Abstract
Perivascular wall tumors (PWTs) are defined as neoplasms deriving from mural cells of blood vessels, excluding the endothelial lining. The spectrum of human cutaneous PWT includes glomus tumor, hemangiopericytoma (HEP), myopericytoma, angioleiomyoma/sarcoma, angiomyofibroblastoma, and angiofibroma. The purpose of this study was to revise clinical presentation, cytology, histopathology, and immunohistology of canine cutaneous PWT with cytology typical of canine HEP. Diagnosis was established on the basis of vascular growth patterns (staghorn, placentoid, perivascular whorling, bundles from media) and immunohistology, including 7 smooth muscle markers and the cell membrane ganglioside of unknown origin recognized by the antibody 3G5 (CMG-3G5). Twenty cases were included. Ages ranged from 6 to 13 years; 12 dogs were males and 8 were females, and there was a prevalence of crossbreeds. Tumors arose from a single site with preferential acral location (10/20). Cytology revealed moderate to high cellularity in all cases, cohesive groups of cells (19/20), capillaries (18/20), and bi- to multinucleated cells (18/20). Six myopericytomas, 5 angioleiomyomas, 2 angioleiomyosarcomas, 2 HEP, 1 angiofibroma, and 1 adventitial tumor were identified. A definitive diagnosis was not possible in 3 cases. Smoothelin, heavy caldesmon, desmin, myosin, calponin, and CMG-3G5 were the most valuable markers to differentially diagnose canine PWT. Similar to reports in humans, canine HEP embodied a spectrum of neoplastic entities arising from different vascular mural cells. Before canine PWTs are assimilated into one prognostic category, a consistent classification and characterization of their biology is necessary. As proposed in humans, HEP should also be considered a diagnosis of exclusion in dogs.
Perivascular wall tumors (PWTs) are defined as neoplasms deriving from the different cellular components of the vascular wall, excluding the endothelial lining. 68 Vessels have a complex wall that is thinnest in the capillary compartment where it is composed of endothelium, pericytes, and basement membrane. It is thickest in large veins and arteries where endothelium, subendothelial lining cells, basement membrane, a medial layer of smooth muscle cells, and an adventitial layer of myofibroblasts and fibroblasts can be found. 4 With the progressive increase of the vascular caliber, subendothelial lining cells convert, via a morphologic continuum, from pericytes in capillaries into myopericyte in the pre- and postcapillary compartments and in mature smooth muscle cells in arteries and large veins. 4, 32, 53, 64 The progressive replacement of pericytes by myopericytes and smooth muscle myocytes in the vascular subendothelial lining is characterized by a progressive increase in the amount and type of cytoplasmic contractile proteins. 19, 46, 63 Accordingly, pericytes express vimentin, and, variably, pan actin 43 and α-smooth muscle actin, 46 while myopericytes are characterized by the additional expression of desmin and calponin, 46 and smooth muscle cells express smoothelin 63 and heavy caldesmon (h-CD). 19 Additionally, human pericytes and melanocytes express a cell membrane ganglioside of unknown origin, recognized by the antibody 3G5. 15, 27
In humans, PWTs have been extensively characterized and are represented by hemangiopericytoma (HEP), 6, 68 myopericytoma, 24, 68 angioleiomyoma/sarcoma (both of subendothelial or medial origin), 24, 68 angiomyofibroblastoma, 17 angiofibroma, 30 glomus tumor, 68 and glomangiopericytoma. 24 HEP derives from pericytes that are capillary subendothelial lining cells that are particularly concentrated in areas of increased blood pressure such as distal extremities. 55 Myopericytoma derives from myopericytes, the intermediate cell type located in the subendothelial lining of arterioles and venules. 32, 46, 53, 64 PWTs reported in dogs are HEP, 23, 25, 29, 41, 48 angioleiomyoma/sarcoma, 10, 39 closely resembling the human counterpart, 39 and glomus tumors. 14 Canine HEPs have been included among the malignant unclassified soft tissue tumors by the World Health Organization classification, 29 and they are described as having a characteristic pericapillary whorling growth. 23, 25, 29, 41, 48 On the contrary, in human, perivascular whorling, as a major morphologic finding, is diagnostic for myopericytoma. 24, 68 Myopericytoma seems not to have been reported in dogs; however, several cases of canine HEP closely resemble human myopericytomas. 24, 68 The differential diagnosis of HEP and myopericytoma in humans is based on histology and immunohistochemistry 24, 68 and their distinction has a clinical significance as HEP is mostly malignant 6, 31, 54, 68 while myopericytoma is generally benign. 42 According to the current literature, the diagnosis of canine HEP is based on the finding of major perivascular whorling associated with a variably and often discordant reported expression of desmin, pan-actin, S100, and CD34. 23, 29, 41, 48 The conflicting immunohistochemical data on canine HEP suggest that “canine HEP” may be a nonspecific term indiscriminately applied to several neoplasms that are actually of different histologic origin.
The aim of this study is to evaluate a spectrum of canine PWT with typical HEP cytologic features 9 in order to delineate their histologic and immunohistochemical profile and thereby further characterize their cellular origin and clinical behavior.
Materials and Methods
Cases
We collected 20 cases of canine PWT from 2002 to 2004. Fresh tissues from excisional biopsies were provided by the surgical unit of the Department of Veterinary Clinical Science of the School of Veterinary Medicine of Milan. Age, gender, and breed of the dogs as well as location and size (largest diameter) of the tumors were recorded. Followup information from attending veterinarians was collected every 6 months (followup collection time, 2 to 4 years).
Cytologic specimens
Cytologic specimens were obtained from all cases by scraping fresh tissue samples. Cytologic evaluation was performed on air-dried smears stained with May-Grünwald-Giemsa (Merck, Darmstadt, Germany), as previously described. 22 PWTs were selected on the basis of the finding of cytologic features previously described as diagnostic for canine HEP. 9
Tissue sampling for histology
Tissues samples were obtained from wedge biopsies or after excisional biopsy. One part of each biopsy was snap-frozen in isopentane cooled in liquid nitrogen. For immunohistochemistry, 5-μm cryosections were air dried, fixed in acetone (3 minutes), and stored at −70°. The remaining part of each biopsy was fixed in 10% neutral buffered formalin and routinely processed for histology.
