Abstract
In an experimental model of transplant rejection, renal transplants were performed on 6 mixed-breed dogs. Capecitabine (CPC) was administered as an oral immunosuppressive agent. All recipients received systemic CPC, cyclosporine (CSA), prednisolone, and famotidine throughout the study. Two dogs developed superficial keratitis, which was characterized by multifocal geographic erosions, superficial corneal epithelial pigmentation, and corneal neovascularization. These clinical signs correlated with the dose of CPC given, whereas other drug doses remained unchanged. After euthanasia, routine histologic sections were stained with hematoxylin and eosin and with alcian blue periodic acid— Schiff for light microscopic evaluation. Ocular histopathologic abnormalities were limited to neovascularization and inflammatory infiltrate of the anterior corneal stroma and abnormal basal cell morphology, disorganization, thinning, and pigmentation of the corneal epithelium. The purpose of this communication is to describe the clinical and histopathologic evidence of CPC corneal toxicity in dogs.
Capecitabine (CPC), an oral antineoplastic and immunosuppressive agent, is a prodrug that is converted to 5-fluorouracil (5-FU) in vivo (Roche, Pharmaceuticals: Xeloda. Product information. http://www.rocheusa.com/products/xeloda/, 2005). CPC is most commonly administered as a chemotherapeutic agent in a variety of human cancers. 7 Adverse effects have included hyperexcitability, altered mentation, seizures, gastric ulceration, and lymphoplasmacytic enteritis. 4, 6 Like 5-FU, eye irritation with corneal deposits has been reported with the use of CPC in humans. However, the histopathology of CPC corneal toxicity has not been described (Roche, Pharmaceuticals: Xeloda. Product information. http://www.rocheusa.com/products/xeloda/, 2005). 1, 7 CPC has also been used as an immunosuppressive agent to counteract canine renal transplant rejection. 4 Ocular adverse effects were noted in this renal study and led to an examination by a veterinary ophthalmologist and veterinary ophthalmic pathologist. The results of those ophthalmic examinations are reported here.
The methods have been reported elsewhere. 4, 6 In brief, 6 adult, female, unrelated, purpose-bred research mongrels were used in a study that evaluated the effectiveness of CPC in preventing renal transplant rejection. Renal transplants were performed with the dogs under general anesthesia. All recipients received gradually increasing doses of CPC(5′-deoxy-5-fluoro-N-[pentyloxy]carbonyl)-cytidine, Xeloda, Hoffmann-La Roche Ltd., Basel, Switzerland), from the day of surgery through the end of the study (50 mg/m2 PO twice a day [BID] to 200 mg/m2 PO BID), except when CPC was discontinued temporarily in 2 dogs because of ocular adverse effects. All recipients also received cyclosporine (CSA) (Cyclosporine oral solution [modified], Pliva Inc., East Hanover, NJ; 4 mg/kg PO BID, adjusted to 250 ng/ml based on 12-hour trough levels), prednisolone (0.25 mg/kg PO BID), and famotidine (PEPCID, 0.5 mg PO once a day) beginning 2 days before surgery. All protocols were approved by the University of Wisconsin-Madison Institutional Animal Care and Use Committee.
Blepharospasm and conjunctival hyperemia were noted in 2 dogs 6 to 8 weeks after surgery and led to an ophthalmic examination on all dogs by a veterinary ophthalmologist, including fluorescein staining, slit-lamp biomicroscopy, funduscopy, Schirmer tear test, tear film break-up time, and tonometry. Ophthalmic examinations in dog Nos. 1 and 2 were performed at least once weekly after the onset of clinical signs. In dogs that did not show clinical evidence of ocular toxicity, ophthalmic findings were recorded at least once (after approximately 12 weeks of continuous treatment with CPC).
