Abstract
An enucleated left eye from a 15-year-old female spayed Labrador Retriever was received by the Comparative Ocular Pathology Laboratory of Wisconsin (COPLOW) for histopathologic evaluation. Routine histologic preparation included staining with hematoxylin and eosin, and with alcian blue periodic acid-Schiff (PAS). At necropsy 9 months later, all grossly abnormal tissues (ipsilateral orbit and lung) were submitted to the COPLOW for histopathologic evaluation. Histopathologic evaluation of the globe revealed extensive invasion of the uvea and sclera by a pleomorphic cell population that formed disorganized cords and exhibited PAS-positive basement membrane material. Necropsy revealed a morphologically similar tumor in the ipsilateral orbit and lung. On immunohistochemical examination, the intraocular tumor stained diffusely immunopositive for vimentin, S-100, and neuron-specific enolase and multifocally, sparsely immunopositive for cytokeratin AE1/AE3. The orbital and thoracic tumors stained positively for vimentin but negatively for cytokeratin AE1/AE3. There are few reports of canine metastatic iridociliary adenocarcinoma in the literature; this is the first with immunohistochemical analysis.
Iridociliary adenomas and adenocarcinomas arise from pigmented or nonpigmented iris or ciliary epithelium, and are the second most common primary intraocular tumor of dogs (second only to melanocytic tumors). 4, 11 A review of canine iridociliary adenomas and adenocarcinomas by Peiffer 11 describes morphologic features and previous reports of these tumors.
Although extraocular epithelial tumors and intraocular metastatic epithelial tumors are usually immunopositive for cytokeratin and immunonegative for vimentin, both normal iridociliary epithelium and iridociliary adenomas are immunohistochemically positive for vimentin and negative for cytokeratin AE1/AE3. Iridociliary epithelial tumors are also characterized by S-100 and neuron-specific enolase (NSE) immunopositivity (Table 1). 5 The term iridociliary adenocarcinoma has been used for iridociliary epithelial tumors that are vimentin and cytokeratin AE1/AE3 positive, invade the sclera, and exhibit features of anaplasia. 5 A recent study found that expression of cytokeratins AE1 and AE3 was higher in scleroinvasive canine iridociliary adenocarcinomas compared with relatively well-differentiated iridociliary adenomas that more frequently expressed cytokeratin 20. 9 To date, no morphologic or immunohistochemical criteria have been associated with decreased survival in canine iridociliary tumors; however, there are few reports of unconfirmed 5 and confirmed metastasis. 6 Metastasis is likely a late-stage phenomenon and is extremely unlikely in the absence of extensive scleral invasion. 10 Enucleation is both diagnostic and most reliably curative in benign cases; surgical excision or evisceration carries the risk of recurrence. 1, 2
Cytokeratin, vimentin, NSE, and S100 immunopositivity of various tissues and intraocular tumors.∗

∗ + = immunopositive; − = immunonegative
† Information listed in rows 1–3 is derived from the experience of the author (RRD) and the veterinary literature (from Ref. 5); immunohistochemistry in rows 4–6 was performed in this study.
The purpose of this report is to describe the histologic features of a canine iridociliary adenocarcinoma that exhibited marked histologic features of malignancy, deeply invaded uveal, scleral, and orbital tissues, and metastasized to the lungs.
An enucleated left globe from a 15-year-old female spayed Labrador Retriever was received by the Comparative Ocular Pathology Laboratory of Wisconsin (COPLOW) for histopathologic evaluation. After sectioning in the dorsoventral plane, the globe was embedded in paraffin, routinely sectioned (5-microns thick), and stained with hematoxylin and eosin (HE) and with alcian blue periodic acid–Schiff (PAS) for evaluation. After euthanasia 9 months later, a full gross necropsy was performed by the referring veterinarian. All grossly abnormal tissues (left orbit and lung) and the grossly normal right eye were submitted to the COPLOW for histopathologic evaluation. These tissues were routinely stained with HE and alcian blue PAS as described above. Immunohistochemistry for vimentin, cytokeratin AE1/AE3, S-100, and NSE was performed on all submitted tissues as previously described. Additional patient history was obtained directly from the referring veterinarian and the owner.