Histology
Morphologic features were evaluated on 4- to 6-μm hematoxylin and eosin stained sections. Vascular growth patterns were recorded 6, 24, 30, 31, 51, 52, 54 as follows: staghorn, placentoid, 34, 35 pericapillary whorling, 6, 24, 31, 51, 52, 54 whorling from media or adventitia, 10, 30, 51 and radiating bundles from tunica media. 51 Additional growth patterns recorded were interlacing or parallel bundles, solid, storiform, myxoid, and Verocay-like (parallel rows of palisading nuclei). The presence of giant cells, necrosis, hemorrhages and multilobulation was also recorded.
Immunohistology
For immunohistochemical and immunofluorescence studies, a panel of 20 monoclonal and polyclonal antibodies was applied to 5 μm frozen sections of the tumors (Table 1). The primary antibodies listed inTable 1 were used to support or confirm the diagnosis of PWT (laminin, smoothelin, myosin, desmin, pan-actin, α-smooth muscle actin, heavy caldesmon, calponin, and CMG-3G5) and to exclude the following soft tissue tumors: epithelioid hemangiosarcoma (Factor VIII and CD31), 5 peripheral nerve sheath tumors (GFAP and S100), 23 melanoma (S100 and Melan A), 56 rhabdomyosarcoma (myoglobin), histiocytic sarcoma (CD18), 1 and biphasic synovial sarcomas (cytokeratin). 13 Most of these antibodies have been previously shown to crossreact with canine tissues. 1, 7, 8, 11, 18, 36, 39, 41, 65 For immunohistochemistry, sections were stained using a streptavidin-biotin peroxidase complex method as previously described. 1 Negative controls consisted of substituting specific antibodies with an isotype-matched irrelevant monoclonal antibody (anti-feline CD22) or omitting the primary antibody. Formalin fixed and frozen sections of canine skin and striated muscle were used as positive controls and to check for the normal distribution of the antigens. Evaluation of the expression of the CMG-G5 antigen was performed on frozen sections using immunofluorescence. For immunofluorescence, frozen tissue sections of the PWT and of normal canine skin were stained with the anti-CMG-3G5 monoclonal antibody (mAB). Samples incubated at room temperature with 3G5 mAB (1:60, 30 min), washed with phosphate-buffered saline for 10 minutes and incubated (1:100, 30 minutes) with rhodamine-labeled anti-mouse IgM, μ chain specific (Organon Teknica/ICN, Eschwege, Germany). Sections were finally stained with DNA fluorochrome Hoechst 33342 (Sigma, St Louis, MO) (0.25 mg/mL, 3 minutes), washed in phosphate-buffered saline (10 minutes) and a coverslip was applied with aqueous mounting media (Dako, Glostrup, Denmark).
List of source, type, and dilution of antibodies applied to canine Perivascular Wall Tumors.

Dako, Glostrup, Denmark; Sigma, St Louis, MO; Novocastra Laboratories Ltd, Newcastle UK; Chemicon International, Inc. Temecula, CA; Zymed Laboratories, San Francisco, CA; Serotec Ltd, Kindkington, Oxford, UK.
Diagnosis
In accordance with previously published histologic and immunohistochemical criteria, cases were assigned to one of the following classes: glomus tumor, 26, 68 HEP, 26, 29, 31 myopericytoma, 24 angioleiomyoma/sarcoma, 30, 51 angiomyofibroblastoma, 37 or angiofibroma. 52
Results
Clinical summary and distribution of lesions
Based on typical cytologic findings, a total of 20 cases of PWT were included in this study. Lesions were observed predominantly in crossbreed dogs (11 cases), Boxers (3 cases), and German Shepherds (2 cases) as well as in an Irish Setter (1 case), Labrador Retriever (1 case), Siberian Husky (1 case), and Miniature Schnauzer (1 case). Age of affected dogs ranged from 6 to 13 years (median age, 9.5). Eight dogs were female and 12 were males, and the female to male ratio was 0.67. All dogs had a single subcutaneous mass. Tumors were excised from the anterior limb (Fig. 1) in 7 dogs (elbow 4/7; radio-ulnar region 1/7; carpus 1/7; unspecified anterior limb region 1/7); the hind leg (knee, tarsus, and digit) in 3 dogs; the thorax in 2 dogs; and the lateral neck, back, sternal region, umbilical region, scrotal region, perineal region, (Fig. 2) and tail in 1 dog each. The largest diameter of the lesions was <2 cm in 2 dogs, 2–5 cm in 3 dogs, 5–10 cm in 4 dogs, and >10 cm in 5 dogs (Fig. 2). In 6 dogs the size of the lesion was not available.
