well tissue-culture plate. Cells were incubated for 4 hours at 37°C in 5% CO2, and serial dilutions of toxin preparations, in 100-μl aliquots, were added to each well. Cells were incubated, as before, for an additional 18 hours. The endpoint was determined as the dilution that caused 50% cytopathic effect.
Toxin purification and biotinylation
Supernatant from dialysis bag cultures of C. difficile was harvested by centrifugation at 15,000 × g for 20 minutes in a Sorvall RC-5B centrifuge (Kendro Laboratory Products, Asheville, NC, USA). Toxins were purified from the supernatant by ion-affinity chromatography on a DEAE-sepharose column (Amersham Biosciences, Upsalla, Sweden) on the basis of established techniques. 21, 30 TcdA was eluted with a linear gradient of 0.05- to 0.25-M sodium chloride (NaCl) in 50-mM tris(hydroxymethyl)aminomethane-hydrochloride (Tris-HCl) at a pH of 7.4. After elution of TcdA, TcdB remained on the column and was recovered with a linear gradient of 0.25- to 0.6-M NaCl in 50-mM Tris-HCl at a pH of 7.4. Toxin activity was determined by CHO-cell assay, and purity was assessed by gel electrophoresis (Fig. 1). To assess potential binding activity of nontarget proteins that may co-elute with TcdA or TcdB, we eluted dialysis-bag supernatant from cultures of nontoxigenic strains of C. difficile from the ion-affinity column under the same conditions as those used to elute TcdA and TcdB. CHO-cell assays demonstrated no toxicity of these preparations. These proteins were subsequently biotinylated and used as an additional negative control in IHC procedures.
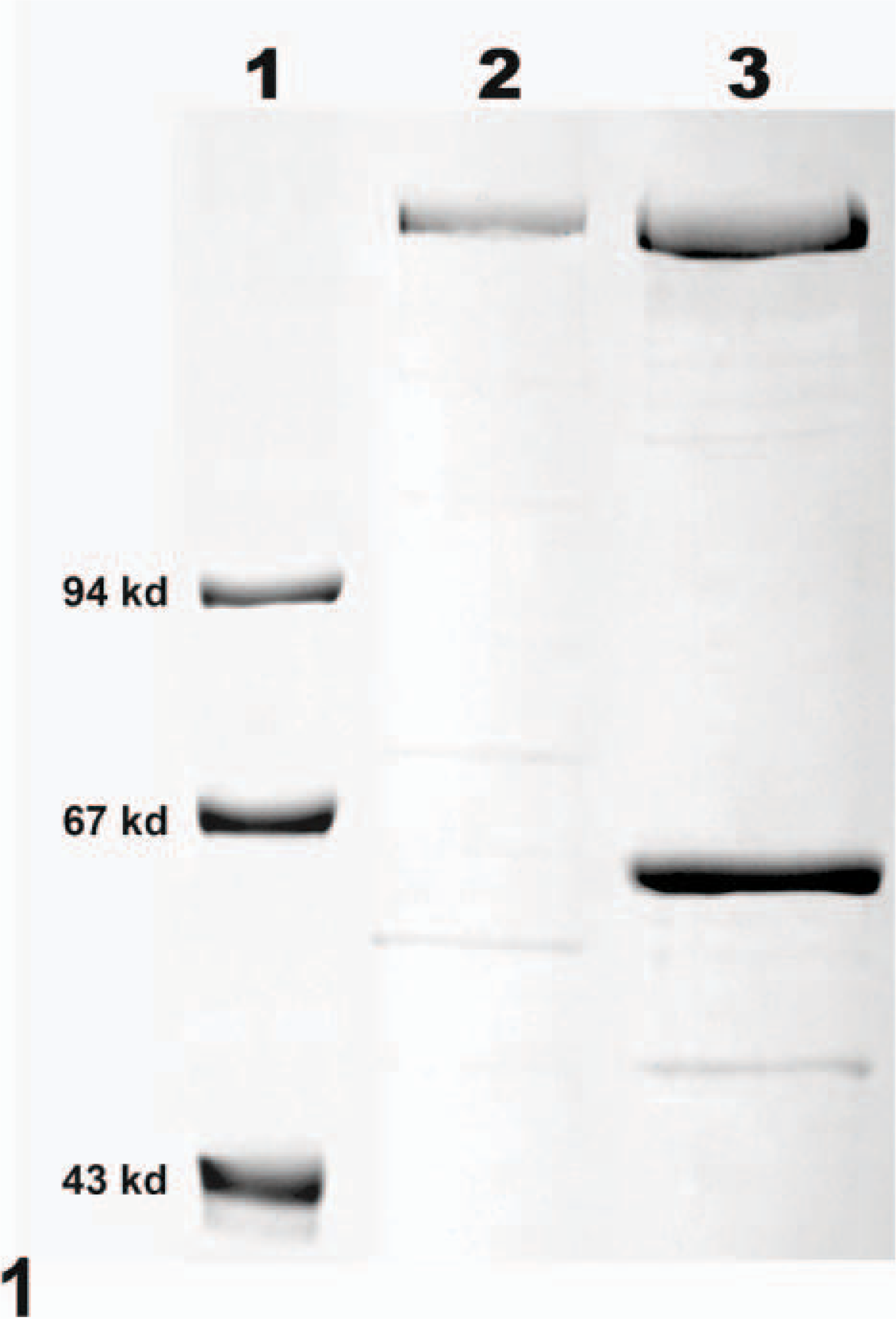
Toxin A (TcdA) and toxin B (TcdB) purified by ion-affinity chromatography. Lane 1 includes molecular-weight markers. Lane 2 demonstrates the band representing TcdA. The upper band in lane 3 represents TcdB. The lower band in lane 3 is an unidentified protein that co-eluted with TcdB.
Biotin (DAKO Corporation, Carpinteria, CA, USA) was conjugated to purified TcdA or TcdB by addition of 25-μg biotin to 1.5-mg toxin per milliliter. After 4 hours incubation at 25°C, 0.2-μl 1 M-ammonium chloride was added for every 25-μg biotin in the conjugation mixture. The mixture was incubated for 10 minutes at 25°C, and the biotinylated toxin was dialyzed overnight in phosphate-buffered saline. CHO-cell assay demonstrated full activity of biotinylated and freeze-thawed toxins. Biotinylated TcdA and TcdB were diluted to 150 μg/ml and stored at −4°C.
Immunohistochemistry
Deparaffinized slides of normal tissues were blocked by avidin and then biotin (DAKO Corporation) for 15 minutes each. Biotinylated TcdA or TcdB (each at a stock concentration of 150 μg/ml) was added at a dilution of 1 : 80 and incubated on the slides for 30 minutes. Streptavidin-horseradish peroxidase and diaminobenzidine (DAB; DAKO Corporation) were used to indicate the location of bound toxin. Negative controls consisted of tissue sections incubated with biotinylated “nontoxic” proteins derived from cultures of a nontoxigenic C. difficile strain (JGS692) and eluted from the ion-affinity columns under the same conditions used to purify TcdA and TcdB.
