Abstract
Ovine herpesvirus 2 (OvHV-2) is one of the gammaherpesviruses in the genus Macavirus that can cause malignant catarrhal fever (MCF) in ungulates. Sheep are the adapted host for OvHV-2 and it is generally assumed that infection is not associated with disease in this species. However, cases of “polyarteritis nodosa” or idiopathic systemic necrotizing vasculitis reported in sheep are similar to vascular lesions in clinically susceptible species with MCF. Using a recently developed in situ hybridization (ISH) method, we were able to identify OvHV-2 nucleic acids within lesions and correlate the viral distribution with systemic necrotizing vasculitis in 9 sheep, including both naturally and experimentally OvHV-2–infected animals. ISH, combined with polymerase chain reaction and histology, identify OvHV-2 as the likely agent responsible for sporadic, MCF-like vascular disease in sheep.
Keywords
Systemic necrotizing vasculitis is a rare, sporadic disease recognized in individuals or clusters of sheep. 9,11,20,22,26 The histologic lesions have been considered typical of “polyarteritis nodosa” (PAN), which also has been reported in pigs, 15 cats, 5 dogs, 23 rats, 21 and humans. 7,10 Many veterinary reports use a definition of PAN derived from human studies, which is a necrotizing arteritis of medium or small arteries without vasculitis in arterioles, capillaries, or venules. 10 In rats, PAN is recognized in naturally occurring or drug-induced hypertension and is used as a model for investigating the correlative human disease. 21 Etiologies for PAN in veterinary species are likely to be multifactorial, as is the case in humans, in whom a distinction between “primary” (idiopathic) and “secondary” PAN is made when an etiologic agent, most commonly a persistent viral infection such as hepatitis B virus, human immunodeficiency virus, or cytomegalovirus, is found within the lesions. 7,10 By extrapolation, this raises the possibility that a persistent viral infection may be the cause of systemic necrotizing vasculitis in sheep. Important differentials for viral-associated vasculitis in sheep include Maedi-Visna, which has been reported to produce a vasculitis but only in association with overt lesions typical of the disease; 6 border disease, which is associated with only mild periarteritis; 27 and bluetongue, in which edema and hemorrhage occur without histologic evidence of vascular inflammation. 16
Natural infections with ovine herpesvirus 2 (OvHV-2) occur in a wide range of animals, including sheep, cattle, bison, deer, moose, and swine. 18 Sheep are considered the adapted host for OvHV-2 and are the natural reservoir source for infection of clinically susceptible species such as cattle and bison. Clinically susceptible species are those that develop the systemic vasocentric disease termed malignant catarrhal fever (MCF). OvHV-2 infection in sheep is usually subclinical, and comparatively little is known about the pathogenesis of OvHV-2 infection in its adapted host species. Hüssy et al 12 quantitatively analyzed tissue distribution of OvHV-2 in normal sheep, and infection of circulating lymphocytes and/or epithelium has been demonstrated in field infections 17 and under experimental conditions. 24 Natural disease in OvHV-2–infected sheep has been proposed, 9,11,20 and experimentally, lambs exposed to a high dose of OvHV-2 can develop an MCF-like syndrome. 3,13 But because most sheep are subclinically infected with OvHV-2, 1,12,14,18 detection of viral DNA or antibodies that confirms infection and/or exposure cannot uncover disease association. Diagnosticians have long lacked any method able to recognize the distribution of OvHV-2 within lesions in formalin-fixed, paraffin-embedded tissues. In this study, we use in situ hybridization (ISH), along with polymerase chain reaction (PCR) and histopathology, to test the hypothesis that OvHV-2 is the agent responsible for sporadic cases of systemic necrotizing vasculitis in sheep.
The ovine vasculitis cases investigated in this study are summarized in Table 1 and Supplemental Table S1. Case Nos. 1 to 8 are naturally occurring cases in 5 independent flocks from 3 countries (Spain, United Kingdom, and United States). All of these cases have been previously described as idiopathic polyarteritis 8,26 or OvHV-2-associated MCF, 20 and they were selected based on the histologic criteria of a systemic (or multiorgan) necrotizing vasculitis. Case No. 9 was a lamb with vasculitis following experimental inoculation of a high dose of OvHV-2 by intranasal nebulization. 13
Description of Cases Evaluated in This Study, Including OvHV-2 PCR and ISH Results.
