Abstract
Clostridia-associated intestinal disease in horses was generally reported to be due to infection with Clostridium perfringens type A, which harbors the cpa-encoded α-toxin. A recent study demonstrated a high incidence of β2-toxigenic C. perfringens in horses suffering or dying from typhlocolitis, suggesting that this novel type of C. perfringens might play an important role in typhlocolitis and possibly other equine intestinal diseases. A retrospective study was conducted to assess the presence of the β2-toxin in tissues of the equine gastrointestinal tract. Monospecific polyclonal antibodies against recombinant β2-toxin were produced in rabbits and used to demonstrate the β2-toxin in sections of the gastrointestinal tract by immunohistochemical methods. Sections from 69 horses were stained and β2-toxin was observed immunohistochemically in 40 animals. Sections from the stomach, small intestine, and large intestine were positive. Immunopositivity for β2-toxin was significantly associated with presence of β2-toxigenic bacteria. This investigation demonstrates local production of β2-toxin and suggests that immunohistochemistry using antitoxin antibodies represents a useful diagnostic method in those cases where isolation of bacteria and polymerase chain reaction typing is not feasible. Although the association between the presence of β2-toxin and development of gastrointestinal disease in horses remains uncertain, the findings of this study indicate that the potential causal relationship warrants further investigation.
Introduction
Bowel disorders of various types are common in the horse and consist mainly of displacements and inflammatory diseases. 8 , 16 , 19 Chronic and acute diarrhea are usually the result of diverse causes such as infectious agents, neoplastic processes, or alimentary disease. 17 , 25 Specific etiologies for clinical signs and intestinal lesions often cannot be identified, and for this reason only descriptive terms are generally used to characterize these diseases. 24 Infectious agents known to cause enterocolitis and diarrhea in horses are most commonly bacteria such as Clostridium sp., Salmonella sp., Rhodococcus equi, and Ehrlichia risticii. 11 , 13 , 21 , 27
Clostridium perfringens and Clostridium difficile are both regularly isolated from horses with enterocolitis or typhlocolitis. 4 , 18 Because of the difficulties involved in reproducing the disease experimentally and therefore fulfilling Koch's postulates, however, the term clostridia-associated enterocolitis is frequently used. 27 Additionally, the isolation of clostridia from the intestine of a horse with diarrhea alone is not regarded as evidence that clostridia are the etiology. 27 Nevertheless, the demonstration of toxigenic strains of Clostridium sp. in animals with clinical and pathologic changes is suggestive of a causal association.
C. perfringens strains are grouped according to the specific toxins they produce, and these toxin profiles are associated with different types of disease and hosts. In foals with clostridia-associated enterocolitis, infections with C. perfringens type A and type C have been reported. C. perfringens type A produces α-toxin (a phospholipase C), and type C produces the α- and ß-toxin (a cytotoxin). 22 One study showed that foals with C. perfringens type A–positive intestinal culture have a better prognosis than foals with a type C-positive culture, indicating that severe intestinal disorders due to C. perfringens are probably not caused by the α-toxin alone. 4 A novel toxin produced by C. perfringens has recently been identified. 9 This protein has functional but not structural similarity to the previously known beta (ß)-toxin and has been designated β2-toxin; it has been shown to be cytotoxic and lethal in mice. 5 , 9 C. perfringens strains harboring the genes cpa, encoding the α-toxin, and cpb2, encoding the β2-toxin, were isolated from the intestines of horses with enterocolitis. Therefore, a causal relationship of β2-toxigenic C. perfringens with these diseases has been postulated. 12
The existence of toxin genes does not necessarily imply production of the toxins themselves. In order to demonstrate the local production of the β2-toxin in the tissue of horses from which β2-toxigenic C. perfringens was isolated, gastrointestinal tissues from 69 horses were stained immunohistochemically for the presence of β2-toxin.
