Abstract
Marked lateral ventricular enlargement associated with atrophic cerebral cortex and periventricular encephalitis is described in a 2–month-old fox affected by disorientation, generalized ataxia, difficulty in walking, circling, and blindness. Clinical conditions progressed to stupor and spontaneous death within a few days. At necropsy, severe inflammatory and necrotizing lesions were observed in periventricular sites associated with diverticula and cleft formation in perithalamic areas and rhinencephalic cortex. Immunolabeling for Toxoplasma gondii, Neospora caninum, Encephalitozoon cuniculi, canine distemper virus, and rabies virus was negative. Given the presence of periventricular and choroidal neutrophilic/mononuclear cell infiltration, it is thought that a bacterial infection may have been the cause of the inflammatory lesions, with internal hydrocephalus secondary to the severe periventricular lesions. A similar condition has been previously reported in the pathogenesis of spontaneously occurring acquired canine hydrocephalus, but no viral or bacterial causes have been investigated to date.
Hydrocephalus is one of the most common developmental disorders in animals. It may be congenital, associated with inborn defects or malformations, or acquired, associated with an inflammatory process or masses, all of which block cerebrospinal fluid (CSF) flow, especially through the mesencephalic aqueduct or lateral apertures of the fourth ventricle. 8 Therefore, hydrocephalus consists of a secondary increase in CSF volume in the ventricular system of the brain and results in compression atrophy of nervous tissue parenchyma. When hydrocephalus is associated with inflammation, diverticula and clefts may be seen in the periventricular white matter of the brain. A similar condition has been reported in dogs; 2– 4, 10, 11 in some cases attributed to bacterial, parasitic, or viral infections that have not been further investigated. 3, 4, 10 In the fox, morphologic and pathogenetic studies of hydrocephalus associated with the most common inflammatory and infectious brain disorders have not been reported.
In this study, a 2-month-old fox was found off the road with evident disorientation and in a poor nutritional state. Clinical examination showed generalized ataxia, difficulty in walking, circling, and blindness. Distemper virus infection was suspected, and the young fox was treated with antibiotic and anti-inflammatory therapy without success. The clinical condition progressed to stupor and spontaneous death within a few days.
A full necropsy was performed after death, and representative tissue samples were removed and fixed in buffered 10% formalin. Half the fresh brain was submitted for direct immunofluorescence for rabies virus (antipurified rabies nucleocapsids rabbit antibody, 1 : 100, Bio-Rad, Tokyo, Japan). The right half of the brain was fixed in buffered 10% formalin and paraffin-wax embedded for routine histologic examination and then was tested for canine distemper virus (anti-CDV monoclonal antibody, 1 : 800, VMRD Inc, Pullman, WA), Toxoplasma gondii (anti- T. gondii polyclonal caprine antibody, 1 : 4,000, VMRD Inc), Neospora caninum (anti- N. caninum polyclonal caprine antibody, 1 : 4,000, VMRD Inc), and Encephalitozoon cuniculi (anti- E. cuniculi polyclonal rabbit antibody, 1 : 1,000, prepared by us). Paraffin-embedded brain sections were also submitted for avidin-biotin-peroxidase complex staining for glial fibrillary acid protein (GFAP, polyclonal antibody, 1 : 500, Dako, Milan, Italy) and periodic acid–Schiff staining (PAS).
At necropsy, no skull enlargement was found, or malformation or reduced thickness of the calvarium. The brain was enlarged and cerebral convolutions flattened. The cerebral cortex was restricted to a thin rim showing a severe atrophic subcortical white matter. Lateral ventricles were 3 times the thickness of the cortex, and their outline was extremely irregular (Fig. 1). Severe disruption of periventricular white matter resulted in diverticula and cleft formation in perithalamic areas and the rhinencephalic cortex (Fig.1a). In the former, the lesions even involved the optic tract. More caudally, damage by necrotizing lesions appeared to extend as far as the white matter of parahippocampal gyrus (Fig. 1b).