Superficial keratitis in 2 dogs (dog Nos. 1, 2) was characterized by multifocal geographic epithelial erosions and rapid, superficial corneal epithelial pigmentation (seeFigs. 1,2). Over the subsequent weeks, both dogs developed unilateral corneal neovascularization and associated mild corneal edema. Schirmer tear tests were borderline low in 1 dog (dog No. 1, ranging from 12 to 15 mm of wetting/60 seconds) and normal in dog No. 2 (>15 mm/60 seconds) over the 6-week time period. 2 Dog No. 1 developed a unilateral, axial, superficial crystalline corneal deposit in 1 eye 8 weeks after clinical signs were initially noted (seeFig. 2). Both dogs showed improvement when CPC was discontinued for 7 to 14 days; clinical signs (conjunctival hyperemia, fluorescein positivity) recurred within 1 week after CPC was reinstituted. CPC had been (re)administered for at least 1 week before euthanasia and necropsy in both dogs. Ophthalmic findings were normal in dog Nos. 3–6 except for slightly decreased tear break-up times in several dogs. 2 Schirmer tear testing in clinically normal dogs treated with CPC ranged from 14 mm/minute (1 eye) to 25 mm/minute.
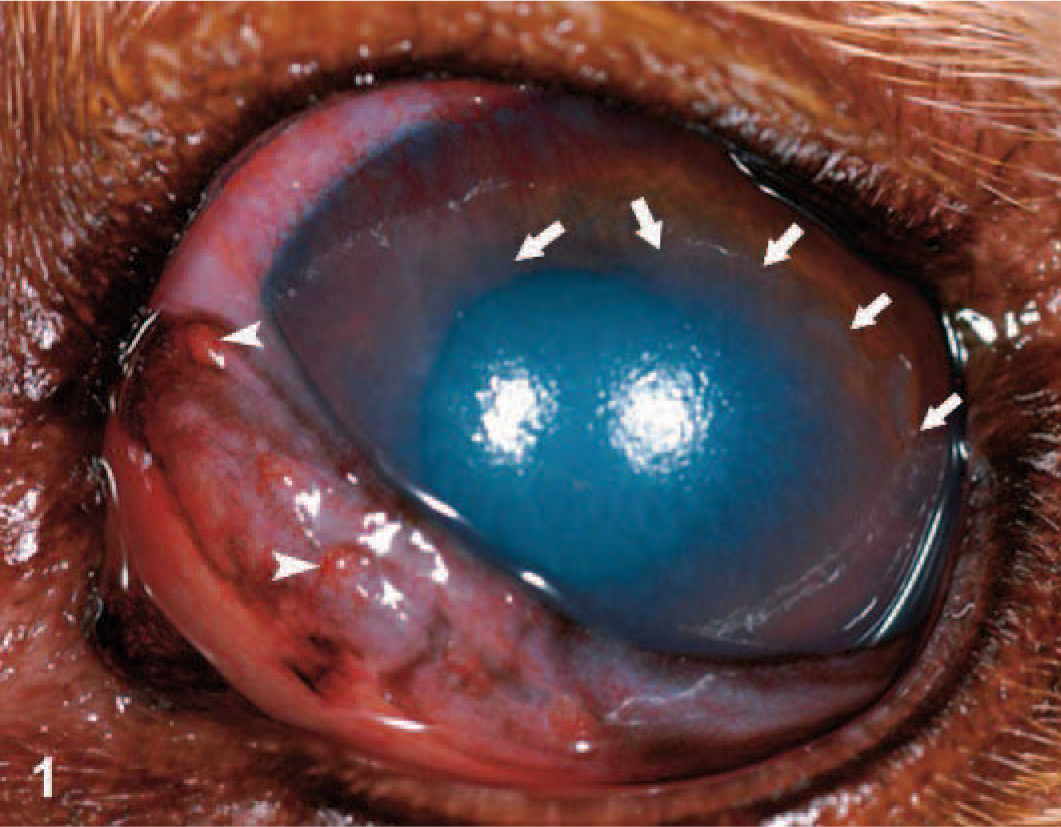
Canine eye (oculus sinister); dog No. 1: Corneal irregularity, corneal edema, and hyperemia of nictitans with follicle formation are evident. White arrows indicate the edge of corneal epithelium; the cornea was fluorescein positive ventral to these arrows. Arrowheads are directed toward conjunctival follicles.