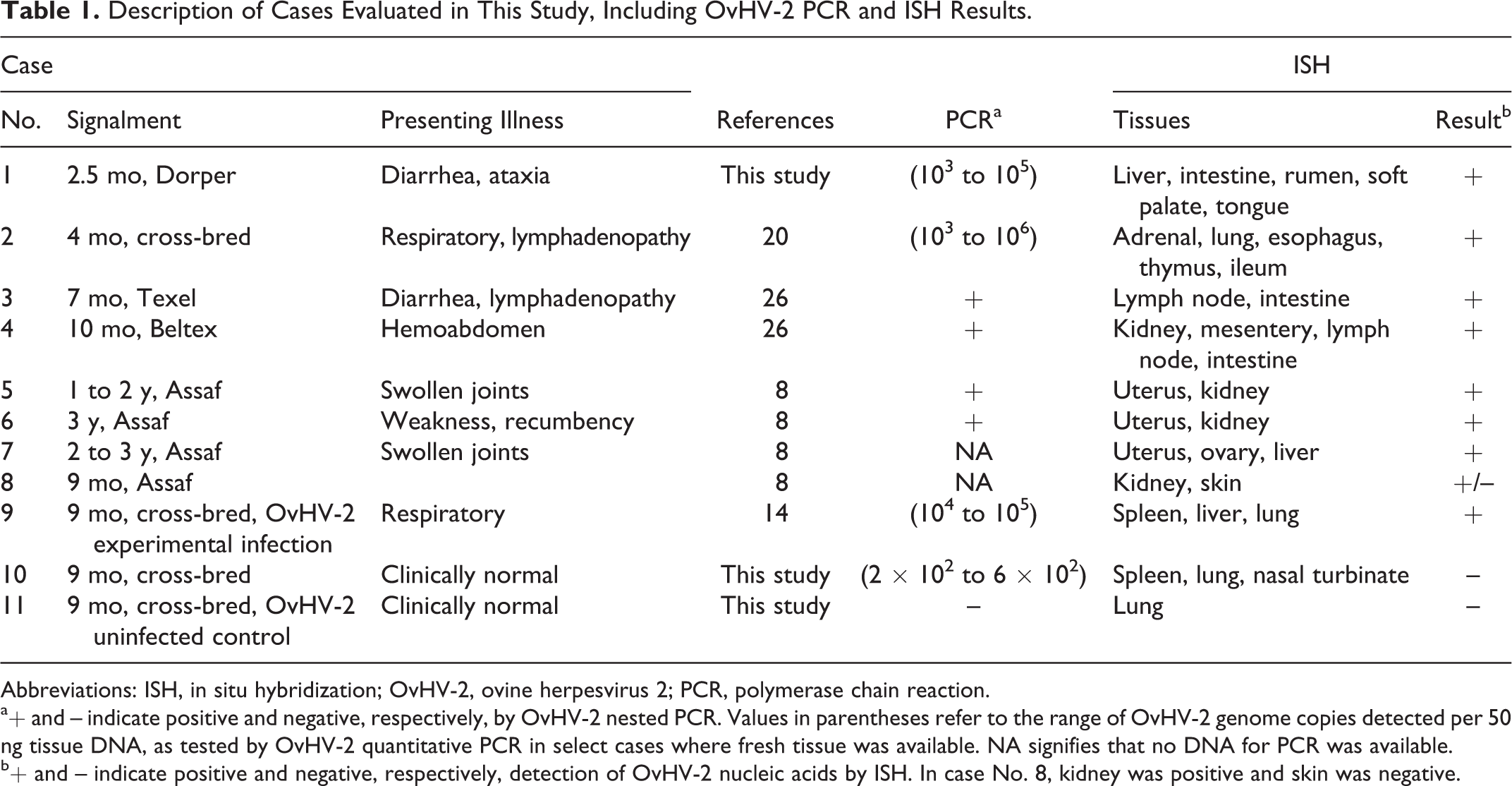
Abbreviations: ISH, in situ hybridization; OvHV-2, ovine herpesvirus 2; PCR, polymerase chain reaction.
a+ and – indicate positive and negative, respectively, by OvHV-2 nested PCR. Values in parentheses refer to the range of OvHV-2 genome copies detected per 50 ng tissue DNA, as tested by OvHV-2 quantitative PCR in select cases where fresh tissue was available. NA signifies that no DNA for PCR was available.
b+ and – indicate positive and negative, respectively, detection of OvHV-2 nucleic acids by ISH. In case No. 8, kidney was positive and skin was negative.