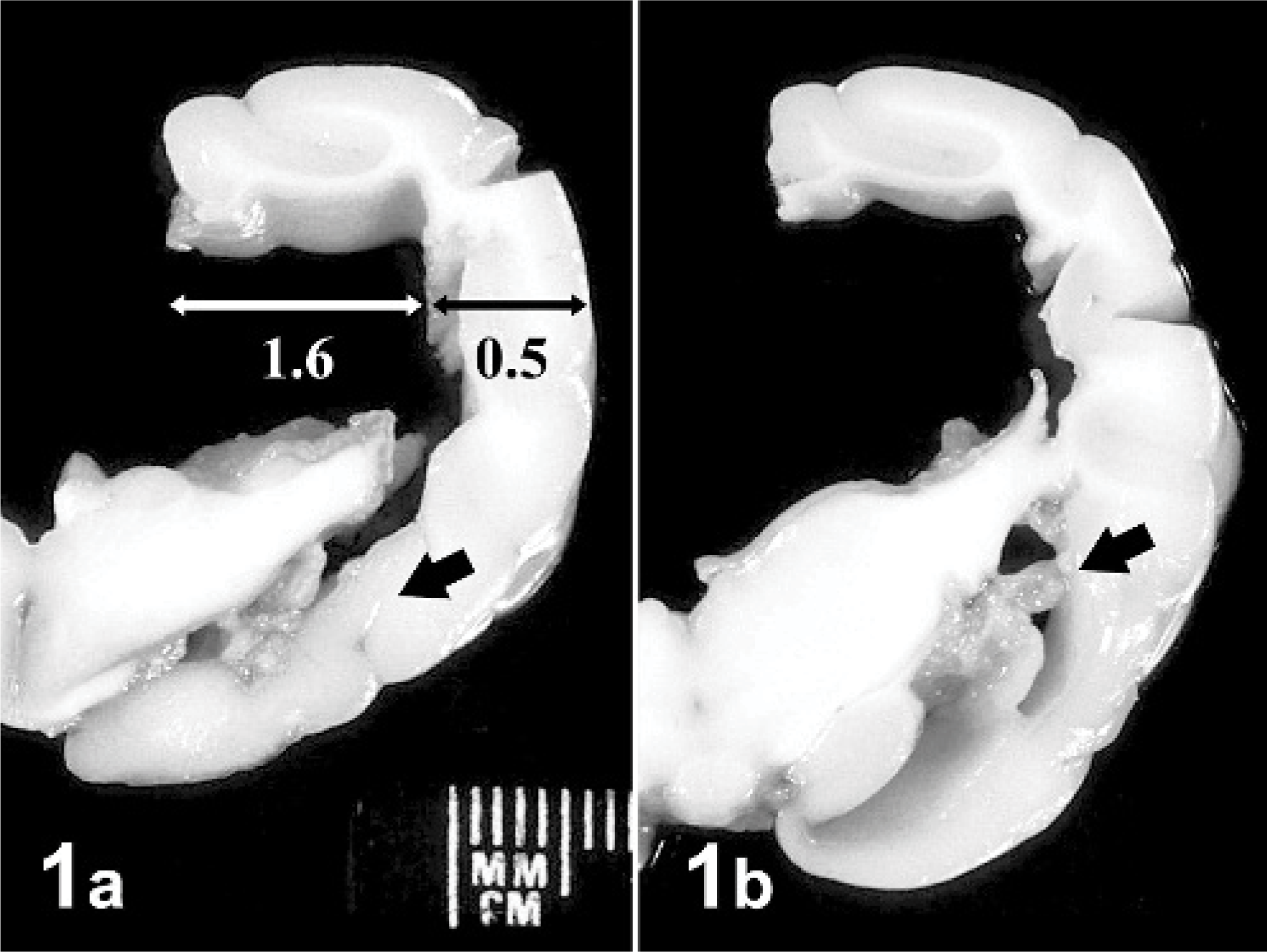
Right half; brain; fox. Severe atrophy of subcortical white matter. The lateral ventricle diameter is 3 times the cortex thickness, and the optic tract appears interrupted by a necrotic lesion (arrow).