A traditional IHC assay was used to detect nonbiotinylated TcdA localized on sections of normal tissues. Sections were blocked by avidin and biotin as before. Untreated TcdA (200 μg/ml) was added at a dilution of 1 : 100 and incubated for 30 minutes. Slides were washed, and a rabbit polyclonal anti-TcdA (100 μg/ml; Lee Labs, Grayson, GA, USA) was incubated on the sections at a dilution of 1 : 500 for 30 minutes. Alternatively, a mouse monoclonal anti-TcdA, BDI515 (100 μg/ml; Biodesign International, Saco, ME, USA) was used at a dilution of 1 : 20 for 60 minutes. Both the rabbit and mouse antibodies were detected by the Envision reagent (Dako Corporation), followed by DAB.
The distribution of α-galactosyl was also determined by a standard IHC procedure, using a mouse monoclonal antibody, M86, with known specificity for this carbohydrate. 8, 10 M86 (5 μg/ml) was applied to slides at a 1 : 4 dilution. The Envision reagent and DAB were used to demonstrate tissue localization of M86. All incubations were at 25°C, washes were included prior to each treatment, and all slides were counterstained with hematoxylin. Negative controls consisted of tissue sections examined by the described IHC method but with the substitution of a mouse monoclonal immunoglobulin-M antibody specific for Cryptosporidium α protein, instead of M86.
Sections were examined by light microscopy, and staining intensity was scored from – (no signal visible) to +++ (intense signal deposition) for each tissue and for surface epithelium and crypts or glands. Values from all individuals were averaged for each site. Tissues from neonatal pigs were examined by each of the aforementioned methods. Direct IHC was performed on adult hamster tissues using biotinylated TcdA and TcdB. Indirect IHC was performed on neonatal hamsters, using the polyclonal antibody specific for TcdA.
Results
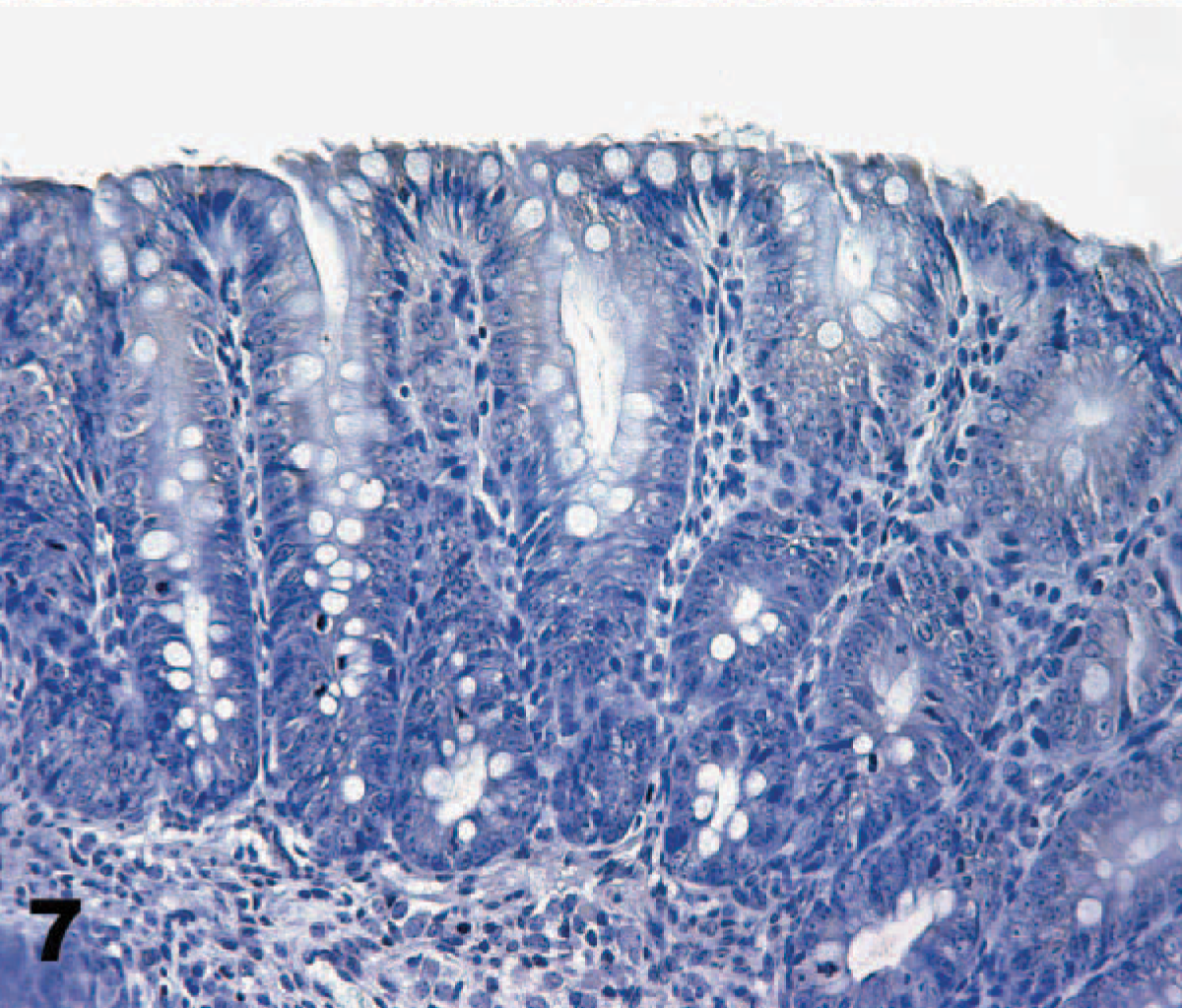
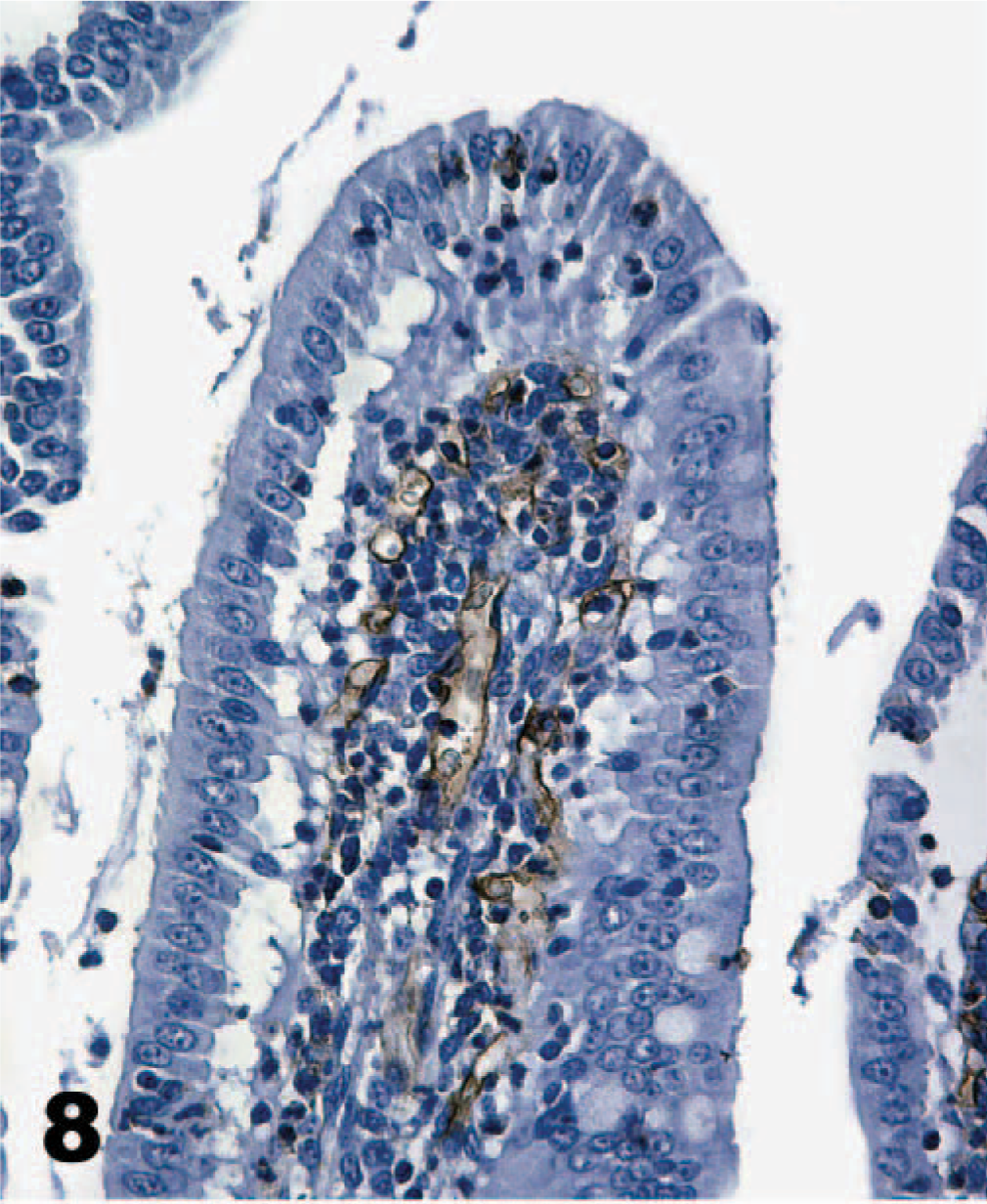
