Abstract
Sheep-associated malignant catarrhal fever (MCF) due to infection with ovine herpesvirus 2 (OvHV-2) is common in commercial herds of American bison (Bison bison). Inability to propagate OvHV-2 in vitro has been a constraint on experimental studies of the disease. We sought to establish whether nasal secretions from sheep that shed OvHV-2 might induce the disease in bison and to define a minimum challenge dose. Fourteen bison were nebulized with sheep nasal sections containing 103–107 OvHV-2 deoxyribonucleic acid (DNA) copies. Most challenged bison (11/14, 78.6%) developed clinical signs at 29–52 days postnebulization (DPN). The mean incubation time was 42.18 (±7.33 SD) DPN. Using real-time polymerase chain reaction, we detected OvHV-2 DNA in peripheral blood leukocytes at 21–31 DPN. All bison that developed MCF had antibodies against the MCF group viruses. Gross and histologic lesions were typical of the acute disease. There was no morphologic evidence of a dose-related difference in the severity or distribution of lesions. This is the first successful reproduction of MCF in bison using a nasal route of exposure. Experimentally challenged bison are more susceptible to MCF, compared with experimentally challenged domestic cattle in a previous experiment. Bison are a pertinent ruminant species in which the pathogenesis of the disease can be investigated.
Sheep-associated malignant catarrhal fever (MCF) is caused by infection with ovine herpesvirus 2 (OvHV-2). The disease causes heavy mortality in commercial bison. 2, 14, 20, 25, 26 Other species are susceptible, including domestic cattle, pigs, and cervid species. 4, 6, 19, 22, 23 MCF is distinctive in the range of species affected, its poorly understood pathogenesis, the multi-systemic nature of lesions, and the limited number of laboratory models available to study the disease. 22 No vaccine exists to protect against MCF. Control is directed toward management practices that limit exposure of clinically susceptible ruminants to sheep. 4, 15 Effective separation is often impractical in range operations in Western states and provinces of North America. 2, 14, 19, 25, 26
A better infection model for MCF using naturally susceptible species is needed to investigate immunologic and genetic strategies to control the disease. The inability to propagate OvHV-2 in vitro necessitated the use of alternative sources of virus for experimental infection studies. Experimental reproduction of MCF in domestic cattle and deer in the past required the use of large volumes of whole blood (300–1,000 ml) or tissues and/or cells from clinically affected animals with naturally acquired infection. 3, 5, 8, 17, 18, 21, 24 Although successful on multiple occasions, reproduction of MCF using blood is unpredictable in clinically susceptible species.
Domestic sheep, particularly lambs aged 6–9 months, intermittently shed large amounts of cell-free, enveloped OvHV-2 in nasal secretions. 9, 10 Epidemiologic studies suggest that natural infection occurs via aerosol. 2, 14, 22 We wondered whether pooled, frozen OvHV-2-rich nasal secretions might be used to induce infection and disease. Recent studies with sheep and cattle confirm that infection and disease can be established in this way using high doses of OvHV-2 (>109 and 108 OvHV-2 genome copies in cattle and sheep, respectively). 12, 27, 28 The work presented here was undertaken to establish whether aerosol exposure of American bison to OvHV-2 might induce MCF and, if so, to define a minimal disease challenge dose.
Materials and Methods
Animals
Sixteen intact male yearling (150–239 kg) American bison (Bison bison) were obtained from a commercial ranch in southeastern Wyoming. Four of these were used in experiment 1. Twelve were used in experiment 2. They were selected by screening groups of animals to identify bison that were seronegative for MCF viruses using a competitive inhibition ELISA (cELISA). 11 All were negative for OvHV-2 deoxyribonucleic acid (DNA) using a seminested polymerase chain reaction (PCR). 1, 13 Clinically healthy bison negative for MCF viral antibody and for OvHV-2 DNA were transferred to the Wyoming State Veterinary Laboratory. They were then tested for MCF viral antibody and OvHV-2 DNA on 2 (experiment 1) or 3 (experiment 2) consecutive samples collected 1 week apart before the challenge studies were performed. Animals were held in an outdoor pen and fed a native grass hay diet. The management of animals was in accordance with an approved animal care and use protocol.
Nasal secretion inocula
Details of nasal inoculum collection and preparation are described elsewhere. 12, 28 In experiment 1, nasal secretions were collected daily from OvHV-2-infected sheep and examined using real-time PCR to determine the viral genome copy number. Sheep whose nasal secretions contained ≥100,000 copies of viral DNA/2 μg total DNA were reswabbed within 5–6 hours of initial sampling. Nasal-secretion inocula were obtained from 2 sheep experiencing shedding episodes, stored in liquid nitrogen, and thawed once at the time of use. A pooled nasal-secretion inoculum that was previously characterized in experimental infection studies using sheep 12, 28 and cattle 27 was used in experiment 2. Nasal secretions were collected daily from OvHV-2-infected sheep experiencing shedding episodes. After secretions were diluted in phosphate buffered saline and clarified, chicken ovalbumen (5%) was added as a cryoprotectant. Secretions were divided and stored in liquid nitrogen. Five preparations from 4 sheep experiencing shedding episodes were combined to generate the pooled inoculum. This pool and its dilutions were frozen and stored in liquid nitrogen. A control inoculum was prepared from nasal secretions collected from OvHV-2-negative sheep.
Experimental inoculation
For nebulization, 1 end of a plastic tube was connected to a commercial nebulizer (Devilbiss Pulmo-Aide nebulizer, Sunrise Medical, Somerset, PA), and the other was inserted 5 cm into the nasal passage of each bison. In experiment 1, 2 bison (Nos. 1, 2) were each nebulized with secretions from a single sheep, containing 4.7 × 107 OvHV-2 genome copies (Table 1). The other 2 (Nos. 3, 4) were each nebulized with secretions from a second sheep, containing 2.7 × 107 viral DNA copies. In experiment 2, 10 animals (Nos. 5–14) were nebulized using pooled nasal secretions containing 1.0 × 103 (n = 2), 104 (n = 2), 105 (n = 2), 106 (n = 2), and 107 (n = 2) genome copies. Two remaining bison (Nos. 15, 16) were each nebulized with nasal secretions derived from OvHV-2-negative sheep. The control bison were held in the pen that contained the inoculated animals.
Clinical and laboratory findings in bison following intranasal inoculation with OvHV-2 containing sheep nasal mucus.