Age, breed, and presenting illnesses in these cases were variable (Table 1), but histologic detection of vasculitis (which was a criterion for case selection) was present in all cases. The tissues examined in each case are listed in Table 1. Each case included a subset of the following tissues: gastrointestinal tract, liver, lungs, adrenal glands, heart, pancreas, spleen, meninges, urinary bladder, gallbladder, kidneys, uterus, and/or mammary glands. Regardless of tissue type, affected tissues shared a primarily lymphocytic vasculitis that targeted small- to medium-sized arteries (Figs. 1a, 2a, 3a, 3c). Two clinically normal sheep were also included in this study; 1 was an OvHV-2 naturally infected but asymptomatic lamb (case No. 10), and the other was OvHV-2 uninfected (case No. 11). OvHV-2 DNA was detected by PCR in all clinically affected animals (case Nos. 1–6 and 9). 2,25 Extracted DNA was not available from case Nos. 7 and 8, and thus no PCR data were obtained. Quantitation of OvHV-2 DNA was estimated by real-time PCR on case Nos. 1, 2, 9, and 10, which were the cases that had fresh tissue available, and results from individual tissues are reported in Supplemental Table S1. Viral quantity ranged from 103 to 106 viral genome copies per 50 ng total DNA (Table 1 and Suppl. Table S1) in clinical animals (case Nos. 1, 2, and 9). The infected but clinically normal animal (case No. 10) was positive by real-time PCR but had a low OvHV-2 genome copy number, ranging from 200 to 600 per 50 ng total DNA, which was quantified independently in 3 tissues (Suppl. Table S1). The uninfected control animal (case No. 11) was OvHV-2 negative by PCR. During this study, a reevaluation of the whole-genome sequencing (Illumina MiSeq, San Diego, CA, USA) that had been