Abstract
An eosinophilic substance (ES) is usually observed in the mouse nasal septum and increases in volume with aging. It has been described as amyloid in textbooks and one report. However, it has been described as “not amyloid” in other reports because there was a negative reaction to Congo red. In this study, the ES was investigated histopathologically and electron microscopically to determine whether it was amyloid or not. The ES was only observed at the interstitium of clear HE-stained nasal glands in the septum, in which 2 kinds of glands were present (dark and clear stained by HE). The volume of the ES was small in young mice and large in older ones. Neither nasal gland degeneration nor inflammation resulted, even if a large amount of the ES was observed. The ES reacted negatively to Congo red but was strongly positive to periodic acid–Schiff reaction with prior diastase treatment. In the electron microscope observation, the ES consisted of amorphous material and collagen, but no nonbranching fibrils. Similar amorphous material was also observed in the nasal gland epithelial cells and was connected to the material in the rough endoplasmic reticulum. The above-mentioned findings indicated that the ES was not amyloid and suggested the ES might consist of not only collagen but also complex carbohydrate, which was produced by the nasal gland epithelial cells.
Amyloid stains as an amorphous, eosinophilic, and hyaline substance with HE, and it is characterized by extracellular deposition in various tissues and organs. 1, 6 Congo red–stained amyloid has a pink or red color, according to the light microscopic examination. Ultrastructurally, amyloid consists of nonbranching fibrils of indeterminate length with a diameter of approximately 7.5–10 nm.
Amyloid deposition is a frequently encountered finding in aged mice of many strains used for toxicologic studies and is sometimes found systemically. 6 Two naturally occurring types of amyloid have been identified in mice, namely AA and AapoAII. 3, 6 AA amyloid is considered secondary or reactive, and AApoAII amyloid, senile. It is known that amyloid deposition and organ predilection in mice are strain-dependent.
In mice, the eosinophilic substance (ES) is observed in the nasal septum and increases in volume with aging. ES has been described as amyloid in textbooks and one report. 2, 5, 7 ES, however, occurs in almost all aged mice and is independent of amyloid deposition at other sites. 2, 7 In addition, there are other reports that ES is not amyloid because it reacts negatively with Congo red. 4, 8 In these reports, it was also reported that ES was strongly positive by periodic acid–Schiff reaction and stained pale blue with Masson trichrome. However, a detailed investigation including an ultrastructural examination was not done. The purpose of this report was to determine whether the nasal ES is amyloid or not. For that purpose, ES was investigated histopathologically and electron microscopically.
Materials and Methods
Jic:CB6F1-Tg rasH2 mice and Jic:CB6F1-nonTg rasH2 mice, purchased from CLEA Japan Inc. (Shizuoka, Japan), and B6C3F1/Crlj mice, purchased from Charles River Laboratories Japan Inc. (Kanagawa, Japan), were used in this investigation. They were control animals used in toxicity studies. Their microbial levels were specific pathogen–free. They were housed in cages in animal rooms that were maintained at a temperature of 22 ± 3°C, a humidity of 55 ± 20%, with 6–20 air changes per hour and a 12-hour light and 12-hour dark cycle. They were given a commercial diet (CE-2, CLEA Japan Inc.) and tap water ad libitum. Their health status was normal during the observation period. The antibody tests for Clostridium piliforme, Ectromelia virus, lymphocytic choriomeningitis virus, mouse hepatitis virus, Mycoplasma pulmonis, and Sendai virus were negative in the monitor mice, which were housed in the same rooms as the investigated mice. Antibody tests for some mouse pathogens like pneumonia virus of mice, mouse minute virus, mouse parvovirus, and Helicobacter spp. were not done.
Thirty-three-week-old Jic:CB6F1-Tg rasH2 mice (73 males and 71 females) and Jic:CB6F1-nonTg rasH2 mice (25 males and 25 females) were sacrificed by exsanguination under ether anesthesia. Tissues from the whole body were removed, fixed in 10% neutral phosphate-buffered formalin, embedded in paraffin, sectioned, and stained with HE for microscopic examination (Table 1). The nasal tissue was decalcified with 10% formic acid formalin for 10 days before processing and was examined at levels I (taken immediately posterior to the upper incisor teeth), II (taken through the level of the incisive papilla), and III (taken through the middle of the second molar teeth). 5
The number of Jic:CB6F1-Tg rasH2, Jic:CB6F1-nonTg rasH2, and B6C3F1/Crlj mice examined in this study.

∗– not applicable.
†Histopathologic examination of the organs/tissues from the whole body.
‡Investigation for the relation between eosinophilic substance and aging, and for the property of several special stains.
§Electron microscopic examination.
Eleven-, 15-, and 33-week-old Jic:CB6F1-Tg rasH2 mice (3 males each) were used in the investigation for the relation between ES and aging and for the properties of several special stains (Table 1). After exsanguination of the mice under ether anesthesia, the nasal tissues were removed, fixed, sectioned according to the above methods, and stained with HE, Congo red, periodic acid–Schiff reaction with prior diastase treatment, Masson trichrome, reticulin silver impregnation, toluidine blue, and Alcian blue (pH 1.0 and 2.5).
