Abstract
To assess the safety of a potent muscarinic receptor antagonist (antimuscarinic) for human clinical testing, repeat-dose oral toxicity studies were conducted in the Beagle dog. Treatment resulted in unexpected gastrointestinal system effects that were likely mediated by the exaggerated pharmacologic effects of the anti-muscarinic on intestinal motility. Dogs developed profound anorexia followed by severe bloody diarrhea, and at necropsy, the intestinal tract was distended, filled with red-tinged fluid, and the mucosa was reddened. Histologically, intestinal lesions consisted of severe epithelial necrosis and mucosal atrophy localized predominantly to the ileum and large intestine. Feces from dogs with diarrhea were culture-positive for
Parasympathetic responses are mediated by the action of acetylcholine on muscarinic acetylcholine receptors (mAChRs) located on postganglionic parasympathetic nerves. Both agonists (parasympathomimetics) and antagonists (antimuscarinics) of the mAChRs are used medically in diverse settings. Antimuscarinics are effective agents for the induction of mydriasis during retinal examination, for the prevention of sinus bradycardia in surgical patients, for the treatment of spasmodic intestinal disorders and overactive bladder, and for the alleviation of bronchoconstriction in chronic obstructive pulmonary disease. 2,4,8,9
Because of the broad tissue distribution of mAChRs, there are several well-characterized examples of exaggerated pharmacology that are expected in preclinical studies with animals given antimuscarinics. 3,5,8,9 These include tachycardia, decreased salivation and lacrimation, dysuria, and intestinal atony and are an exaggeration of the desired clinical effect (e.g., dysuria is an exaggerated version of improved control for overactive bladder). Pharmacologically mediated side effects may limit the clinical utility of the antimuscarinic and may result in discontinuation of their use by patients.
To assess the safety of a potent antimuscarinic (PNU-171990A) for human clinical testing, an oral toxicity study was conducted in the Beagle dog. The antimuscarinic was given to 4 dogs/sex/group once daily orally through gastric gavage for 17 days at doses of 0, 30, 60, and 120 mg/kg/day. Toxicity was assessed by evaluating changes in clinical signs, body weight, food consumption, electrocardiograms, ophthalmology, hematology, clinical chemistry, and urinalysis. A complete necropsy was conducted on all dogs at the end of the dosing period, selected organs were weighed, and tissues were evaluated histologically. Feces were collected using anaerobic culturettes from three high-dose dogs that had bloody diarrhea. Aerobic cultures were established using MacConkey for
Clinical signs in dogs given 30 or 60 mg/kg/day were considered to be the result of the pharmacology of mAchR antagonism and included sedation and decreased activity at ≥30 mg/kg/day and increased heart rate, unsteady gait, tremors, mydriasis, and difficult urination at ≥60 mg/kg/day. In addition to the above clinical signs, dogs given 120 mg/kg/day had diarrhea with or without blood. The diarrhea was first observed on day 14 of the study and was preceded by markedly decreased food consumption, approximately 50%. Dogs that developed diarrhea eventually refused to eat dog chow and were provided canned food.
Blood was collected for hematologic and biochemical analyses pretest and on day 15. Hematologic and clinical chemistry findings were generally restricted to dogs with diarrhea. Hematologic findings included increased platelets, decreased relative and absolute reticulocytes, and increased prothrombin time and activated partial thromboplastin time. Bone marrow in histologic sections and smears prepared at necropsy was characterized by an increased myeloid to erythroid (M : E) ratio. The increased M : E ratio was because of a marked decrease in erythroid precursors instead of an increase in myeloid lineages. The bone marrow hypocellularity, increase in M : E ratios, and decrease in peripheral reticulocytes were judged to be at least partly a result of profound anorexia and weight loss because high-dose dogs without anorexia and weight loss did not have bone marrow or peripheral reticulocyte changes. Increased platelet counts, only observed in dogs with bloody diarrhea, may have been a consequence of increased release from bone marrow and splenic stores as a consequence of intestinal blood loss.