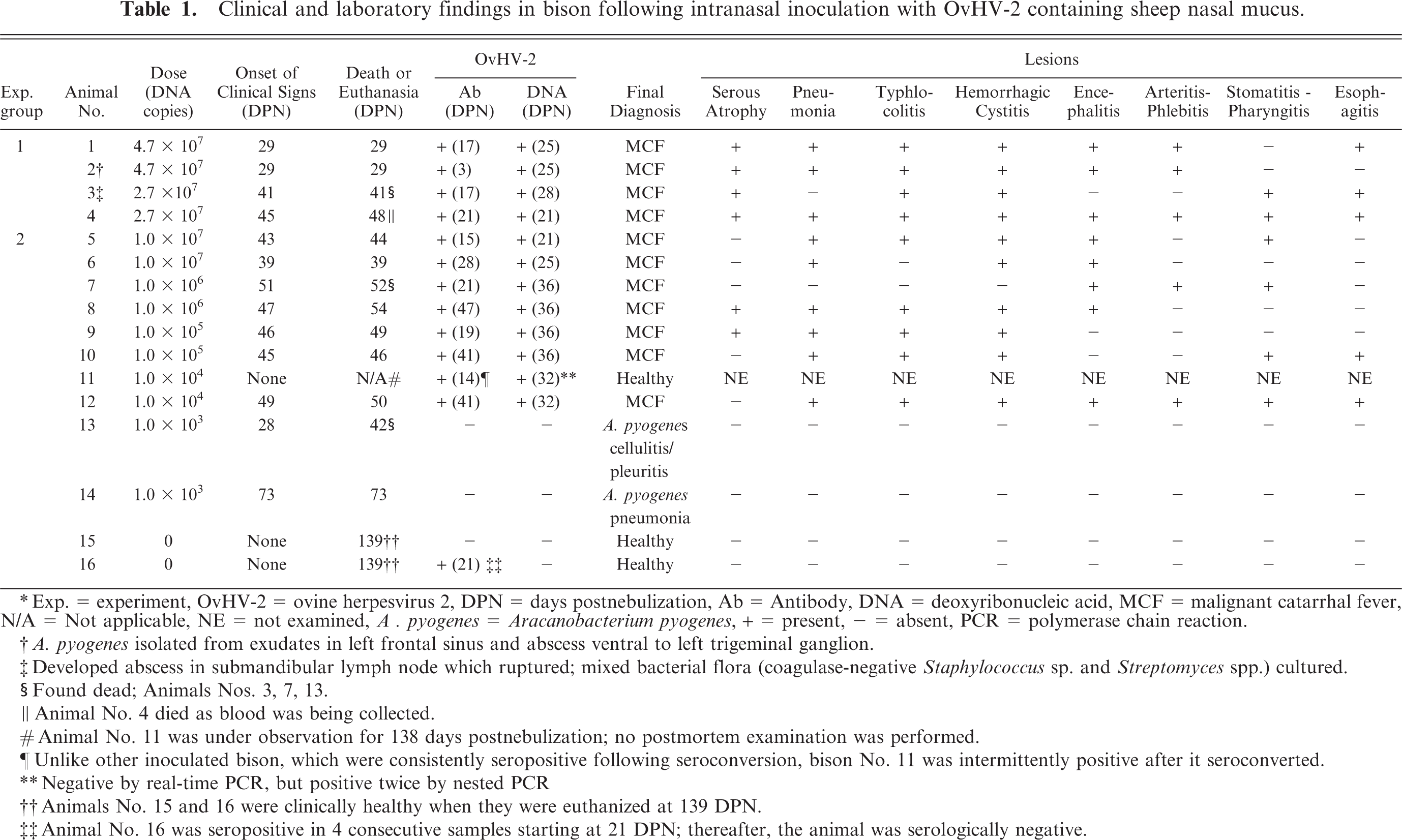
∗ Exp. = experiment, OvHV-2 = ovine herpesvirus 2, DPN = days postnebulization, Ab = Antibody, DNA = deoxyribonucleic acid, MCF = malignant catarrhal fever, N/A = Not applicable, NE = not examined, A . pyogenes = Aracanobacterium pyogenes, + = present, − = absent, PCR = polymerase chain reaction.
† A. pyogenes isolated from exudates in left frontal sinus and abscess ventral to left trigeminal ganglion.
‡ Developed abscess in submandibular lymph node which ruptured; mixed bacterial flora (coagulase-negative Staphylococcus sp. and Streptomyces spp.) cultured.
§ Found dead; Animals Nos. 3, 7, 13.
∥ Animal No. 4 died as blood was being collected.
# Animal No. 11 was under observation for 138 days postnebulization; no postmortem examination was performed.
¶ Unlike other inoculated bison, which were consistently seropositive following seroconversion, bison No. 11 was intermittently positive after it seroconverted.
∗∗ Negative by real-time PCR, but positive twice by nested PCR
†† Animals No. 15 and 16 were clinically healthy when they were euthanized at 139 DPN.
‡‡ Animal No. 16 was seropositive in 4 consecutive samples starting at 21 DPN; thereafter, the animal was serologically negative.
Sample collection
Following nebulization, blood was collected once (experiment 1) or twice weekly (experiment 2). Serum antibodies were monitored using a cELISA that detects MCF group viral antibody in plasma. 11 Results were expressed as percentage inhibition derived by the formula: 100 − [mean optical density at 450 nm (OD450) of test serum × 100 / mean OD450 of negative control serum]. Serum was considered positive when it had an inhibition value of ≥25%. OvHV-2 DNA in peripheral blood leukocytes (PBLs) was quantified using an OvHV-2-specific real-time PCR. 7 Clinical observations of animals were made 3 times a day.
Pathology
Bison that developed clinical signs consistent with a terminal illness were euthanized using intramuscular xylazine, followed by a lethal intravenous dose of pentobarbital. Bison that died were examined postmortem as soon as they were found. A comprehensive set of tissues was collected at necropsy from 15 animals and fixed in 10% neutral buffered formalin. One animal (No. 11) that did not develop disease during the 138-day observation period was not examined postmortem. Following fixation, tissues were dehydrated, embedded in paraffin, sectioned at 4–6 μm on a microtome, stained with HE, and examined. Samples of tissues, including turbinate, trachea, lung, lymph node, spleen, kidney, intestine, and brain, were collected fresh from each animal at necropsy and examined by real-time PCR for OvHV-2 DNA. Aerobic microbiology was performed as needed.
Results
Experiment 1: establishing susceptibility
This was a preliminary study to determine whether bison exposed via the respiratory route to OvHV-2 in nasal secretions from sheep would become infected and develop MCF. MCF viral antibody was detected in all 4 bison beginning at 3–21 days postnebulization (DPN). OvHV-2 DNA was detectable in PBLs by real-time PCR beginning at 21–28 DPN (Table 1). Viral DNA levels in PBLs increased during infection and reached peak levels of approximately 105 copies/0.5 μg PBL DNA (Fig. 1). OvHV-2 DNA was detected in tissues of all 4 bison at necropsy (data not shown). Three developed clinical signs at 29, 29, and 45 DPN. The remaining animal died without demonstrating clinical signs at 41 DPN (Table 1). Affected bison exhibited weakness, fever (rectal temperatures >39.4°C), and depression and stood separate from others in the group. One animal (No. 2) exhibited hypermetria.