Histologically, severe inflammatory and necrotizing lesions were observed in periventricular sites associated with diverticula of the capsula interna and periventricular white matter, involving the optic tract and rhinencephalic cortex as well. They were characterized by malacia and loss of the ependymal layer, inflammatory and gitter cell infiltration, marked endothelial reaction, and microvascular proliferation in adjacent nervous parenchyma (Fig. 2). An apparent subependymal astrogliosis was confirmed by marked GFAP immunoreaction. In the diencephalon, distinct neuronal loss was found with numerous reactive astrocytes (Fig. 3). A number of necrotic neuronal cells became encrusted with basophilic material, consisting of calcium and iron salts. Large amounts of exudates containing mainly neutrophils and cellular debris were found in the lateral ventricle, while in the third ventricle and mesencephalon canal, numerous phagocytic cells were observed. These cells contained brownish PAS-positive pigments, consistent with lipofuscin. Suppurative choroiditis was also observed in the lateral and fourth ventricles (Fig. 4). Immunolabeling for T. gondii, N. caninum, E. cuniculi, canine distemper virus, and rabies virus was negative.
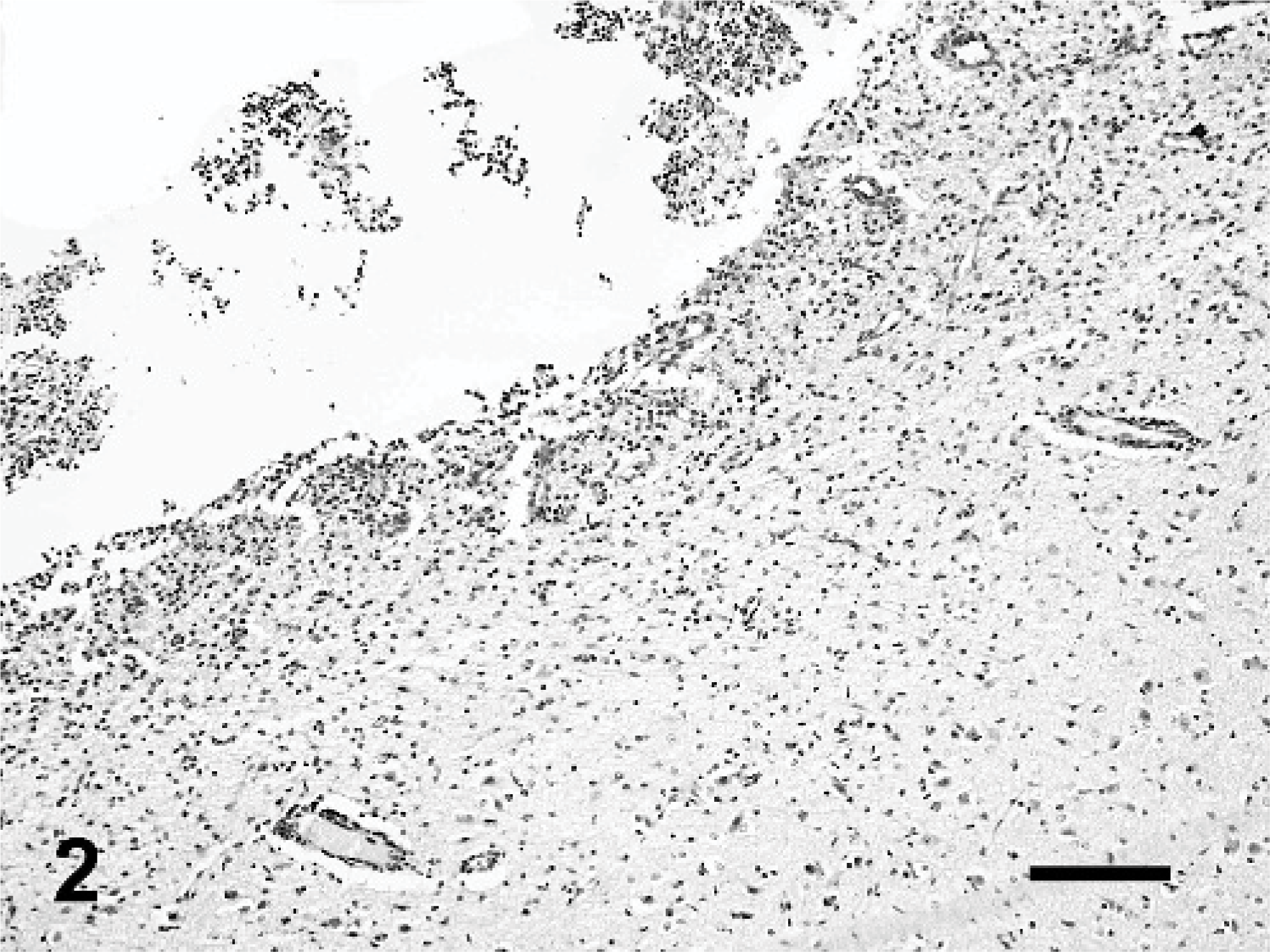
Lateral ventricle; brain; fox. Severe necrotizing ependymitis. The ependymal layer is completely absent and replaced by a marked gliosis and vascular reaction developed in the subependymal white matter. HE. Bar = 102 μm.