Clinical chemistry changes were consistent with hepatocellular membrane leakage (alanine aminotransferase and aspartate aminotransferase increases), and intestinal malabsorption and inflammation (decreased albumin and globulins and increased fibrinogen) in high-dose dogs with diarrhea. Alkaline phosphatase and γ-glutamyltransferase were increased moderately in dogs that were anorectic and had diarrhea, consistent with cholestasis. Histologic evidence of minimal cholestasis was seen in only one high-dose dog. No hepatocellular necrosis was observed; hepatocytes in anorectic dogs contained large, clear vacuoles, morphologically consistent with lipid. The hepatocellular vacuolation was likely secondary to anorexia.
The principal light microscopic finding attributed to treatment with the antimuscarinic was limited to high-dose dogs. These dogs had necrotizing inflammation of the ileum, cecum, and colon. In severely affected dogs, lesions were diffuse, more prominent in the large intestine, and characterized by necrosis of the epithelium lining the villi, crypts, and/or glands with subsequent attenuation of epithelium and decreased crypt and gland density. Attenuated and atypical epithelial cells lined intact crypts and glands, both of which were plugged with mucus, neutrophils, and cell debris (Figs. 1, 2). Focal areas of the superficial mucosa were effaced by dense aggregates of neutrophils, and the intestinal mucosa was congested without hemorrhage. In less severely affected dogs, the lesions were multifocal, and inflammation predominated over necrosis. Mucus admixed with gram-positive rods and gram-negative bacteria was observed in affected areas of the ileum and large intestine; however, bacterial colonization of the intestinal epithelium was absent. Feces collected from two dogs demonstrating clinical signs of bloody diarrhea were positive (3+ growth) for CP or bacilli consistent with CP. The fecal sample from one of these dogs also had
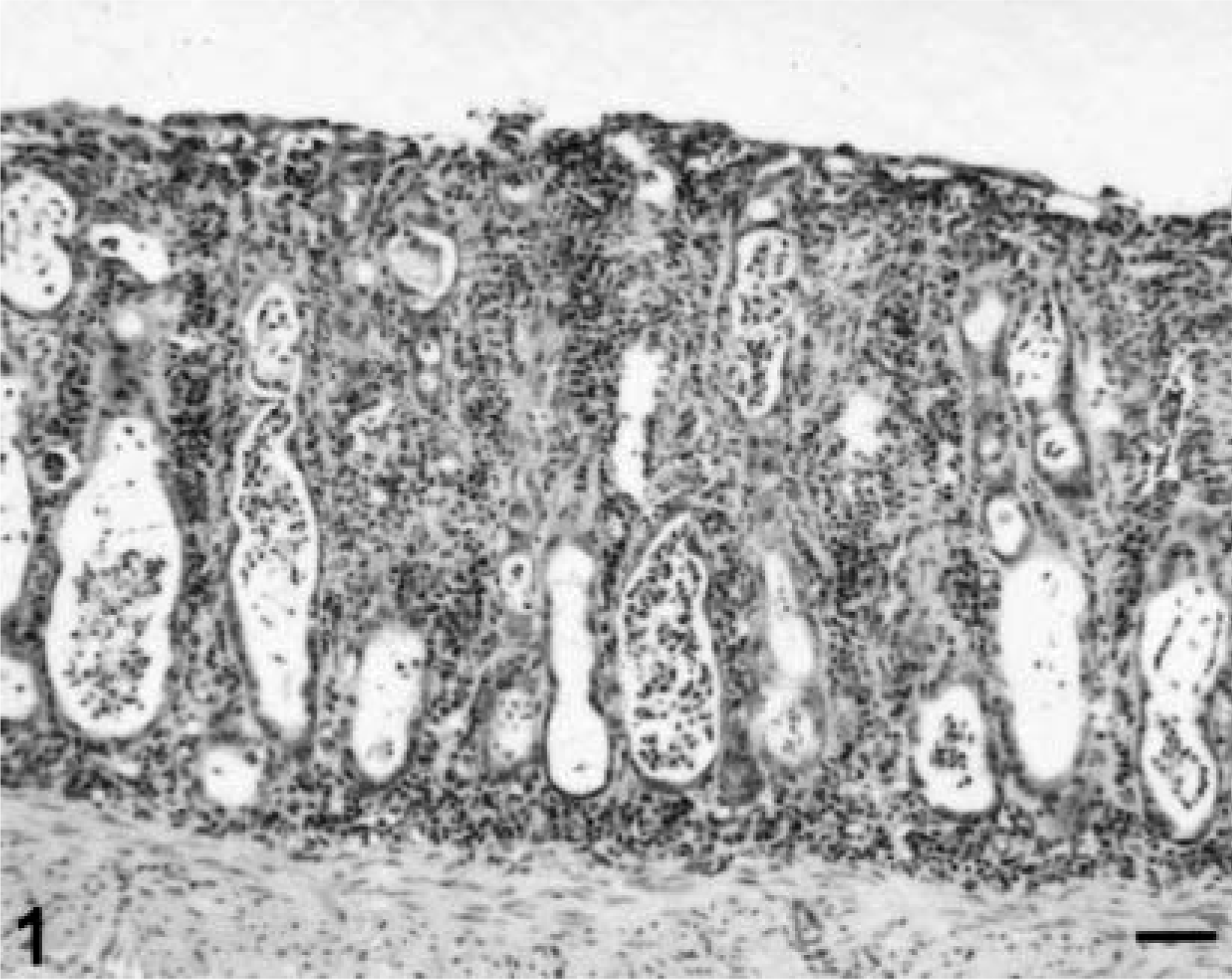
Colon; Beagle dog, high-dose. The mucosa is characterized by an attenuated superficial epithelium and decreased number of dilated glands filled with neutrophils and cellular debris. The lamina propria is hypercellular. Hematoxylin, phyloxine, and eosin. Bar = 70 µm.
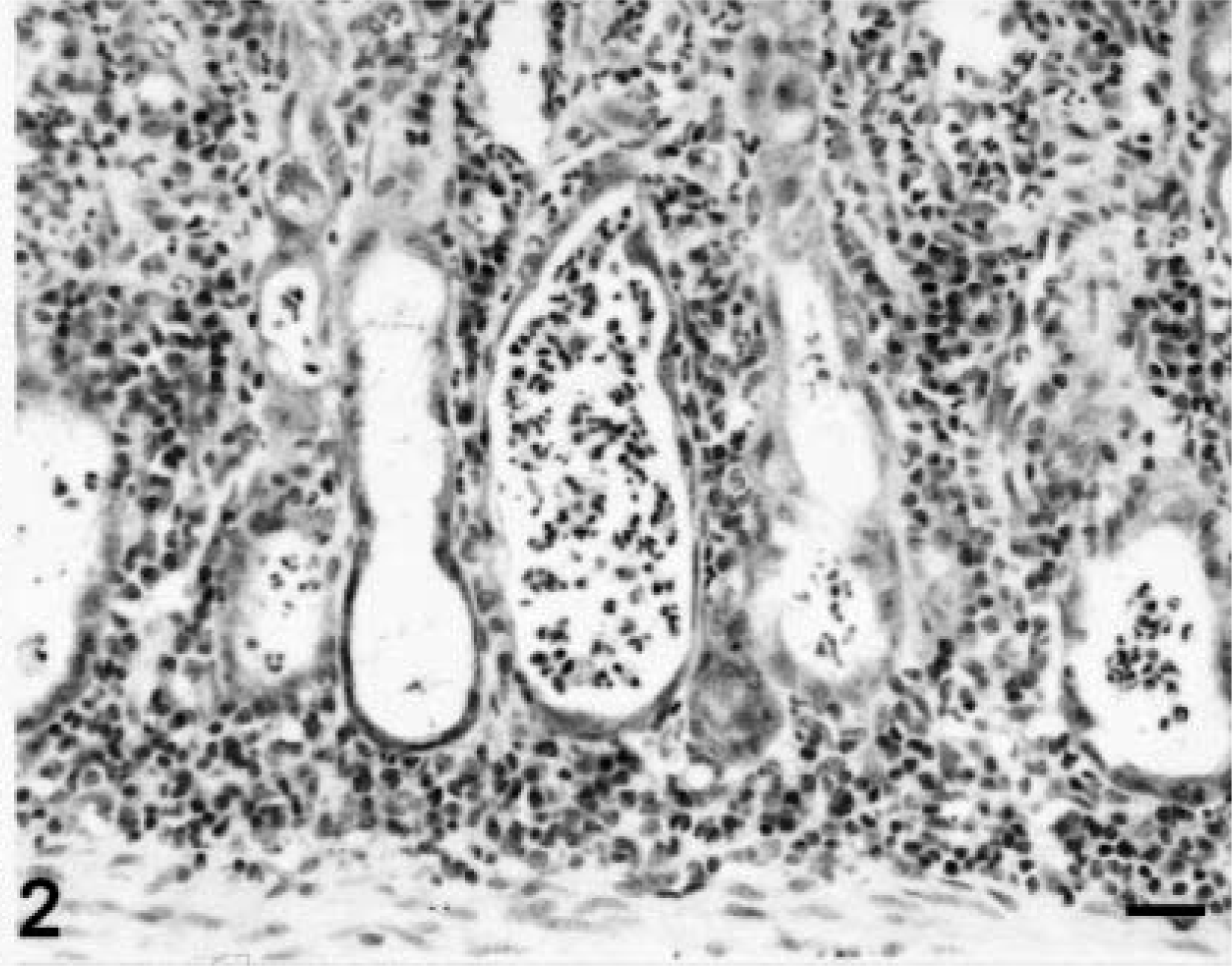
Colon; Beagle dog, high-dose. Glands are markedly dilated, lined by attenuated epithelial cells, and filled with neutrophils and cellular debris. The lamina propria has increased numbers of lymphocytes and plasma cells. Hematoxylin, phyloxine, and eosin. Bar = 30 µM.
