Abstract
Toxin A (TcdA), secreted by toxigenic strains of Clostridium difficile, produces lesions typical of C. difficile–associated disease (CDAD) in susceptible mammal species. Porcine colon explants maintained for 2 hours with TcdA developed severe lesions characterized by cell swelling, swelling of mitochondria and other organelles, distension of cytoplasmic vesicles, expansion of paracellular spaces, apoptosis, and necrosis. Severity of lesions was proportional to the dosage of toxin. No lesions were present in uninoculated control tissues after 2 hours. Receptor-mediated endocytosis is the keystone event in the pathogenesis of the toxin, and susceptibility of a given species is thought to depend on the presence of receptors in intestinal epithelial cells. The fate of TcdA applied to viable colon explants was determined by transmission electron microscopy in an anti-toxin-labeled gold assay. At 5 minutes postinoculation, the presence of TcdA was indicated at the membrane of microvilli or in the cytoplasm of epithelial cells. TcdA was also indirectly observed within endosomes or attached at their margin. A 30-minute inoculation period was associated with many more gold particles labeling structures inside the cell, although some were still attached to microvilli. Within the cell, most TcdA was associated with mitochondria of epithelial cells, but some gold particles decorated the nuclei. Endothelial cells of the lamina propria had evidence of TcdA at both their lumenal and basal aspects, as well as in the cytoplasm and, occasionally, nuclei. Gold particles also labeled the lumen of such vessels as well as leucocytes in blood vessels and the lamina propria.
Keywords
Introduction
Clostridium difficile is an anaerobic, gram-positive, spore-forming bacillus associated with diarrhea and enterocolitis in a variety of mammals and is the most common bacterial cause of antibiotic-associated diarrhea in humans. 18,19 The principal virulence factors, toxins A and B (TcdA and TcdB), are large, AB-like toxins. They belong to a group of toxins with similar topology and function, the large clostridial toxins (LCTs). TcdA and TcdB share significant sequence homology and have similar domains, including an amino-terminal enzymatic domain, a hydrophobic region, and a carboxyl-terminal domain characterized by a series of repetitive peptides. 42,44,45 The carboxyl-terminus is the receptor binding portion and differs significantly between the two toxins. The carboxyl terminus of TcdA recognizes several carbohydrate receptors on the plasma membrane of susceptible cells. These include Galα1-3β1-4GlcNAc-R (α-Gal epitope), Galβ1-4{Fuα1-3}GlcNAc (Lewis X), Galβ1-4GlcNAcβ1 (Lewis Y), Galβ1-14GlcNAcβ1-3Galβ1-4(Glc) (Lewis I), and sucrose-isomaltase. 25,33,34,41 Receptor binding is followed by endocytosis, and acidification of the endosome is essential for full toxin activity. 9,16,36 A receptor for TcdB has never been identified but is hypothesized to exist on the basolateral aspect of intestinal epithelial cells.
The amino-termini of TcdA and TcdB contain the enzymatic domain and exhibit similar activity and substrate specificity. 7,8,43 Both inactivate Rho-subtype GTPases by glucosylating a threonine residue at 35/37. 10,21,22 These GTPases are a group of signal transduction molecules essential for the regulation and maintenance of actin filaments. Dysregulation of actin filaments leads to loss of tight junctions and cell rounding, both directly associated with the loss of mucosal function as an osmotic barrier.
TcdA is also associated with mitochondrial damage of epithelial cells and colocalizes with mitochondria within 5 minutes of cell entry. 12 Mitochondrial colocalization and damage by TcdA occur prior to the blockade of Rho-dependent pathways. Disruption of mitochondria is associated with decreased production of adenosine triphosphate (ATP), generation of reactive oxygen intermediates, and production of cytokines such as IL-8. 13 Although the effects of TcdA on mitochondrial function have been catalogued, the direct interactions of TcdA with the mitochondria remain uncertain. TcdA-induced apoptosis is associated with the generation of reactive oxygen species by the mitochondria and activation of pro-apoptotic caspases but is also dependent on the inactivation of Rho-subtype GTPases. 3 Both caspase 8 and 9 are involved in TcdA-induced apoptosis, incriminating both the extrinsic and intrinsic apoptosis pathways.
A number of intestinal cell types other than mucosal epithelial cells are directly affected by TcdA, including neutrophils, mast cells, monocytes, and neurons. 4,5,49 Pathogenesis of the disease is complicated by the interactions of affected cell types by inflammatory mediators and release of cytotoxic inflammatory compounds that can contribute directly to damage of host tissues. 35
Proinflammatory mediators are associated with vascular changes in vivo, and much of the vascular leakage is supposedly leukocyte dependent. 26 However, both TcdA and TcdB are directly toxic to endothelial cells, and such toxicity could contribute to the effusion and edema associated with C. difficile –associated disease (CDAD) if the toxins gain access to endothelial cells. 30 Virtually all cell types are somehow involved in the complex cascade of events associated with CDAD. Which of these associations are due to the direct interaction of the toxins with the target cells, and which are due largely to the actions of intermediate participants is often difficult to elucidate.
Typhlocolitis and neonatal diarrhea due to C. difficile were only recently reported in pigs. 46 Previous attempts to isolate C. difficile from pigs were unsuccessful, although it was eventually cultured from 8-week-old pigs coinfected with Salmonella typhimurium. 20 The susceptibility of pigs to CDAD was further cast in doubt by intestinal loop studies in which C. difficile toxins directly injected into the lumens did not result in significant lesions. 37 There have since been additional reports of CDAD in pigs, and experimental inoculation of colostrum-deprived pigs resulted in disease and lesions identical to those of spontaneous cases. 38 Nonetheless, there is a need for better characterization of the susceptibility of pigs to CDAD and the host-tissue response to infection. This is important as a potential model for better understanding the disease in humans and other species and to better explore prevention strategies for the swine industry.