Materials and Methods
Animals
Gastrointestinal sections from 69 horses were evaluated immunohistochemically for clostridial β2-toxin. This retrospective study included horses that had been submitted for postmortem examination to the institute during 1997 through 2002 (horse Nos. 1–7, 9–56, 68, 69) and slaughtered horses (horse Nos. 8, 57–67). All necropsy cases from which histologic samples were collected and intestinal content was taken at the time of necropsy and cultured for the presence of pathogenic bacteria were included. Two horses (horse Nos. 34 and 35) were included in the immunohistochemical study because they originated from the same farm as horse No. 4; and all three horses died within a week. Information regarding age, sex, breed, clinical signs, postmortem interval, and gross findings was obtained from records. Most horses were adults (n = 51) or juvenile (n = 11), and only seven horses were less than 6 months old. The sex distribution was 38 females and 31 males. Horses belonged to the common breeds present in Switzerland, namely Franche Montagne (also called Freiberger horses, 18), Swiss Warmblood (17), ponies (8), and Selle Française (7). The remainder represented various breeds, including Holstein, Andalusian, Friesian, Hannovarian, or thoroughbred. For seven horses, no specific breed was recorded. Most horses were euthanatized because of clinical signs compatible with gastrointestinal disease including colic and/or diarrhea with poor prognosis. Six horses had been subjected to a laparotomy in order to resolve a small intestinal volvulus or a colonic torsion (horse Nos. 9, 11, 19, 33, 49, and 52). On three occasions, horses (n = 7) originating from three pastures were found dead without prior clinical signs and were delivered to the Department to exclude malicious poisoning or accidental intoxication (horse Nos. 4, 34, and 35, horse Nos. 15 and 16, horse Nos. 18 and 37). Five horses were killed for other reasons and had undergone a scintigraphy (horse Nos. 26, 28, and 55), humerus fracture (horse No. 27), or splenic malignancy (horse No. 56). The postmortem interval varied between 1 hour and occasionally more than 24 hours. All horses were necropsied, and reported gross findings were variable, ranging from no significant macroscopic changes to massive congestion, edema, and hemorrhage of various sections of the intestinal tract. Edema, often associated with an increased redness, was the most commonly reported gross change and occurred in various parts of the gastrointestinal tract. The presence of liquid contents throughout the intestinal tract without grossly recognizable changes of the intestinal walls was found in some horses. The reports of histologic examination of hematoxylin and eosin–stained sections ranged from no significant microscopic changes to massive necrosis. In three horses (horse Nos. 12, 17, and 24), tissues were autolyzed. The samples from the slaughtered horses were collected within 30 minutes after death (horse Nos. 8 and 57–67), and no changes were reported; histologically, the tissues from these horses were unremarkable.
Bacterial culture and Polymerase Chain Reaction (PCR) analysis
The intestinal contents of horses (all except for horse Nos. 34 and 35) were tested for the presence of pathogenic bacteria. Contents of the jejunum/ileum and colon were inoculated on 5% blood agar plates and incubated overnight at 37 C under anaerobic conditions. Colonies with zones of double hemolysis were considered to be C. perfringens, and their identity was confirmed by PCR using the α-toxin gene (see below). Gastric contents of the control slaughter horses were also analyzed for the presence of C. perfringens. To further type the C. perfringens isolates by their toxin genes, PCR was employed as previously described, using several colonies of each primary culture. Samples were tested for presence of the cpa, cpb, cpb2, etx, and cdtA genes, which encode the α-, β-, β2-, ∊- and ι-toxins, respectively. 2 , 3 , 12
Immunohistochemistry
Immunohistochemistry was used for the demonstration of clostridial β2-toxin in specific tissues. Monospecific polyclonal anti-β2-toxin antibodies were obtained by immunization of rabbits with purified detoxified recombinant β2-toxin. Recombinant β2-toxin was produced in Escherichia coli BL21 (DE3) harboring plasmid pJEFbeta2M1 containing the coding part of the cpb2 gene in expression vector pETHIS-1. The recombinant β2-toxin fusion protein containing N′- and C′-terminal polyhistidine tails was subsequently purified by Ni2+ chelate affinity chromatography as described earlier for other bacterial toxins. 23 The specificity and sensitivity of the antibody was tested by western blotting using equine feces from which β2-toxigenic bacteria were isolated. Immunohistochemistry was conducted as described. 1 In short, sections from all paraffin blocks containing stomach or intestines (usually two to four blocks per horse) were deparaffinized, and the rabbit anti-β2-toxin antibody was used at a final dilution of 1:1,000 overnight at 4 C. A positive reaction was visualized with 3-amino-9-ethylcarbazole (AEC, DAKO, Zug, Switzerland), which produces a reddish-brown stain after application of the secondary biotinylated anti-rabbit antibody (DAKO). Additional controls for immunohistochemistry consisted of intestines from goats and dogs with enteric disease due to C. perfringens. The goat isolates had been analyzed by PCR and were positive for the α-toxin and ∊-toxin genes but negative for the β2-toxin gene.