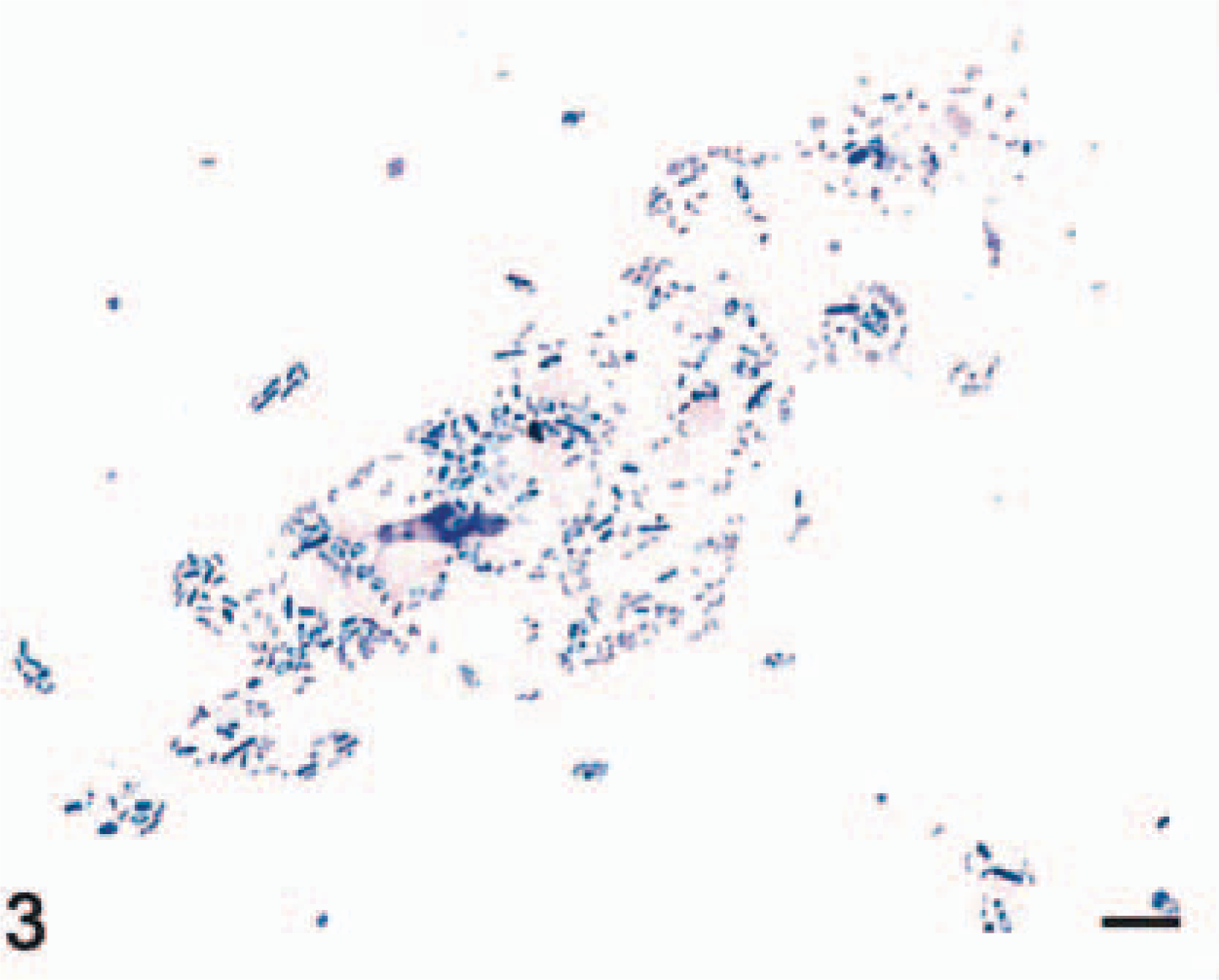
Fecal Smear; Beagle dog, high-dose. Gram-positive bacterial rods and spores are present. Gram's stain. Bar = 30 µM.
Antimuscarinic effects in the central nervous system, heart, salivary gland, intestinal smooth muscle, and urinary bladder were expected in this study. At doses given to the dogs, the plasma drug levels of PNU-171990A were large multiples of the muscarinic receptor binding affinity (Ki) for guinea pig muscarinic receptors in the cerebral cortex (3.6 nM), heart (8.5 nM), and parotid gland (21 nM). Decreased pupillary responses to light and/or mydriasis and heart rate increases were the most sensitive indicators of the antimuscarinic effects of PNU-171990A in dogs.
Clinical signs of bloody diarrhea, positive bacterial cultures for CP, and histologic findings of necrotizing inflammation in the ileum and large intestine suggested that affected dogs had clostridial infections secondary to drug treatment. However, the dense bacterial colonization, colonic coagulation necrosis, and hemorrhage reported in dogs with canine gastrointestinal hemorrhage syndrome caused by CP were not observed in this case.
1,7
Aerobic cultures did not identify any other potential bacterial pathogens (
Persistent decreases in intestinal motility caused by the effects of this antimuscarinic likely precipitated the clinical disease in these dogs. CP is a normal enteric bacterium that under certain conditions can proliferate and cause clinical disease. These conditions include stress associated with hospitalization and surgery, changes in intestinal function because of sudden modifications in diet, altered local immunity, intestinal mucosal damage (i.e., parvoviral enteritis), and modified intestinal flora (i.e., antibiotic therapy).
To our knowledge, the intestinal effects observed in dogs in this study have not been described with other antimuscarinics. 3,5 Antimuscarinics have well-known effects on the gastrointestinal tract including decreased gastric acid secretion and gastrointestinal motility. 8,9 Exaggerated pharmacologic effects on the gastrointestinal tract that occurred with high doses of this antimuscarinic likely created a microenvironment suitable for proliferation of pathogenic bacteria and the clinical syndrome observed in these dogs.
Footnotes
Acknowledgements
We thank Gary Cockerell and Steve Vonderfecht for their review of the manuscript.