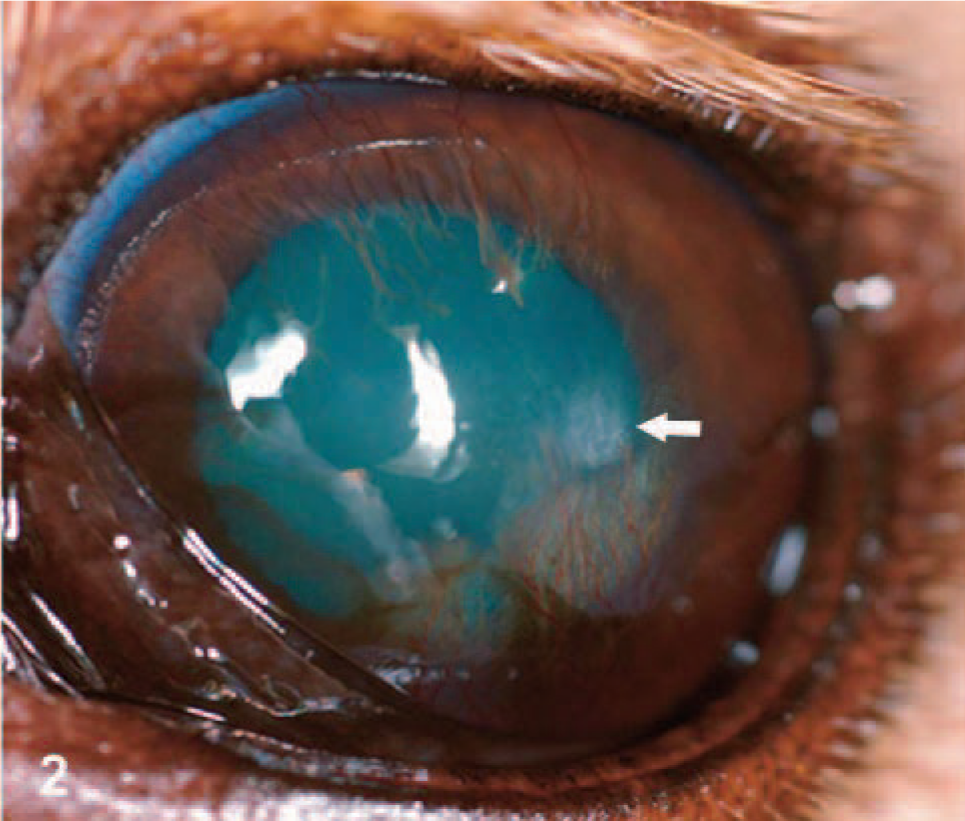
Canine eye (oculus sinister); dog No. 1: Six weeks after Fig. 1. After CPC was discontinued, corneal healing was characterized by neovascularization, pigmentation, and an axial, superficial crystalline corneal deposit. The white arrow is directed at the corneal deposit. Neovascularization and pigment are present ventral to the deposit.
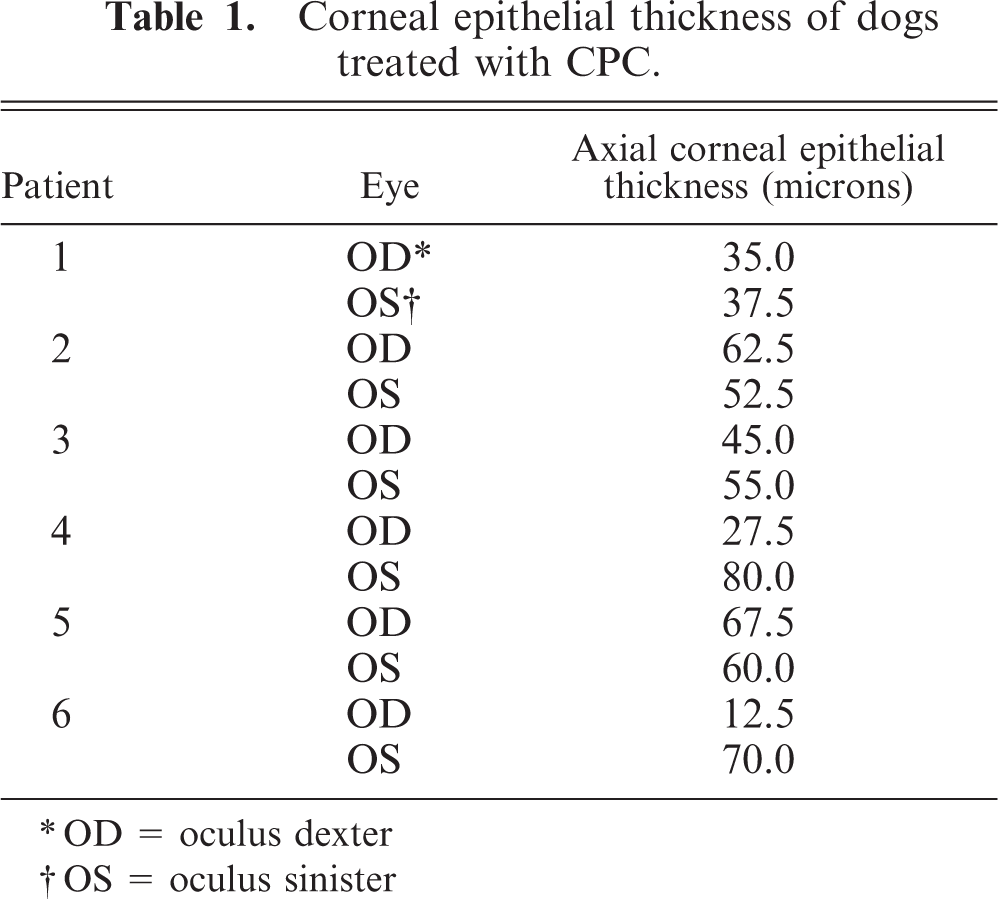
At necropsy (14 weeks after surgery), Davidson's fixed globes (2 parts 37% formalin, 3 parts alcohol, 1 part glacial acetic acid, 3 parts tap water) were vertically cut, paraffin-embedded, sectioned 5 microns thick, and stained with hematoxylin and eosin and with alcian blue periodic acid–Schiff for light microscopy. Corneal histopathology and axial corneal epithelial thickness from 5 normal young adult Beagle dogs were examined for comparison. Axial corneal epithelial thicknesses in normal controls ranged from 57 to 65 microns. Reported normal corneal thicknesses range between 25 and 40 microns for carnivores. 2 Ocular histopathologic abnormalities in affected dogs were limited to the corneal epithelium and the anterior corneal stroma. Epithelial abnormalities noted from histopathologic sections