Materials and Methods
Antibodies
A mouse monoclonal antibody, BDI515 (Biodesign International, Saco, ME), was used in the immunoaffinity chromatography of TcdA. Its isotype was IgG, and it was specific for TcdA without recognizing any of the related toxins. An immunoaffinity-purified rabbit polyclonal antibody, M-C663-50L (Lee Laboratories, Grayson, GA), raised against TcdA, was used for all immunoelectron microscopy procedures. Goat antirabbit antibodies, linked to 6-nm colloidal gold beads (Aurion, Wageningen, The Netherlands), were also used.
Animals
Clinically normal 12- to 24-hour-old pigs were selected based on negative culture for C. difficile and negative enzyme-linked immunosorbent assay (ELISA) for TcdA and TcdB (Techlab, Blacksburg, VA). Intestinal explants collected from the pigs were initially placed in wash media consisting of Dulbecco’s modified Eagle’s medium (DMEM)/ F-12 (Cambrex, Walkersville, MD) with amikacin (40 μg/ml; Fisher Scientific, Fairlawn, NJ), kanamycin (0.6 mg/ml; Sigma-Aldrich, St. Louis, MO), streptomycin (2 mg/ml; Fisher Scientific), and vancomycin (100 μg/ml; Sigma-Aldrich). They were then maintained in nutrient media, DMEM/ F-12 (Cambrex, Walkersville, MD) with amikacin (20 μg/ml), streptomycin (2 mg/ml), and vancomycin (50 μg/ml). Both types of media were prewarmed to 37°C and pregassed with 5% CO2 before initiation of experiments.
Pigs were euthanized by CO2 and were placed in dorsal recumbency in a laminar flow hood. The abdomen was disinfected with surgical scrub and opened by a midline incision; the ascending colon was freed using sterile technique. The colon was placed in a sterile tray, irrigated with wash media, linearized by alternate blunt and sharp dissection, and opened by a longitudinal incision on the antimesenteric surface. Fecal material was gently flushed from the mucosal surface with wash media, and 0.5-cm square sections were trimmed with a razor blade. The tissue samples were maintained in wash media until they were added to 6-well tissue culture plates, 25 to 30 minutes after euthanasia. Sterile instruments were used to ensure that no exogenous bacteria were introduced to the explants.
Tissue Processing
Tissue samples used exclusively for ultrastructural studies were fixed in 4% formalin with 1% glutaraldehyde in phosphate-buffered saline (PBS; 4F1G). After approximately 24 hours, the tissues were rinsed in 0.1% PBS (pH 7.4) and postfixed in 1% osmium tetroxide. They were then rinsed in distilled water and dehydrated through graded alcohol. They were incubated in propylene oxide followed by 50% propylene oxide with 50% epon. Tissues were embedded in epon and cured at 75°C for 8 hours. Thin sections were poststained with saturated aqueous uranyl acetate and Reynold’s lead citrate.
Samples generated for immunostaining were fixed in 1% formalin with 0.1% glutaraldehyde in PBS (1F0.1G). Tissues were fixed for 45 minutes, rinsed in distilled water, and stained with 2% aqueous uranyl acetate for 30 minutes. They were then dehydrated through graded alcohol and embedded in LR-white (Ted Pella, Redding, CA). Thin sections were placed on nickel grids that had been immersed in 1.25% Formvar (Ted Pella) in chloroform, such that grid bars were coated but the open area was uncoated. This optimized adhesion of sections to the grids but eliminated the problem of background staining of colloidal gold on the hydrophilic plastic coating. Grids were blocked by immersion in 0.1% bovine serum albumin (BSA) with 0.1% Tween-20 (Electron Microscopy Sciences, Ft. Washington, PA) in PBS (pH 7.4) for 10 minutes. They were then immersed in a 1:50 dilution of the rabbit polyclonal anti-TcdA and incubated at room temperature for 2 hours. Labeled grids were washed 3 times with PBS (pH 7.4) and labeled with the secondary antibody (1:20) linked to colloidal gold. After another wash step, they were fixed in 0.5% glutaraldehyde. The grids were washed in distilled water and poststained for 10 minutes with saturated uranyl acetate. At each step, grids were completely immersed to ensure that both sides of the sections were labeled and washed.
All sections were examined with a Jeol transmission electron microscope (Japan Electron Optical Laboratories, Peabody, MA) with an accelerating voltage of 60 to 80 kV. The distribution of gold-labeled particles was quantified using sterological techniques. 29 Briefly, photos from randomly selected fields were superimposed with test-line grids to count the number of sites at which grid lines intersected events of interest (immunogold labeling) or not. A relative labeling index (RLI), defined as the number of expected gold particles divided by the number of observed gold particles, was generated for intracellular compartments, and the X-squared test was used to compare expected and observed distributions of gold particles.
Experimental Protocol
Five treatment groups were used in the ultrastructural study of TcdA-associated lesions. Individual samples in the respective groups were treated with 2.5, 1, 0.5, 0.25, or 0 μg/ml of TcdA. Four replicates were performed in each treatment group, and the tissue explant for each treatment was randomly selected from 1 of 2 pigs. Organ culture media were prewarmed to 37°C and pregassed with 0.5% CO2. Each tissue culture well contained 7.5 ml of media with toxin at the concentration designated for the respective treatment groups. Tissues were incubated for 2 hours, then removed for fixation in 4F1G.
The immunoelectron microscopy study was performed in the same manner, with minor variation in the treatment groups. One treatment group consisted of samples incubated in 5 μg/ml TcdA for 5 minutes, and 3 additional treatment groups consisted of samples incubated in 10 μg/ml TcdA for 5, 30, or 60 minutes. Control tissues were incubated in organ culture media, without toxin, for 5 minutes. After the requisite incubation period, tissues were placed directly in 1F0.1G.