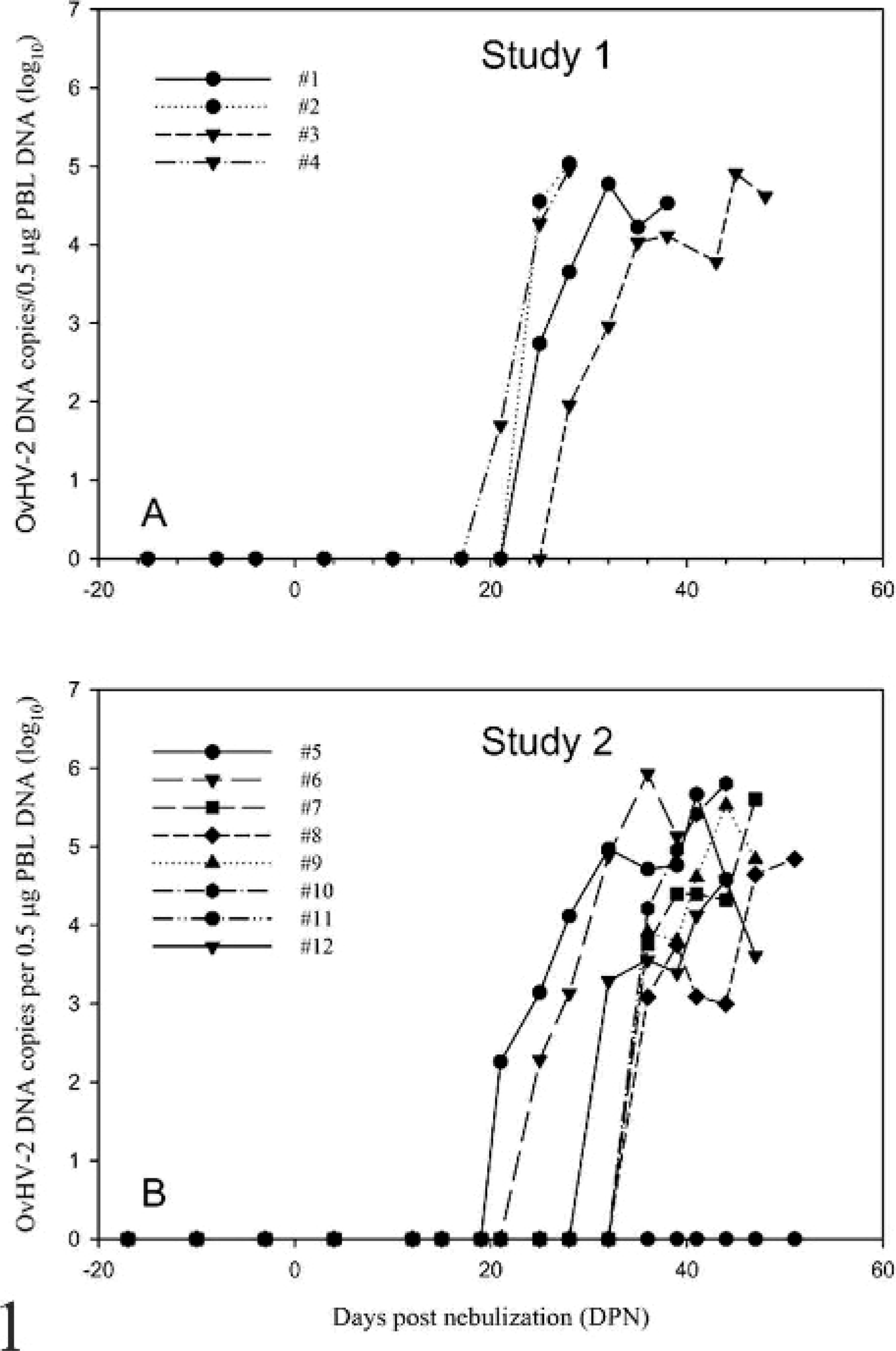
OvHV-2 deoxyribonucleic acid copy number in studies 1 and 2. In both studies, there was an abrupt increase in copy number between 21 and 31 days postnebulization.
Gross lesions consisted of serous atrophy of fat, diffuse fibrinous typhlocolitis, multifocal hemorrhagic cystitis, and erosive-ulcerative stomatitis, pharyngitis, laryngitis, esophagitis, reticulitis, rumenitis, omasitis, and abomasitis. Not all lesions were present in each animal (Table 1). When present, lesions varied from mild to severe. Additionally, 1 bison (No. 2) had a broken left horn with moderate purulent sinusitis and an abscess ventral to the left trigeminal ganglion. Arcanobacterium pyogenes was isolated from both sites. Histologic examination revealed that 3 of 4 animals had mild-to-moderate disseminated arteritis-phlebitis (Nos. 1, 2, and 4). Three of 4 had mild, nonsuppurative encephalitis with gliosis (Nos. 1, 2, and 4). Encephalitis in the animal with hypermetria (No. 2) was comparable in severity and distribution with that in the other bison. Gross and histologic lesions corresponded to published accounts of naturally occurring acute MCF in bison. 20, 25, 26
A distinctive lesion present in bison that has not been described in the natural outbreaks was multifocal necrotizing bronchitis-bronchiolitis with interstitial pneumonia and necrosis of alveolar walls. Grossly, this was manifested as consolidation with purple discoloration of pulmonary parenchyma (Fig. 2). No more than an estimated 1% of parenchyma was involved. Histologically, this corresponded to lobular areas of intra-alveolar proteinaceous and/or fibrin exudation (Figs. 3, 4), intra-alveolar hemorrhage, and loss of type I pneumocytes. Atelectasis was present to varying degrees. Focal to locally extensive degeneration and sloughing of bronchial-bronchiolar epithelium was evident in affected lobules (Fig. 5).
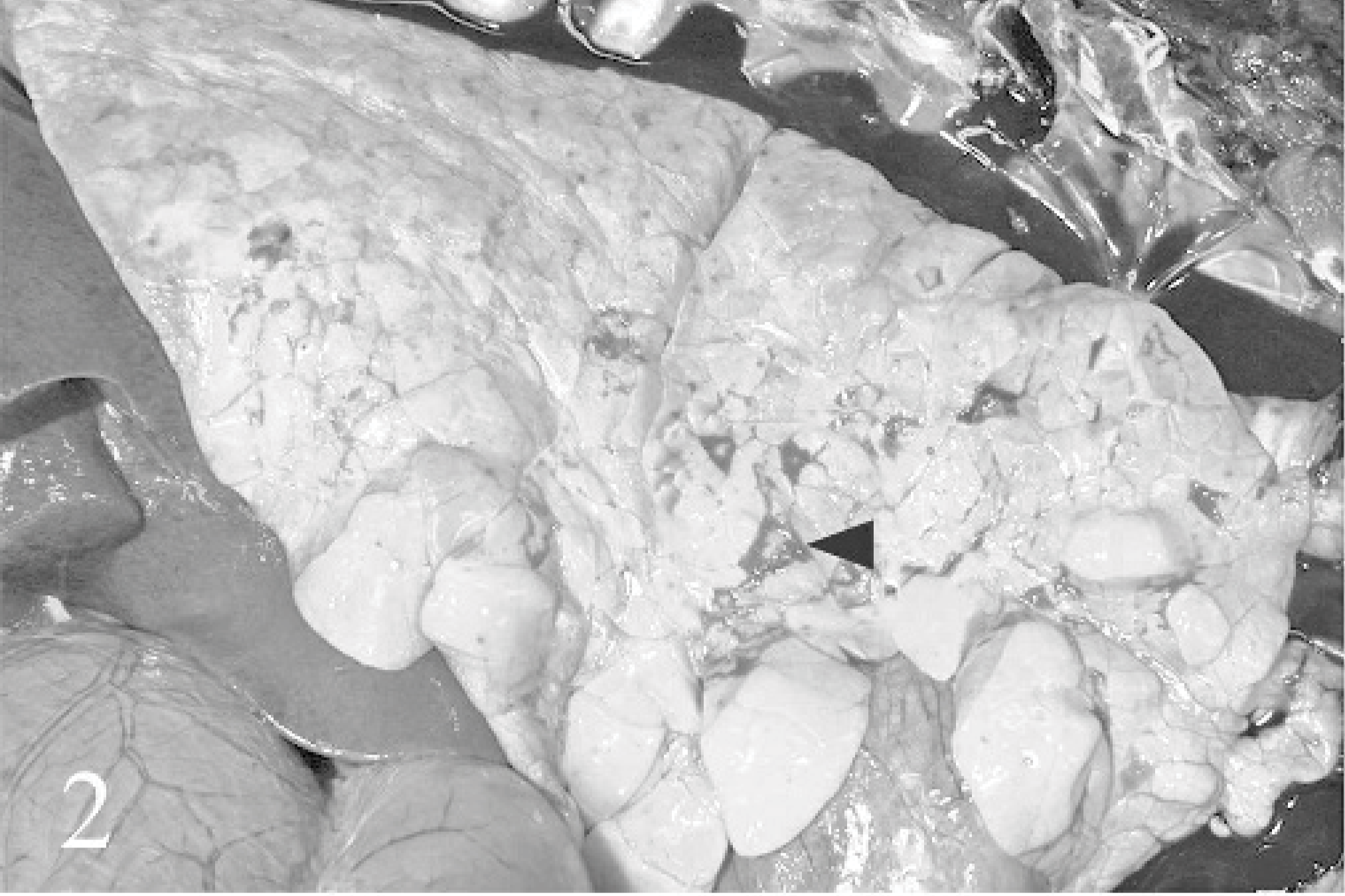
Lung; bison No. 1. Discolored, consolidated pulmonary parenchyma corresponds histologically to foci of peribronchiolar pneumonia (arrowhead). Such areas occurred in all lobes but involved no more than an estimated <1% of parenchyma. HE.