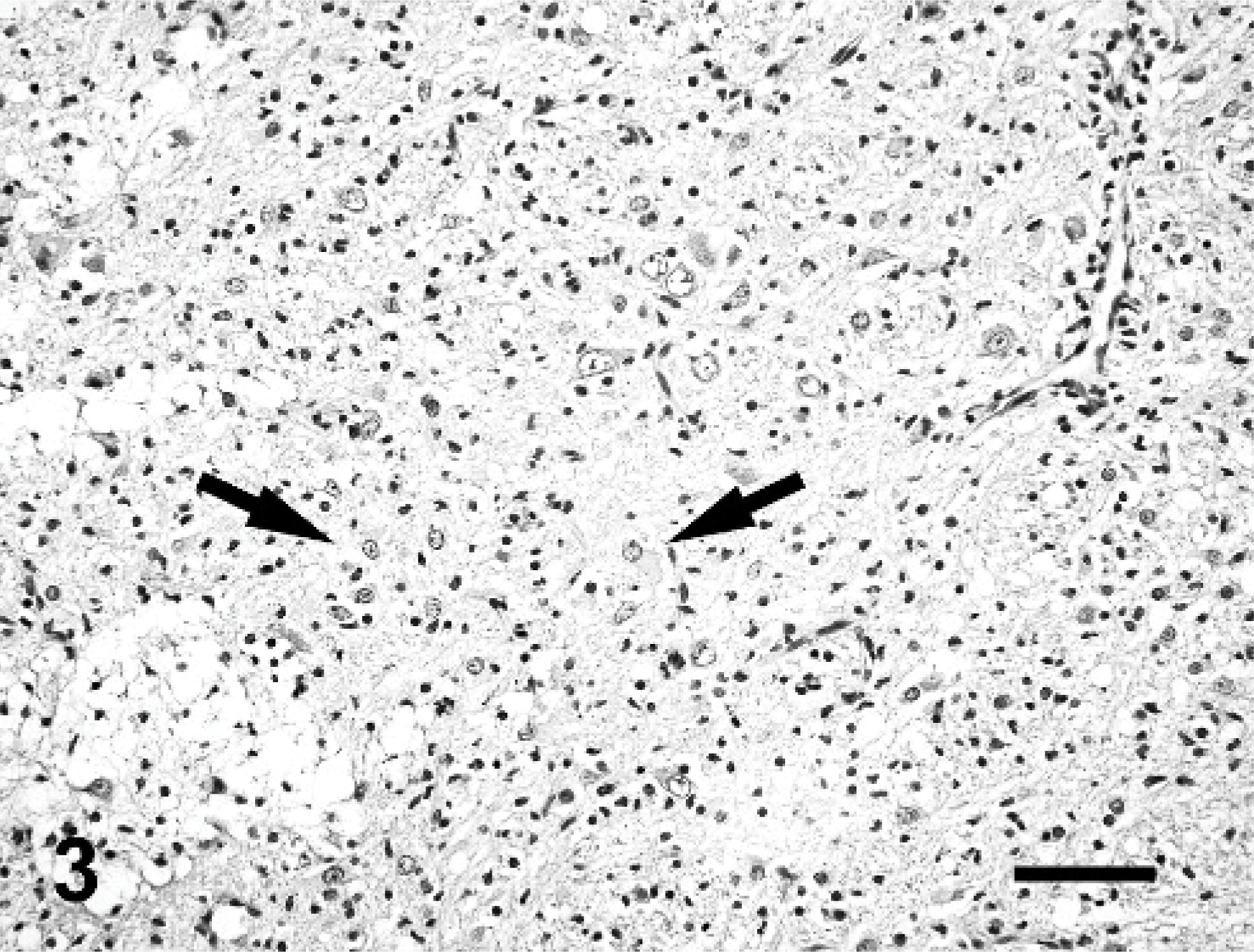
Diencephalon; brain; fox. Microfoci of astrogliosis are found in association with loss of neurons. Some reactive astrocytes are consistent with gemistocytes (arrows). HE. Bar = 58 μm.