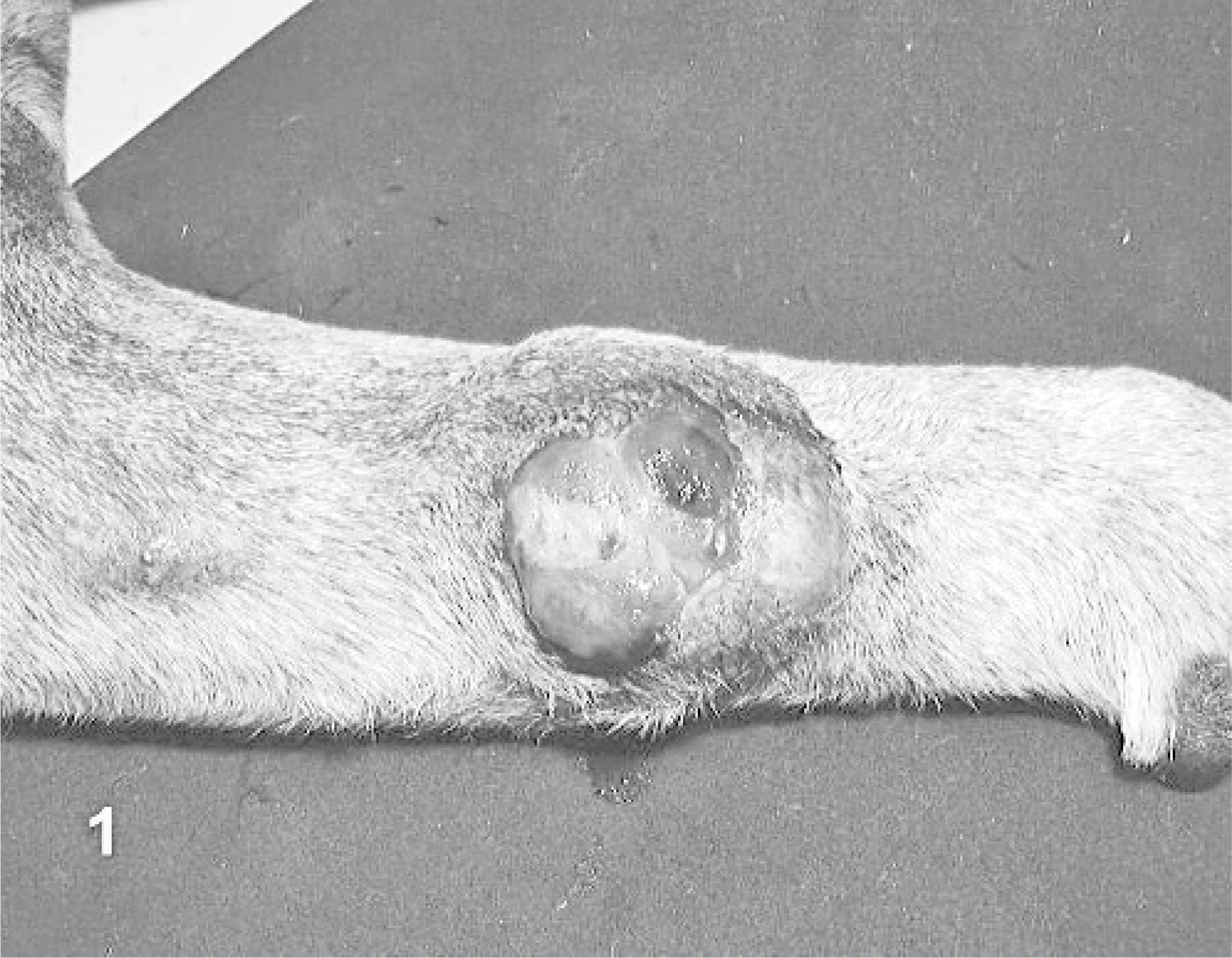
Dog No. 12. Ulcerated, 4.5-cm cutaneous angioleiomyoma in the radioulnar region.
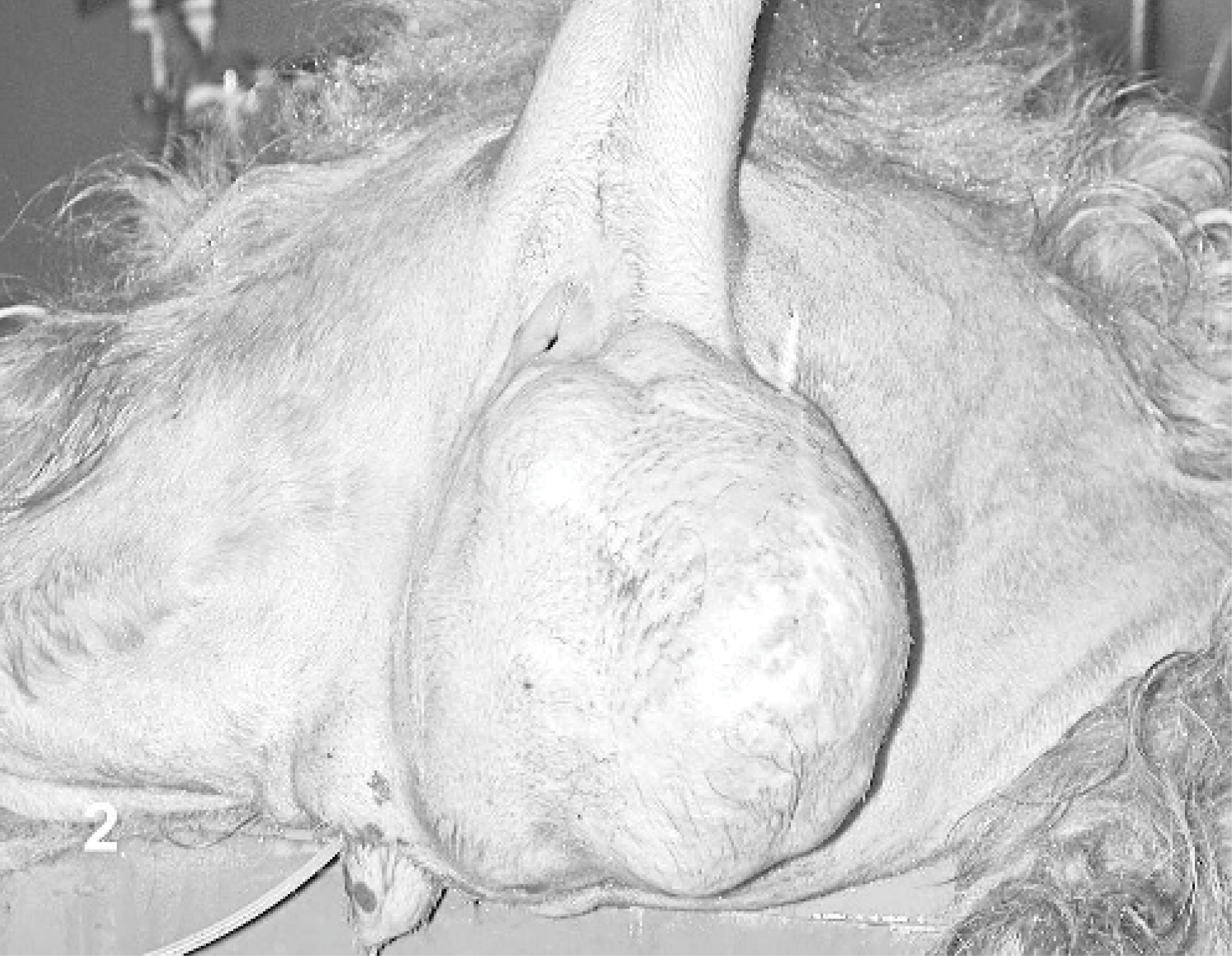
Dog No. 1. Large (> 20 cm), multilobulated hemangiopericytoma in the perineal region.