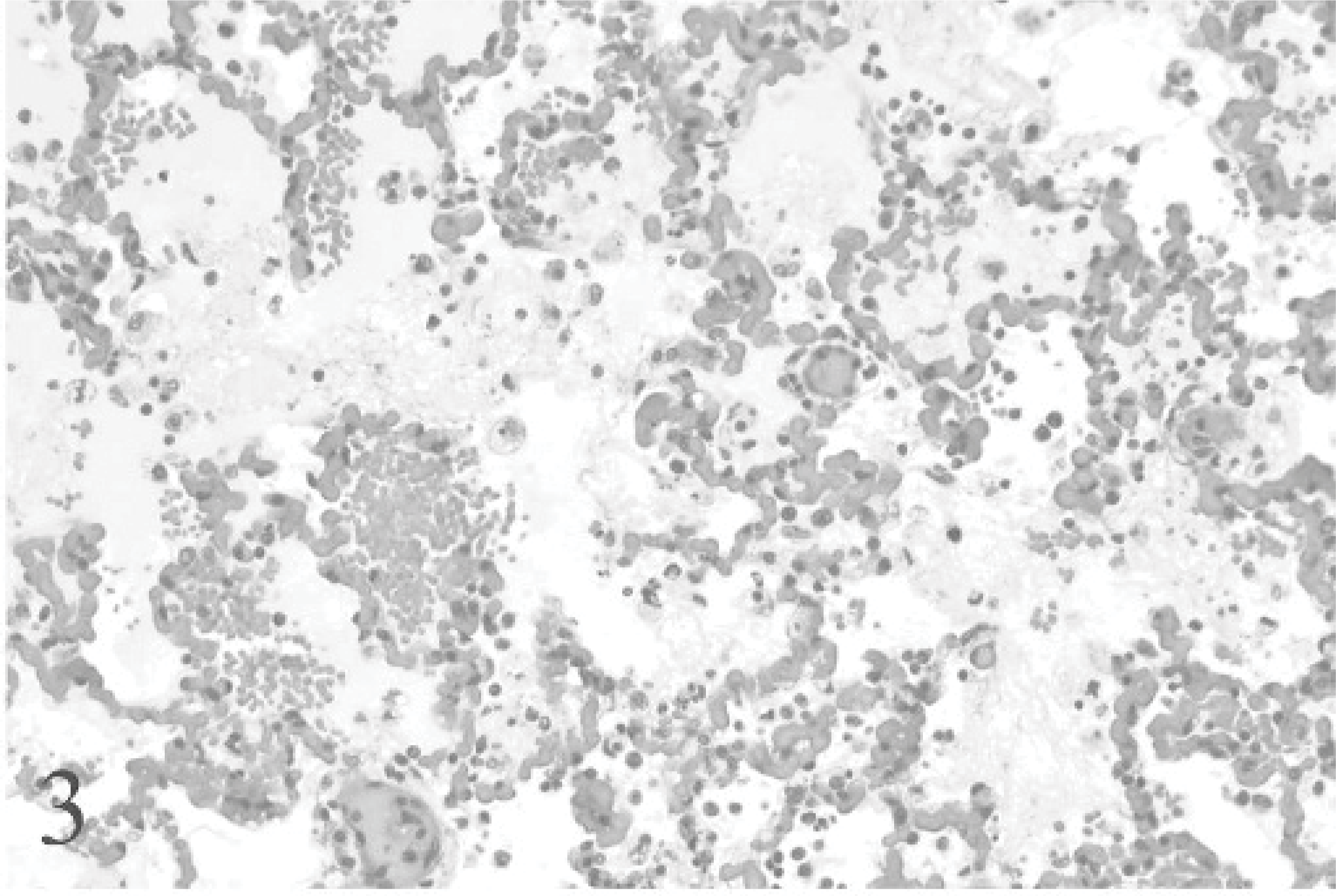
Lung; bison No. 1. This and Figs. 4, 5 illustrate different areas of affected parenchyma in 1 bison: macrophages, proteinaceous material, and some fibrin-filled alveoli. HE.

Lung; bison No. 1. Alveoli are flooded with fibrin, accompanied by macrophages and neutrophils. There is loss of type I pneumocytes. HE.
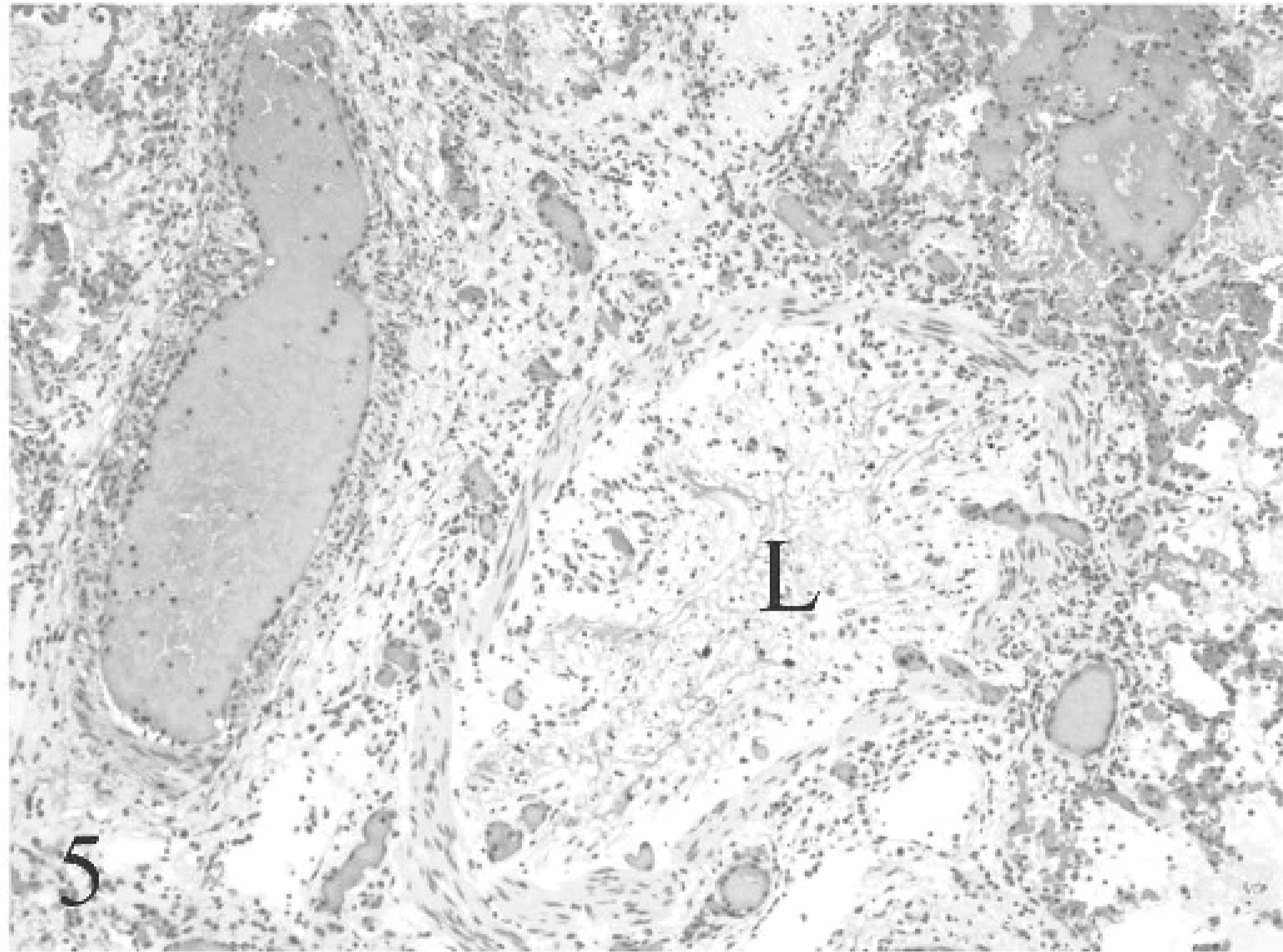
Lung; bison No. 1. Diffuse loss of bronchiolar epithelium is seen. Fibrin and mucin fill the lumen (L). HE.
