Abstract
A juvenile, wild-caught prairie falcon (Falco mexicanus) kept for falconry was presented to a veterinary hospital for intermittent opisthotonos and torticollis. Clinical examination, complete blood count, serum biochemistry panel, and fecal analysis were unremarkable. Clinical signs did not resolve, and the bird was euthanized 6 mo after the appearance of clinical signs. Autopsy revealed a mild, unilateral hydrocephalus and nematodes within the thoracic air sac. Histopathology demonstrated mild, unilateral hydrocephalus; scattered glial nodules; meningeal nematode sections; and meningeal and intraventricular embryonated eggs. Morphology and molecular characterization were consistent with the air sac nematode Serratospiculum or Serratospiculoides spp. Air sac nematode infection can be associated with air sacculitis or pneumonia in falcons. Aberrant migration of air sac filariid nematodes Serratospiculum or Serratospiculoides spp. into the nervous system resulting in clinical disease is rare, but should be included in the differential diagnosis of neurologic diseases in falcons.
Keywords
Filariid nematodes of the Serratospiculum and Serratospiculoides genera (order Spirurida, infraorder Spiruromorpha, superfamily Diplotriaenoidea, family Diplotriaenidae) parasitize the air sacs of birds as an incidental or clinically significant infection.12,15 Serratospiculiasis has been reported in a number of falconiformes, including the prairie falcon (Falco mexicanus), peregrine falcon (F. peregrinus), saker falcon (F. cherrug), gyrfalcon (F. rusticolus), common kestrel (F. tinnunculus), lanner falcon (F. biarmicus), New Zealand falcon (Falco novaeseelandiae), northern harrier (syn. hen harrier, (Circus), northern goshawk (Accipiter gentilis), Cooper’s hawk (Accipiter cooperii), and black kite (Milvus migrans). Non-falconiformes species may also be infected, including the rook (Corvus frugilegus) and great tit (Parus major).2,5,9,13
Serratospiculum spp. have an indirect lifecycle, with an avian definitive host and an arthropod intermediate host. 12 Adult nematodes reside within the air sacs of birds, where eggs are deposited and passed through the lungs to be coughed up and swallowed by the bird. Embryonated (L1) eggs are passed in the feces and are subsequently ingested by the intermediate host, which includes beetles (family Tenebrionidae) and a species of woodlouse (family Eubelidae).3,12 Larval stages L1–L3 develop within the intermediate host, after which L3 larvae encapsulate within the adipose tissue. After consumption of an infected insect by the definitive host, L3 larvae are released from encapsulation and penetrate the proventriculus and ventriculus, migrating directly to the adjacent air sacs. Within the air sacs, L3 larvae undergo 2 additional molts before developing into the adult stage.9,12 Pigeons and other prey species have been proposed as possible paratenic hosts important in the spread of the parasite in some populations.7,14
Lesions associated with serratospiculiasis are restricted to the air sacs and lower respiratory tract. Large numbers of adult nematodes and eggs may be present intraluminally or intramurally within the air sacs, accompanied by eggs in the lower respiratory tract.5,8,9,12,13 Serratospiculiasis is often incidental, but can be a cause of morbidity and mortality in raptors, especially wild raptors taken into captivity.12,18 Common clinical signs associated with serratospiculiasis include dyspnea, lethargy, anorexia, reduced flight performance, pharyngeal plaques, and vomiting.12,13,17,19 Severe serratospiculiasis has been reported in multiple species as a primary cause of death or justification for euthanasia.1,5,6,8 Microscopically, serratospiculiasis manifests as air sacculitis characterized by fibrous thickening, mononuclear inflammation of variable severity, multifocal necrosis associated with embedded nematodes and eggs, and epithelial and mesothelial hyperplasia and sloughing.4,5,8,13 Air sac lesions are variably accompanied by bronchopneumonia to bronchointerstitial pneumonia with a mixed inflammatory infiltrate, occlusion of airways by eggs and edema fluid, and variable multifocal necrosis and fibrosis associated with the parasite eggs.5,8,13 Mesothelial squamous metaplasia and hyperplasia, serosal mononuclear infiltration, and the presence of nematodes and eggs within the proventriculus have been reported. 4
A juvenile, wild-caught, male prairie falcon (F. mexicanus) kept for falconry in central Utah was presented clinically with intermittent episodes of opisthotonos and torticollis. Opisthotonic episodes varied in duration of 4–5 s up to ~15 min (see Supplemental Videos 1 and 2) and occurred multiple times daily. A video camera was used to monitor the bird remotely, and no factors precipitating the opisthotonic events were detected. The falcon’s body weight was adequate, and appetite was normal. The falcon was molting, and plumage was in good condition. On clinical examination, the falcon’s mentation, posture and gait, proprioception, and sensory responses were unremarkable, apart from the spontaneous episodes of opisthotonos. Results of a complete blood count and serum biochemistry panel (Antech, Irvine, CA) were unremarkable. Based on the clinical signs, West Nile virus infection, avian paramyxovirus (avian avulavirus) infection, lead poisoning, head trauma, and polioencephalomalacia were considered as differential diagnoses. The owner declined further test procedures, except testing for West Nile virus. An oral swab was negative for West Nile virus RNA by real-time PCR. Intramuscular, followed by oral, supplementation with thiamine (10 mg/kg) was initiated. The episodes of opisthotonos continued, and slowly increased in frequency according to the owner. The falcon was euthanized 6 mo after the initial appearance of clinical signs.