Gross morphology
On cut section, 10 dogs had a solid to multilobulated, tan to white surface (Fig. 3). In 8 tumors, mixed, multilobulated, solid to myxoid areas were evident. Dog No. 13 was characterized by a large mass composed of a mesh of branching vessels with multiple nodular thickening of the walls (Fig. 4). Dog No. 1 had a diffuse myxoid aspect. No correlation among gross aspect and diagnosis was possible.
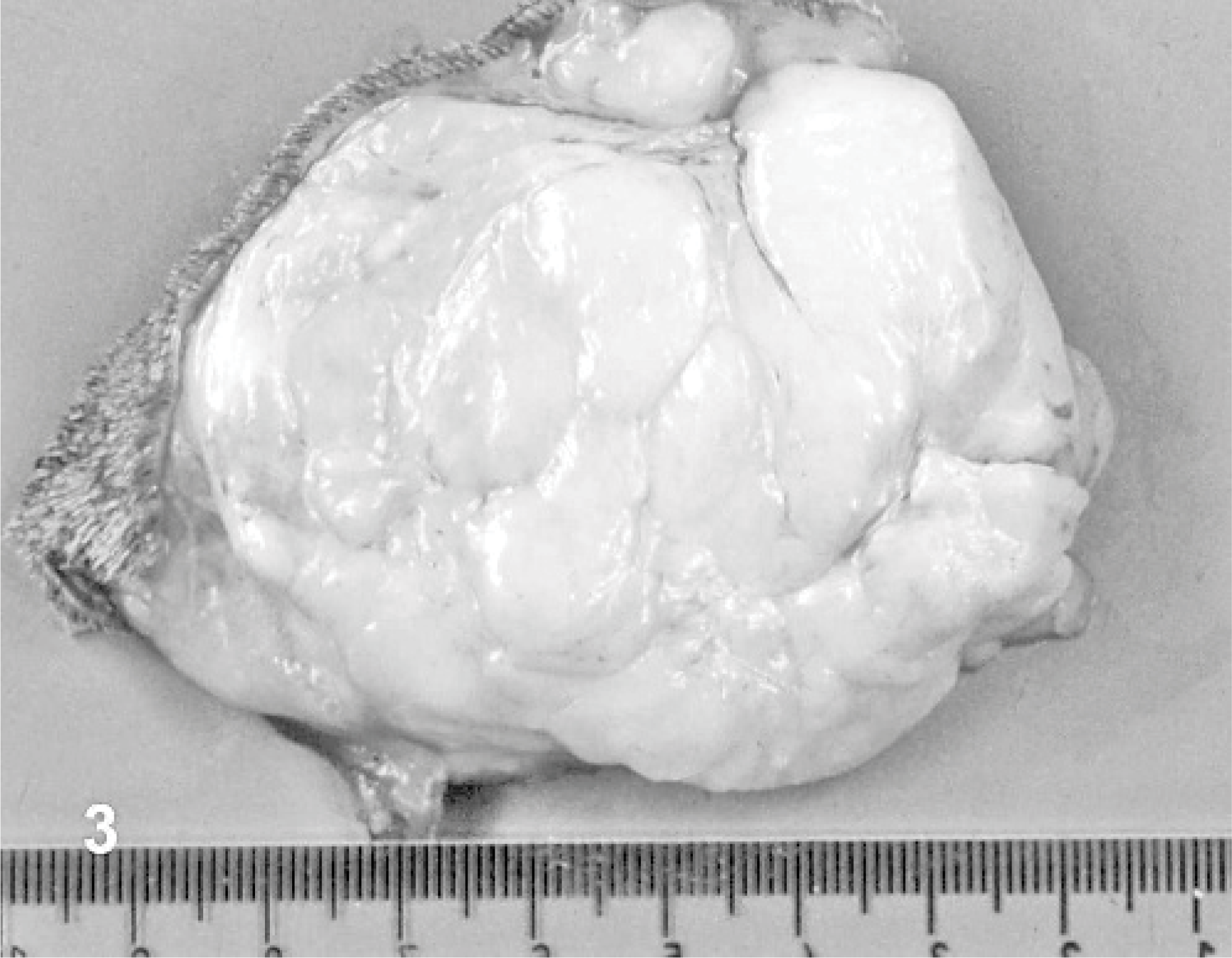
Dog No. 3. Cut section of a 7.5-cm multilobulated cutaneous myopericytoma of the elbow.