Experiment 2: defining minimum infectious dose
Seven bison (Nos. 5–10; 12) nebulized with inocula containing 104–107 OvHV-2 copies seroconverted at 15–47 DPN. An eighth animal (No. 11, inoculated with 104 OvHV-2 copies) had intermittently detectable MCF viral antibody, beginning at 14 DPN. Eight bison had detectable viral DNA by real-time PCR in PBLs, beginning at 21–36 DPN (Table 1). In 7 of 8 challenged bison (Nos. 5–10, 12), OvHV-2 DNA copy numbers in PBLs increased and reached similar peak levels as seen in experiment 1 (Fig. 1). The mean interval between the first appearance of viral DNA in PBLs and onset of clinical signs was 12.9 days (SD = 6.9 days). OvHV-2 DNA was detected in multiple tissues of all 7 bison at necropsy. The remaining animal (No. 11) was intermittently positive only by nested PCR beginning at 32 DPN; this animal was not examined postmortem. Both bison nebulized with the inoculum containing 103 DNA copies died of intercurrent disease at 42 days (No. 13) and 73 days postinoculation (No. 14) without developing evidence of infection based on serologic titer, the presence of OvHV-2 DNA in peripheral blood leukocytes or in tissues at necropsy, or lesions consistent with MCF.
Seven bison nebulized with inocula containing 104–107 OvHV-2 DNA copies (Nos. 5–10, 12) developed clinical signs consistent with MCF or were found dead at 39–51 DPN (Table 1). Bison No. 11, which received an inoculum containing 104 OvHV-2 copies, remained healthy for the duration of the study. Gross and histologic lesions in the 7 animals were consistent with acute MCF and with changes found in experiment 1 (Figs. 6– 10). 18, 20, 25, 26 There was no obvious difference in severity or distribution of lesions in bison given different doses of OvHV-2. Six of 7 bison with MCF had lobular bronchointerstitial pneumonia as seen in the pilot study (Figs. 2– 5).
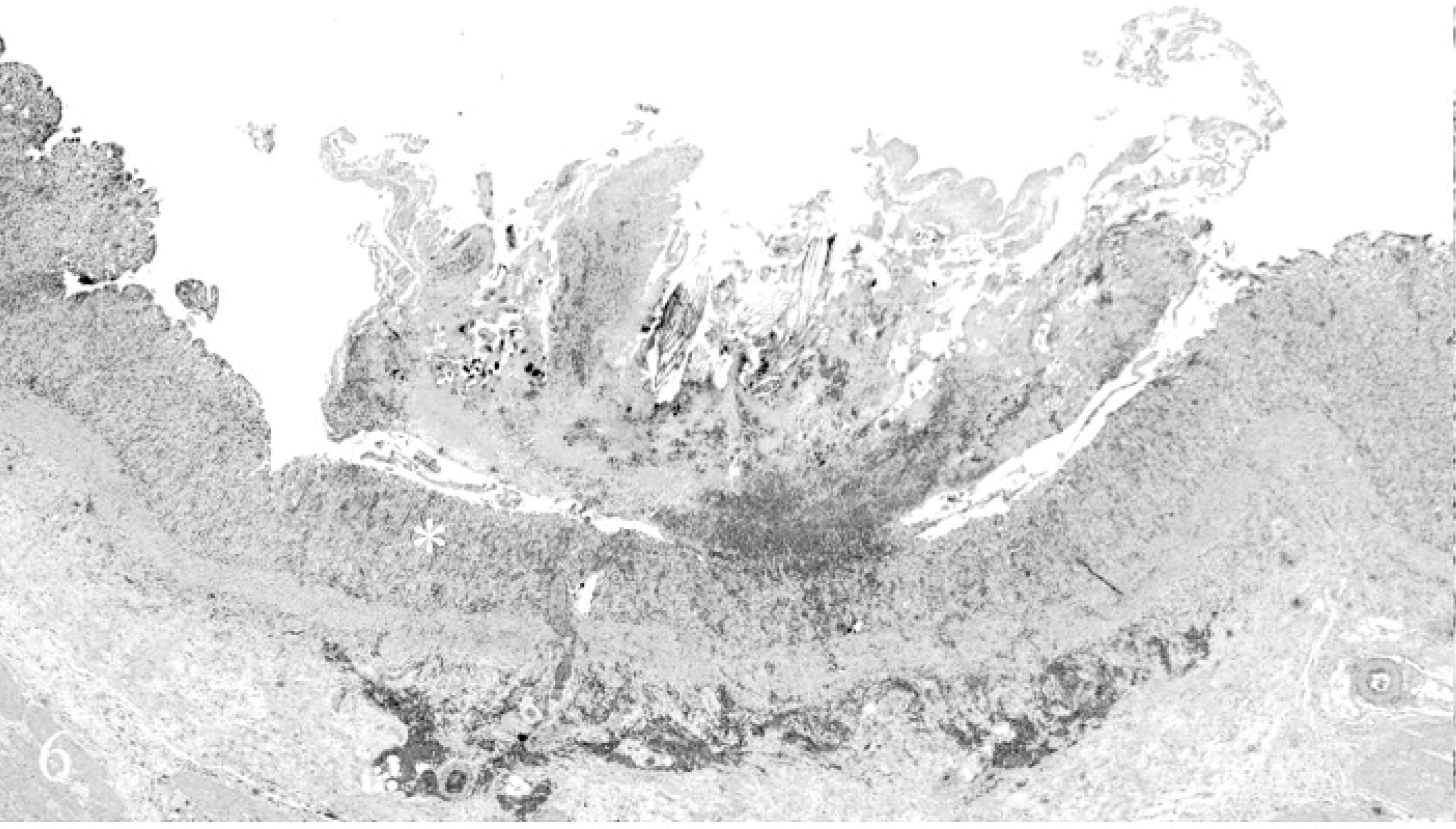
Cecum; bison No. 4. Discreet focal ulceration of cecal mucosa is seen. The area marked with an asterisk is shown at higher magnification in Fig. 7. HE.