Direct IHC with biotinylated TcdA demonstrated extensive binding of toxin throughout the mucosa of the neonatal porcine gastrointestinal tract. Specific binding to mucosal epithelial cells was limited to the brush border; the cytoplasm and nuclei of labeled cells were unstained. Brush border binding was differentiable from any edge effect because individual cells exhibited intense staining while rare cells in the midst of positively stained cells were negative (Figs. 2, 3). In addition, some regions of the intestinal tract, particularly the colon, had superficial cells that were stained, but those deep in the crypts or glands were unstained (Fig. 4). The most intense binding of TcdA was in the jejunum (Fig. 5), where cells of the villi and crypts were all intensely stained. Epithelial cells of the gastric mucosa were lightly stained by the biotinylated TcdA. Duodenal crypt cells were variably stained, and staining was generally more intense on villus surfaces of the duodenum. Strong binding to villus enterocytes in the ileum was also seen, but binding in the crypts was moderate (Fig. 6). TcdA slightly bound epithelial cells in the cecal glands and on the luminal surface. Moderate staining (++) occurred in the ascending and descending colon, but only on the brush border of cells lining the lumen or in the superficial portion of colon glands (Fig. 4). Biotinylated TcdB did not stain any of the tissues (Fig. 7). Negative controls consisting of the biotinylated nontoxic proteins, eluted under the same conditions as TcdA and TcdB, also did not bind any portions of the tissues.
Colon: neonatal pig. Toxin B (TcdB) did not bind to cells in any sections of the tissues. Biotinylated TcdB, streptavidin-peroxidase-DAB method, hematoxylin counterstain.
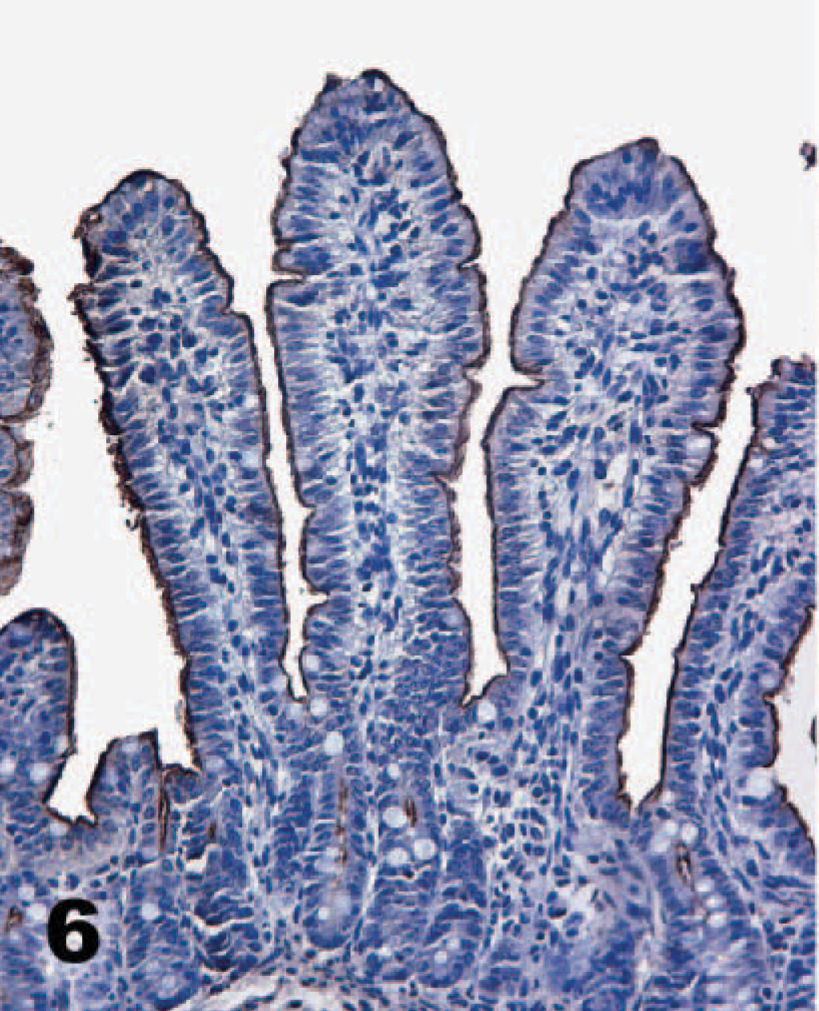
Ileum; neonatal pig. Toxin A (TcdA) binds villus enterocytes in the ileum, but staining extends only a short distance into the crypts. Biotinylated TcdA, streptavidin-peroxidase-DAB method, hematoxylin counterstain.