For electron microscope observation, 6- and 42-week-old B6C3F1/Crlj mice (2 females each) were used (Table 1). After exsanguination of the mice under ether anesthesia, small pieces of the nasal septum in levels I–II were fixed in 2.5% glutaraldehyde, postfixed in 1% osmium tetroxide, and embedded in epoxy resin. Ultrathin sections were stained with uranyl acetate and lead citrate. The samples were observed under an electron microscope (H-7600, Hitachi, Tokyo, Japan).
The animals were cared for according to the principles outlined in the guide for the care and use of laboratory animals prepared by the Japanese Association for Laboratory Animal Science and our institution.
Results
The ES was observed in the dorsal portion to the vomeronasal organ of the nasal septum between levels I and II in all 33-week-old Jic:CB6F1-Tg rasH2 mice and Jic:CB6F1-nonTg rasH2 mice (Fig. 1a). Two kinds of nasal glands were in the nasal septum in all mice (Fig. 1b). One of them stained clearly with HE at the ventral area only. The other one stained dark with HE in the nasal lateral area as well as the septum and was observed in the upper portion of the septum. ES was only observed at the interstitium of the clear HE-stained nasal glands (Fig. 1b). It was confirmed that there were no amyloid depositions in other organs or tissues of any of the mice examined.
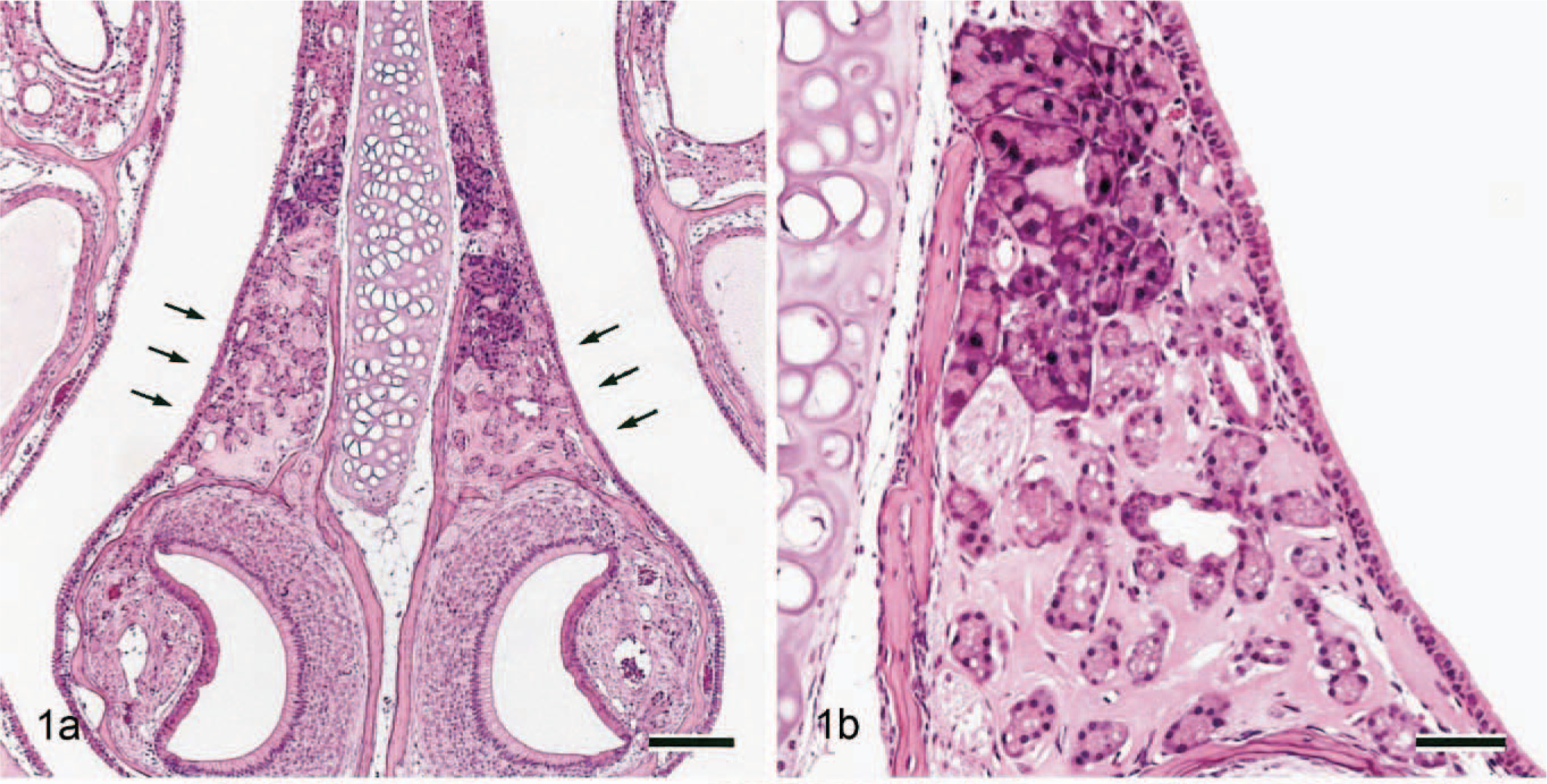
Nasal cavity (level I); 33-week-old Jic:CB6F1-Tg rasH2 mouse.