previously obtained from splenic tissue of one of the clinically affected lambs (case No. 4) 26 indicated a match to the OvHV-2 BJ1035 reference sequence (accession number NC_007646.1) with 6.77% average coverage of the virus genome. No other viral sequences were detected through this mapping analysis.
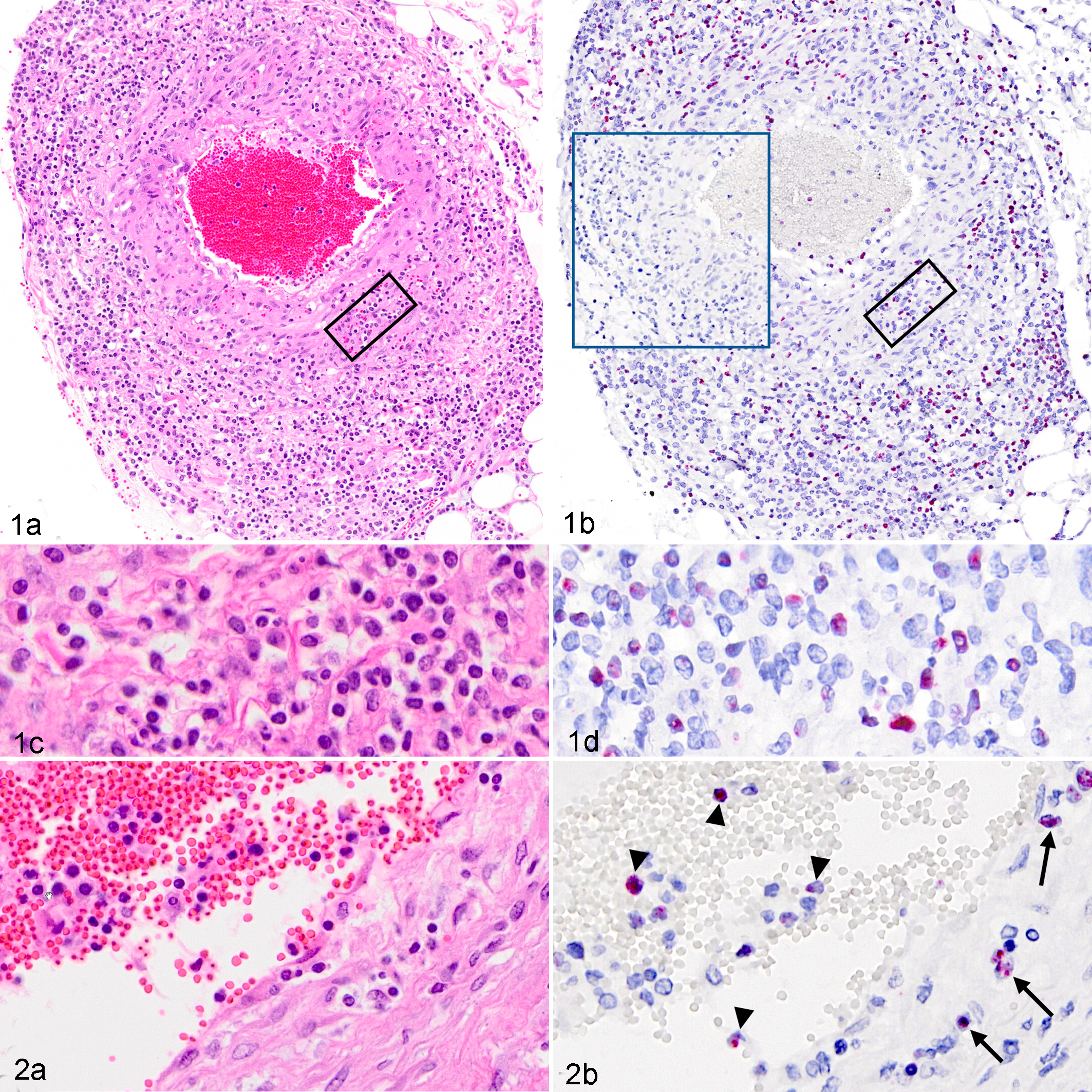
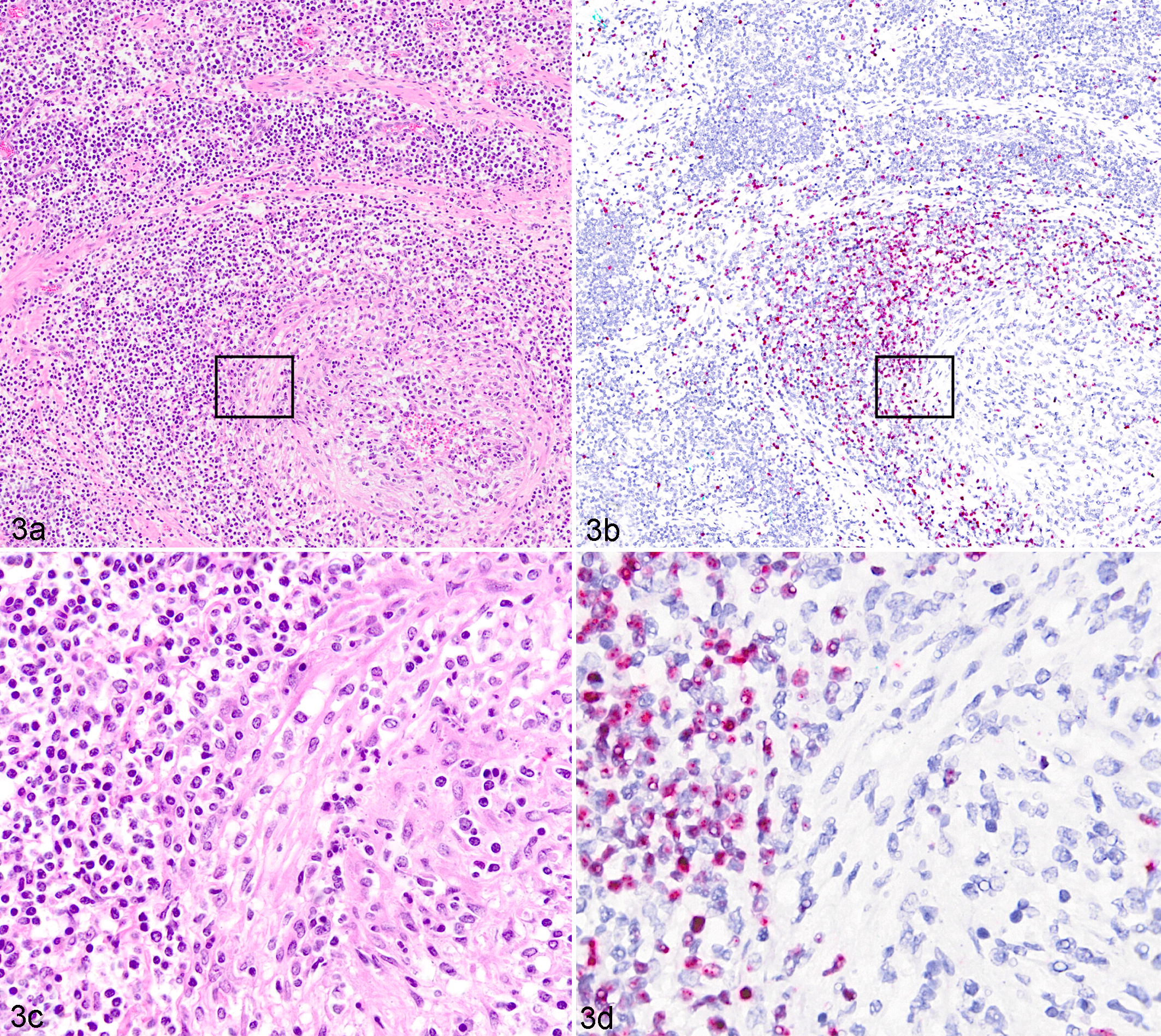
To investigate the distribution of OvHV-2 within tissues, a recently developed ISH was used to detect viral nucleic acids in formalin-fixed, paraffin-embedded sections. 19 Tissues were selected based on the presence of vasculitis, with consideration for the quality of the section and time of tissue fixation (all samples were in 10% formalin for <7 days). Colorimetric ISH was performed as previously described. 19 Briefly, a RNAscope 2.5 Red assay kit (cat. 322360; ACD, Newark, CA, USA) and an OvHV-2–specific probe, V-OvHV2-orf25-orf50 (cat. 501091; ACD), were used per manufacturer instruction. Negative controls included an unrelated (DapB, bacterial gene) probe and tissue sections from an OvHV-2–negative animal (case No. 11). Slides were counterstained with hematoxylin and examined on a 40× objective with bright-field illumination and digitalized using an Olympus VS120 scanner (Center Valley, PA, USA). All clinically affected animals, including natural infections (case Nos. 1–8) and the experimental infection (case No. 9), had ISH-detectable OvHV-2 nucleic acid in all tissues tested (Figs. 1–3 and Suppl. Figs. S1–S3), excepting a single ulcerated section of haired skin in case No. 8 (Table 1). Tissues tested included the mesentery (Fig. 1 and Suppl. Fig. S1), kidney (Fig. 2), lymph node (Fig. 3), liver, spleen, lung (Suppl. Fig. S2), uterus (Suppl. Fig. S3), and