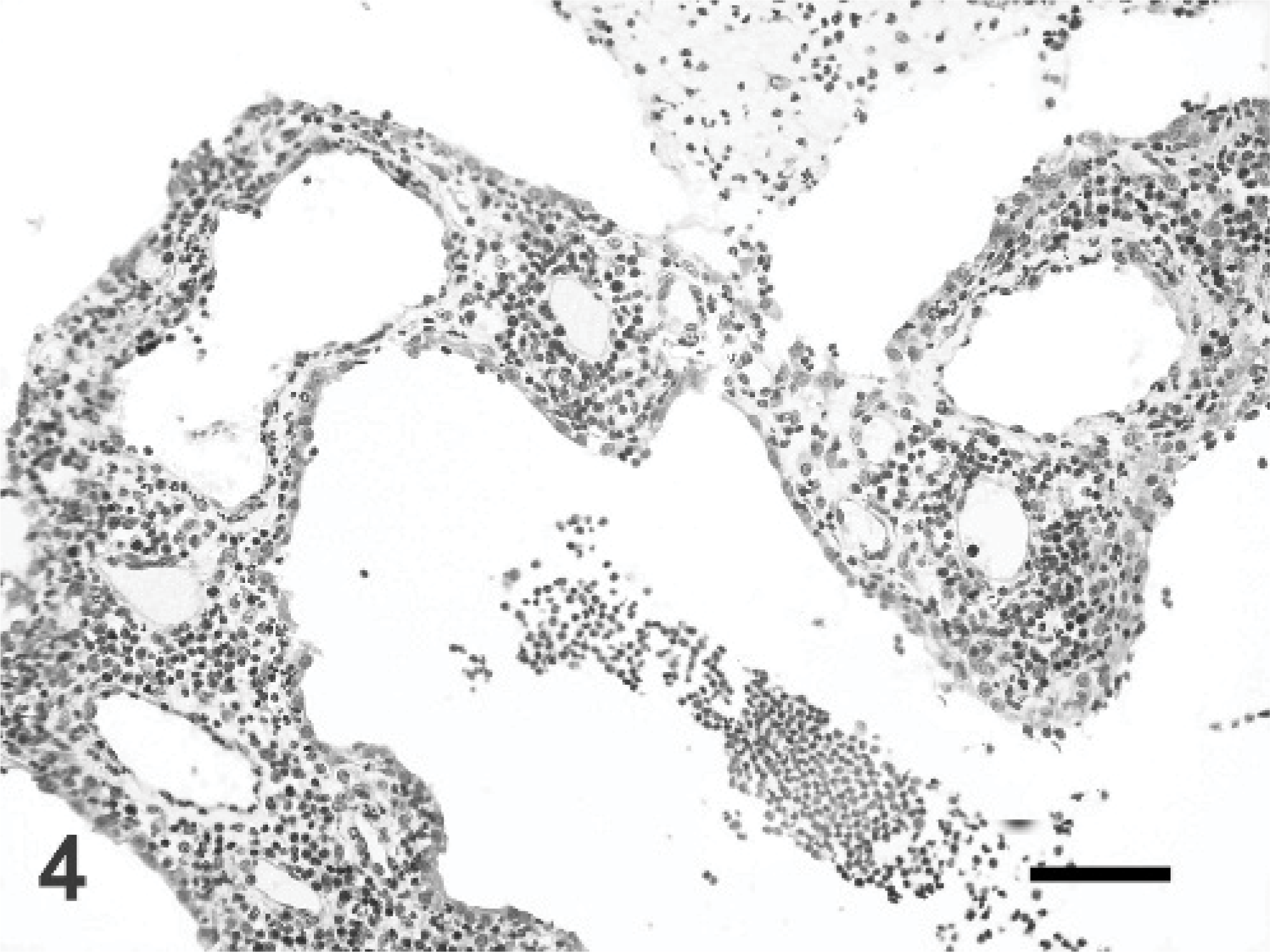
Lateral ventricle; brain; fox. Choroid plexus is severely infiltrated by inflammatory cells, mostly consistent with neutrophilic granulocytes and lymphocytes. HE. Bar = 52 μm.