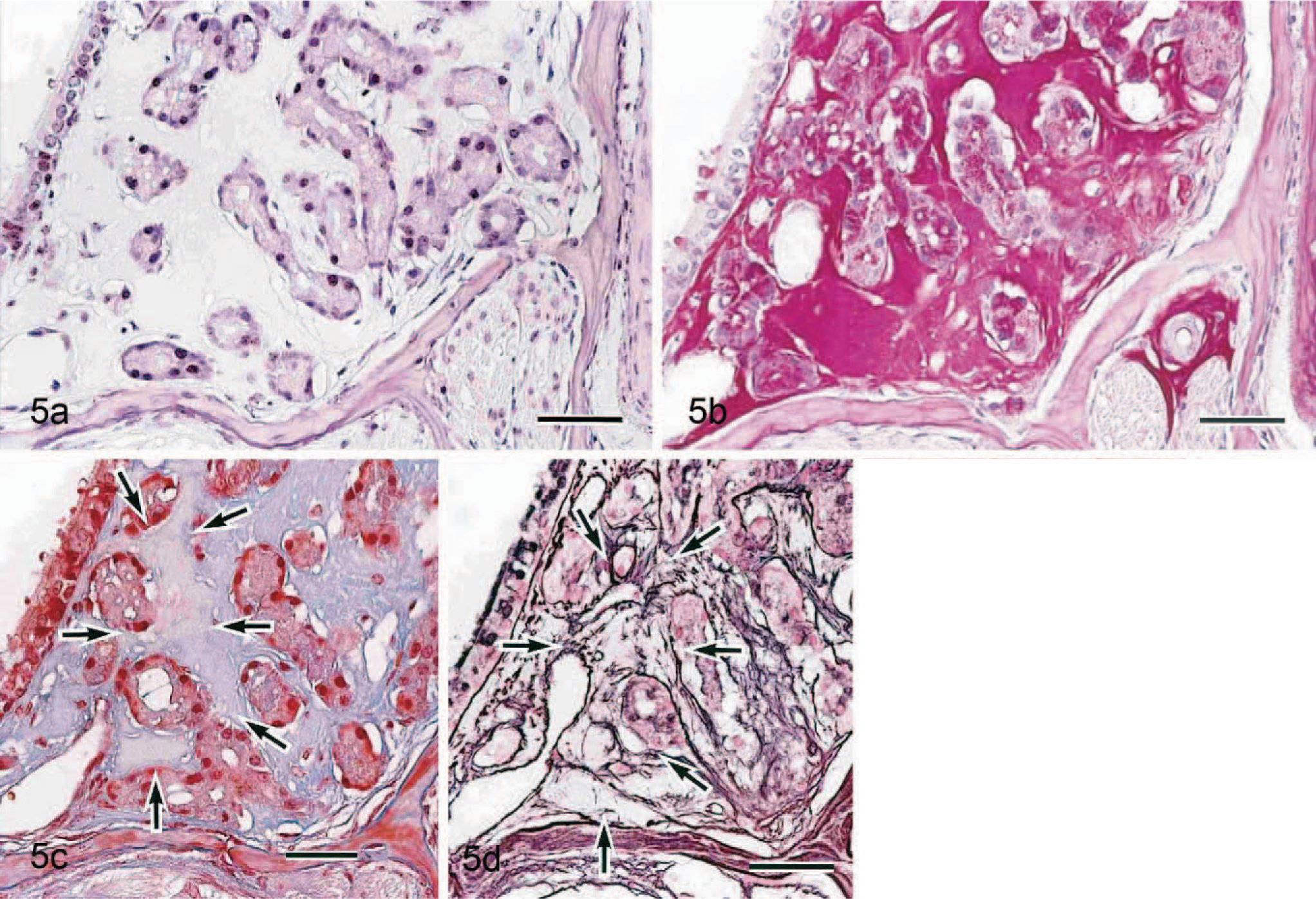
The ES volume was very small in 11-week-old mice but increases with age (Figs. 2–4). Neither nasal gland degeneration nor inflammation was caused even if a large amount of ES was observed, although a slight separation of the nasal glands and slight expansion of the septum width were observed. The 11-week-old mice also had clear HE-stained nasal glands. ES reacted negatively with Congo red (Fig. 5a), toluidine blue, or Alcian blue (pH 1.0 and 2.5) but was strongly positive with periodic acid–Schiff reaction with prior diastase treatment (Fig. 5b). It stained pale blue but also included some pale red areas with Masson trichrome (Fig. 5c). In the pale red areas with Masson trichrome, reticulin or collagen fibers were sparse according to reticulin silver impregnation (Fig. 5d). No differences were found on the properties with the special stains between 11- and 33-week-old mice. Strongly periodic acid–Schiff reaction-positive granules were observed in the clear HE-stained nasal gland epithelial cells (Figs. 5b, 6). Just like ES, they reacted negatively with Congo red (Fig 5a), toluidine blue, or Alcian blue. The dark HE-stained nasal gland epithelial cells also had periodic acid–Schiff-positive granules, but the reaction was much weaker (Fig. 6).