Statistics
The correlation between immunohistochemistry and growth of C. perfringens and immunohistochemistry and PCR results was tested using Fisher's Exact Test (InStat version 3, GraphPad Software, San Diego, CA).
Results
This retrospective study included 69 horses. The immunohistochemical evidence for the presence of β2-toxin and the results of bacterial culture and PCR analysis for the C. perfringens β2-toxin gene are listed in Table 1. Distributions of age, sex, breed, clinical signs, and postmortem interval were similar among the horses submitted to the Department for necropsy and which were either positive or negative for β2-toxin by immunohistochemistry.
Results of immunohistochemistry (IHC) for β2-toxin in 69 horses, bacterial culture and PCR for the presence of the β2-toxin gene, cpb2, in C. perfringens isolates.∗
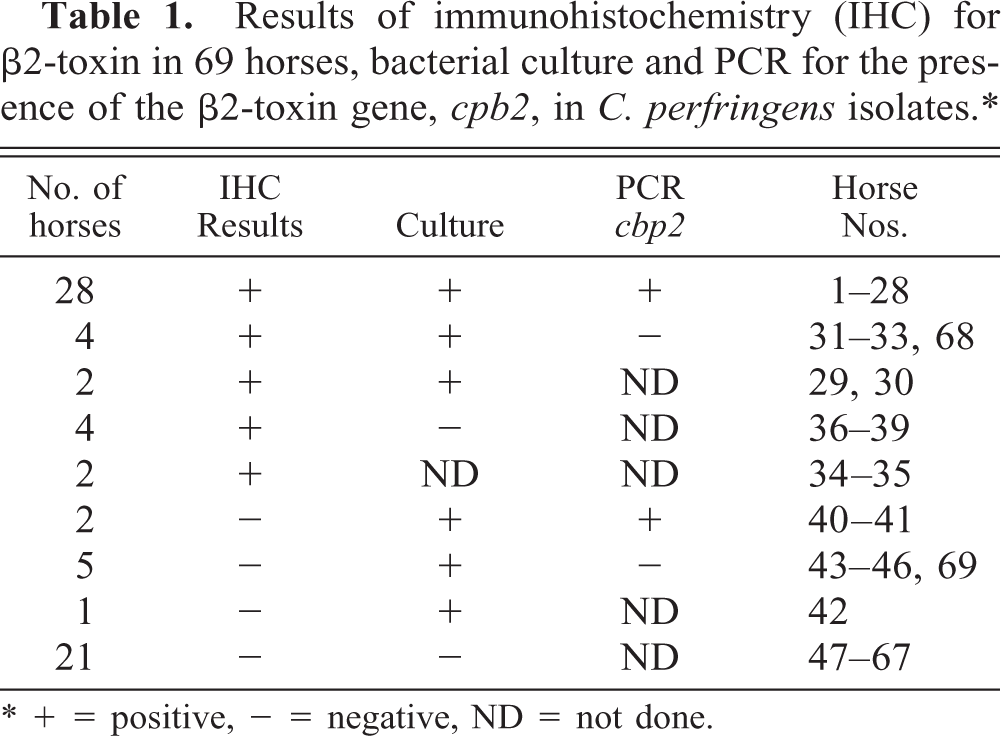
∗ + = positive, - = negative, ND = not done.