Four immunohistochemistry, paraffin-embedded tissues were cut at 4 microns; mounted on charged slides; deparaffinized; and hydrated for 3 minutes each in 3 xylene baths, 3 absolute alcohol baths, and one 95% alcohol bath. Slides were then rinsed in tap water and treated for 5 minutes at room temperature with 3% H2O2. Heat-induced epitope retrieval was used with a buffer at pH 6.0 at 97°C for 20 minutes in a vegetable steamer, then cooled outside the steamer for 20 minutes. The slides were then placed in phosphate-buffered saline solution (PBS, pH 6.0). On a Biogenex Optimax Plus auto stainer, slides were treated with BioGenex Power Block Universal Blocking Reagent (San Ramon, CA) for 10 minutes at room temperature to block nonspecific background staining. Slides were then incubated at room temperature for 30 minutes with the primary antibody (seeTable 1), PBS washed, incubated with biotinylated secondary mouse/rabbit antibody for 20 minutes at room temperature, then PBS washed again. Tissues were incubated with streptavidin-peroxidase conjugate at room temperature for 20 minutes, then PBS washed. Slides were treated with a 0.05% 3-3′-diaminobenzidine tetrahydrochloride solution with 1% H2O2 for 5 minutes. Slides were removed from the stainer and placed in tap water. Slides were then counterstained with Harris' hematoxylin for 2 minutes, dehydrated through progressive ethanol concentrations (50, 75, 95, and 100%) to xylene, and coverslipped (secondary antibody, streptavidin conjugate, and reagents from LSAB+Kit, Dako, Carpenteria, CA). Positive and negative controls accompanied all cases.
This 15-year-old patient had a history of 4 prior tumors. Histopathology of these revealed an oral epulis, a subcutaneous lipoma, an incompletely excised subcutaneous peripheral nerve sheath tumor with low metastatic potential, and a perianal gland adenoma that was completely excised. The patient also had a several-year history of unilateral cataract and a 6-month history of uveitis and secondary glaucoma in the left eye. Ultimately, an intravitreal gentamicin injection was performed in an attempt to control the glaucoma. Two months later, a circumferential thickening of the iris was noted clinically and precipitated enucleation.
On gross examination, upon sectioning, much of the globe was internally lined and infiltrated by a tan mass (Figs. 1, 2). A pleomorphic amelanotic cell population formed disorganized cords and exhibited PAS-positive basement-membrane material, consistent with an epithelial tumor. Neoplastic cells extended from the iris base amid iridal necrosis to infiltrate full thickness through the limbus. Neoplastic cells exhibited a large nuclear to cytoplasmic ratio, prominent single-to-multiple nucleoli, marked anisocytosis and anaplasia (Fig. 3). The mitotic rate was 8 in 10 high-power fields. The ocular tumor expressed diffuse vimentin immunopositivity and few intensely positive areas of cytokeratin immunopositivity, making up between 5 and 10% of neoplastic tissue (Figs. 4– 6) (Table 1). A collagen-rich neoplastic spindle-cell membrane filled the posterior chamber, cradled the lens, lined the inner aspect of the choroid, and infiltrated the sclera around the optic nerve. There was extensive lens capsular wrinkling in the absence of evidence for lens capsule rupture, consistent with hypermature cataract.
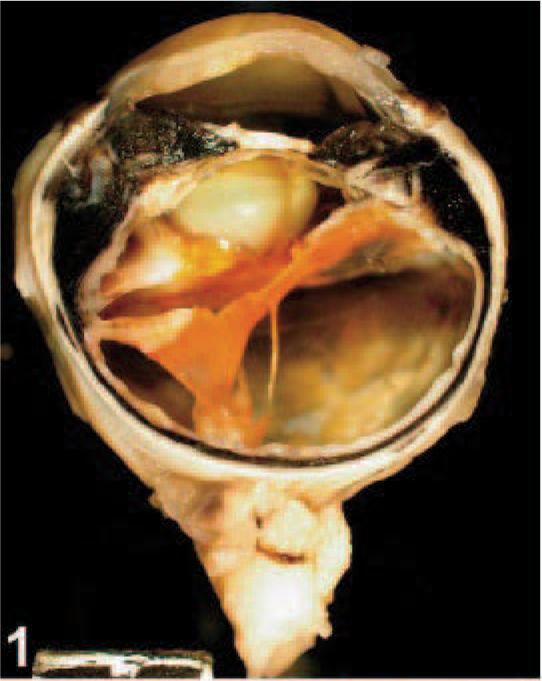
Eye; canine. Gross photograph. The globe was internally lined and infiltrated by a tan mass. Formalin fixation. Ruler = 1 cm.