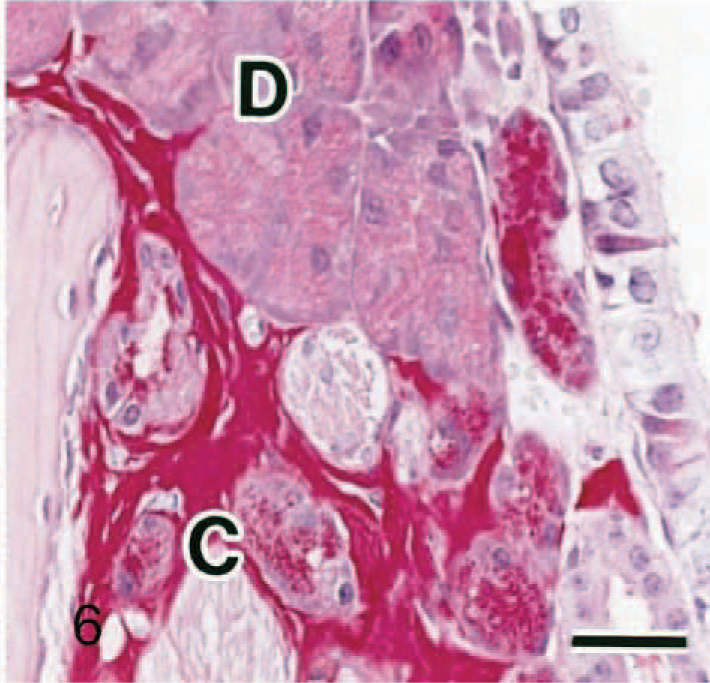
Nasal cavity (level I); 33-week-old Jic:CB6F1-Tg rasH2 mouse. Periodic acid–Schiff reaction with prior diastase treatment. Bar = 25 μm. Strongly positive granules are observed in the clear HE-stained nasal gland epithelial cells (C), just like eosinophilic substance. Note dark HE-stained nasal gland epithelial cells (D).
Nasal cavity (level I); 33-week-old Jic:CB6F1-Tg rasH2 mouse. Special stains. Bar = 40 μm.
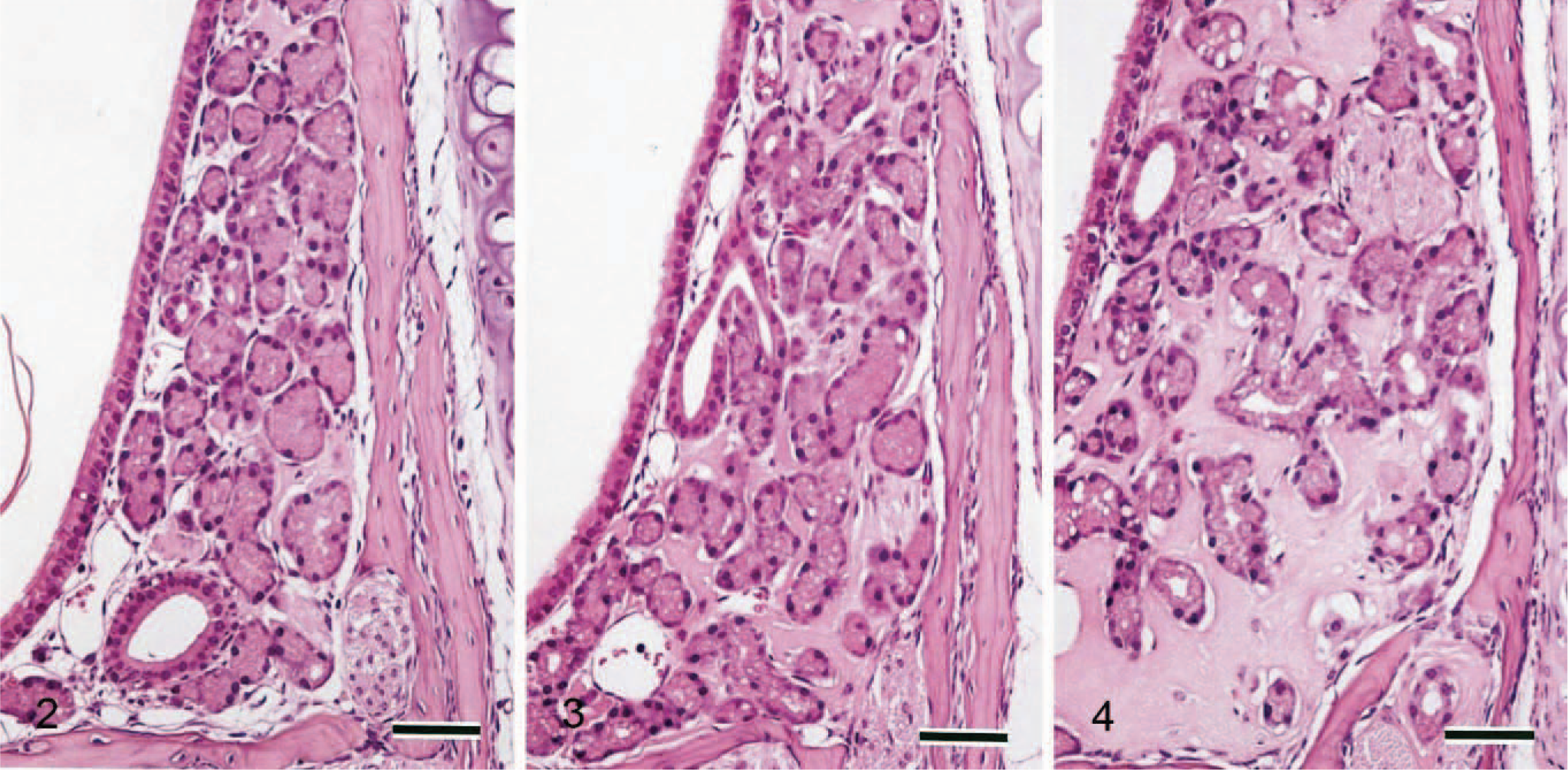
Nasal cavity (level I); 11-(
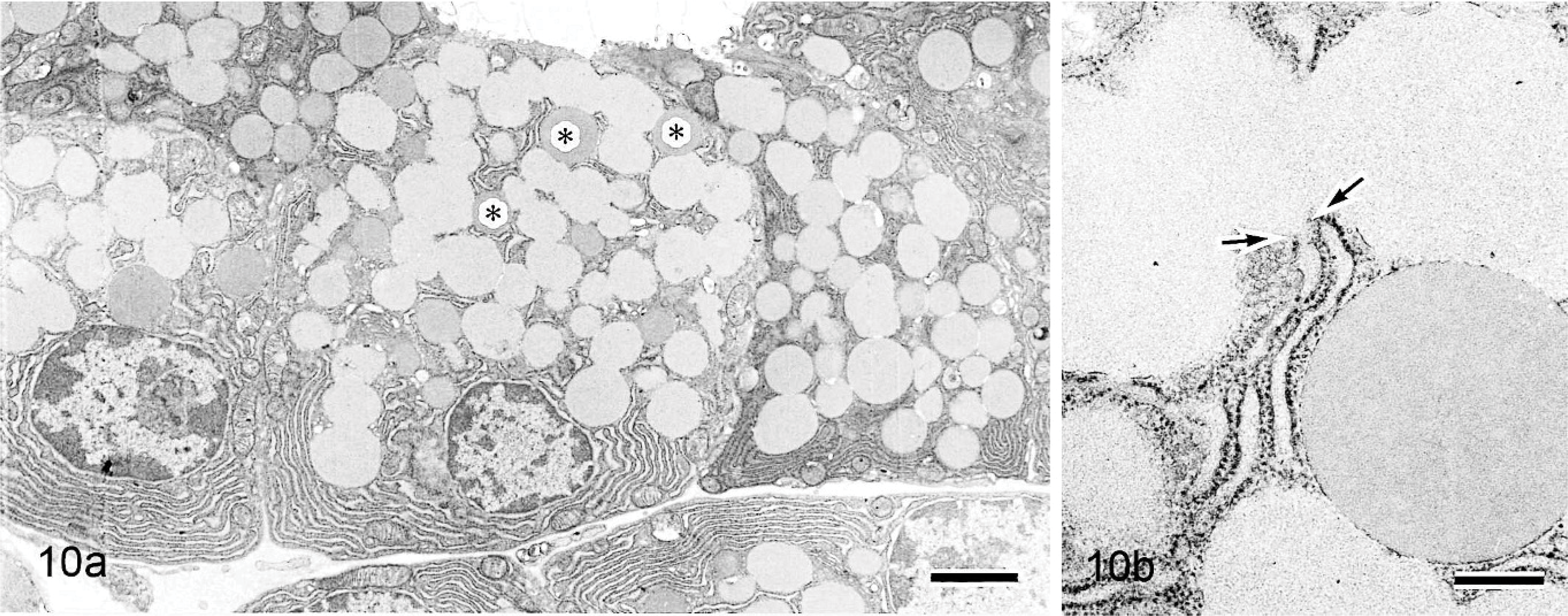
Electron microscopically, large amounts of amorphous material and collagen fiber were observed at the interstitium in 42-week-old mice (Fig. 7), but no nonbranching fibrils were present there (Fig. 8). Similar amorphous material was also present in the nasal gland epithelial cells (Fig. 7) but not in the interstitial mesenchymal cells such as fibroblasts. The 6-week-old mice had areas in which the amorphous material was present only in the clear HE-stained nasal gland epithelial cells but not at the nearby interstitium (Figs. 9a, 10a). The amorphous material was very similar and connected to the material in the rough endoplasmic reticulum of the nasal gland epithelial cells (Figs. 9b, 10b).
Nasal cavity (level I); 6-week-old B6C3F1/Crlj mouse. Electron micrographs.