Bacterial Culture
Fecal samples from all animal subjects were swabbed and streaked for isolation directly on taurocholate, cycloserine, cefoxitin, fructose agar (TCCFA), and C. difficile selective agar (CDSA; brain heart infusion, C. difficile antimicrobic supplement CC, sodium taurocholate, and bovine blood) as previously described. 11,48 After 48 to 72 hours, suspect C. difficile colonies were picked and transferred to brain heart infusion agar with yeast extract (0.5%), cysteine hydrochloride (0.05%; BHI-CYE), and bovine blood (2%). Identification of C. difficile was based on colony morphology, odor, weak α hemolysis, and a positive L-proline aminopeptidase assay (REMEL, Lenexa, KS). For toxin production, cultures of C. difficile strain VPI 10463 (ATCC, Manassas, VA) were maintained for 5 to 7 days in dialysis tubing (molecular weight cutoff, 10,000) suspended in BHI-CYE broth.
Toxin Purification
Supernatant from dialysis bag cultures of C. difficile was harvested by centrifugation at 15,000 × g for 20 minutes. Toxins were purified from the supernatant by ion affinity chromatography on a DEAE-sepharose column (Amersham Biosciences, Uppsala, Sweden). Fractions of 5 ml were collected on a GradiFrac rotary fraction collector (Amersham Biosciences), and protein elution was monitored by a UV-1 ultraviolet monitor (Amersham Biosciences). TcdA was eluted with a linear gradient of 0.05 to 0.25 M NaCl in 50 mM Tris-HCl at a pH of 7.4. Fractions of interest were pooled and the column eluate was concentrated by centrifugation in a centrifugal filter device with a molecular weight cutoff of 100,000 daltons (Millipore, Bedford, MA). Concentration of the toxin was determined by a Bradford assay per the manufacturer’s instructions (Bio-Rad, Hercules, CA). Toxin activity was determined by Chinese hamster ovary (CHO) cell assay, and purity was confirmed by sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE). TcdA collected by ion affinity chromatography was further purified by immunoaffinity chromatography using the monoclonal antibody, BDI515. The antibody was immobilized to an agarose column (Carbolink Gel, Pierce, Rockford, IL) by coupling the carbohydrate groups in the Fc region to terminal hydrazide groups on the column matrix. Binding and elution of TcdA from the column were performed as per the manufacturer’s instructions. Purity of the toxin was determined by SDS-PAGE, using a 7.5% polyacrylamide gel and an electrophoretic current of 200 mV. The gel was stained with 0.2% Coomassie brilliant blue, R-250 (Pierce).
Activity of C. difficile toxin samples was tested by overlay on CHO cells. CHO cells were maintained in Iscove’s modified Dulbecco’s medium (IMDM; Cambrex, Walkersville, MD) with 10% fetal bovine serum (FBS; Intergen Co., Purchase, NY) and 100 mg/ml gentamycin (Sigma-Aldrich). For toxicity assays, cells were trypsinized, washed, and suspended at a concentration of 2 × 105 cells/ml; 100 ml of suspension was dispensed into each well of a 96-well tissue culture plate. Cells were incubated for 4 hours at 37°C in 5% CO2, and then serial dilutions of toxin preparations, in 100-μl aliquots, were added to each well. Cells were incubated, as before, for an additional 18 hours. The endpoint was determined as the dilution that caused 50% cytopathic effect (CPE).
Results
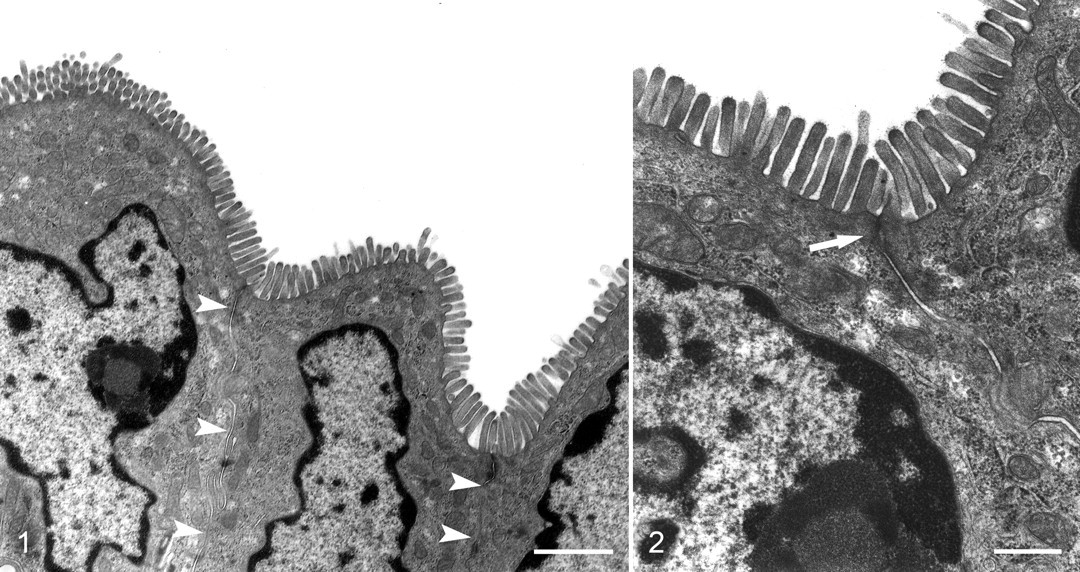
Untreated control tissues were maintained in organ culture media, at 37°C, for 2 hours with minimal development of lesions. Rare, isolated, necrotic cells were present, but the majority of the epithelial cells were normal. Microvilli were of uniform length, and cells remained tightly apposed with no obvious loss of intracellular adhesion complexes and little expansion of the potential space between cells (Figs. 1 and 2).