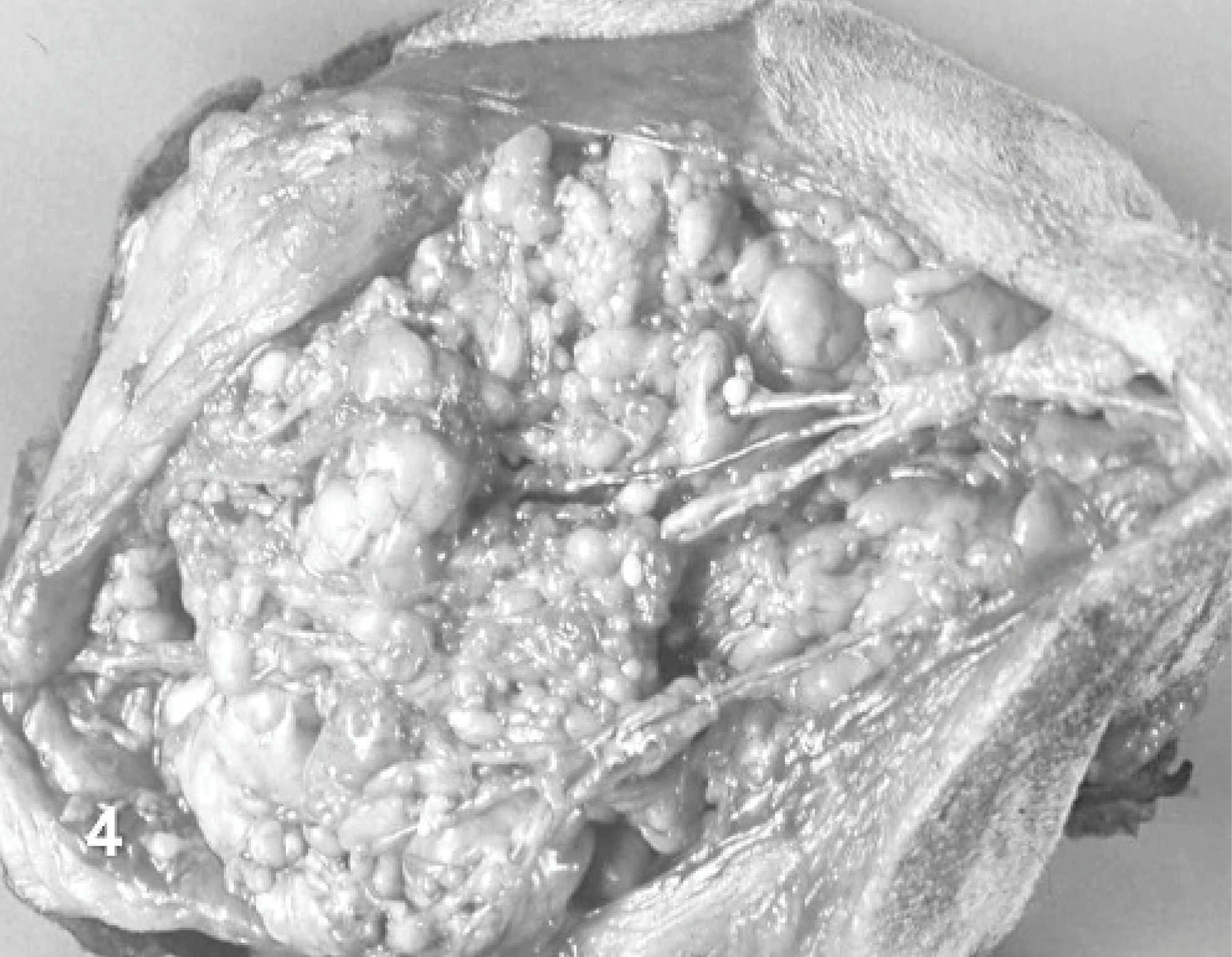
Dog No. 13. Cut section of an angioleiomyoma excised from the anterior limb composed of a mesh of branching vessels with multiple nodular thickenings of the walls.
Cytology
Cytologic features of the PWTs were as previously reported 9 (Fig. 5). Scrapings were highly cellular and composed of mesenchymal cells arranged in cohesive clusters and clumps. The mesenchymal cells were spindle shaped to stellate, had indistinct cytoplasmic membranes, and exhibited a variable amount of pale basophilic cytoplasm that often contained numerous nonstaining vacuoles. Binucleated and multinucleated giant cells, along with normal capillaries (Fig. 6), were commonly seen. Cytologic features were highly indicative of PWT as a group but did not discriminate among the different PWT histologic subtypes.
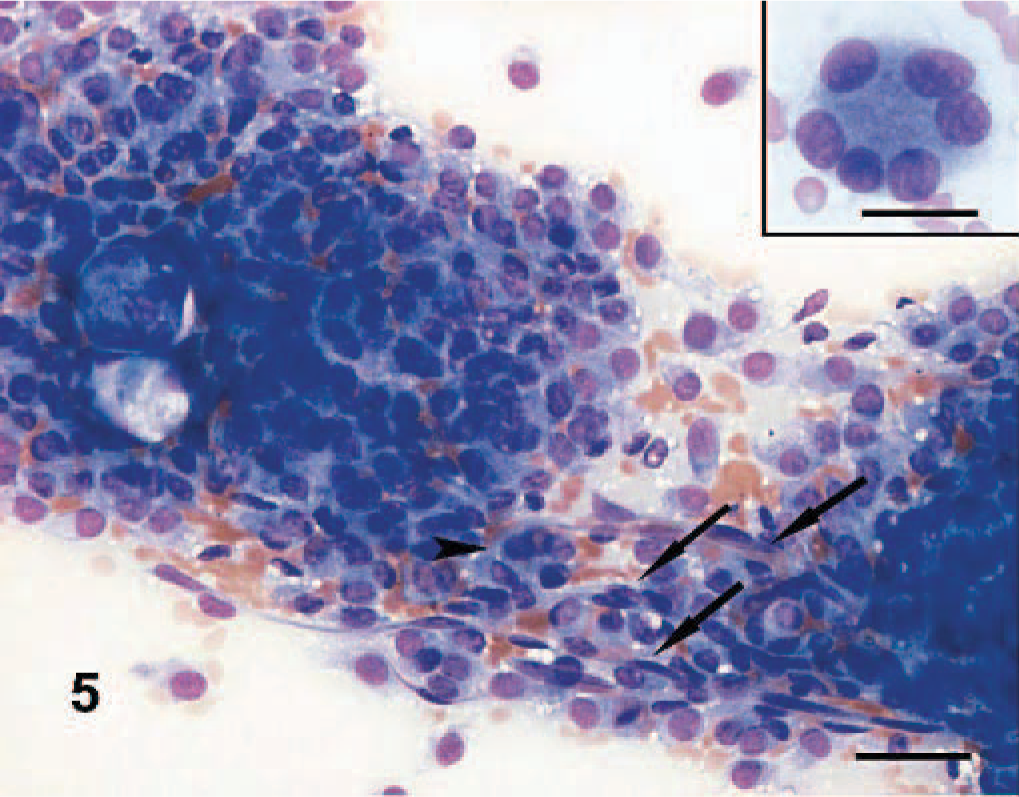
Dog No. 5. Myopericytoma: highly cellular cytologic sample composed of large groups of cohesive spindle cells. Multinucleated giant cells (arrowhead) and capillaries (arrows) are present. Scraping, May-Grünwald Giemsa stain. Bar = 30 μm. Inset: multinucleated neoplastic giant cell with peripheralized nuclei (crown cell). Bar = 20 μm.