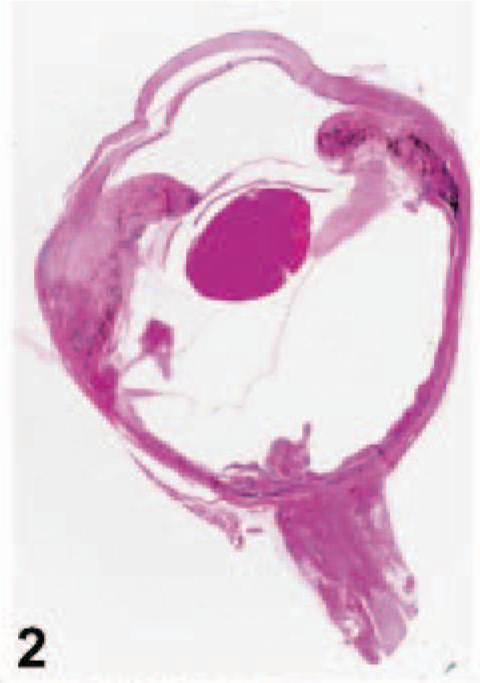
Eye; canine. Subgross photograph. HE.
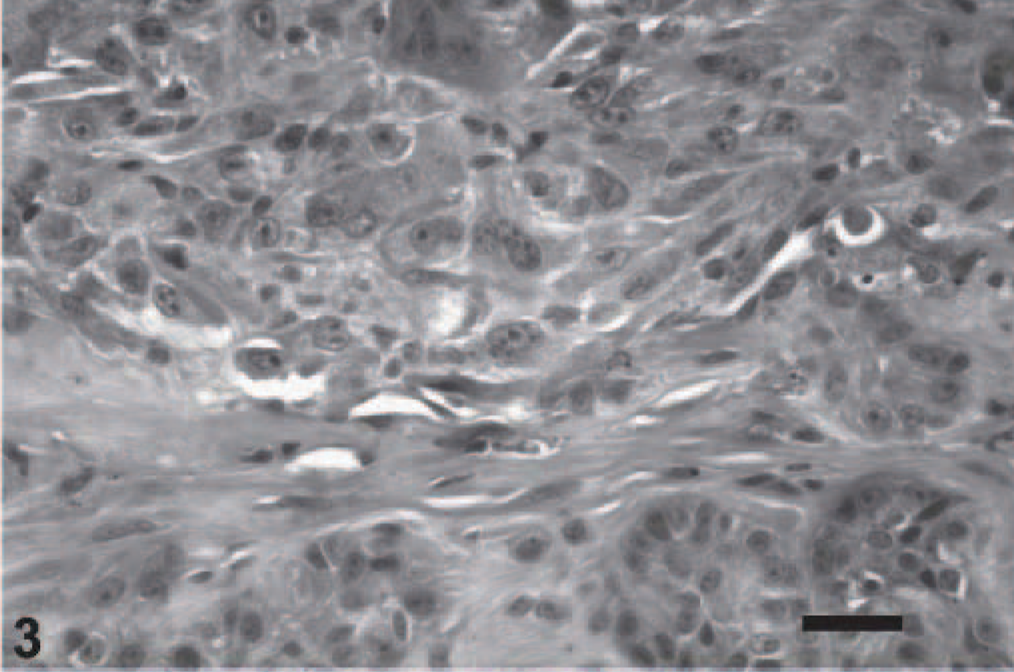
Uvea; canine. Anaplastic, pleomorphic neoplastic cells form invasive clusters, extending through the limbal sclera. HE. Bar = 50 microns.