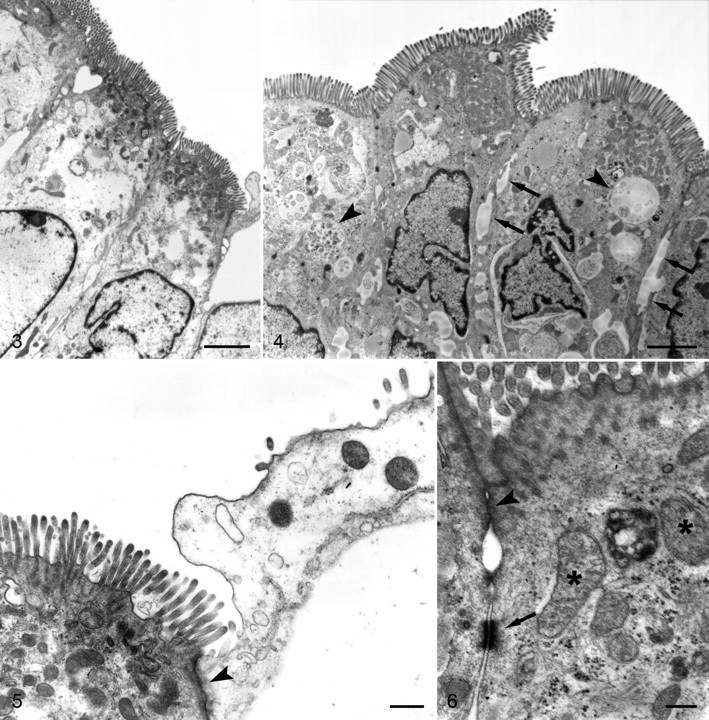
Tissues treated with as little as 0.25 μg/ml TcdA developed significant lesions in all cells. The most common changes included rarefaction of the cytoplasm, expansion of paracellular spaces, swelling of organelles, accumulation of autophagic vacuoles, and blebbing of microvilli (Figs. 3–6). Moderately affected cells had more prominent cytoplasmic rarefaction at the basal aspect of the cells; organelles in the apical cytoplasm were closely packed (Fig. 3). In most sections, the tight junctions appeared to be intact. Individual cells with more serious lesions were often interspersed among more moderately affected cells or were partially extruded from the mucosal epithelium (Fig. 5).
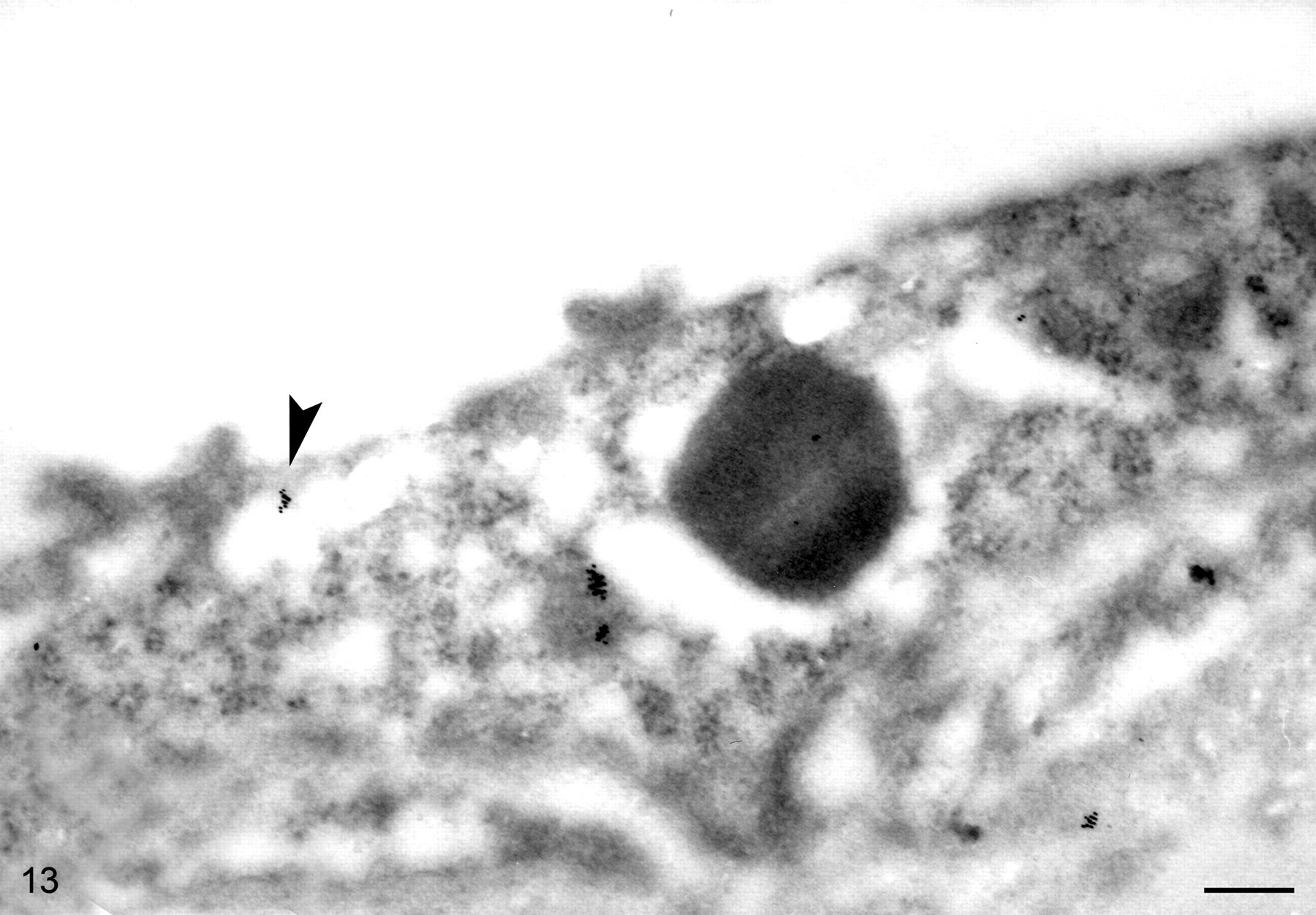
Immunoelectron microscopy of colon explants after 5-minute inoculation with 10 µg/ml TcdA. This image is of an endothelial cell with multiple aggregates of gold beads in the cytoplasm. One group of beads appears to label TcdA in a vesicle adjacent to the lumen (arrowhead). (Bar = 200 nm).