At autopsy examination, the falcon was in good body condition, with adequate pectoral musculature (keel score 3.5 of 5), adequate subcutaneous and abdominal adipose tissues, and body weight of 509 g. The left caudal thoracic air sac was mildly thickened and opaque, and contained ~30 white nematodes measuring ~0.5–1.0 mm diameter and up to 14 cm long (Fig. 1). Samples of brain, air sac, trachea, lung, esophagus, ingluvium, proventriculus, ventriculus, small intestine, liver, spleen, heart, skeletal muscle, kidney, adrenal gland, and testis were collected for microscopic examination. Tissues were fixed in 10% neutral-buffered formalin, trimmed, processed routinely for paraffin embedding, sectioned at 5 µm, and stained with hematoxylin and eosin.
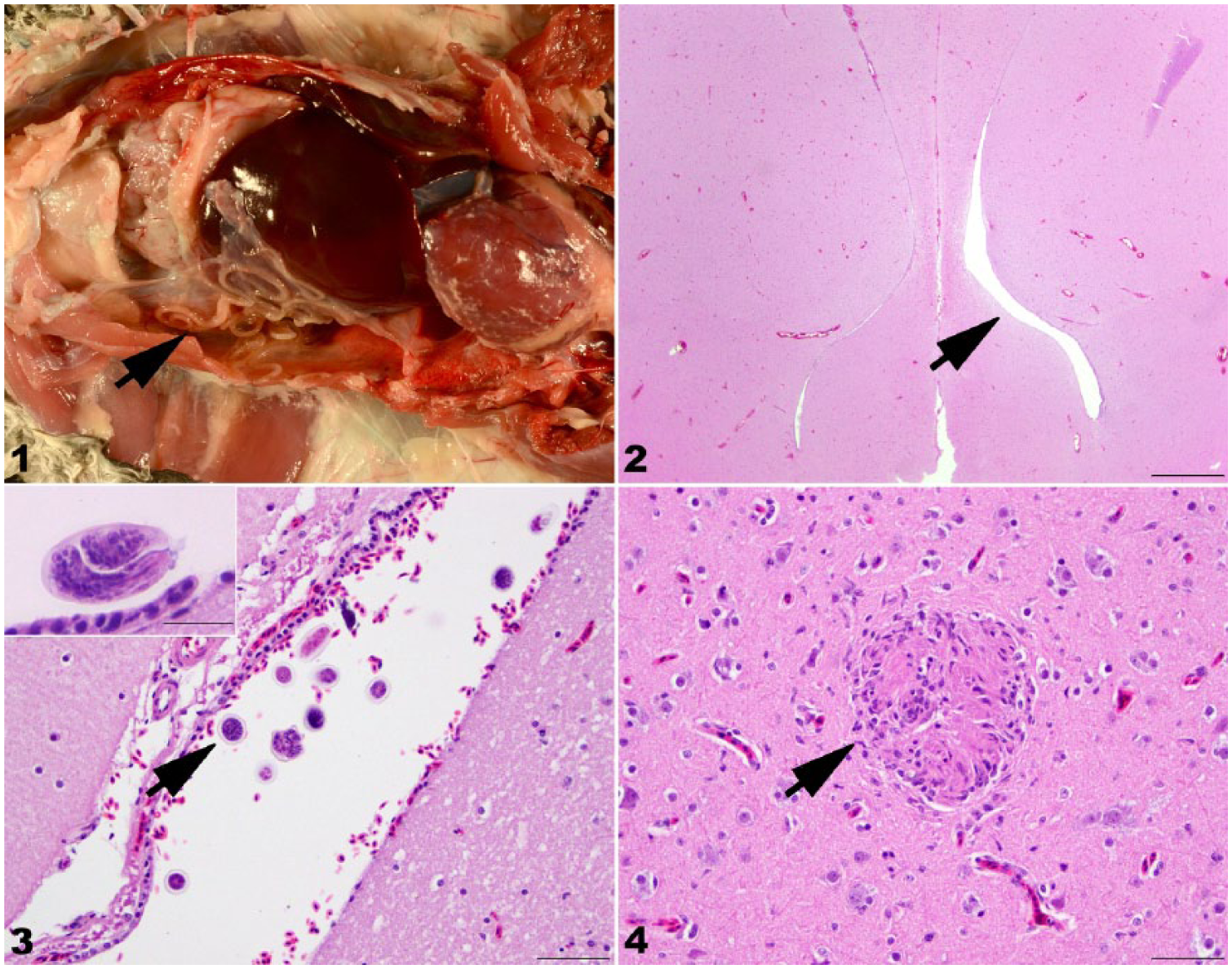
Serratospiculiasis in a prairie falcon (Falco mexicanus).