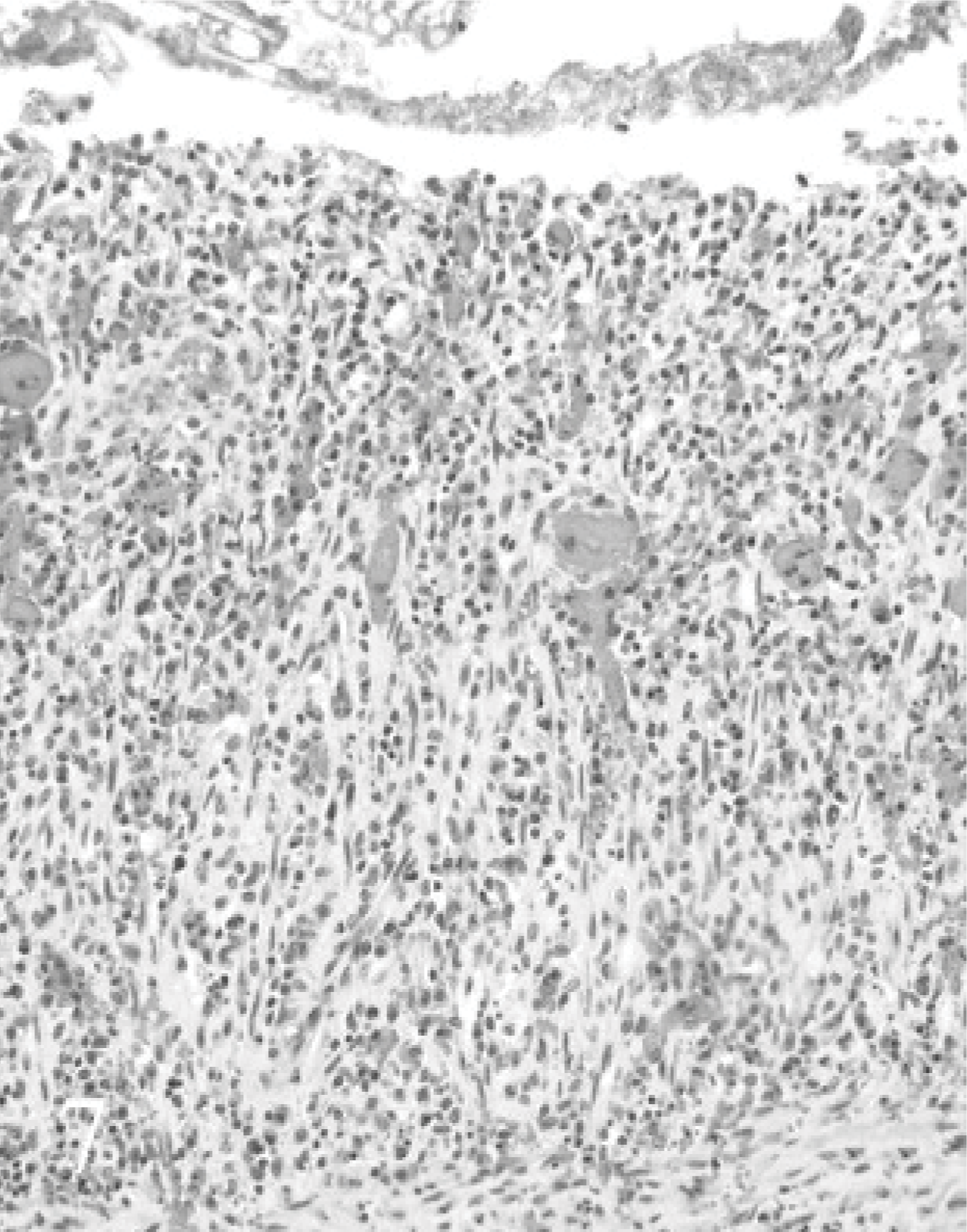
Cecum; bison No. 4. Cecal mucosa adjacent to focal ulcer shown in Fig. 6 is seen. Note complete loss of cecal enterocytes in crypts. HE.
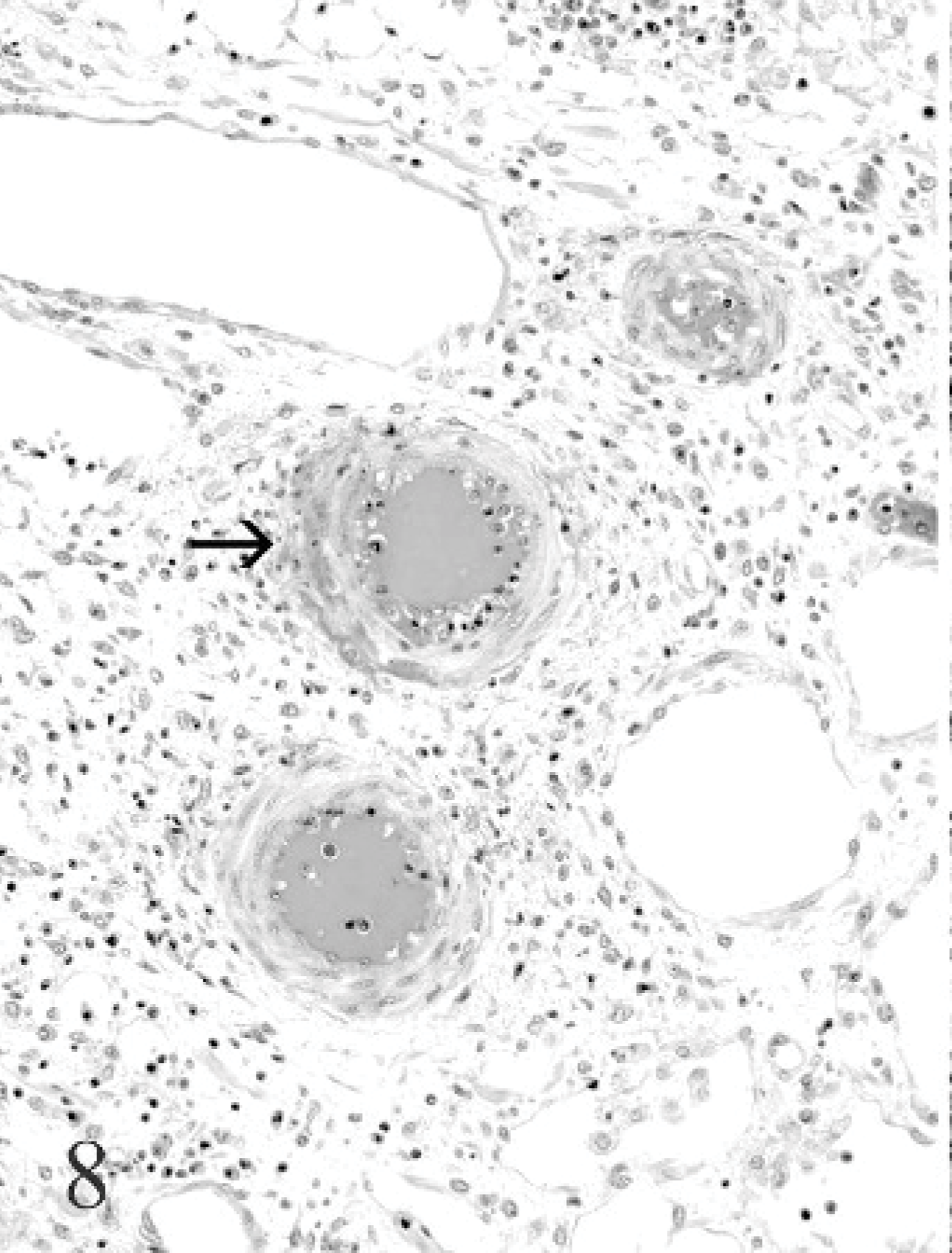
Lymph node; bison No. 4. Focal arteritis with fibrinoid necrosis of tunica media (arrow) in the hilar area of the lymph node is seen. HE.