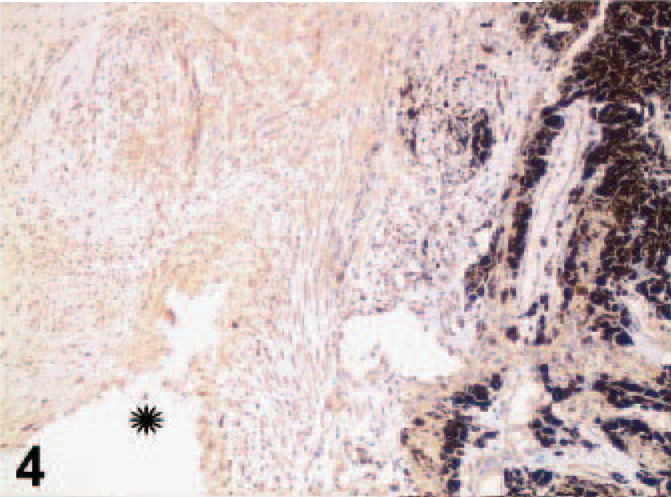
Uvea; canine. Vimentin, streptavidin-biotin indirect immunoperoxidase immunohistochemistry.
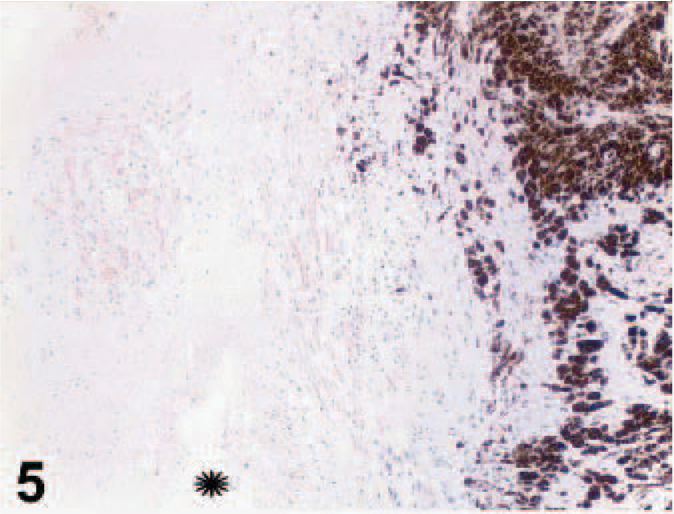
Uvea; canine. Same magnification asFig. 4. Cytokeratin AE1/AE3, streptavidin-biotin indirect immunoperoxidase immunohistochemistry.
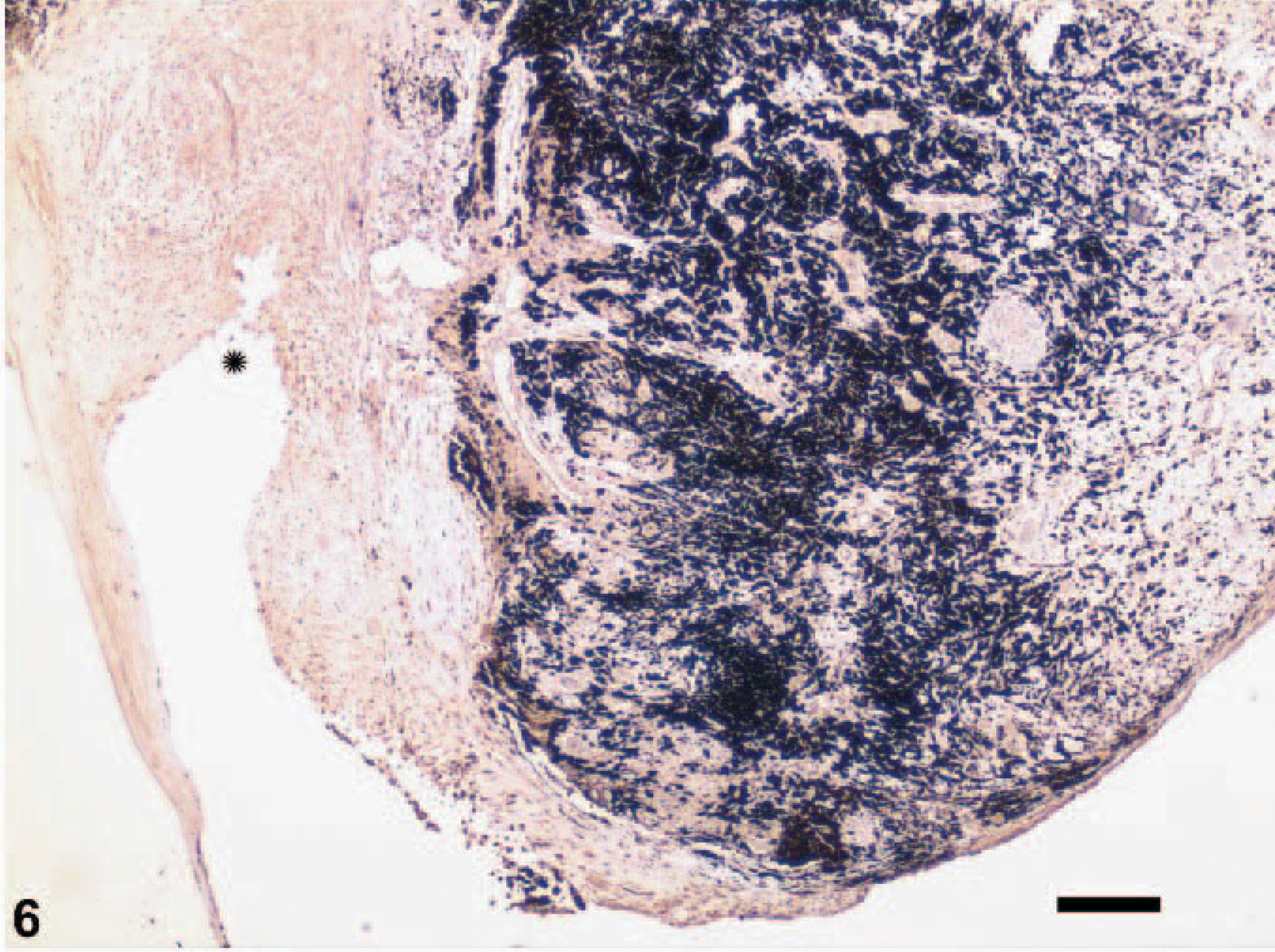
Uvea; canine. Neoplastic tissue extends from the pigmented iris (right) to the lens capsule (left). This image is at lower magnification thanFigs. 4 and 5, showing diffuse immunoreactivity of the tumor. The star is in the same anatomic location inFigs. 4– 6. Vimentin, streptavidin-biotin indirect immunoperoxidase immunohistochemistry. Bar = 150 microns.
Nine months after enucleation, the patient experienced lethargy and nasal discharge ipsilateral to the enucleation site. Thoracic radiographs revealed multiple pulmonary nodules. The patient was humanely euthanatized. At necropsy, all organs were within normal limits, except for the left orbit and lungs. The right eye was histologically normal. Orbital tissue was heavily infiltrated by an anaplastic neoplasm composed primarily of spindle-shaped cells. Orbital neoplastic cells were vimentin, S-100, and NSE positive