Jejunum; neonatal pig. Toxin A (TcdA) binds very intensely to the brush border of villus enterocytes as well as epithelial cells in the deepest portions of the crypts. Biotinylated TcdA, streptavidin-peroxidase-DAB method, hematoxylin counterstain.
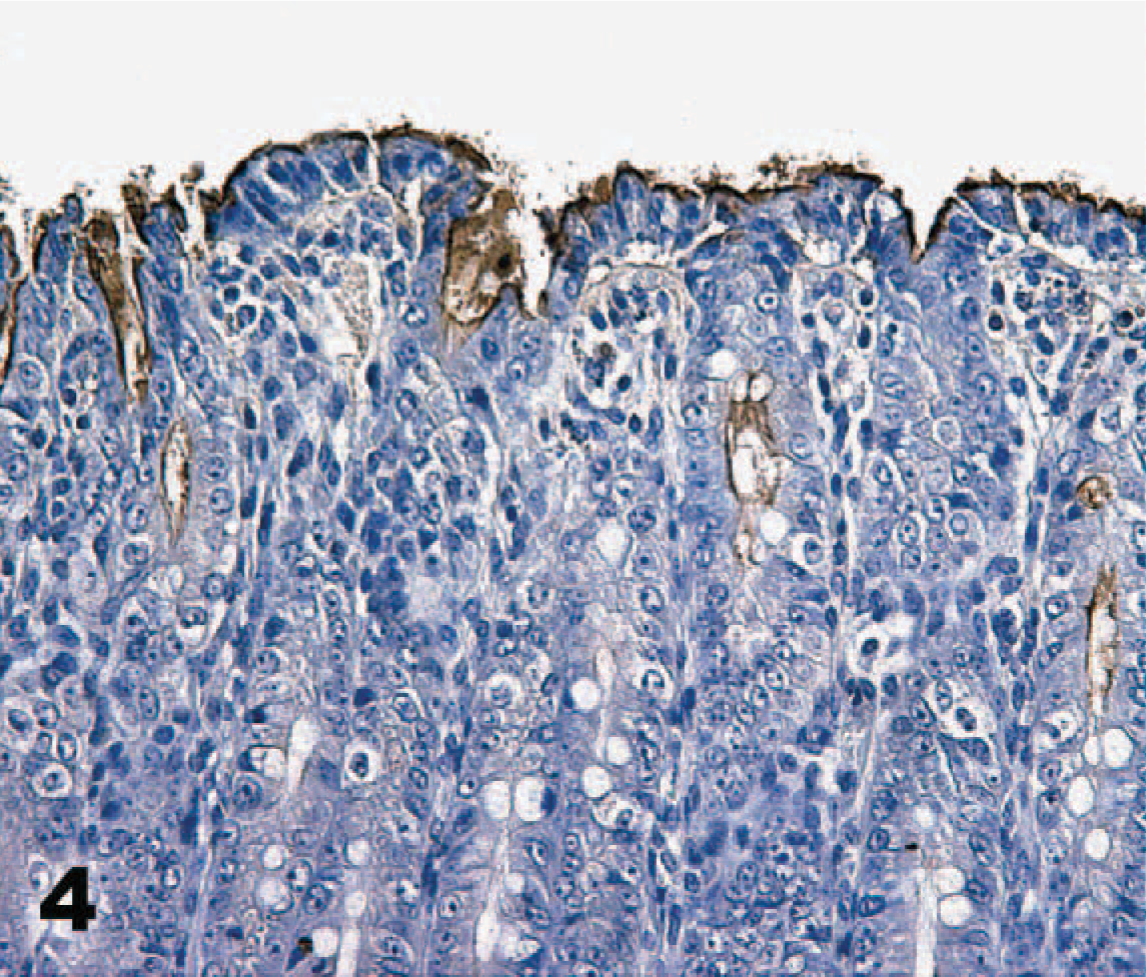
Colon; neonatal pig. Toxin A (TcdA) binds intensely to the brush border of epithelial cells lining the lumen, but binding is diminished in deeper portions of the colon glands. Biotinylated TcdA, streptavidin-peroxidase-DAB method, hematoxylin counterstain.
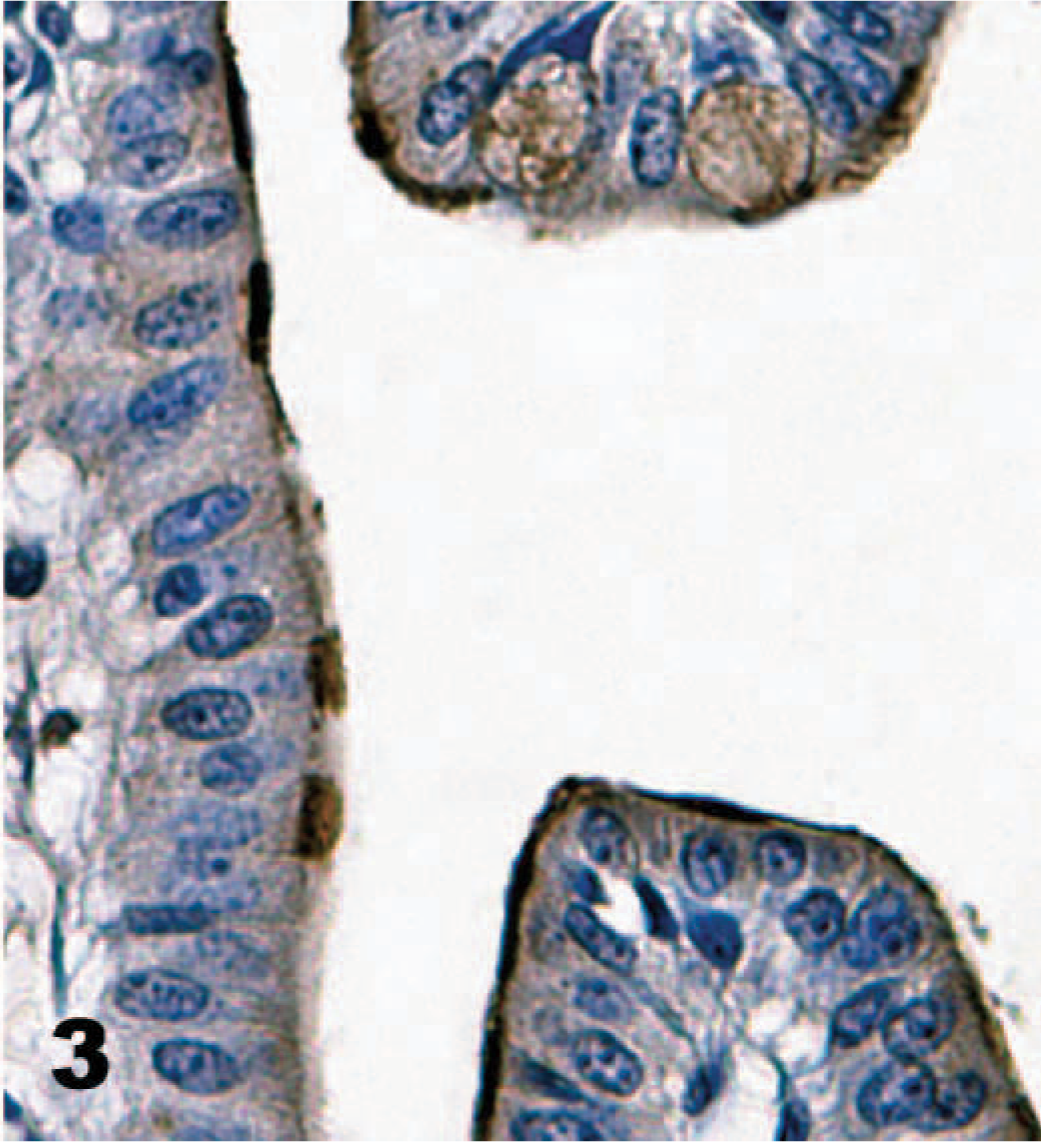
Ileum; neonatal pig. The brush border is variably labeled by biotinylated toxin A (TcdA) with dense staining of individual cells adjacent to cells not stained. Biotinylated TcdA, streptavidin-peroxidase-DAB method, hematoxylin counterstain.