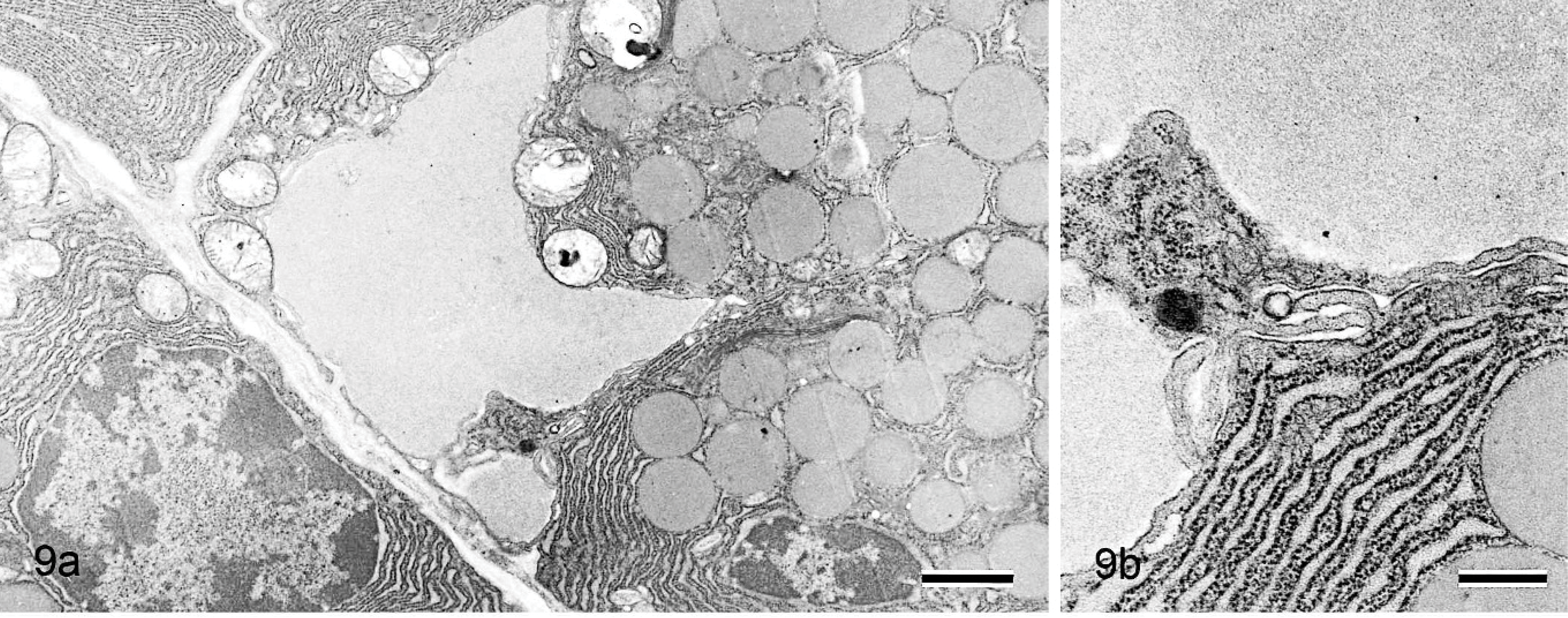
Nasal cavity (level I); 6-week-old B6C3F1/Crlj mouse. Electron micrographs.
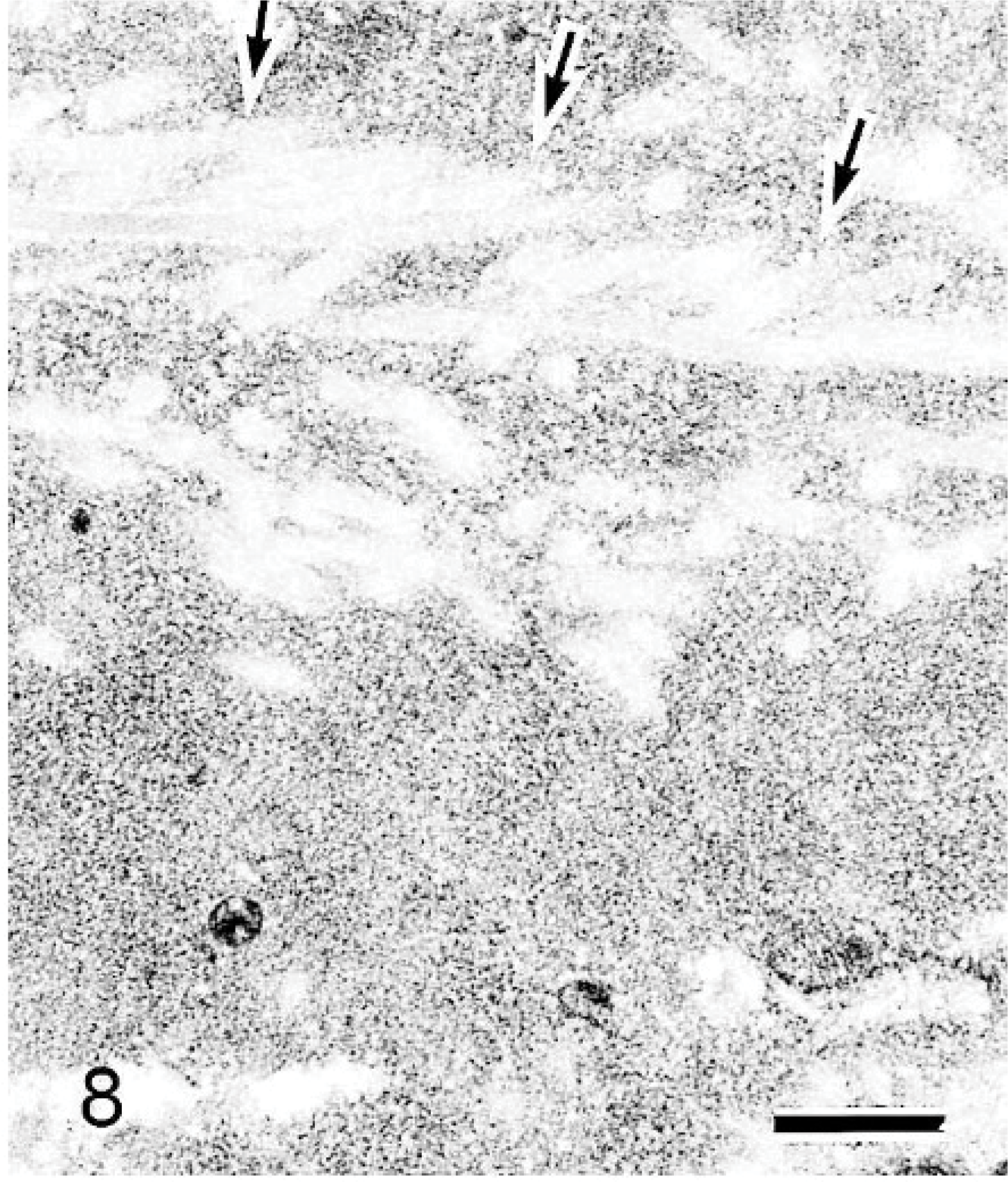
Nasal cavity (level I); 42-week-old B6C3F1/Crlj mouse. Electron micrographs. No nonbranching fibrils are present in amorphous material. Collagen fibers are also present. The collagen fibers stained faintly and are observed as clear spaces indicated by the arrows. Bar = 0.4 μm.