Bacterial culture and PCR
C. perfringens was found in the intestine of 42 horses (63%, Table 1); in two cases bacteriologic culture was not performed. In 25 of the horses C. perfringens was not found by standard bacteriology. Thirty-nine of the C. perfringens isolates were analyzed by PCR for the presence of toxin genes. The gene cpa, encoding the α-toxin, was detected in all samples, confirming the species C. perfringens in these isolates. The cpb gene, encoding the ß-toxin, was not found in any of these samples. Furthermore, none of eight randomly chosen C. perfringens isolates harbored either the etx (∊-toxin) or the cdtA (ι-toxin) gene. In contrast, the cpb2 gene, encoding the α-toxin, was detected in 30 of the 39 intestinal samples (Table 1). Five horses for which a culture of the intestinal contents revealed growth of C. perfringens also had a positive result for C. difficile. C. difficile was detected in intestinal contents of 7 horses in which no C. perfringens was found. From one horse, Salmonella enteridis was isolated, and in three horses, Actinobacillus equuli was found in parenchymal organs (liver, kidney, and/or spleen), indicating bacterial sepsis.
Immunohistochemistry
A total of 246 different sections (usually two to five from each horse) were examined for the presence of β2-toxin by immunohistochemistry. Immunohistochemical evidence for the β2-toxin was provided by a reddish-brown stain, often around bacteria (Figs. 1, 2). At least one of the sections of the gastrointestinal tract examined was positive for the β2-toxin in 40 horses. Immunohistochemical evidence for the β2-toxin was generally found in several sections from the same tissue. Stomach sections (pars glandularis) were positive in 24% of the cases examined (8/33). The small intestine was positive for the β2-toxin by immunohistochemical staining in 48% (56/115) and the large intestine in 35% (35/98) of the cases. The caprine and canine samples were negative for the β2-toxin.
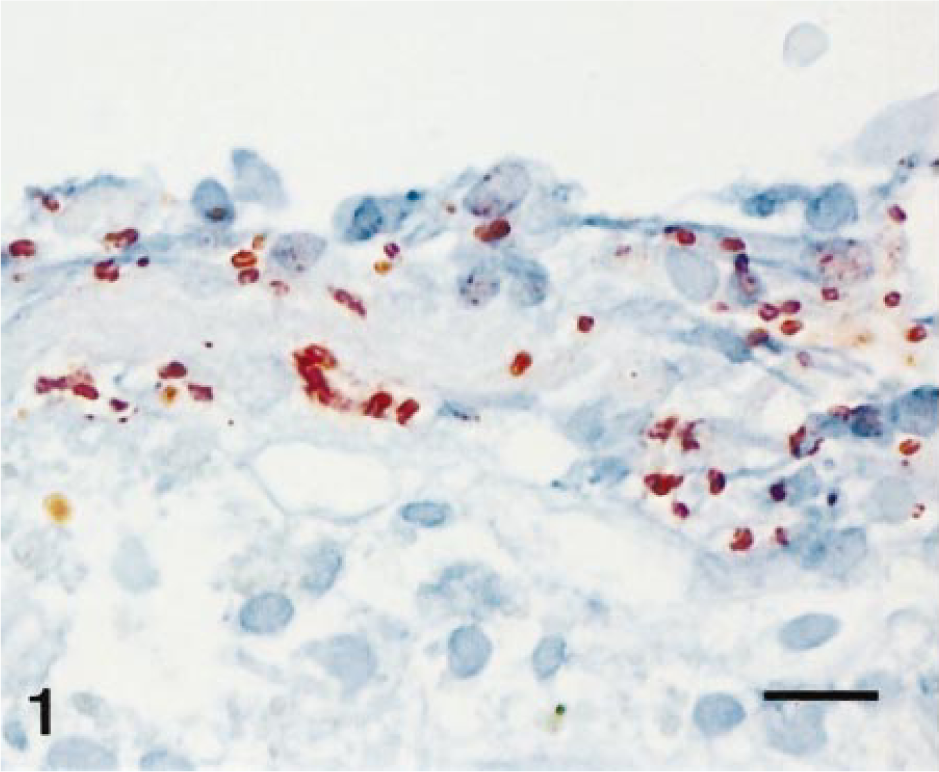
Large intestine with focus positive for β2-toxin (ABC-immunohistochemistry for β2-toxin); horse No. 13. Bar = 30 µm.