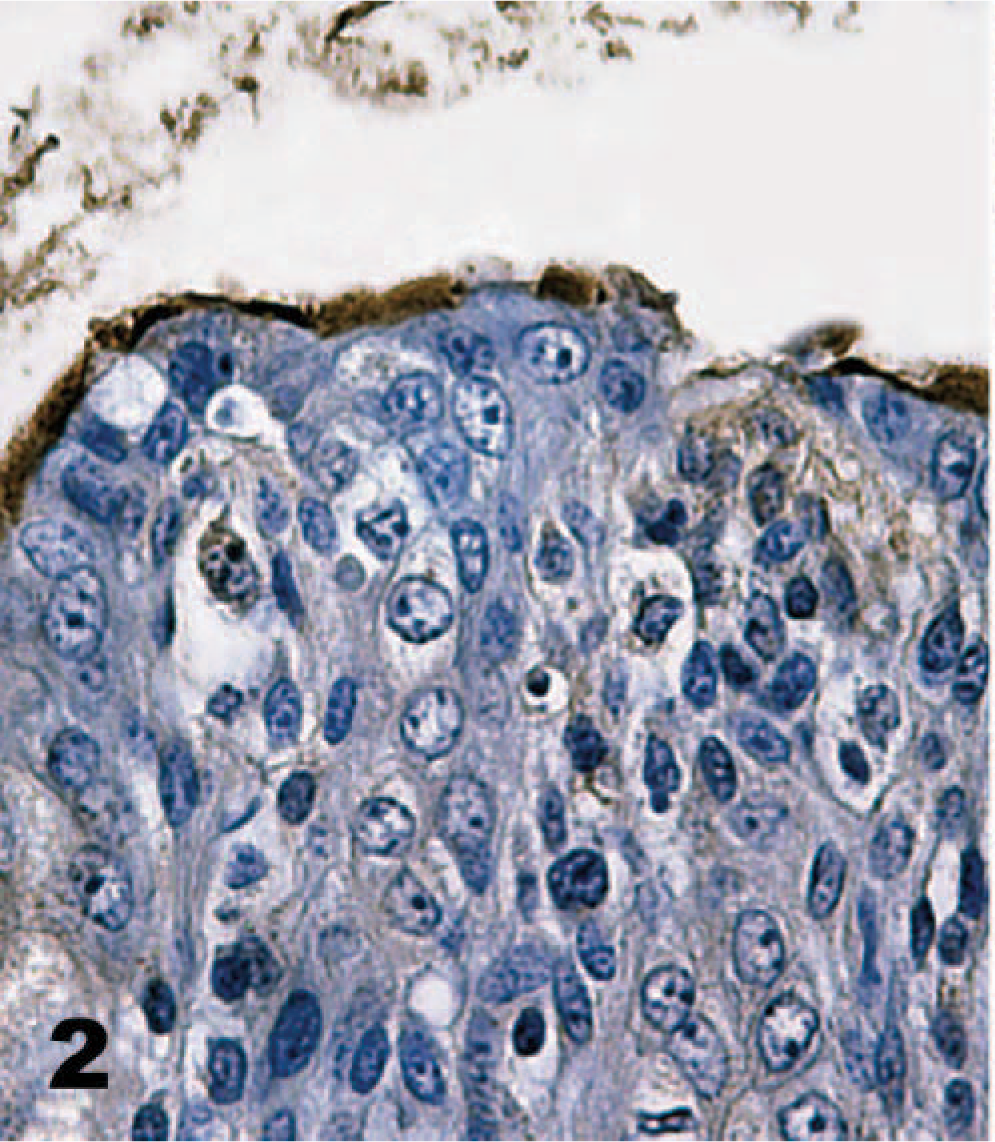
Colon; neonatal pig. Biotinylated toxin A (TcdA) generally binds the brush border of epithelial cells. However, individual colonocytes are unstained. Biotinylated TcdA, streptavidin-peroxidase-DAB method, hematoxylin counterstain.
IHC using polyclonal antibodies to identify unlabeled toxin bound to tissue sections generated the same results as direct IHC experiments using biotinylated toxins. The same relative intensity of toxin binding was demonstrated throughout the gastrointestinal tract. Indirect IHC, with a monoclonal antibody against TcdA for detection, failed to stain any of the bound toxin.
