Abstract
A neurologic disease affected a colony of endangered Fennoscandian arctic foxes (Alopex lagopus) kept in captivity for breeding purposes. Several outbreaks of disease occurred between 1994 and 2004. The clinical signs included ataxia, indications of anosmia, blindness, and abnormal behavior. The disease was characterized by severe necrotizing encephalitis affecting mostly the cranial cerebrum, basal ganglia, and olfactory bulbs. Investigations to identify the etiology of the disease included testing for several infectious agents known to cause encephalitis in carnivores. Tests for Toxoplasma gondii, Encephalitozoon cuniculi, Neospora caninum, canine distemper virus, rabies, adenovirus type 1, Borna disease virus, and Listeria monocytogenes were negative. The colony was closed, and the cause of the disease remains undetermined.
The population of arctic foxes (Alopex lagopus) in Fennoscandia (Norway, Sweden, Finland, and Kola Peninsula) decreased dramatically during the last century because of overhunting. In the mid-19th century, the Swedish breeding population was composed of about 4,700 individuals. Presently, the Fennoscandian arctic fox is threatened with extinction, and only approximately 150 adults remain in the wild. A captive program, aimed at preserving the arctic fox, was initiated in 1992. Wild-caught foxes were kept at a Swedish breeding center for endangered species to produce offspring for release in selected areas. However, many of the captured arctic foxes and their offspring developed a severe neurologic disease associated with necrotizing encephalitis. The first case occurred in 1994 with several episodes of disease until 2004, when the last remaining fox was also affected. A group of 12 blue foxes (Alopex lagopus), the domestic counterpart of the arctic fox, had been kept at the breeding center until 1992, before the arrival of the arctic foxes. All of the blue foxes died of a respiratory and neurologic disease, with clinical signs similar to those shown by the arctic foxes. The arctic foxes were never housed in the same enclosure as that occupied by the blue foxes. The present study describes the clinical and histopathologic features of this apparently novel condition in the arctic fox and the diagnostic investigations conducted in searching for the etiology.
Eight arctic foxes were included in the study. The age, sex, body weight, origin, length of clinical disease, and date of euthanasia are summarized in Table 1. All foxes were euthanized at end-stage disease. Attempts to determine the cause of the disease included virologic, parasitologic, bacteriologic, serologic, and immunohistochemical investigations. The agents for which the foxes were tested and the types of analyses used are shown in Table 2. A postmortem examination was conducted. Samples for histopathology included the brain, spinal cord (5 foxes), liver, kidney, spleen, lungs, skeletal muscle, heart, and lymph nodes. The tissues were fixed in 10% buffered formalin, processed and embedded in paraffin wax, sectioned at 5 mm and stained with hematoxylin and eosin (HE). Selected brain sections were stained with luxol fast blue (for myelin) and with biotinylated lectin a with an avidin-biotin complex detection system b to identify macrophages and activated microglial cells. Selected brain sections were immunostained for infectious agents (Table 2) and for an astrocyte marker for glial fibrillary acidic protein (GFAP). Sections (5-6 μm) were rehydrated, immersed for 20 min in 1.5% hydrogen peroxide to inhibit endogenous peroxidases, and blocked for 20 min with 2% bovine serum albumin. The primary antibodies and the dilutions used were: in-house-developed monoclonal antibodies for Toxoplasma gondii (1:1,000), in-house-developed rabbit hyperimmune sera for Neospora caninum (1:250), Encephalitozoon cuniculi (1:1,000) and Borna disease virus (BDV) (1:15,000), and commercially available rabbit anti-bovine GFAP b (1:500) and monoclonal antibodies for canine distemper virus (CDV) c (1:400). The sections were counterstained with Mayer hematoxylin and cover-slipped. Each immunostain was accompanied by a positive reference control and a negative (primary antibody omitted) control of each section.
Virus isolation was attempted in A-72 cells d in 3 foxes (2, 5, and 7). Additionally, virus culture from brain, spinal cord, trigeminal ganglia, cerebrospinal fluid, lungs, kidneys, liver, and spleen of fox 7 was attempted in mink lung primary cell culture, e OL-cells (human oligodendroglial) cell culture, f bovine turbinate primary cell culture, g and in the following cell lines: Vero, d C6 (Rat glial tumor cell line), d and Madin-Darby canine kidney cell line. d Polymerase chain reaction (PCR) analysis was used for the detection of BDV 4 and E. cuniculi. For E. cuniculi, standard nested PCR procedures were applied using the following primers: EC1: 5′-CAC CAG GTT GAT TCT GCC TGA C-3′, EC2: 5′-CCT CTC CGG AAC CAA ACC CTG-3′, EC3: 5′-TAT TCT CTG GGG CTA AGC CA-3′, and EC4: 5′-CTG ATC CCC CGT ATC CCG TC-3′.