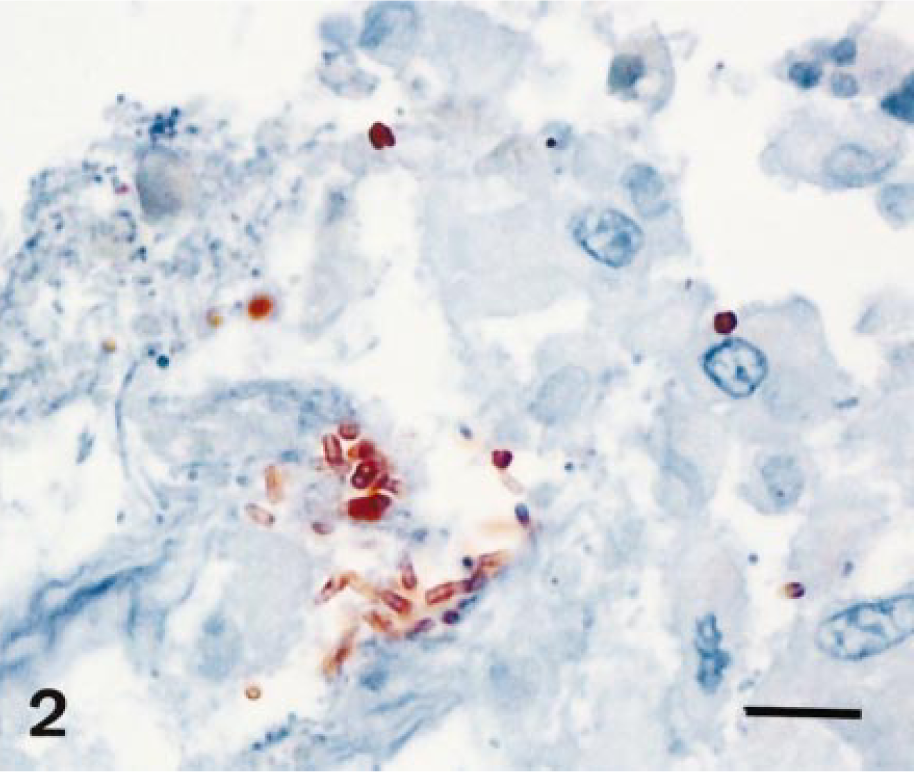
Small intestine with focus positive for β2-toxin (ABC-immunohistochemistry for β2-toxin); horse No. 16. Bar = 25 µm
The β2-toxin was demonstrated in a total of 40 horses; these included 28 horses (70%) from which β2-toxigenic bacteria were isolated and typed by PCR. Additionally, four horses (10%) were positive for the β2-toxin, with no β2-toxigenic bacteria being typed by PCR, as were two (5%) horses with a culture positive for C. perfringens that were not typed. Four horses (10%) from which no C. perfringens was isolated and two horses (5%) from which no cultures were taken were positive for β2-toxin by immunohistochemistry.
The Beta2-toxin was not demonstrated immunohistochemically in 29 horses. Among these were two horses (7%) from which β2-toxigenic C. perfringens was isolated and five horses (17%) with C. perfringens type A (no β2-toxin gene). One horse (3%) with a culture positive for C. perfringens, which was not further typed, was also negative for the β2-toxin. Finally, 21 horses (73%), from which no C. perfringens was isolated were also negative for the β2-toxin by immunohistochemistry. This group included 10 of the 11 slaughtered horses.
There were statistically significant correlations between immunohistochemistry and growth of C. perfringens (P < 0.0001) and immunohistochemistry and detection of β2-toxigenic C. perfringens (P = 0.0037).
Discussion
C. perfringens infection is associated with gastrointestinal disease in the horse, and to date α-toxin- and β-toxin-producing strains type A and C were isolated from such cases. 4 , 27 C. perfringens strains containing the newly described β2-toxin gene were identified in horses with typhlocolitis by bacterial culture and PCR-based toxin gene-typing of C. perfringens and were correlated with the disease. 12 The current study was conducted to assess the presence of the β2-toxin and its localization in the digestive tract of horses. This retrospective study demonstrated the frequent presence of the β2-toxin in intestinal tissue sections in association with the presence of β2-toxigenic C. perfringens strains.