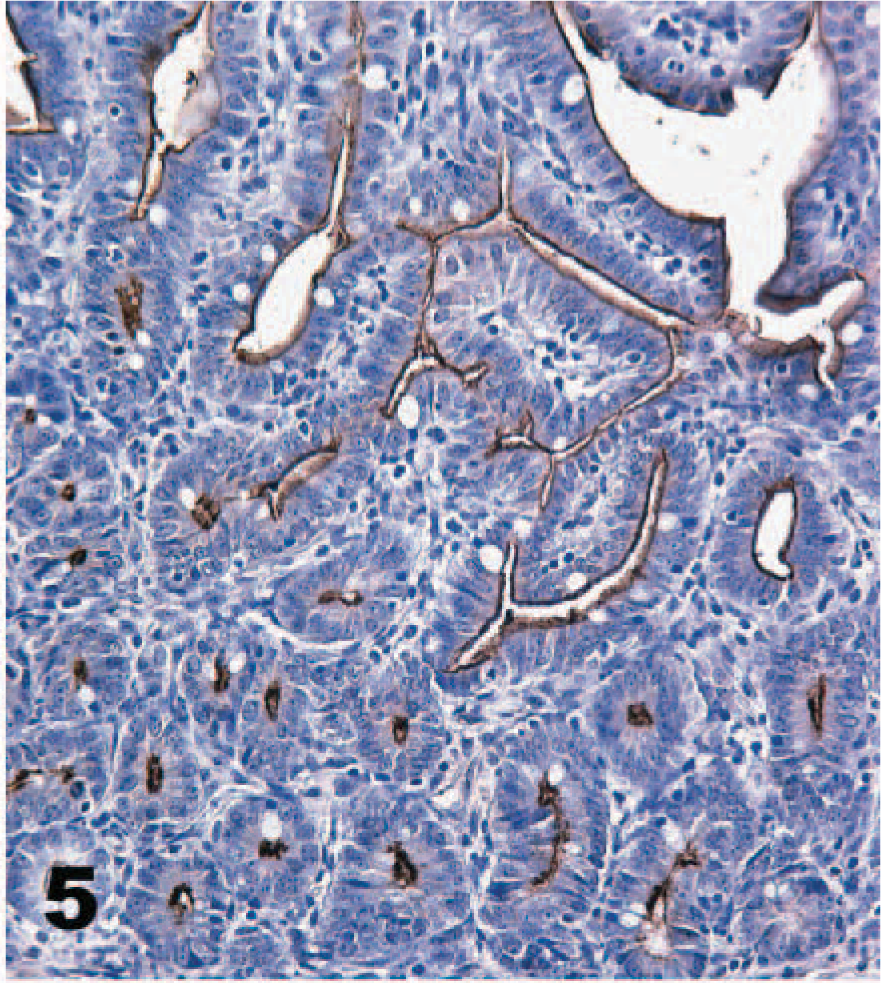
A monoclonal antibody against α-galactosyl specifically stained the brush border of enterocytes, but the stain distribution was substantially different from that of TcdA binding (Table 1). Most cells labeled by TcdA, as detected by either direct or indirect IHC, were not labeled by the α-galactosyl antibodies. For example, the brush borders of duodenal enterocytes and colonic epithelial cells were not stained by the α-galactosyl technique, though there was specific staining of endothelial cells (Figs. 8, 9). Biotinylated TcdA bound the brush border of the colon epithelial cells but did not bind endothelium (Fig. 4). The only cells labeled by both methods were villus enterocytes in the jejunum and ileum. Staining of these cells by the α-galactosyl technique was less intense (++) than that observed with the TcdA techniques, and staining of the crypts did not occur. The α-galactosyl antibodies also stained cells of the pancreatic duct, but these were not bound by TcdA.
Immunohistochemistry of Clostridium difficile toxin attachment and receptor sites in pig tissues.†

∗TcdA = toxin A, Abs = XXX, TcdB = toxin B, Duod. = duodenum.
†Relative intensity of immunohistochemical signal is indicated for mucosal epithelial cells in each segment of the gastrointestinal tract and in additional, select tissues.
‡No binding is indicated by a score of −. Maximal binding is indicated by a score of +++.
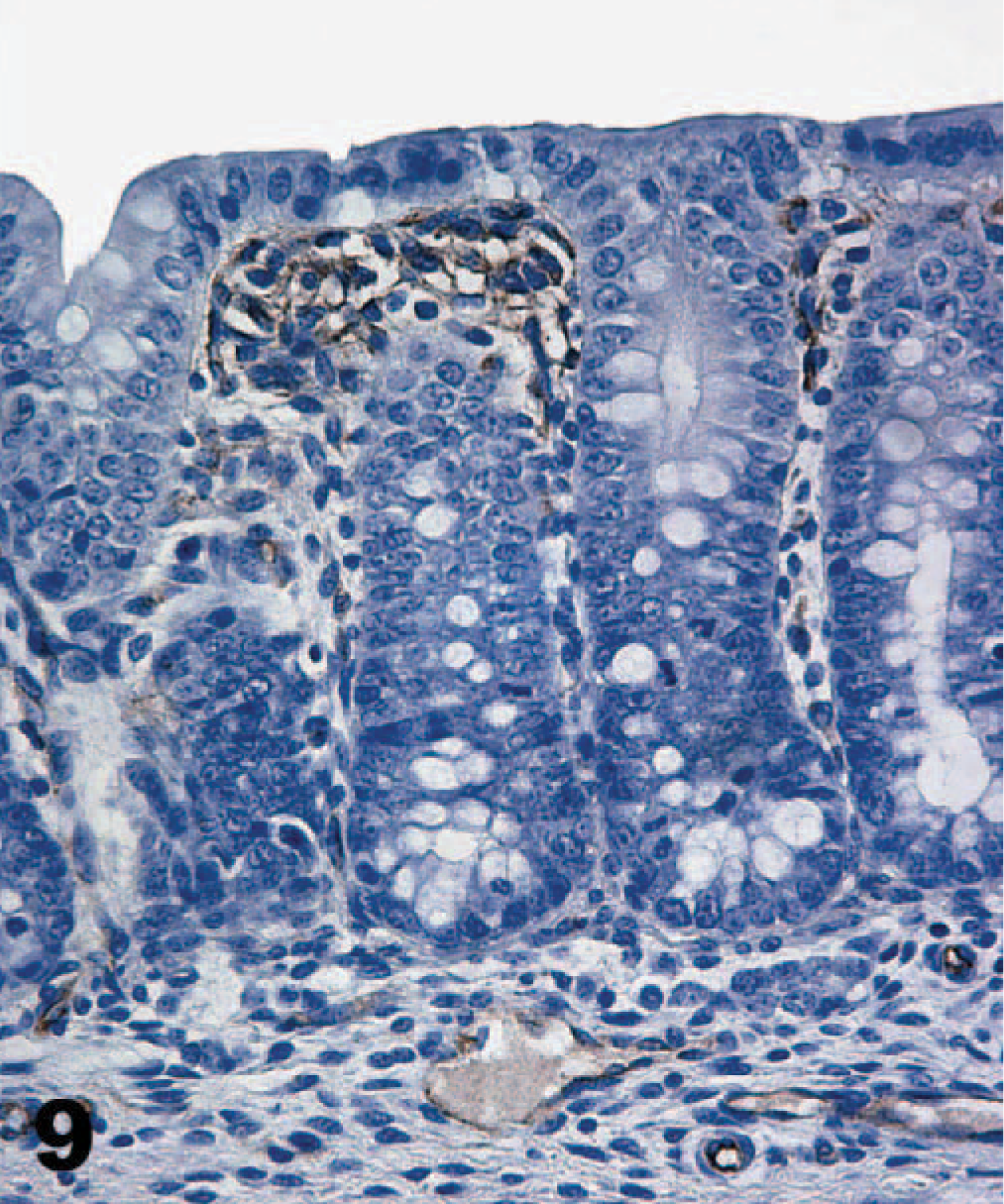
Colon; neonatal pig. The α-galactosyl epitope is not stained on the brush border of colon epithelial cells, but endothelial cells in the lamina propria and submucosa are positively stained. M86, streptavidin-peroxidase-DAB method, hematoxylin counterstain.