Background data for the arctic foxes in the study.

NR = not recorded.
The following parameters were determined from a blood sample obtained before euthanasia in 4 foxes (3, 5, 7, and 8): hemoglobin concentration, hematocrit, total and differential leukocyte counts, urea, creatinine, and aspartate aminotransferase, creatine kinase (CK), alanine aminotransferase (ALAT), alkaline phosphatase, gamma glutamyltransferase, and glutamate dehydrogenase activities.
The early clinical signs observed in the arctic foxes included mild dry cough and balance disturbance. Further signs of disease became apparent in 2 weeks to 3 months. The abnormal movements and ataxia became more marked, and the foxes started to show decreased sense of smell and blindness and could not find their food. Behavioral changes included loss of fear and lack of awareness of people and circling. One fox slept for prolonged periods. Intermittent diarrhea and loss of nutritional condition were also observed. In late-stage disease, the foxes had difficulty swallowing and were observed to clutch objects tightly. Their nutritional condition deteriorated. The foxes did not improve clinically after treatment with antimicrobial drugs, corticosteroids, and B-vitamins and were euthanized when terminally ill. The longest clinical course was 6 months (fox 1). Several episodes of disease occurred during the years, with several foxes affected simultaneously.
Laboratory investigations to determine the cause of necrotizing encephalitis in arctic foxes. Agents for which the foxes were tested, type of analysis, and number of foxes tested. All tests were negative unless specified.

IHC = immunohistochemistry; PCR = polymerase chain reaction; SN = serum neutralization test; FAVN = fluorescent antibody virus neutralization.
One of the foxes (3) had a titer of 1/1,620.
At the postmortem examination, gray soft areas were observed on the cut surface of the frontal lobes of the brain in 1 fox (4) (Fig. 1A). One fox (5) had hyperemia of the mucosa of the urinary bladder, associated with the presence of more than 20 nematodes, identified as Capillaria plica.
Histopathology of the brain revealed severe nonsuppurative meningoencephalitis with multifocal necrosis in all the foxes (Fig. 1B). The inflammatory reaction was characterized by dense infiltrates of macrophages and activated microglial cells in the cerebral gray matter (Fig. 1C, 1D) with lesser lymphocytes and plasma cells, especially in the perivascular spaces and meninges. Perivascular cells were prominent, sometimes narrowing the lumen of the vessels. Degenerate neurons undergoing phagocytosis were interspersed among the inflammatory cells (Fig. 1C). In some of the foxes, the necrosis was extensive, involving the cerebral gray and white matter and resulting in cavities (Fig. 1B). The rarified areas contained cellular debris mixed with foamy macrophages, lymphocytes, and gemistocytes. Gemistocytes are large bi- or multinucleate acidophilic cells with homogenous cytoplasm and vesicular nuclei (Fig. 1E). They are reactive hypertrophic astrocytes and stain strongly with GFAP (Fig. 1F). In 1 case (fox 1), intensely eosinophilic structures consistent with Rosenthal fibers, filamentous inclusions of astrocytes that are usually found in association with long-lasting intense fibrillary gliosis, were observed in the brain. In the white matter there was multifocal gliosis. No indications of demyelination were observed. The most severely affected areas were the frontal cerebrum, olfactory bulbs, and basal ganglia. Lesser inflammatory changes were observed in the thalamus, midbrain, and medulla oblongata; they were mild or absent in the spinal cord and were not observed in the cerebellum.
Seven of the 8 foxes had inflammatory lesions in the lungs, discrete perivascular mononuclear cell infiltration to more widespread, but still mild, interstitial pneumonia. In addition to mononuclear cells, eosinophilic granulocytes were usually present in the inflammatory infiltrates in the lung. In the urinary bladder of 2 foxes (5 and 6) there was eosinophilic cystitis. Fox 3 had intraepithelial Capillaria eggs in the mucosa of the larynx, associated with local inflammation. Parasitic lesions consistent with Capillaria infestation also occurred in the nasal turbinates and trachea of foxes 7 and 8. No significant histopathologic lesions were observed in the other organs examined.
The results of analyses for the detection of various infectious agents were negative (Table 2) except for 1 test that showed presence of antibodies against Toxoplasma gondii (titer 1:1,620) in 1 fox. Hematology and clinical chemistry revealed leukopenia, lymphopenia, and decreased hematocrit and increased CK and ALAT activity levels in 3 foxes (3, 7, and 8) with decreased hemoglobin concentrations in 2 (3 and 8).