and cytokeratin negative (Table 1). Multiple pulmonary nodules were within and surrounding blood vessels, consistent with a metastatic neoplasm (Fig. 7). These neoplastic cells were anaplastic and morphologically similar to the cells within the orbit (also vimentin, S100, and NSE positive and cytokeratin negative).
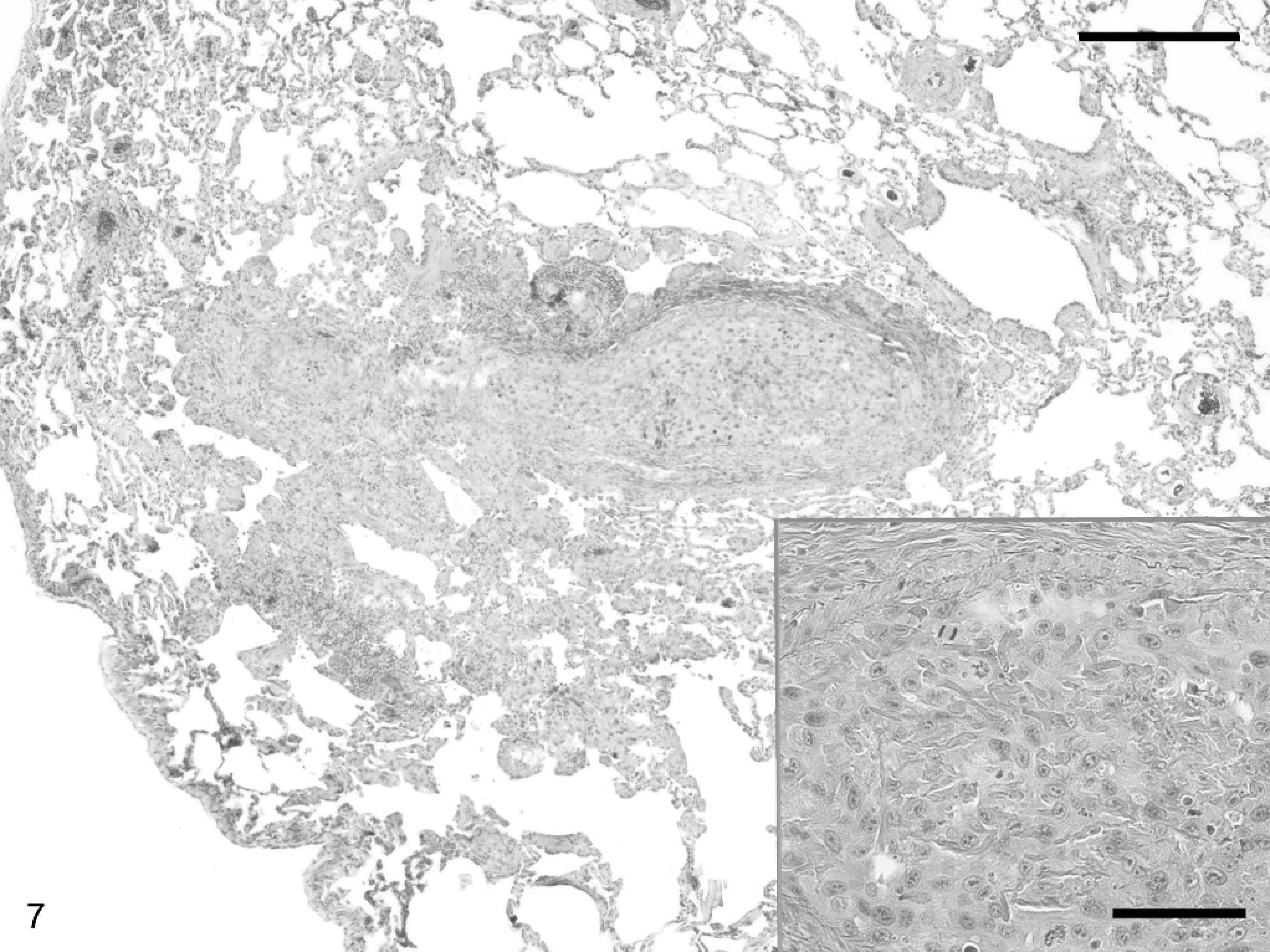
Lung; canine. Larger image is lung, inset at higher magnification. Multiple pulmonary nodules are present within and surrounding blood vessels. Neoplastic cells are morphologically similar to the intraocular tumor. HE. Bar (larger image) = 150 microns, Bar (inset) = 60 microns.
The distinction between iridociliary adenomas and adenocarcinomas is not clear within the human and veterinary literature. In humans, iridociliary epithelial tumors are much less common than in dogs, and histologic features of malignancy vary widely. In 1 study, human iridociliary adenocarcinomas with orbital extension metastasized to the parotid gland and distant bones. 10 A subset of human iridociliary tumors is associated with a history of trauma (including surgical trauma), intraocular inflammation, or phthisis bulbi. 3, 7
The only known incident of trauma in this case was a gentamicin intravitreal injection. The gentamicin-induced intraocular inflammation would have caused further compromise of the blood-ocular barrier and could have affected tumor morphology. In this case, the intraocular tumor showed sparse, strong, cytokeratin-immunopositive areas that were not maintained within the morphologically similar orbital or pulmonary tumors.
Extraocular epithelial tumors are generally immunopositive for cytokeratin and immunonegative for vimentin. However, rare epithelial tumors are vimentin positive, suggesting a potentially more aggressive, mesechymal-cell variant. 8 Loss of cytokeratin reactivity and gain of vimentin reactivity was reported in metastatic epithelial tumors as part of a malignant transformation. 12 The loss of cytokeratin immunoreactivity in the extraocular tumors in this case was most likely because of tumor cell anaplasia.
Footnotes
Acknowledgements
The authors would like to thank the owners of this patient, Dr. Kelly Carver and Dr. Jennifer Chilton, for allowing the necropsy, performing the necropsy, and perusing the manuscript. Immunohistochemistry was accomplished with the expertise of Lona K. Barsness, Anne Slattery, and Sandra Cheasty, HT (ASCP).