Upon trimming of the brain for histopathology, it was noted that the right lateral ventricle was mildly dilated up to 1.5 mm across (unilateral internal hydrocephalus). No other significant macroscopic changes were observed.
On microscopic examination, the wall of the left caudal thoracic air sac was diffusely, mildly widened by moderate numbers of infiltrating lymphocytes and plasma cells. Longitudinal and cross-sections of nematodes were present within the air sac lumen. The nematodes were characterized by a thick cuticle, lateral chords, coelomyarian–polymyarian musculature, a digestive tract lined a single layer of cuboidal epithelial cells, and gravid uteri containing thick-shelled, embryonated eggs. In the brain, the right ventricle was mildly dilated and contained a few nematode eggs (Figs. 2, 3). One nematode cross-section was observed in the meninges. Within the cerebral neuropil were a few small scattered glial nodules (Fig. 4). No significant microscopic lesions were observed in other tissues.
The nematodes were grossly and microscopically consistent with Serratospiculum sp. or Serratospiculoides sp. The 18S region was amplified by PCR to confirm the nematode species. 10 The 18S ribosomal RNA (rRNA) gene amplicons were sequenced (Eurofins Genomics, Louisville, KY), and amplicon forward and reverse sequences were visually scanned for quality and miss-calls (BioEdit, http://www.mbio.ncsu.edu/BioEdit/bioedit.html) to view the chromatograms and select contiguous sequences. The resulting sequence was compared to those in GenBank (National Center for Biotechnology Information, Bethesda, MD) and the European nucleotide archive (European Bioinformatics Institute, Hinxton, UK) using BLASTN. The amplicon showed 99% nucleotide homology (1,532 of 1,548 base pairs) with Serratospiculum tendo 18S rRNA. A diagnosis of respiratory serratospiculiasis with central nervous system (CNS) aberrant migration and associated unilateral internal hydrocephalus was made.
Clinical signs of serratospiculiasis attributable to aberrant migration of this parasite have been reported once. Pelvic limb deficits in a prairie falcon were reported to be caused by Serratospiculoides amaculata adult nematode and embryonated eggs within the ventral subarachnoid space of the synsacral vertebral canal, and embryonated eggs within the lumen of synsacral meningeal blood vessels. 6 The authors speculated that the nematode could have reached this site via the diverticulum iliolumbale of the abdominal air sacs, or alternatively via aberrant vascular larval or subadult migration. Chronic, asymmetric myelitis and Wallerian degeneration of the dorsal funiculi of the distal thoracolumbar and proximal synsacral spinal cord, along with the presence of embedded embryonated eggs within the white matter, supported the possibility of previous migration of the adult nematode through the white matter. In another prairie falcon, embryonated eggs were noted within the proventricular wall, pulmonary lymphatic vessels, and hepatic veins, and their intravascular location suggested vascular migration. 4
In our case, changes within the air sac were consistent with typical subclinical serratospiculiasis. The presence of a nematode and eggs within the meninges, eggs within the ventricular system, and mild unilateral hydrocephalus were unusual, and suggest the possibility that the nematode or eggs impaired cerebrospinal fluid outflow and led to unilateral hydrocephalus. How the nematode reached the meninges is unknown, but the presence of a few small foci of gliosis in the cerebrum suggests that an adult nematode or nematode larva may have migrated through the brain.
Opisthotonos has been reported previously in falcons and tentatively attributed to thiamine deficiency, as some affected birds respond to thiamine supplementation.11,18 However, the falcon in our report was supplemented with thiamine after the onset of clinical signs without clinical improvement, and the autopsy did not reveal any lesions supportive of thiamine deficiency. 16 Therefore, opisthotonos is tentatively attributed to the unilateral hydrocephalus.
The 9 species in genus Serratospiculum are classified based on the length of their spicules.4,12,20 One species, Serratospiculum amaculata, was reclassified as Serratospiculoides amaculata, and is the only species reported to infect falcons in North America.4,12 Partial sequencing of the 18S rRNA of the nematode in our case showed 99% homology with Serratospiculum tendo. The availability of molecular data to identify the species of Serratospiculum is unfortunately limited to the 18S rRNA gene region of S. tendo, and it remains uncertain if the nematodes were Serratospiculoides amaculata or Serratospiculum tendo. Nematode identification based on morphologic features was attempted but inconclusive, given that spicules were not observed in the 6 specimens examined. Morphologic identification of these closely related nematode species can be difficult, and molecular data are presently scarce. 7 Although rare, aberrant migration of Serratospiculum spp. or Serratospiculoides spp. into the CNS should be included in the differential diagnosis of neurologic diseases in falcons.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors prepared this article as part of routine duties of employment and received no outside financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