Congenital hydrocephalus is one of the most common developmental disorders of the central nervous system (CNS) in animals and is often associated with malformations that produce obstruction of the ventricular pathway with secondary CSF volume increase. 9 As to the pathogenesis of acquired hydrocephalus, masses as well as inflammatory processes may result in blockage of CSF flow and/or resorption in the ependymal lining or subarachnoid space. 9 Rarely, acquired hydrocephalus can also result from an overproduction of CSF, as in choroid plexus papilloma in people. In the fox, the exact pathogenesis of hydrocephalus has not yet been investigated. On the contrary, the list of possible etiologies of inflammatory encephalopathies is quite extensive in this species and includes viral, bacterial, and parasitic agents, most of which affect the dog as well.
In the case under study, we excluded the possibility that the hydrocephalus was congenital, owing to the absence of an increased volume and/or reduced thickness or deformation of the calvarium. We further excluded perinatal trauma since the nervous tissue lesions were localized deep in the brain, especially in the periventricular white matter, and were not associated with parenchyma hemorrhages.
As for necrotizing periventriculitis, T. gondii, N. caninum, E. cuniculi, canine distemper virus, and rabies virus infections were excluded as the cause, given both the morphologic findings and negative immunostaining reactions. Toxoplasmosis, neosporosis, and encephalitozoonosis are not commonly associated with acquired hydrocephalus in puppies. 8 Moreover, they produce random multifocal lesions characterized by primary blood vessel involvement with secondary necrotizing foci, where microglia and macrophages are prevalent, sometimes in association with organisms. Although canine distemper virus infection can also produce necrotizing lesions of the ependyma and adjacent tissue, 5 it is characterized by multifocal demyelination with perivascular cuffings in more chronic stages. As for a rabies virus infection, histology shows a widespread neuronal degeneration and marked microglial cell proliferation associated with perivascular mononuclear cuffings and intracytoplasmic inclusion bodies. Cryptococcal ependymitis was also excluded because granulomatous lesions and fungal elements were not observed. Although studies of the prevalence of serum antibodies to canine adenovirus and canine herpesvirus in the European Red Fox have demonstrated that this species could be considered susceptible, no cases of encephalitis have been reported in the literature. 7 In our case, canine adenovirus and herpes virus type 1 infections were excluded given the absence of the typical lesion pattern and distribution. The lack of hemorrhagic encephalopathy related to vasculitis, thrombosis and hemorrhagic, or ischemic infarcts in the brain also led us to exclude parvovirus infection. Parainfluenza virus in the dog 1 and reovirus type 1 in mice 6 have been reported to cause acquired hydrocephalus secondary to ependymitis in fetal or neonatal animals, but so far they have not been reported as infective agents in the fox.
In the case under study, it is thought that a perinatal bacterial infection could have been the cause of the inflammatory lesions, based on the presence of ependyma and choroid plexus neutrophilic/mononuclear cell infiltration, but unfortunately no bacteriologic examination was done on the brain tissue or CSF. Indeed, many bacterial agents are known to affect the CNS, including the choroid plexus, ependyma, and meninges, which represent the main organs involved in CSF dynamics. 8
In the present case, the severe widespread necrotizing and inflammatory ependymal and periventricular lesions were considered the primary cause of the internal hydrocephalus and not the result of any initial intraventricular CSF volume or pressure changes. Therefore, the infection hypothesized might have impaired secretory and absorptive ependymal functions hindering either the normal fluid dynamics of transependymal flow or the diffusion gradients. The severe inflammatory choroidal infiltration in the forth ventricle would also suggest that obstruction of lateral apertures should be considered in the pathogenesis of hydrocephalus. Furthermore, the association of necrotizing lesions and increased intraventricular pressure might have contributed to the formation of diverticula and clefts in perithalamic areas and the rhinencephalic cortex. The involvement of the optic tract accounted for the blindness noted in the fox.
To our knowledge, while internal hydrocephalus associated with periventricular encephalitis has been previously reported in the pathogenesis of spontaneously occurring acquired canine hydrocephalus, it has not been investigated in the fox until now.
Footnotes
Acknowledgements
We thank Dr. E. Pozio (Istituto Superiore di Sanità, Rome, Italy) for E. cuniculi immunolabeling and Dr. G. Ricci for technical assistance.