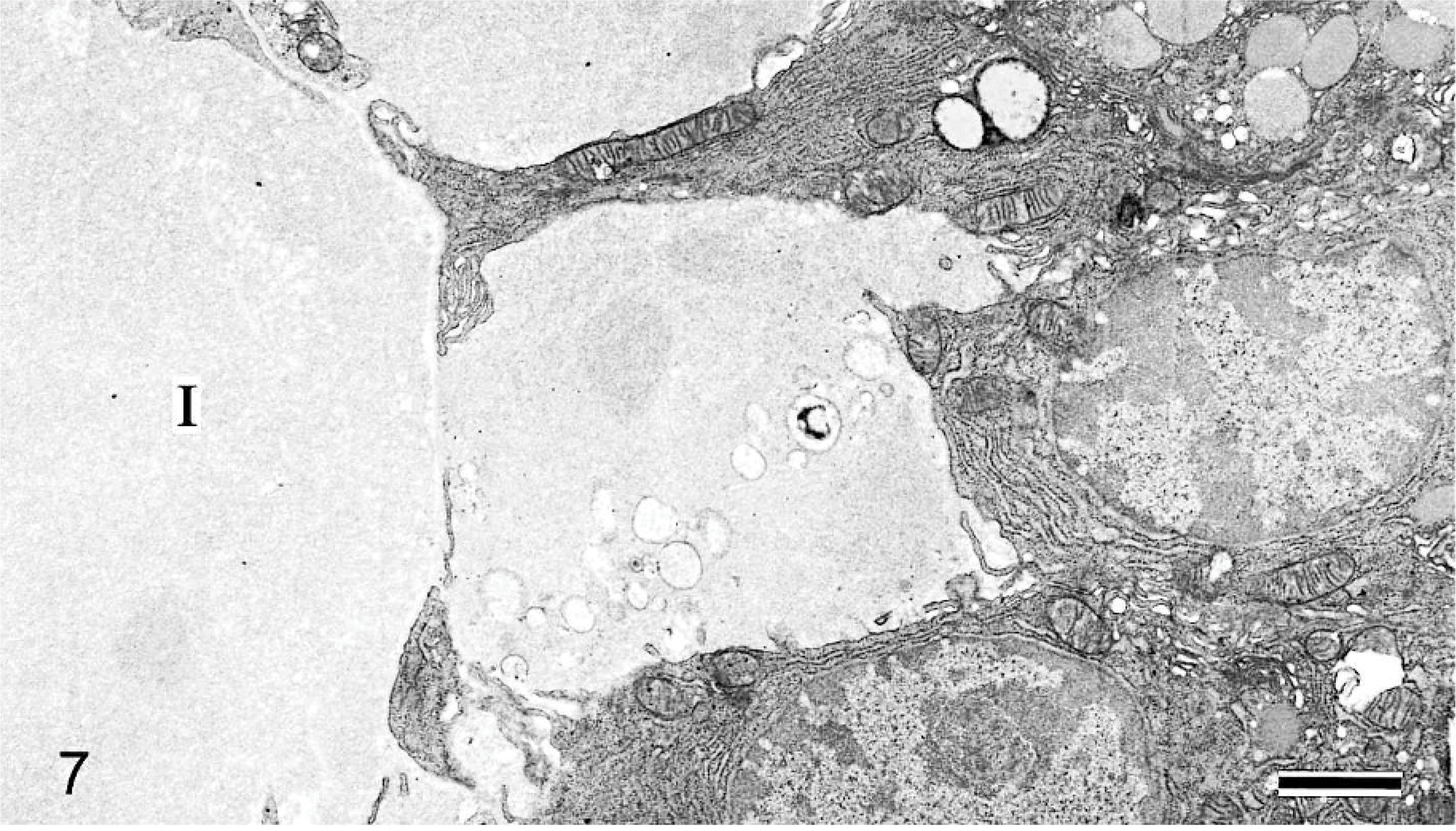
Nasal cavity (level I); 42-week-old B6C3F1/Crlj mouse. Electron micrographs. Amorphous material is observed at the interstitium (I), and similar material is also present in the nasal gland epithelial cells. Bar = 1.2 μm.