intestines, and in all cases, the nuclei of a subset of lymphocytes were hybridization targets for the OvHV-2 probe. Lymphocytes containing viral nucleic acid (ISH positive) transmigrated the arterial walls, expanded the adventitia, and surrounded multifocal arteries (Figs. 1b, 1d, 2b, 3b, 3d). The histologic character of the lymphocytes was mixed, with some small and some having open, large nuclei (Fig. 1c,d). OvHV-2 nucleic acid was also detectable in circulating lymphocytes within the lumen of arteries (Fig. 2a,b). As with other MCF-susceptible animals, the inflammation was predominantly CD3+ lymphocytes (clone CD3-12, IgG1rat; generous gift from the Moore laboratory; Suppl. Figs. S1b, S2b and De Virgilio et al 7 and Zakarian et al 27 ). In addition to their arteriocentric distribution, ISH-positive cells were also scattered in sinusoids and rarely cords of lymph nodes (case Nos. 3–4; Fig. 3b,d), the lamina propria of the intestine (case Nos. 1, 3, and 4), hepatic portal areas (case Nos. 1, 7, and 9), within the interstitium around individual renal glomeruli (case Nos. 4–6 and 8), and multifocally expanding the uterine interstitium and intercalating within glandular cells (case Nos. 5–7; Suppl. Fig. S3a,b). Sequential sections of all OvHV-2–positive tissues (eg, Fig. 1b, blue boxed insert) were negative when probed with an unrelated probe (all tissues; eg, Fig. 1b, blue boxed inset). The lung, liver, and spleen of the uninfected animal (case No. 11) probed with the OvHV-2 probe were negative. No ISH-positive signal was detected in any tissues from the OvHV-2–infected, clinically normal sheep (case No. 10), although these same tissues were quantitatively PCR positive with a lower genome copy number than affected animals (Table 1 and Suppl. Table S1).