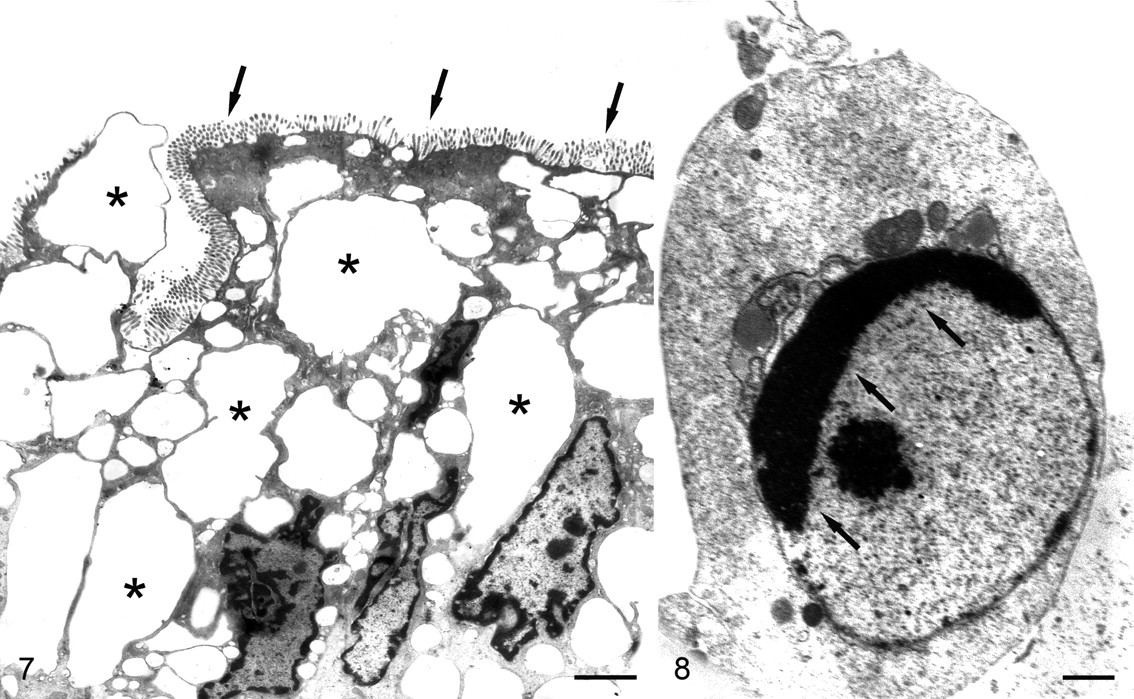
A dosage of 0.5 μg/ml TcdA was correlated with more severe swelling of organelles, a greater frequency of necrosis, and extrusion of necrotic epithelial cells at the lumenal aspect of the mucosa (Fig. 7). Tight junctions and desmosomes were still visible, even at the margins of some necrotic cells. Most cells exhibited features of coagulative necrosis, including cell swelling and karyorhexis, but some cells had features of apoptosis, such as formation of apoptosomes and fragmentation of nuclei with chromatin crescents (Fig. 8 ).
At TcdA dosages of 1 and 2.5 μg/ml, cell swelling increased in severity, affected all cells in the mucosa, and was primarily due to enormous distension of cytoplasmic vesicles. Surprisingly, microvilli often persisted on such cells, although there was abundant blebbing. Only in cells that were in advanced stages of apoptosis or necrosis were microvilli consistently lost, significantly blunted, or severely misshapen. As with low-dosage groups, extrusion of necrotic cells at the lumenal surface was common. Cells were present at various stages of necrosis or apoptosis; apoptosis appeared to be more common than in tissues at lower dosages.
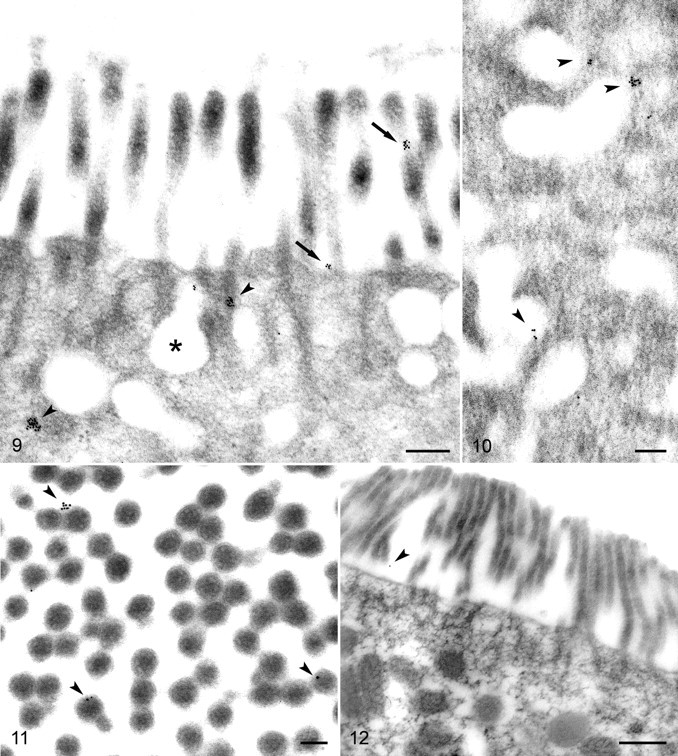
Immunoelectron microscopy specifically indicated TcdA bound to the surface of microvilli and within the cytoplasm of epithelial cells after 5 minutes (Figs. 9 and 10 ). Particles attached to microvilli specifically labeled TcdA at the plasmalemma. Transverse sections of microvilli demonstrated specific adherence of beads to the surface of the microvilli (Fig. 11 ). Colloidal gold did not label control tissues as it did the TcdA-treated samples (Fig. 12 ). Only rare beads adhered to the control tissues, and these occurred singly or occasionally in pairs, whereas beads in treated tissues most often occurred in clusters consistent with the multiple epitopes available on toxin molecules and primary antibodies.