Duodenum; neonatal pig. The α-galactosyl epitope appears to be restricted to endothelial cells, and none can be identified on the brush border. M86, streptavidin-peroxidase-DAB method, hematoxylin counterstain.
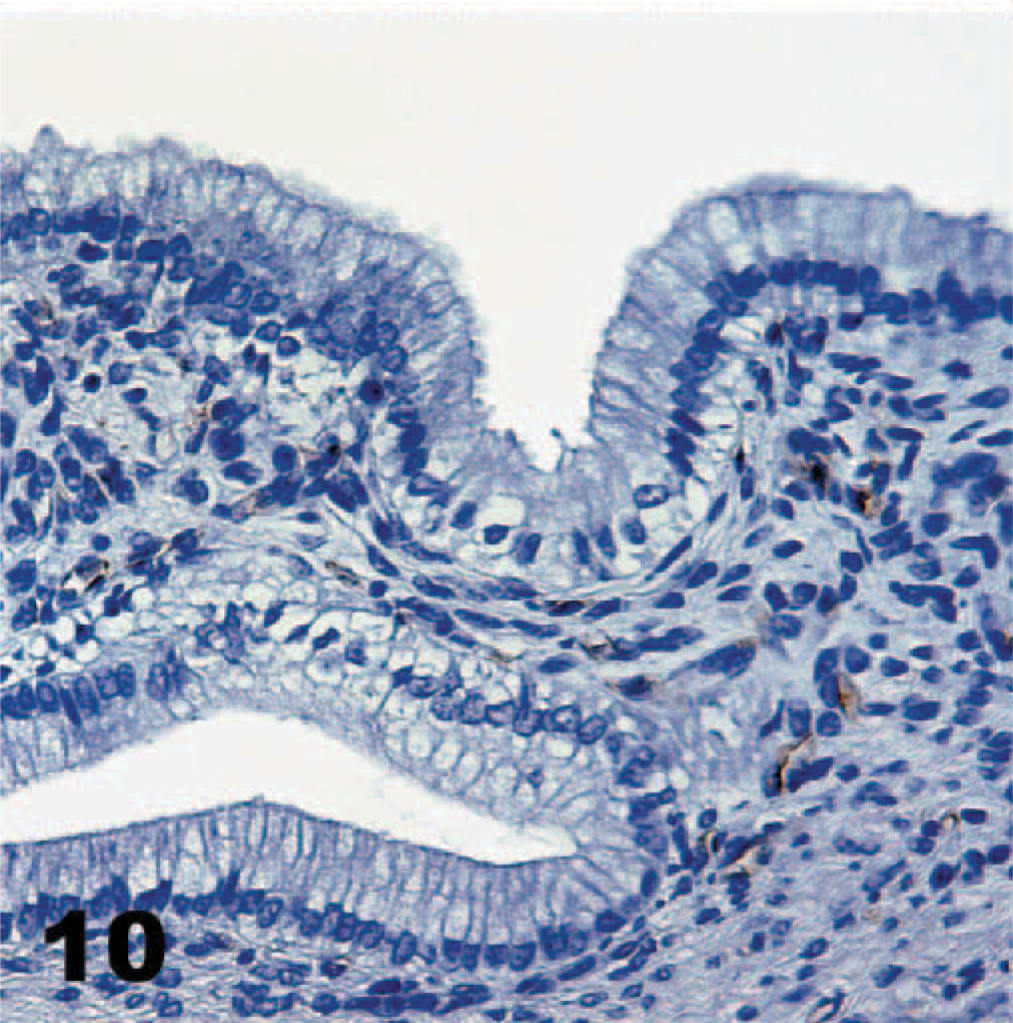
As in other porcine tissues, α-galactosyl antibodies recognized endothelium in the gallbladder, but epithelial cells were not stained (Fig. 10). The only cell type, other than those of the gastrointestinal mucosa that TcdA bound, was gall bladder epithelium (Fig. 11). Intense staining of all the epithelial cells occurred, but only in the cytoplasm, not at the cell surface. Endothelial cells in the submucosa of the gallbladder were not stained by the TcdA technique.
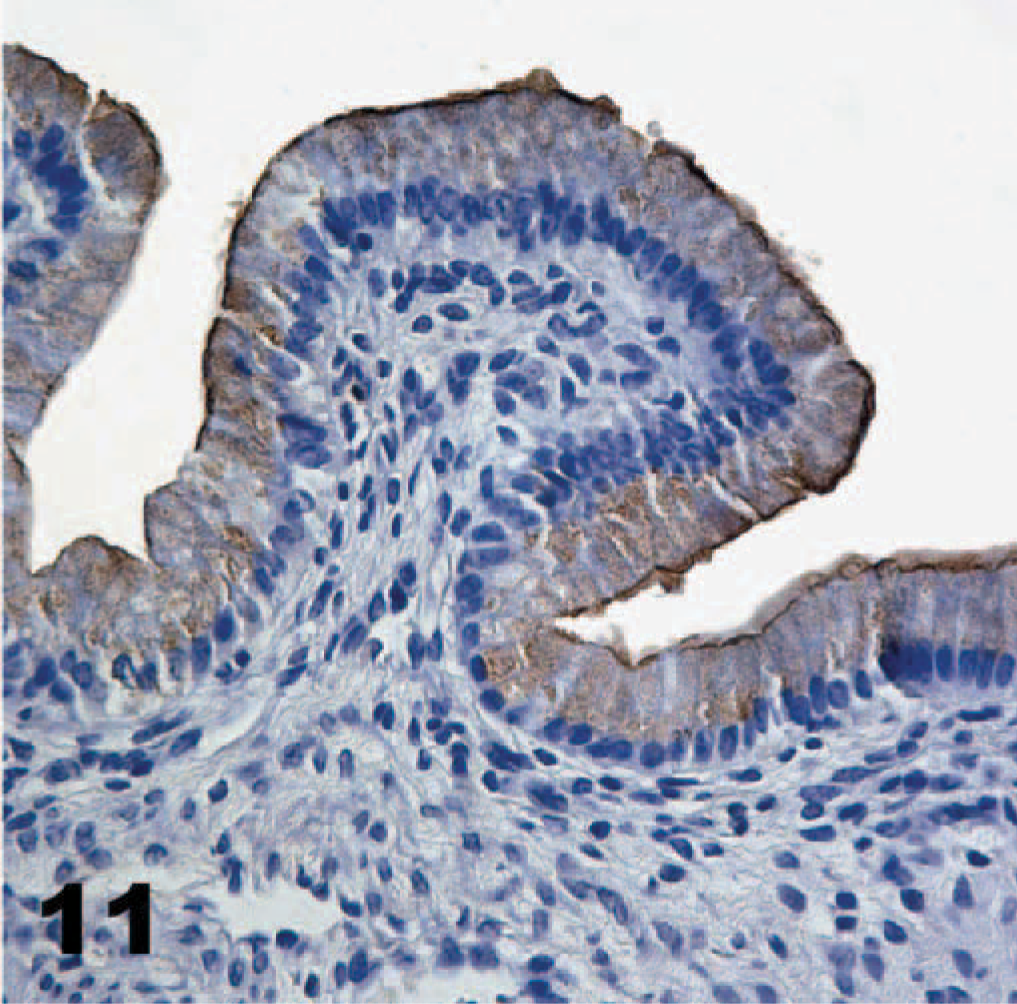
Gall bladder; neonatal pig. Toxin A (TcdA) is bound throughout the cytoplasm of gall bladder epithelial cells, but no other cell types are bound. Biotinylated TcdA, streptavidin-peroxidase-DAB method, hematoxylin counterstain.