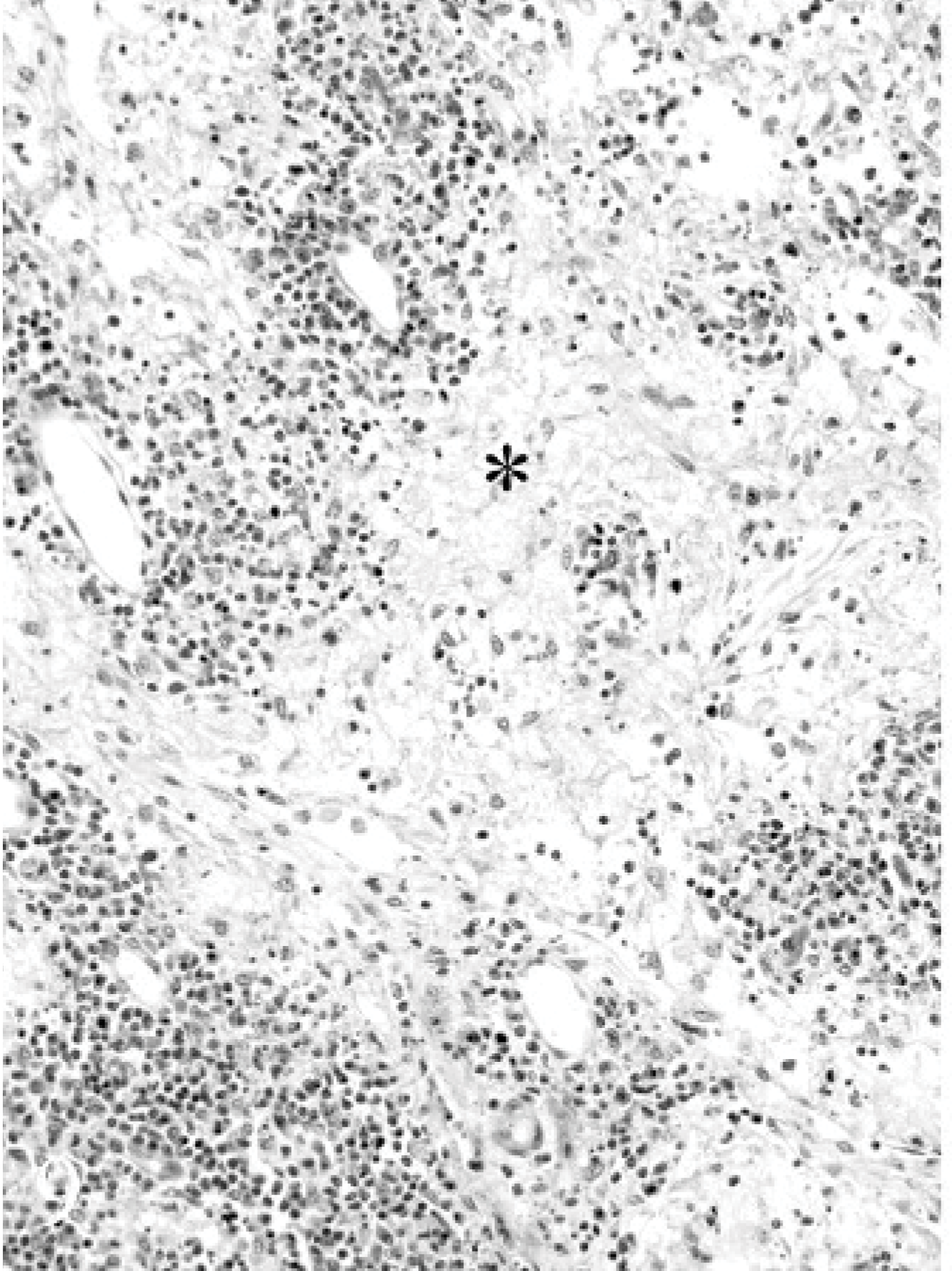
Lymph node; bison No. 4. Medullary sinusoids are filled with fibrin (asterisk). Multiple foci of karyorrhexis are present. HE.
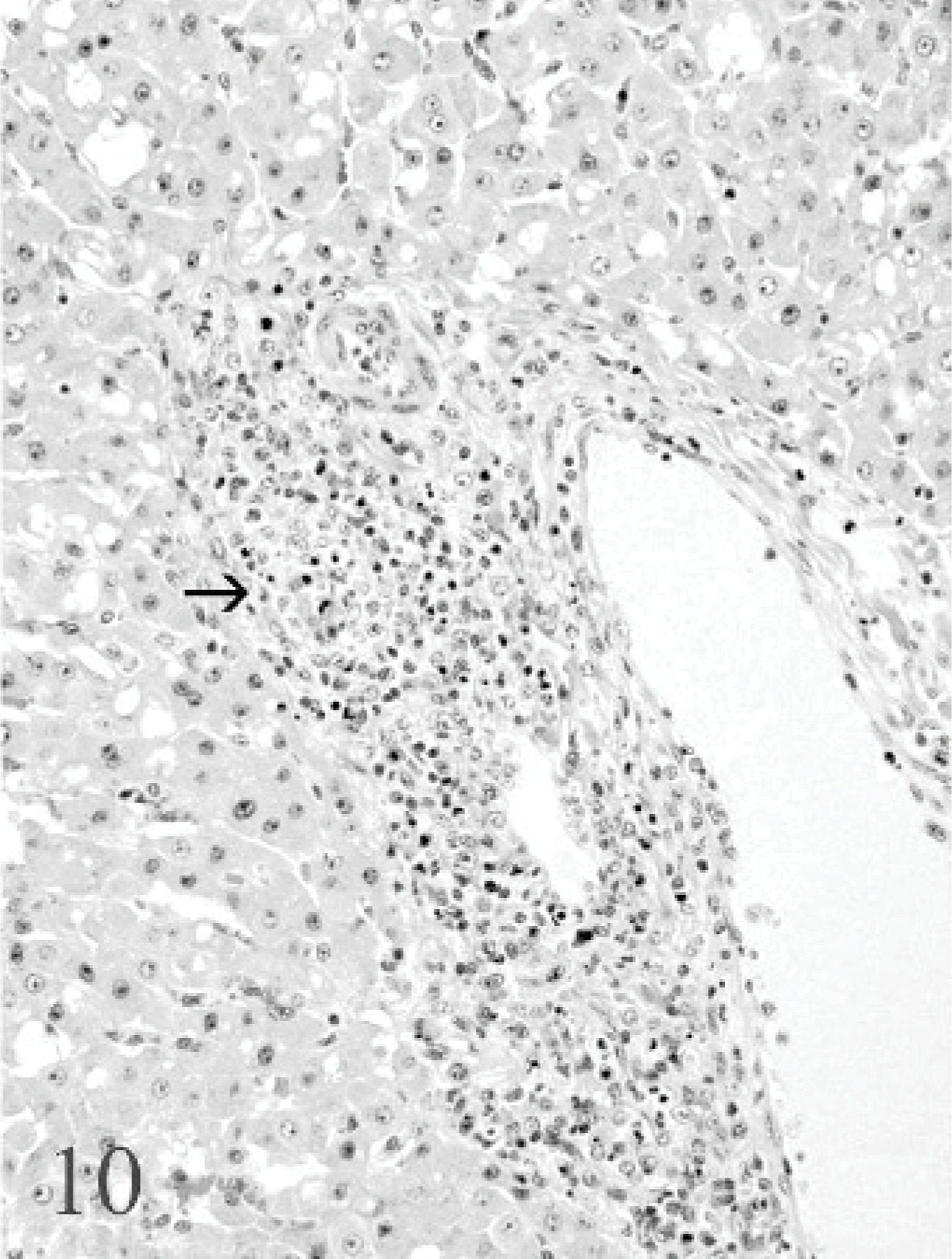
Liver; bison No. 8. Lymphocytic portal hepatitis with degeneration of bile duct epithelium (black arrow) is seen. There is focal endophlebitis (white arrow). HE.