The previously undescribed disease reported in this study of arctic foxes resulted in the termination of a breeding colony. Part of the study was retrospective and based on archival material, which restricted the availability of samples for further testing. The cause of the disease was not identified with the testing that was conducted.
In general, necrotizing encephalitis can be caused by bacterial, viral, or protozoan infection; ischemic and hypoxic lesions; vasculitides; several drugs; toxins; and metabolic disorders. 8 None of these causes could be ruled out. However, since the foxes had received a balanced diet and were housed in safe enclosures (with no access to toxins) it was considered that a toxic or nutritional cause was less likely. Additionally, the histopathologic features suggested an infectious etiology. Some of the known infectious causes of encephalitis in Alopex lagopus are T. gondii, 9,16 canine adenovirus type I (fox encephalitis), 12 rabies virus, 3 and E. cuniculi. 1 In other fox species, encephalitides caused by CDV 19 and BDV 6 have been described. Additionally, there is serological evidence of infection of foxes with tick-borne encephalitis 18 and La Crosse virus. 2 Further agents known to cause encephalitis in carnivores include N. caninum, Listeria monocytogenes, and West Nile virus. Several of these agents were investigated in this study, but none of them appeared to have caused the disease, and the lesions in the brains of the present arctic foxes showed some differences from those described in these encephalitides. For instance, adenoviral fox encephalitis is characterized by vasculitis with disseminated hemorrhages, often associated with intranuclear inclusions in endothelial cells, and minimal or absence of inflammation in the brain parenchyma. 17 The classic form of canine distemper affects the cerebellar white matter and the optic nerve in particular, with demyelination as a prominent feature. 7 Toxoplasma gondii and N. caninum cause granulomatous encephalomyelitis, sometimes associated with necrosis. Although one of the foxes had antibodies to T. gondii, the relevance of this finding as the cause of the encephalitis is doubtful. Toxoplasma was not detected in the brain, and no lesions in peripheral organs were suggestive of toxoplasmosis. Alopex lagopus are highly susceptible to encephalitozoonosis and develop nonsuppurative meningitis, arterial and periarterial inflammatory changes, granulomas, and sometimes cystic lesions in the brain. 1,13 The absence of renal lesions (an organ usually affected in encephalitozoonosis) and failure to detect parasites in the brain reduces the probability of encephalitozoonosis. Serology for E. cuniculi did not indicate positive cases. BDV and rabies virus produce nonsuppurative encephalomyelitis with intraneuronal inclusion bodies; malacia is not observed. No inclusion bodies were observed in the foxes. Rabies is considered to be an unlikely cause of the encephalitis because Sweden is free of rabies. Encephalitis caused by herpesviruses share several similarities with the present cases in arctic foxes. Classical herpes simplex virus in humans causes necrotizing encephalitis, which progresses to cavitation in the chronic phase. 10 Bovine herpesvirus type 5 causes necrotizing encephalitis in cattle 5 and is also associated with upper respiratory infection. 11,15 Further investigation of the study cases for herpesvirus is ongoing.