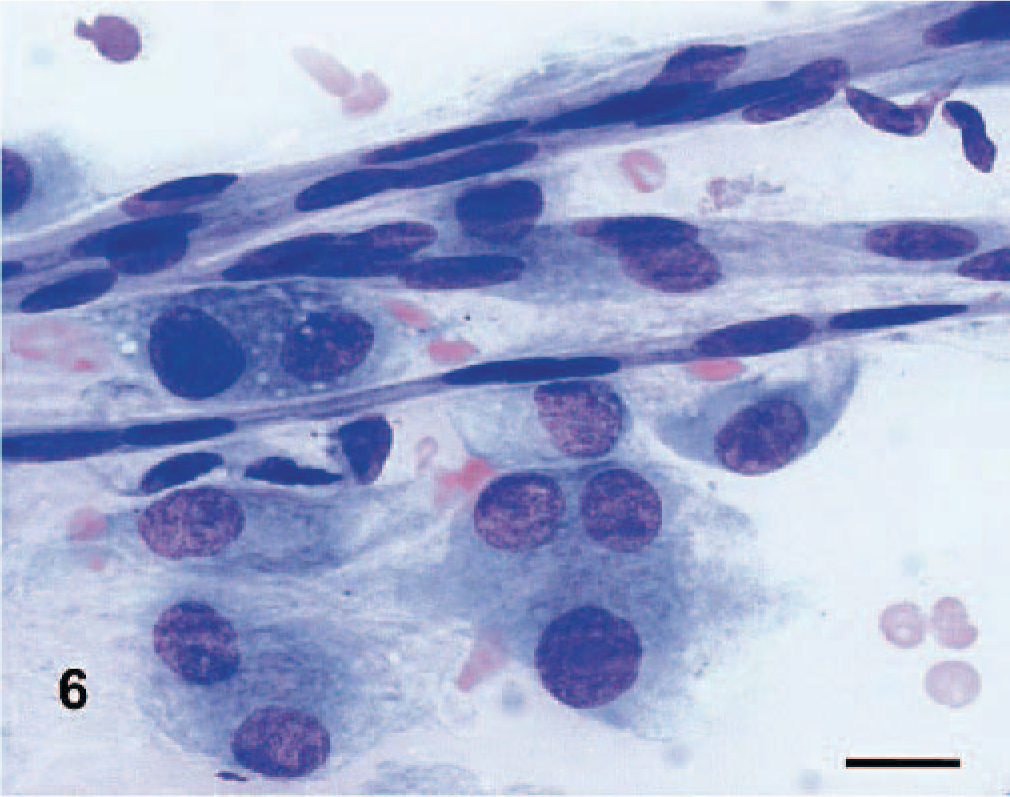
Dog No. 8. Presence of bi- to multinucleated giant cells and capillaries in a myopericytoma. Scraping, May-Grünwald Giemsa stain. Bar = 15 μm.
Histology
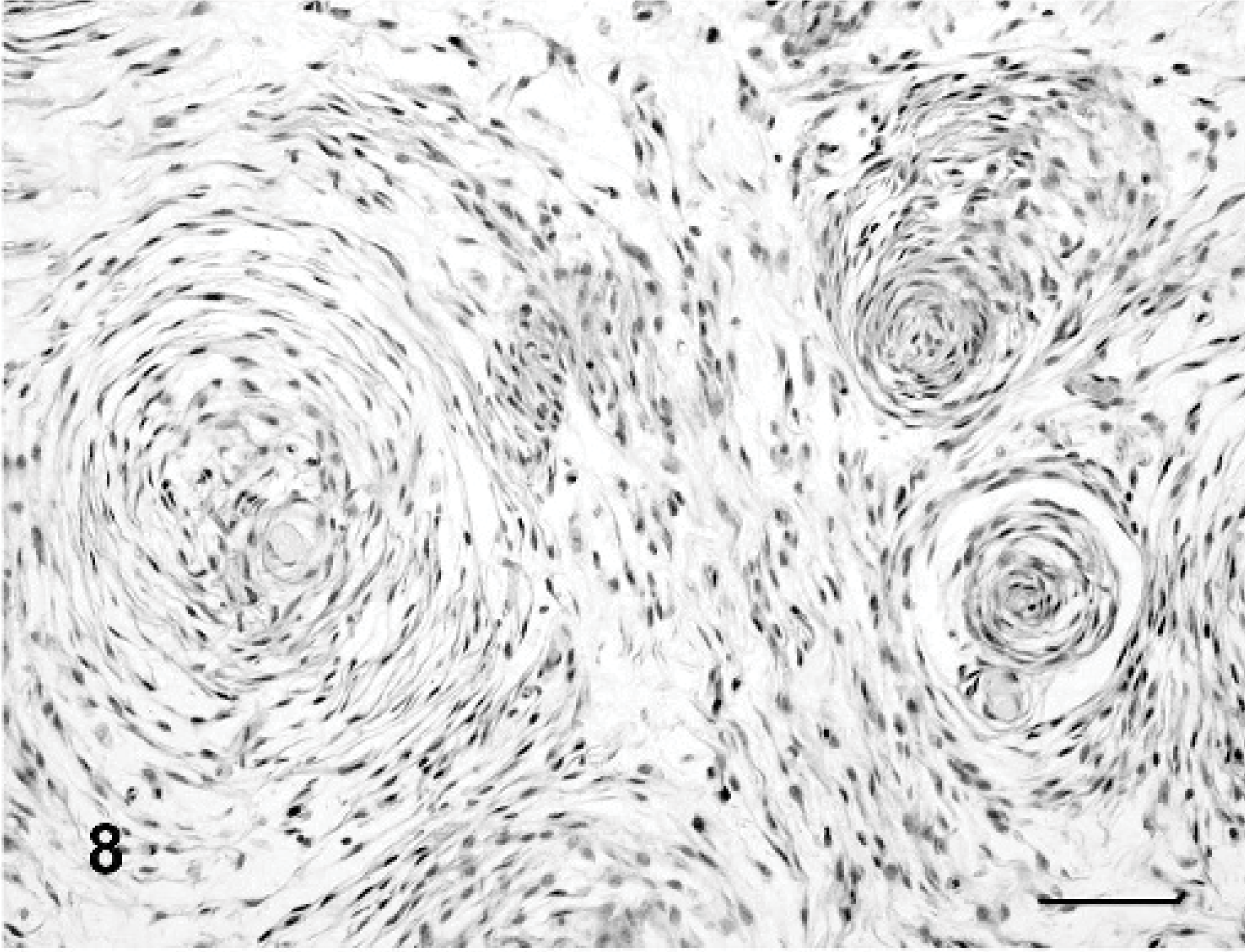
Histological findings are summarized inTable 2. In 17 of 20 dogs, 1 or 2 vascular growth patterns were present (Figs. 7– 10). Neoplastic satellite nodules were observed in 12 of 20 dogs. Necrosis (11/20) and hemorrhages (12/20) were common. Multinucleated neoplastic giant cells were observed in 18 of 20 dogs.