Discussion
In mice, the ES was observed in the nasal septum. There are 2 different opinions concerning ES, amyloid or not, based on very few descriptions. 2, 4, 5, 7, 8 Amyloid reacts positively with Congo red according to light microscopy and consists of nonbranching fibrils of indeterminate length and a diameter of approximately 7.5–10 nm ultrastructurally. 1 In our investigation, ES reacted negatively with Congo red and did not consist of nonbranching fibrils. In addition, ES was observed in all mice examined, although no amyloid was deposited in other organs or tissues. These findings indicated that ES in the mouse nasal septum was “not amyloid,” as Haines et al. 4 and Monticello et al. 8 have reported.
Haines et al. 4 reported that ES reacted positively with periodic acid–Schiff reaction and pale blue with Masson trichrome, so ES might include collagen. The same findings were observed in this investigation as well. Furthermore, it was found that ES consisted of not only collagen but also amorphous material in the electron microscopic findings. The chemical composition of the amorphous material remains to be determined in detail but might contain a complex carbohydrate except for osteoglycan because ES reacted positively with periodic acid–Schiff reaction with prior diastase treatment and negatively with toluidine blue or Alcian blue.
Electron microscopically, the amorphous material was observed not only at the interstitium but also in the glandular cells. In young mice, there were areas observed in which the amorphous material was present only in the nasal gland epithelial cells but not in the nearby interstitium. The amorphous material was very similar and connected to the material in the rough endoplasmic reticulum of the nasal gland epithelial cells. These findings indicated that the nasal gland epithelial cells produced the amorphous material. Unfortunately, it was not clear in this investigation how the amorphous material was released to the interstitium.
Two kinds of glands were present in the nasal septum in mice. One of them stained clear for HE, the other stained dark for HE. To our knowledge, there has been no description concerning this. However, the 2 kinds of glands in the nasal septum are thought to be common findings in mice regardless of the strain because we have also observed this finding in B6C3F1 mice and CD-1 mice in routine examinations (data not shown). ES was only deposited at the interstitium of the clear HE-stained nasal glands. Hence, ES might relate to the clear HE-stained glands. We could also say that the clear HE-stained glands were not degenerative glands caused by ES deposition because they were present in young mice, which had a very small amount of ES.
The ES volume was very small in young mice but became larger and larger with aging. If there were no ES elimination mechanisms, the nasal septum would burst due to the extraordinary amount of ES deposition in older mice. In this investigation, the rupture of the nasal septum was not found, although separation of the nasal glands and/or expansion of the septum width was observed in older mice. Hence, ES might be eliminated from the interstitium by some mechanisms—for example, phagocytosis by macrophages, resorption by the nasal glandular cells, and so on.
A very common lesion in nasal tissue is intracytoplasmic hyaline inclusions, and it stained eosinophilic with HE in aging mice. 5, 7, 8 They occur not only at the septum but also in multiple sites, often at the junction of respiratory and olfactory epithelia in the dorsal medial meatus. They stain negatively with periodic acid–Schiff reaction and are also observed in rats. 8 On the basis of these findings, it is clearly understood that the intracytoplasmic hyaline inclusions have no relationship with ES.
Strain-specific high incidences of unusual hyalinosis in the gastric, biliary, and respiratory tracts have been reported in mice with the background 129S4/SvJae strain. 9 In the nasal tissue, the respiratory epithelium is affected the most, with olfactory epithelial involvement occurring primarily near the olfactory/respiratory transition areas characterized by the form of eosinophilic inclusions. It is clearly understood that the hyalinosis in mice with the background 129S4/SvJae strain was quite different from ES.
In our investigation, mainly Jic:CB6F1-Tg rasH2 mice were used, which are transgenic mice carrying a human prototype c-Ha-ras gene with its own promoter region. It remains to be determined whether there are some differences concerning the incidences of ES between in Jic:CB6F1-Tg rasH2 and the equivalently aged mice of the other strains. However, no differences were observed concerning the depositing site and the HE-staining property between the 2 strains in our investigation (Jic:CB6F1-Tg rasH2 mice, Jic:CB6F1-nonTg rasH2 mice) and the 3 strains described in textbooks and reports (CD-1 mice, B6C3F1 mice, B6;129 mice). Hence, ES in Jic:CB6F1-Tg rasH2 mice and Jic:CB6F1-nonTg rasH2 mice would not be a strain-specific substance, but a substance similar to that in CD-1 mice,2 B6C3F1 mice,5 and B6;129 mice,4 which has been reported.
In conclusion, according to the histopathologic and electron microscopic investigation, it was indicated that the ES was not amyloid, and it was suggested that the ES might consist of not only collagen but also complex carbohydrate, which is produced by the nasal gland epithelial cells. The significance of ES is not clear, but it might be a physiologic phenomenon because ES was deposited in all mice and without degeneration or inflammation in the nearby nasal tissue.
Footnotes
Acknowledgements
We thank Mr. Takayoshi Ito for his excellent technical assistance. We also thank Mr. Steve Yamakami for language editing.