consisted of sparse pigmentation (dog No. 1), abnormal basal cell morphology and epithelial disorganization (dog Nos. 1–4, 6) and epithelial thinning (Table 1,Figs. 3–6). Anterior stromal abnormalities consisted of anterior stromal neovascularization, fibroblast accumulation, and mild lymphoplasmacytic inflammatory infiltrate in dog Nos. 1 and 2. The only histopathologic finding that could be associated with the crystalline deposit in dog No. 1 was clusters of reactive fibroblasts within the superficial corneal stroma. Histopathologic abnormalities in clinically normal dogs treated with CPC were limited to epithelial thinning and epithelial disorganization.
Corneal epithelial thickness of dogs treated with CPC.
OD = oculus dexter
OS = oculus sinister
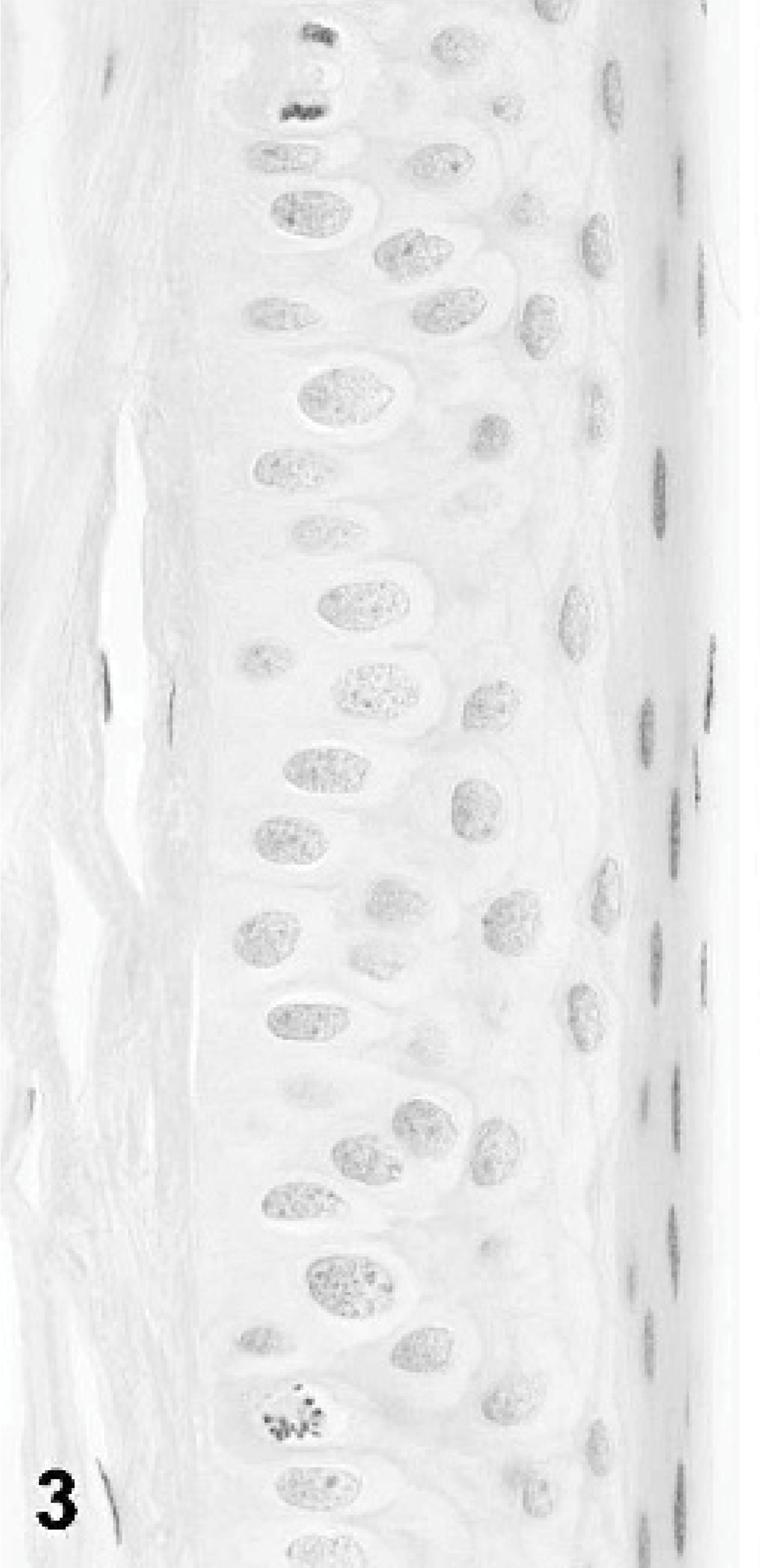
Canine anterior cornea (oculus sinister); dog No. 3. This dog was clinically normal and histopathologically exhibited only mild epithelial disorganization. In this image, the epithelium is relatively normal. HE.

Canine anterior cornea (oculus dexter); dog No. 1. Abnormal basal cell morphology, epithelial pigmentation, and epithelial thinning are evident. HE.
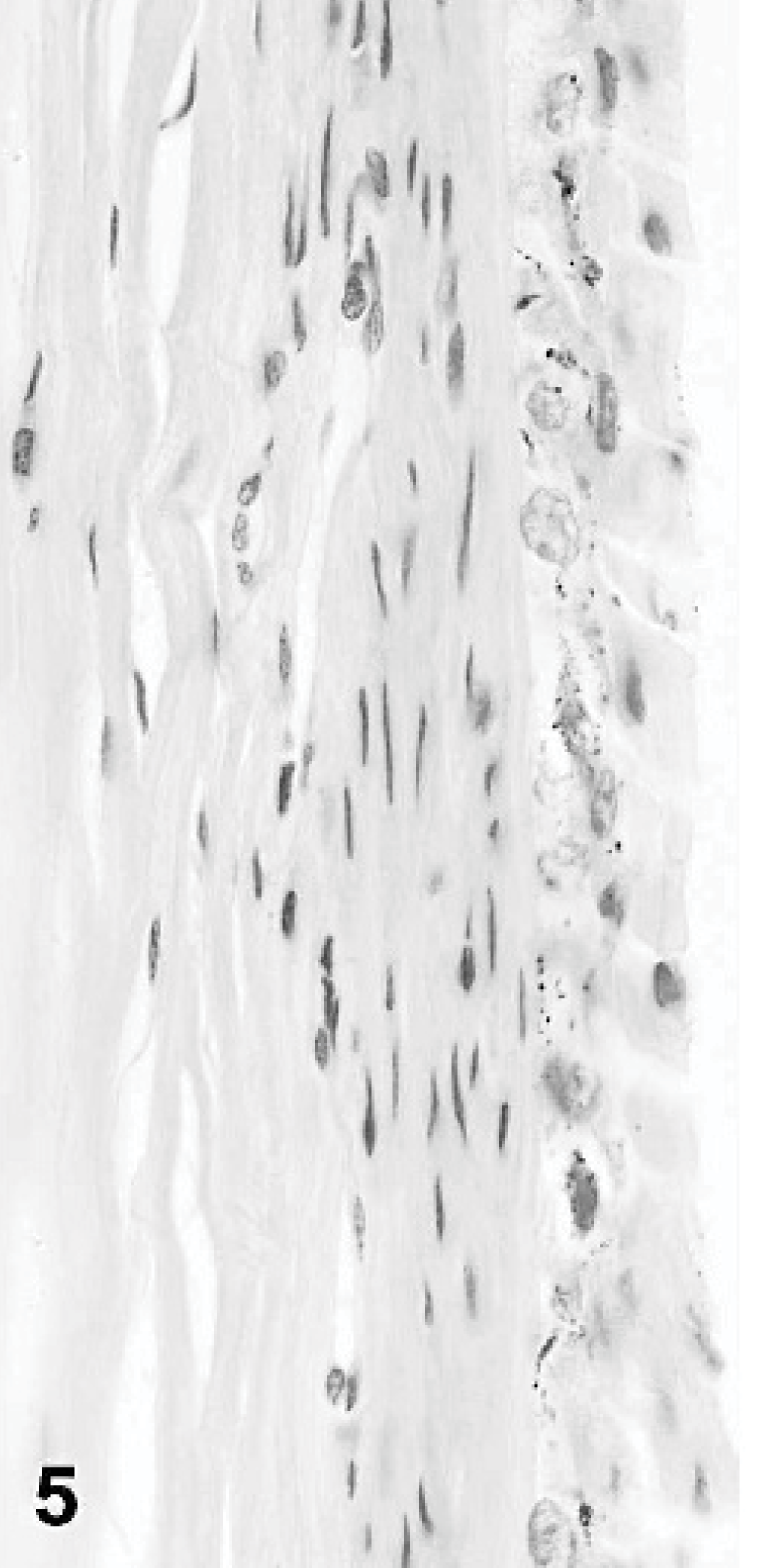