Morphology and phenotype of canine Perivascular Wall Tumors.∗
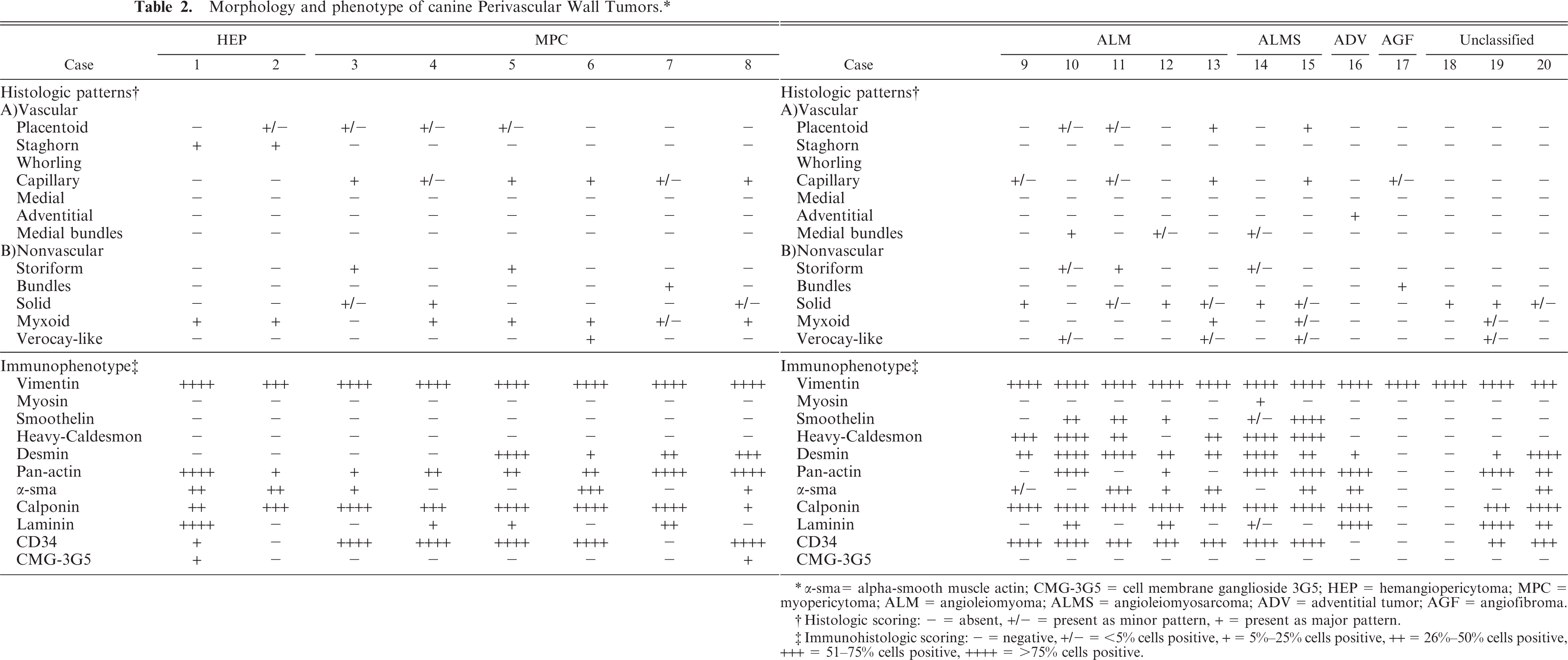
α-sma = alpha-smooth muscle actin; CMG-3G5 = cell membrane ganglioside 3G5; HEP = hemangiopericytoma; MPC = myopericytoma; ALM = angioleiomyoma; ALMS = angioleiomyosarcoma; ADV = adventitial tumor; AGF = angiofibroma.
Histologic scoring: − = absent, +/− = present as minor pattern, + = present as major pattern.
Immunohistologic scoring: − = negative, +/− = <5% cells positive, + = 5%–25% cells positive, ++ = 26%–50% cells positive, +++ = 51–75% cells positive, ++++ = >75% cells positive.