It is generally assumed that sheep are well-adapted hosts for OvHV-2 and that infection is quite common in clinically normal sheep. 1,14 This creates a challenge with suggesting a diagnosis of MCF in sheep since PCR detection of OvHV-2 DNA by PCR alone in sheep is insufficient evidence of causality. Using OvHV-2–specific ISH, we demonstrate that vasculitis is directly associated with the presence of OvHV-2 nucleic acid. Colocalization of viral nucleic acids within lymphocytes in lesioned tissues supports the hypothesis that OvHV-2 causes an MCF-like disease in sheep. It is notable that higher levels (by quantitative PCR) of OvHV-2 DNA were present in tissues of sheep with vasculitis compared to the levels detected in unaffected persistent carriers (Suppl. Table S1), suggesting that ISH-positive results associated with vasculitis and high levels of OvHV-2 DNA can be used to confirm cases of MCF in sheep.
Two historic reports that did not invoke OvHV-2 specifically as a cause but described an “idiopathic” polyarteritis or polyarteritis nodosa (PAN) in sheep were included in this study, 8,26 and all 6 cases tested were positive by OvHV-2–specific ISH. Immunohistochemical assessment, whether this study (case No. 1; Suppl. Fig. S1b) or previously performed (case Nos. 3–8), 8,26 demonstrated that transmural arterial infiltrates were predominantly lymphocytic (Suppl. Fig. S1b), with fewer macrophages and plasma cells. The character of the inflammation correlates well with PAN lesions in humans where T-cell–mediated immune mechanisms are important in the development and perpetuation of the lesions. 4 Our data indicate that OvHV-2 plays a role in the development of vasculitis in sheep and can be an etiological agent in certain cases of PAN, but we cannot rule out the possibility of a comicrobial pathogen. Our data broaden the potential range of MCF disease to include the adapted host species.
Supplemental Material
Supplemental Material, DS1_VET_10.11770300985818795166 - Systemic Necrotizing Vasculitis in Sheep Is Associated With Ovine Herpesvirus 2
Supplemental Material, DS1_VET_10.11770300985818795166 for Systemic Necrotizing Vasculitis in Sheep Is Associated With Ovine Herpesvirus 2 by Patricia A. Pesavento, Rahul B. Dange, M. Carmen Ferreras, Akbar Dasjerdi, Valentin Pérez, Anna LaRoca, Julio Benavides Silván, Santiago Diab, Kenneth Jackson, Ida L. Phillips, Hong Li, Cristina W. Cunha, and Mark Wessels in Veterinary Pathology
Footnotes
Acknowledgements
We thank Shirley Elias (Washington State University) for technical assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received the following financial support for the research, authorship, and/or publication of this article: Support funding to the Li laboratory for this project is from USDA-ARS CWU 2029-3200-037-00D. In the Pesavento laboratory, the study was supported by Boehringer Ingelheim Vetmedica and the Bernice Barbour Foundation for research in naturally occurring infectious disease.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