At 5 minutes postinoculation, a small number of gold beads were visible in endosomes, but most of the intracellular particles at this time point were free in the cytoplasm. Some beads were attached at the periphery of endosomes (Fig. 10). These particles could indicate toxin molecules still attached to the membrane or recently translocated to the cytoplasm. Blood vessels in the superficial lamina propria had a moderate number of beads labeling the lumen, as well as the surfaces and cytoplasm of endothelial cells (Fig. 13 ).
There was too much variability within each section to objectively enumerate the beads attached to cells, but a subjective assessment indicated that the tissues incubated with 10 μg/ml toxin had more attached beads than tissues incubated with 5 μg/ml toxin.
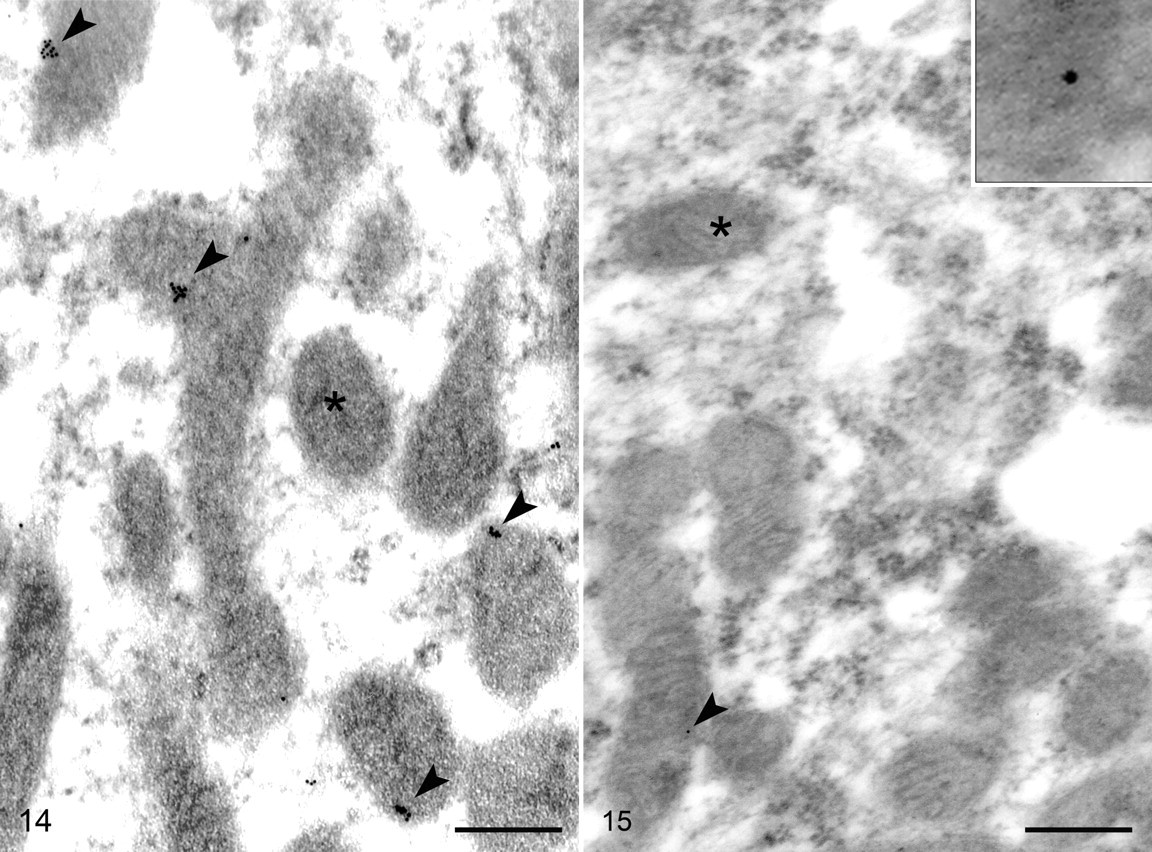
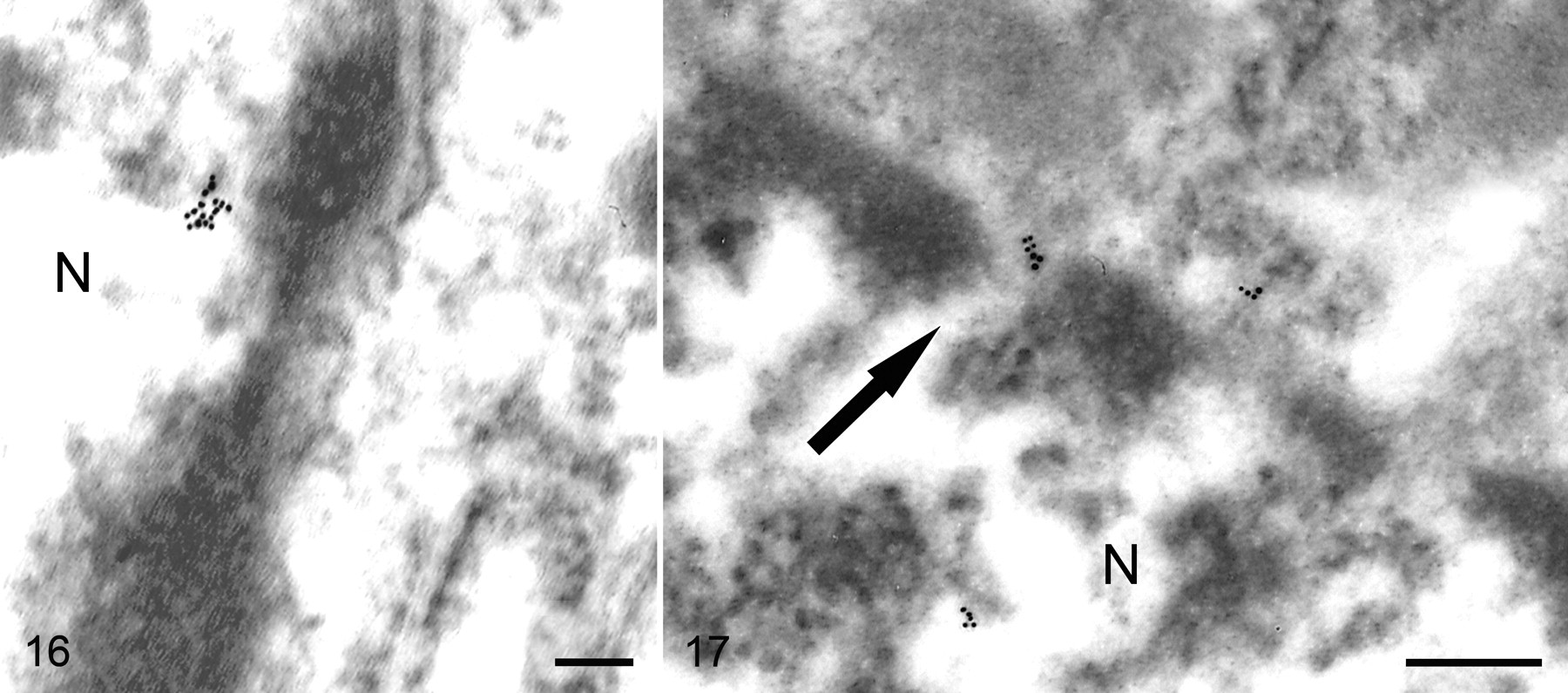
At 30 minutes postinoculation, a moderate number of beads still labeled microvilli but far fewer than at 5 minutes. The majority of the beads were intracellular, and most of those were associated with mitochondria. Stereological analysis indicated an RLI of 2.79 for the mitochondria as compared to an RLI of 0.45 for the cytoplasm, indicating a preferential association of immunogold particles for the mitochondria (P < .001). The majority of mitochondria-associated particles were attached at the margin, but occasionally they appeared in the mitochondrial matrix (Fig. 14 ). At this time point, TcdA was also identified in the nuclei of epithelial cells and other cell types in the lamina propria (Fig. 15 ), but the predilection for association of TcdA with nuclei was much lower than that for the mitochondria (RLI = 1.26; P < .1). TcdA was rarely associated with the nuclear membrane and seemed to be randomly dispersed in the interior. Rarely, beads in the vicinity of the nuclear membrane were associated with nuclear pores (Fig. 16 ). Small vessels at the 30-minute postinoculation interval were again labeled by beads localized to the cytoplasm of endothelial cells, but they also occurred in the lumen and at the basal aspect of endothelial cells (Fig. 17 ).
Discussion