Gall bladder; neonatal pig. Only endothelial cells are labeled for α-galactosyl. M86, streptavidin-peroxidase-DAB method, hematoxylin counterstain.
Hamster tissues, both neonatal and adult, exhibited binding characteristics similar to those of pig tissues (Table 2). The duodenum of the adults had more intense staining (+++) than that noted for neonatal pigs, with equal staining along the villi and in the crypts. Staining was more intense on the surface of the cecal (++) and colonic (+++) mucosa, but the glands were not stained at all. Gall bladder epithelium was stained just as it was in the pigs. Neonatal hamsters had analogous staining, but the signal was consistently weaker. There was also some staining of cecal and colon glands, not evident in the adult hamsters. Gall bladder epithelium was not examined in the neonates. Hamster tissues were not examined by the α-galactosyl technique.
Immunohistochemistry of Clostridium difficile toxin attachment and receptor sites in hamster tissues.†

∗TcdA = toxin A, TcdB = toxin B, α-TcdA Abs = XXX, Duod. = duodenum.
†Relative intensity of immunohistochemical signal is indicated for mucosal epithelial cells in each segment of the gastrointestinal tract and in additional, select tissues.
‡No binding is indicated by a score of −. Maximal binding is indicated by a score of +++.
Discussion
Specific staining for TcdA was achieved by the use of biotinylated TcdA or polyclonal antibodies against TcdA. Intensity of toxin binding does not correlate with the distribution of lesions occurring in spontaneous cases of porcine CDAD. 24, 31 Naturally occurring lesions are generally limited to the cecum and colon, but toxin binding was much more intense in the small intestine, especially the jejunum. Experimental gavage of pigs with TcdA, however, generated lesions that were much more severe in the small intestine than elsewhere in the gastrointestinal tract (M.K. Keel, unpublished data). This distribution of lesions could reflect the greater density of receptors on those tissues, but unintended effects of anterograde administration of the toxin cannot be ruled out.
The limitation of lesions to the cecum and colon, in spontaneous cases, may reflect the natural distribution of vegetative C. difficile and production of toxin, rather than specific susceptibility of those tissues. Inoculation of naive hamsters with C. difficile led to greater concentrations of the bacteria in the distal small intestine, cecum, and colon. 23 Highly virulent strains of C. difficile most readily adhered to the cecum of hamsters, the typical site of lesions in that species. 3 Within each segment of the intestine, initial lesions are limited to those cells possessing receptors, as indicated by toxin binding (M.K. Keel, unpublished data). Lesions in naturally occurring cases of disease may progress to involve much deeper tissues. 13 However, the pathogenesis of CDAD is multifactorial, and much of the damage to deeper epithelial cells, lamina propria, and even submucosa, probably results from inflammation. 20
TcdA binding to tissue sections could not be documented by an IHC employing a monoclonal antibody specific for TcdA. However, the antibody may have recognized epitopes in or near the receptor-binding portion of the protein. Such sites would likely have been masked in the bound toxin. The monoclonal antibody did not cross-react with TcdB. This also is consistent with antibody binding near the receptor portion of the molecule because other regions of the 2 toxins were well-conserved.
The well-documented abundance of α-galactosyl on pig tissues and often-repeated claims that it is a receptor for TcdA in other species both led to the hypothesis that it is the dominant receptor on porcine tissues. However, there is significant disparity in the binding of TcdA to mucosal epithelium and the distribution of this carbohydrate; toxin binding was much more extensive than the distribution of α-galactosyl. IHC indicated that α-galactosyl was limited to the surface of villus enterocytes in the jejunum and ileum only. Specific binding was also present on endothelial cells and ductal epithelial cells of the pancreas. TcdA, in contrast, bound neither of those cell types. In addition, TcdA labeled the cytoplasm of gall bladder epithelial cells, though M86 did not (Fig. 11).
Neonates of most species are resistant to CDAD, even though C. difficile readily colonizes their large intestines. 5, 11, 23 This has prompted the hypothesis that newborns of those species lack adequate toxin receptors until later in life. Neonatal pigs seem to have abundant toxin receptors and are known to be susceptible to the disease. The IHC assay was repeated with newborn hamsters, who do not develop CDAD as neonates. 23 Significant binding of toxin occurred throughout the intestine, suggesting that resistance to the effects of C. difficile colonization may be due to other factors. Very young hamsters, less than 4 days of age, are resistant to colonization by C. difficile, possibly providing some protection from CDAD. 23 Neonatal pigs, however, are readily infected with C. difficile. 31 IHC of tissues from adult hamsters indicated a slightly more intense binding of TcdA. As in neonatal pigs, TcdB did not bind any tissues in neonatal or adult hamsters.
A receptor for TcdB has never been identified on the luminal surface of intestinal epithelial cells, and there is evidence that an intact mucosa is resistant to the effects of TcdB. 18 Only with mucosal trauma or concomitant exposure to TcdA was TcdB reportedly able to exert a significant effect. A prominent hypothesis to explain this scenario is that the receptor for TcdB is restricted to the basolateral aspect of the cell. After damage to epithelial cells or loss of intracellular attachments due to TcdA action, the receptors are exposed to TcdB. However, in the IHC experiment, TcdB had access to all aspects of cells because the tissues were sectioned. Still, no binding was detected even though the biotinylated toxin was fully active in cell culture assays. TcdB may gain access to the cell through another route, perhaps pinocytosis.
Demonstration of abundant widespread receptors on intestinal surfaces of neonatal pigs provides additional evidence of their specific susceptibility to C. difficile toxins. The receptors for TcdA in the pig remain to be identified, but α-galactosyl does not seem to be significant to its binding.
Footnotes
Acknowledgements
We thank Sharon Dial, Woodson Tucker, Matt McDaniel, Mike Riggs, and Debbie Schaeffer for their assistance with the IHC techniques. We also thank Uri Galili, of Rush University, for donating the monoclonal antibody specific for α-galactosyl.