Canine anterior cornea (oculus sinister); dog No. 1. Epithelial thinning and anterior stromal inflammatory infiltrate are evident. HE.

Canine anterior cornea (oculus sinister); dog No. 2. Marked epithelial thinning is present. HE.
The mechanism of action of 5-FU and CPC ocular toxicity is not fully understood. In humans, fluorouracil can be measured in tears after intravenous injection. It has been shown in humans that the effects of CPC vary greatly, an effect that may relate to individual susceptibility, drug dosage, or to enzymatic deficiencies that promote serious adverse systemic effects. 5 In both these dogs and in human patients, ocular toxicity of CPC is ameliorated by the discontinuation of the drug. 7 Further study is needed to investigate the presence of CPC in tears, the mechanism of toxicity, the presence/involvement of limbal stem cells, and possible treatments to prevent or ameliorate CPC toxicity. The limitations of this study include a limited number of dogs and the administration of multiple medications throughout the study. Although systemic famotidine, CSA, and prednisolone were given in this study, CSA and famotidine are widely used in dogs without clinical evidence of ophthalmic toxicity. Although prednisolone may cause delayed corneal wound healing, it does not generally cause clinical keratopathy. 1 Decreased tear break-up times suggest a qualitative tear film deficiency that may be related to abnormalities in conjunctival goblet-cell function. 2 The importance of qualititative/quantitative tear film abnormalities or blinking frequency (not measured) in this study cannot fully be quantified. Although the dogs used in this study were raised solely for research purposes in pathogen-free conditions, another possible etiology that cannot be excluded is canine herpesvirus. 3 The clinical and histopathologic findings in this study and the clinical findings in previous human reports suggest that the primary toxic effects of CPC are on the corneal epithelium. 7 To the authors' knowledge, histopathologic evidence of ocular CPC toxicity has never been reported in any species. This study emphasizes the need for full necropsy with examination of ocular structures in all experimental studies.
Footnotes
Acknowledgements
Funding was received from the Comparative Ophthalmic Research Laboratories and the SVM Companion Animal Fund. None of the authors have any financial interest in any of the products used in this study.