C. difficile toxins cause necrosis of epithelial cells in intestinal mucosa through a complex reaction involving many host cell types and numerous intercellular mediators. 23 This report demonstrates the susceptibility of pig colon explants to TcdA and documents the associated ultrastructural lesions. The specific attachment of toxin A to epithelial cells and the endocytosis and intracellular translocation of the toxin are also described. This is the first time the intracellular localization of the toxin has been reported in intact mucosa.
In vivo studies demonstrate that glucosylation of Rho-subtype proteins by TcdA leads to disorganization of tight-junction structural proteins. 31 This perturbation of tight-junction function is thought to be the premise for altered epithelial barrier function. 14,15 The increased paracellular accumulation of fluid in tissues inoculated with TcdA in this study is consistent with permeability of the apical aspect of the mucosa. However, there was no clearly defined loss or degeneration of tight junctions, even in cells with severe lesions. Observation of the tight junctions in transverse sections might not reveal focal damage to the structures, or functional changes could occur in tight junctions that are not easily resolved by the techniques used. Ultrastructural studies of human polarized intestinal epithelial cells treated with TcdA yielded the same results described here. 15
Other ultrastructural lesions associated with TcdA exposure were severe and dose dependent. Cell swelling seen in the treated colon explants may be associated with alteration of cytoskeletal structure, as has been reported for both TcdA and TcdB. 15,39,40,47 The organization of F-actin in the apical cytoplasm is negatively affected by TcdA and may be involved in the cell swelling described here. Microfilament arrays or derangement of microfilaments were generally not evident. The only exception was the disorganization of microfilaments, associated with the terminal web, coeval with the loss or derangement of microvilli. The presence of filaments forming in the perinuclear cytoplasm and within the nucleus due to TcdA has been reported in CHO cells but was not evident in these experiments. 27 However, our fixation protocols differed from those of the earlier research, and the significance of the absence of the filaments in this study is uncertain.