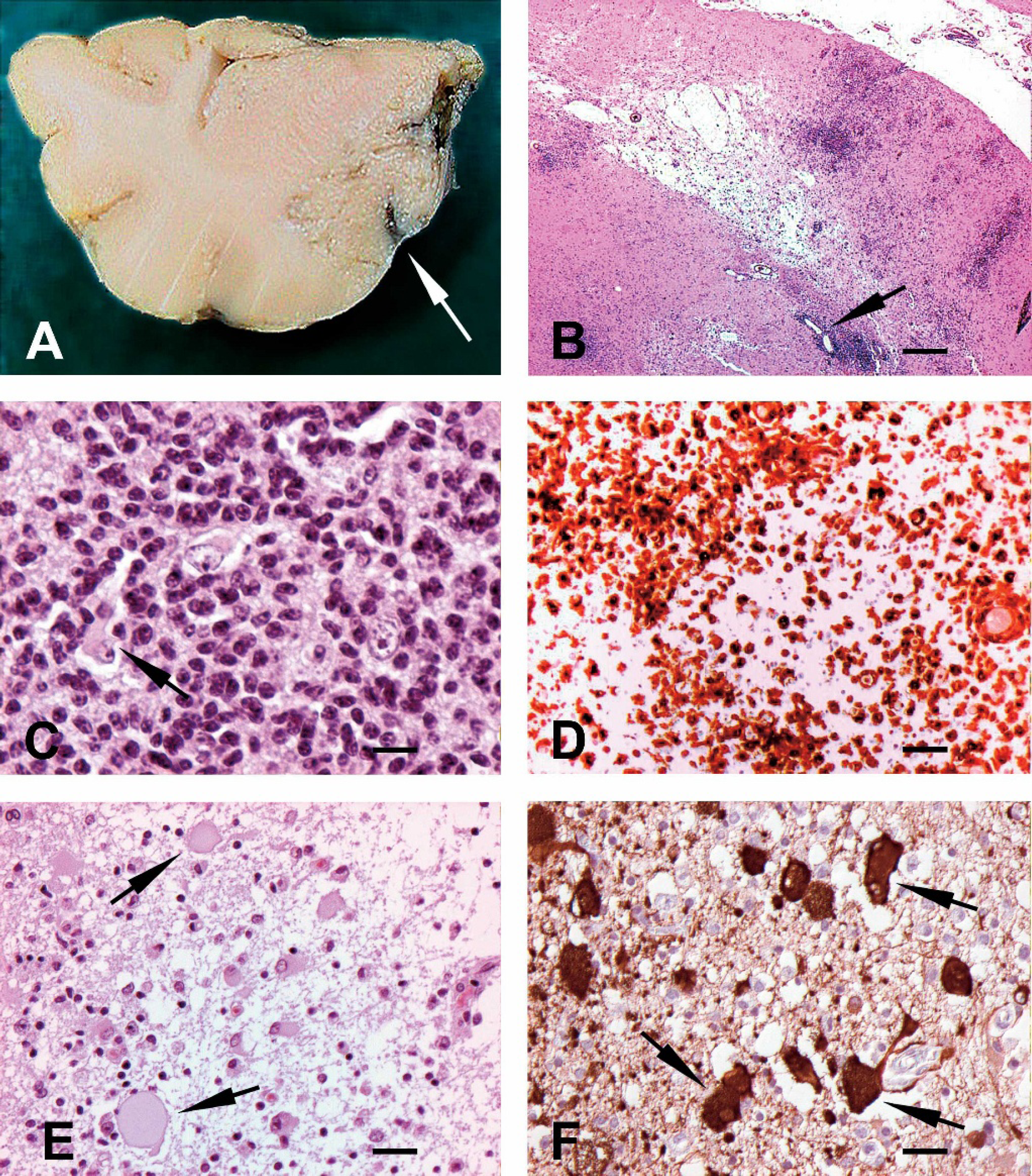
The brain of arctic foxes with necrotizing encephalitis.
The prominent reactive astrocytosis, including the presence of gemistocytes and Rosenthal fibers, is not unique to a specific neurological disease, but often accompanies chronic reactive fibrillary astrogliosis with extensive damage of brain tissue. No link between the respiratory signs, the mild pulmonary inflammation, and the encephalitis could be established. Whether the arctic foxes trapped in the wild were already infected on arrival or contracted an infection at the breeding center could not be established. It is not known if the blue foxes with the respiratory and neurological condition had lesions similar to those in the arctic foxes, as no pathology data from the blue foxes could be obtained. No analyses had been conducted on the blue foxes to determine the cause of the disease. No arctic foxes were released from the breeding center to the wild.
In conclusion, the encephalitis described in this study is probably a novel disease entity, which may constitute a threat to the preservation of the Fennoscandian arctic fox. It is possible that blue foxes are also affected. The cause of the disease could not be determined.
Acknowledgements. We thank Ewa Westergren and Birgitta Dillner for the immunohistochemical reactions; Caroline Bröjer, Henrik Ulhorn, Carl Hård af Segerstad, and Anders Lager for the necropsies on some of the foxes; Katinka Bélak for the bovine herpesvirus positive control material and antibodies for the immunohistochemistry; and Ian Lipkin at University of California, Los Angeles, Berkeley for providing the oligodendrocyte-cell culture. Financial support came from the European Commission (LIFE 03 NAT/S/000073-Saving the endangered Fennoscandian Alopex lagopus-SEFALO).
Footnotes
a.
Vector Laboratories, Burlingame, CA.
b.
DakoCytomation, Glostrup, Denmark.
c.
VMRD Inc., Pullman, WA.
d.
American Type Culture Collection, Rockville, MD.
e.
Danish Institute for Food and Veterinary Research, Lindholm, Denmark.
f.
University of California, Berkeley, CA.
g.
National Veterinary Institute (SVA), Uppsala, Sweden.