The 2 bison nebulized with the lowest dose of inoculum (103 OvHV-2 DNA copies) died of intercurrent disease (Table 1). One (No. 13) developed Aracanobacterium pyogenes cellulitis at 28 DPN and died at 42 DPN. Disseminated A. pyogenes and associated inflammation was present in the lungs, lymph nodes, and pericardial sac. The other animal (No. 14) became ill at 73 DPN and had severe A. pyogenes lymphadenitis with terminal dissemination. Lesions consistent with MCF were absent in both animals. OvHV-2 DNA was not detected in tissues. Neither animal inoculated with the lowest dose of inoculum (Nos. 13, 14) nor 2 given the control inoculum (Nos. 15, 16) had detectable OvHV-2 DNA in PBLs. Three of these 4 bison (Nos. 13–15) were seronegative throughout the study. In the remaining bison (control No. 16), 4 consecutive samples starting at 21 DPN were cELISA-positive. Thereafter, antibody was undetectable. Both controls remained healthy throughout the study period (Table 1). Postmortem examination did not reveal gross or histologic lesions of MCF, and no OvHV-2 DNA was detected in their tissues (Table 1).
Discussion
Reliable experimental reproduction of sheep-associated MCF has long been problematic. The use of large amounts of fresh blood derived from naturally affected cattle, bison, or deer, as well as the problems that attend long-term storage of such inocula, made it difficult to perform replicate experimental studies of MCF. Inocula derived from OvHV-2-shedding sheep have advantages over the use of blood obtained from animals with spontaneous MCF. First, nasal exposure represents an important form of natural transmission. 2, 14 Second, inocula can be stored frozen and used for replicate experiments. Third, multiple studies can be performed in different species using various concentrations of virus derived from a standardized inoculum.
The work reported here documents that MCF can be induced in a proportion of bison exposed to defined, pooled nasal secretions containing ≥104 viral DNA copies of OvHV-2. Concentrations required to induce MCF in bison are at least 1,000 times less than those that induced MCF in cattle (108 OvHV-2 genome copies). 27 This corroborates suggestions that American bison are more susceptible to clinical MCF than cattle. 14 The interval between experimental exposure and onset of clinical signs (or death) corresponds to estimates of the incubation period in naturally exposed bison. Two recent studies reported that the incubation period following exposure to sheep peaked at 60–70 days postexposure 2 and at 41–55 days post-mean exposure. 14 Until now, MCF has been reproduced experimentally only once, when a bison calf developed the disease 25 days after inoculation with blood from an infected calf. 18 In the current study, the mean incubation period was 42 days with a range of 29–51 days. OvHV-2 DNA levels increased and were maintained at high levels during the latter part of the incubation period in all bison that developed terminal MCF. The mean interval between detecting OvHV-2 DNA and onset of clinical signs was about 2 weeks. The detection of high concentrations of OvHV-2 DNA (>500 copies/0.5 μg PBL DNA) in PBLs reliably augured that bison would soon develop terminal disease.
The 2 bison given the lowest dose of inoculum (103 OvHV-2 genome copies) did not become infected during the 42- and 73-day observation period respectively, as indicated by lack of seroconversion, absence of detectable OvHV-2 DNA in PBLs or tissue, and absence of lesions consistent with MCF. One of 2 bison given an inoculum containing 104 viral DNA copies became infected yet did not develop disease. Collectively, these data suggest that there may be a dose at which bison may be infected without developing MCF. This may account for the high proportion of bison in some commercial herds (∼30%) that are seropositive for MCF group virus, yet are free of disease. The current antibody test and PCR do not guarantee that all latently infected animals can be identified. This poses a challenge when animals are screened to detect uninfected animals for transmission studies. Bison No. 16 was probably infected at the time he was enrolled into the study, and infection was probably missed by initial cELISA and PCR screenings. It is also likely that bison No. 2 was previously infected, since seroconversion was detected at 3 DPN, though antibody titer increased continuously after nebulization. Use of multiple screenings at 2-week intervals for several months should help ensure the negative status of animals in future experiments.
Bison, unlike cattle, generally die of acute MCF without displaying florid bilateral keratoconjunctivitis or purulent rhinitis (the head and eye form of MCF). 20, 25, 26 A common observation in herds and feedlots in which MCF outbreaks occur is that bison die with few or no premonitory signs. In the present study, 3 bison with MCF died without detectable clinical signs. The absence of clinical signs in animals with advanced lesions suggests that bison are stoic and will attempt to mask clinical signs of MCF until near death.
All animals that developed MCF had lesions indicative of acute disease. In spite of a relatively long incubation period (up to 51 DPN), there was no evidence of chronic MCF as it occurs in domestic cattle. 19 A tentative diagnosis of MCF can be made on the basis of gross and histologic lesions in the oral cavity, pharynx, larynx, forestomachs, abomasum, cecum and colon, and bladder. Unlike cattle, arteritis-phlebitis cannot be relied upon in bison to serve as diagnostic stigmata of MCF. 16 Lymphadenomegaly, a prominent and helpful feature of MCF in cattle, is generally minimal in affected bison. When it occurs, its principal basis is fibrin effusion in sinusoidal spaces, accompanied by necrosis of lymphoid cells, vasculitis, and lymphadenitis. The marked paracortical lymphoid expansion, which can be such a prominent feature of MCF in cattle, was not evident in the current study.
A distinctive form of bronchointerstitial pneumonia with atelectasis was seen in bison in both experiments. It resembled the type of pneumonia seen in cattle and sheep following experimental nebulization with OvHV-2. 12, 27 We have not recognized this in bison dying of naturally acquired MCF. It likely reflects the delivery of infectious material in lower airways. In the absence of a method to readily detect OvHV-2 antigen in tissue, we were unable to confirm this. Nevertheless, it suggests that lungs may be a useful target tissue to use in attempting to develop a visible histologic marker for viral antigen or DNA early in the course of disease.
In summary, acute MCF was induced in 11 of 14 challenged bison (78.6%) using nasal secretions containing OvHV-2. A minimum infectious dose that consistently induces disease, based on these studies, is 105 OvHV-2 DNA copies.
Footnotes
Acknowledgements
This work was supported by the US Department of Agriculture-Agricultural Research Service CWU 5348-32000-024-00D and CSREES grant 2001-35204-10151. We thank Rod Rogers for animal care; Brian Parrie for assistance with necropsies; Paula Jaeger, Leslie Kerr, and Mercedes Thelen for histologic sections; Dr. Ken Mills and Amy Boerger-Fields for bacteriology support; and Jan Keller, Shirley Elias, and Lori Fuller for technical assistance.