In most horses from which β2-toxigenic C. perfringens was isolated, the β2-toxin could be observed immunohistochemically within the tissues; this finding indicates local production of the toxin. To date, only the presence of β2-toxigenic C. perfringens has been detected, whereas local production of the toxin has only been assumed. 12 The results of our study seem to indicate that toxin production occurs in the majority of horses from which β2-toxigenic C. perfringens has been cultured from the gastrointestinal tract. This finding implies that immunohistochemistry using anti-β2-antibodies represents a possible diagnostic alternative in those cases in which bacterial culture and PCR typing are not feasible.
C. perfringens strains harboring the β2-toxin gene, cpb2, have been considered to play an important role in the development of typhlocolitis in horses. 12 Clostridial infections are also suspected of playing a role in hemorrhagic fibrinonecrotic duodenitis–proximal jejunitis, also known as anterior enteritis. 7 In our study, the percentage of small intestinal samples that showed a immunohistochemical reaction for the β2-toxin was even higher than the percentage of large intestinal samples. This finding suggests that involvement of the β2-toxin in small intestinal disease is also be possible. However, the causal relationship between the presence of the β2-toxin and lesions in the small or large intestine is still uncertain. The β2-toxin may have had a causal or contributory role in the pathogenesis of gastrointestinal disease in these cases; such a role has been described in guinea pig intestine under experimental conditions. 9 Alternatively, progressive gastrointestinal disease in the horse may change the microbial environment, thus potentially favoring the growth of β2-toxigenic C. perfringens and subsequent toxin production. Vascular compromise, as occurs in intestinal torsion, might also facilitate proliferation of anaerobic bacteria. A scenario wherein a pre-existing condition enhances the growth of β2-toxigenic C. perfringens with subsequent toxin production is also conceivable, possibly resulting in complications and death subsequently. Results from another study implicated gentamicin as a possible inducer of β2-toxin production, and it is conceivable that additional β2-toxin in an already compromised animal can lead to fatal outcomes. 12
The presence of the β2-toxin in gastric sections was surprising. An agonal reflux from the small intestine in these cases was considered to be unlikely because the toxin was located deep in the mucosa and not just superficially, thus suggesting local production.
C. perfringens was rarely isolated in slaughter horses. This finding parallels previous studies that reported that healthy horses do not harbor C. perfringens in their gastrointestinal tract in detectable amounts or if they do, they do so only in very low numbers. 6 , 14 Similar findings are reported from another study in which C. perfringens was isolated less frequently from healthy foals than from foals with diarrhea. 20 Furthermore, C. perfringens isolated from healthy horses was mainly of type A, containing solely the cpa (α-toxin) gene, indicating that pathogenic equine C. perfringens contained additional toxins. 14 Possibly, some cases of clostridia-associated diseases in horses assumed to be caused by type A strains were associated with β2-toxigenic strains. Such an assumption is supported by observations in other species. In a group of over 100 lambs from different farms with clostridial dysentery, all contained C. perfringens harboring the α-toxin gene cpa and at least one additional toxin gene. 10 In diarrheic piglets, isolates of C. perfringens harboring only the α-toxin gene were relatively rare compared with isolates of C. perfringens harboring the α-toxin gene and one of the genes encoding the ß- or β2-toxin. 15 A prevalence of β2-toxigenic clostridia was also found in dogs with diarrhea, demonstrating the significance of this gene. 26 Additionally, plasmid-encoded toxin genes like the β2-gene may be lost by subculturing, which could lead to underestimation of its presence in enteric diseases. 5 , 9
Beta2-toxigenic C. perfringens is implicated in gastrointestinal disease in various animal species, but so far only the presence of the toxin gene has been demonstrated, and its role in the pathogenesis of the disease is speculative. 12 , 15 , 26 As our report demonstrates, further experimental studies under controllable conditions are needed to substantiate and define the correlation between gastrointestinal disease and β2-toxin production by ß2-toxigenic C. perfringens. Immunohistochemistry of the gastrointestinal tract for the toxin represents a potential method to investigate the association between the toxin and intestinal lesions and will facilitate such experimental studies.
Footnotes
Acknowledgements
We thank M. Bozzo, E. Garchi, M. Krawinkler, E. Rohrer, Y. Schlatter, and C. Suter for excellent technical assistance. We extend special thanks to Dr. Edward Stevens for a critical review of the manuscript.