The fate of cells in this study included apoptosis and necrosis, either with condensation of cell contents or, rarely, cell lysis. Both necrosis and apoptosis were most severe at higher dosages of toxin. Severity was not always uniform in the epithelial layer, and necrotic or apoptotic cells were frequently attached to less severely deranged cells. This may reflect a greater susceptibility of individual cells due to age, differentiation, metabolic activity, or a greater density of toxin receptors. Immunohistochemistry assays demonstrated significant heterogeneity among the ability of individual cells to bind toxin. 24 This greater density of bound toxin is presumably due to a greater density of receptors. As cell lesions due to C. difficile toxins are dose dependent, a greater density of TcdA receptors may contribute to the susceptibility of individual cells.
The specific binding of TcdA to the brush border of porcine enterocytes described here is consistent with reports of TcdA binding to the brush border of other species with known susceptibility to C. difficile. 25,34 At 5 minutes postinoculation of colon explants, TcdA was bound to microvilli, but some was seen in endosomes and even more was translocated to the cytoplasm. Five minutes reportedly is sufficient time for internalization of toxin in viable cells. 12,27 Many beads were also attached immediately adjacent to endosomes, and these may indicate toxin that is partially translocated. Other researchers report that TcdA internalization was due to pinocytosis and the association of bound toxin with coated pits. 27 Beads were not observed in coated pits in this study, but the fixation methods were probably not optimal for their observation.
After 30 minutes, fewer particles were adhered to microvilli, and more beads decorated intracellular structures. Most particles were localized to the margins of mitochondria, but there was insufficient resolution to determine whether the particles were adhered to the outer membrane or other structures. Rarely, beads did appear to be within mitochondria, but again, it could not be determined if they were in the intermembrane space, attached to cristal membranes, or were in the internal matrix. Using confocal microscopy and fluorescent antibody techniques, other researchers also observed internalization of TcdA and localization to mitochondria. 12 In those studies, TcdA specifically caused early mitochondrial dysfunction, including the production of reactive oxygen intermediates, decreased cellular concentration of ATP, and release of cytochrome C. Such changes occurred prior to and independent of the glucosylation of GTPases, and TcdA exerted its effects directly on isolated mitochondria.
TcdA was also detected inside nuclei, although the localization there was not preferential over other intracellular compartments. Rarely, immunogold particles were localized to nuclear pores, but the frequency of association with nuclear pores was not great enough to draw any conclusions about their involvement in entry of the toxin. Nuclear localization has been described for a number of other viral proteins and bacterial toxins. These use a variety of mechanisms such as transport vesicles derived from endocytosis, association with a host protein that has a nuclear localization sequence, or expression of a domain within the alien protein, analogous to host nuclear localization sequences. 2,32,50
Vascular changes are very important to the pathogenesis of CDAD in the host, and endothelial cells are known to be directly influenced by the effects of C. difficile toxins as well as cytokines released by local inflammatory cells. 1,17,26,30 TcdA was labeled at the basal aspect of endothelial cells, in vesicles, and in the cytoplasm, consistent with uptake of TcdA from the lamina propria and transport into endothelial cells. Aggregates of colloidal gold also indicated the presence of TcdA in the lumen of vessels. TcdA within blood vessels could have been transferred by endothelial cells or may have entered postcapillary venules or lymphatic vessels along an osmotic gradient. All blood vessels examined were located in the superficial lamina propria.
Spontaneous cases of CDAD in pigs, as well as pigs experimentally intoxicated with TcdA or TcdB, often developed systemic lesions, including hydrothorax, pleural effusion, and subcutaneous edema. 46 The fluid accumulating in body cavities was a high-protein effusion with abundant fibrin, suggesting a direct effect of C. difficile toxins on regional blood vessels (M. K. Keel, unpublished data). The general outcome is hypotensive shock. The demonstration of TcdA localizing to endothelial cells supports the hypothesis that a direct interaction is important to the pathogenesis of such lesions.
Leucocytes, including macrophages, monocytes, mast cells, and neutrophils, were also labeled by colloidal gold, indicating TcdA in the cytoplasm and in nuclei. A direct link between TcdA and leucocytes has been confirmed in multiple reports, and the interactions generally result in a proinflammatory response that exacerbates lesions and disease associated with CDAD. 4 –6,28 No molecular targets of TcdA have been identified on the surface of leucocytes. Within the cells, they presumably have the same activity as in epithelial cells, and alterations in actin filaments have been reported in neutrophils. 4 The immunoreactivity of these cells for TcdA reveals the same types of interactions seen in epithelial cells.
This report confirms the specific susceptibility of porcine colonic mucosa to TcdA. Severe intracellular lesions were associated with inoculation with TcdA, although they were generally nonspecific. Immunoelectron microscopy studies confirmed the specific binding of TcdA to the apical plasmalemma of enterocytes. As indicated by indirect assays, bound TcdA was internalized within endosomes and rapidly transferred to the cytoplasm. Within the cytoplasm, TcdA localized primarily to mitochondria. The specific association of the two is uncertain, but it is consistent with reports of mitochondria-mediated apoptosis in TcdA-treated cells. TcdA also occurred in blood vessels of the lamina propria as well as various leucocytes. These reports confirm, by ultrastructural visualization, the reports of various functional assays as to the nature of the interaction between TcdA and intestinal epithelial cells, as well as various cells of the lamina propria.
Footnotes
Acknowledgements
We would like to thank Dave Bentley for guidance with electron microscopy and imaging. Also, this work would not have been possible without the assistance of Dawn Bueschel, who provided excellent advice and help with everything in the laboratory related to C. difficile. This research was supported, in part, by a grant from USDA-CSREES-NRI.
The authors declared that they had no conflicts of interests with respect to their authorship or the publication of this article.
The authors declared that they received no financial support for their research and/or authorship of this article.