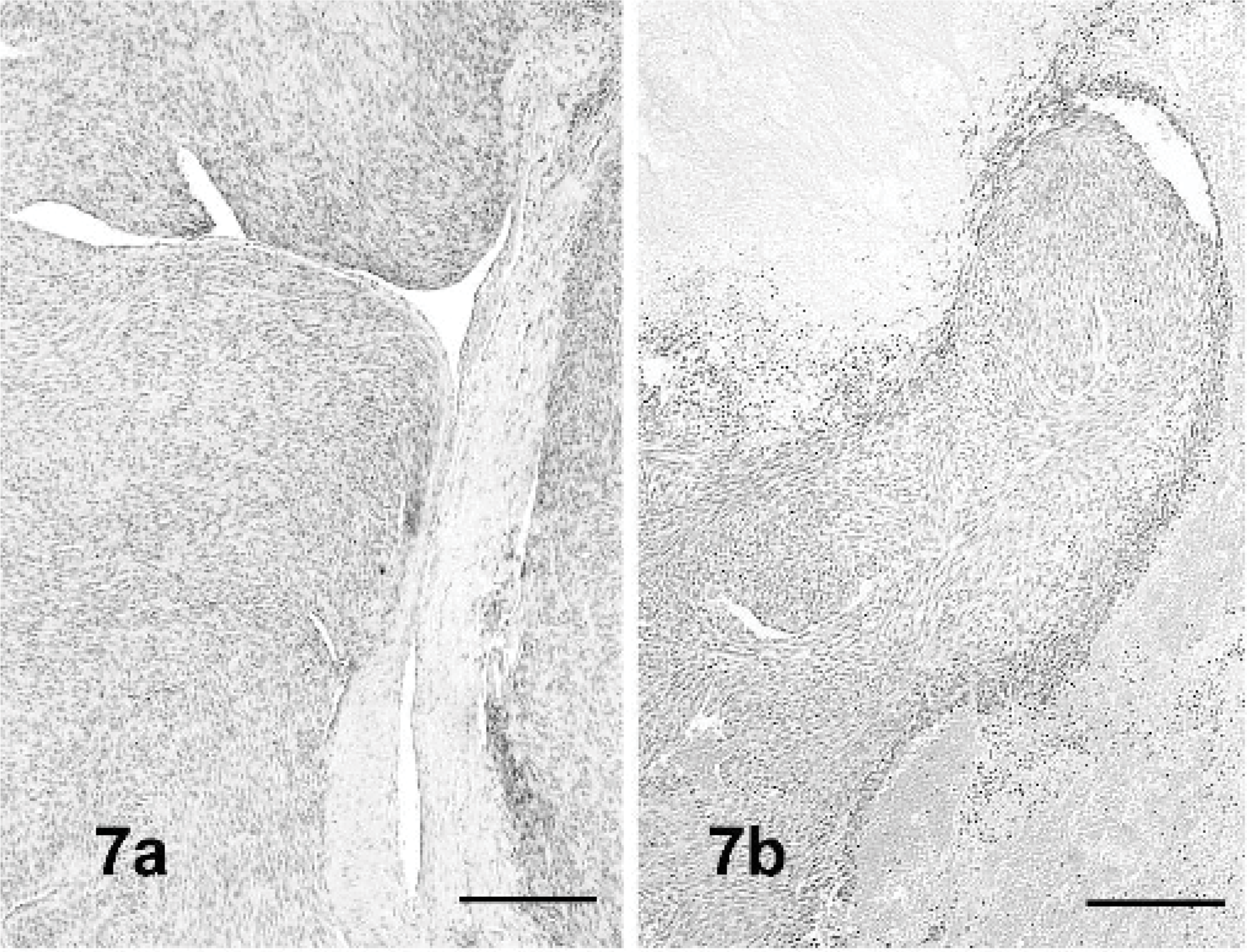
Dog Nos. 12 and 14. Growth patterns of angioleiomyomas. Neoplastic leiomyocytes originating from media of a large artery (7a) or from the subintimal lining of medium-sized vessels (7b). Hematoxylin and eosin (HE) stain. Bar = 120 μm.
Myopericytoma, dog No. 6. Perivascular concentric arrangement of neoplastic cells. Perivascular whorling is associated with myxoid areas. HE stain. Bar = 70 μm.
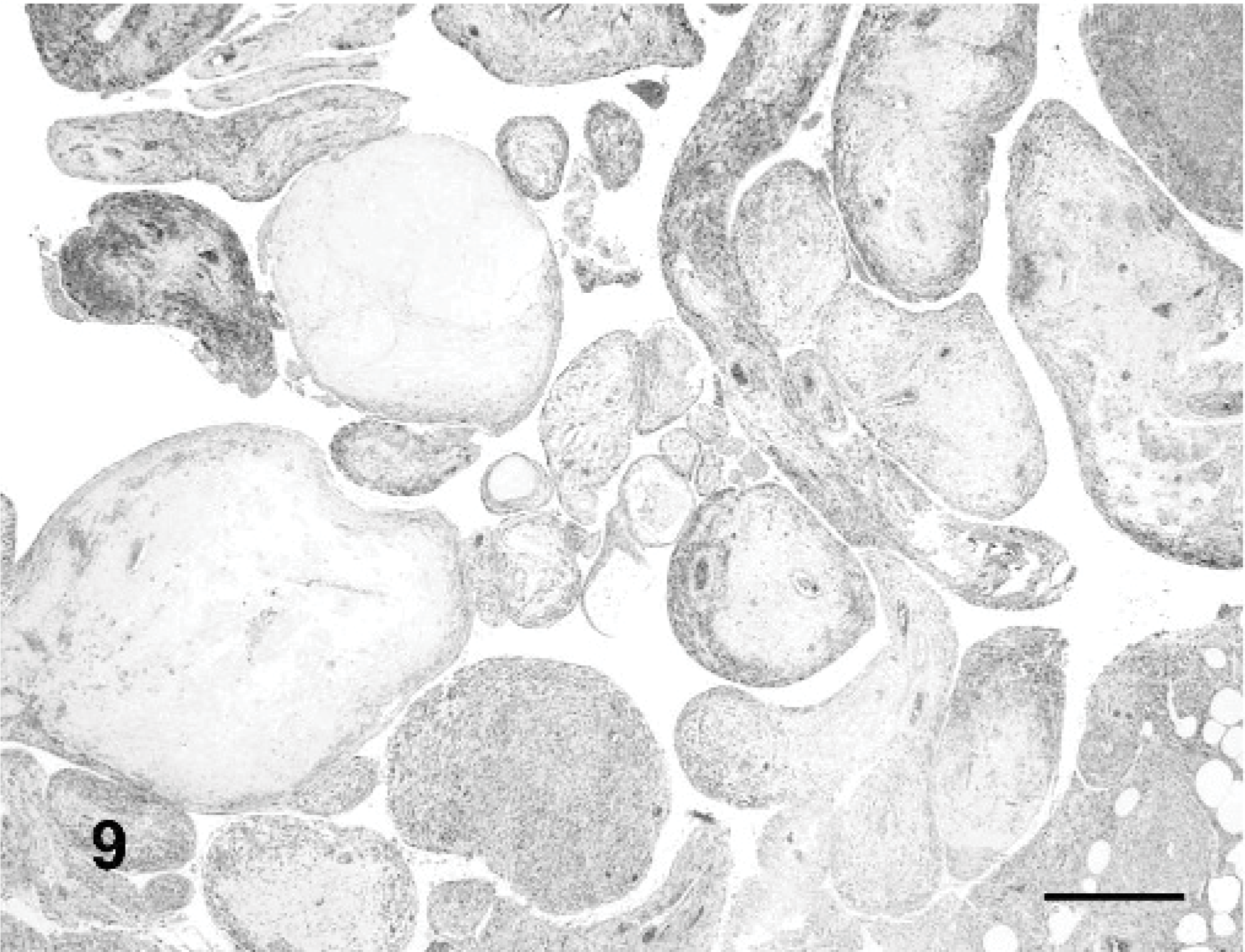
Myopericytoma, dog No. 1. Intravascular placentoid growth pattern. HE stain. Bar = 70 μm.
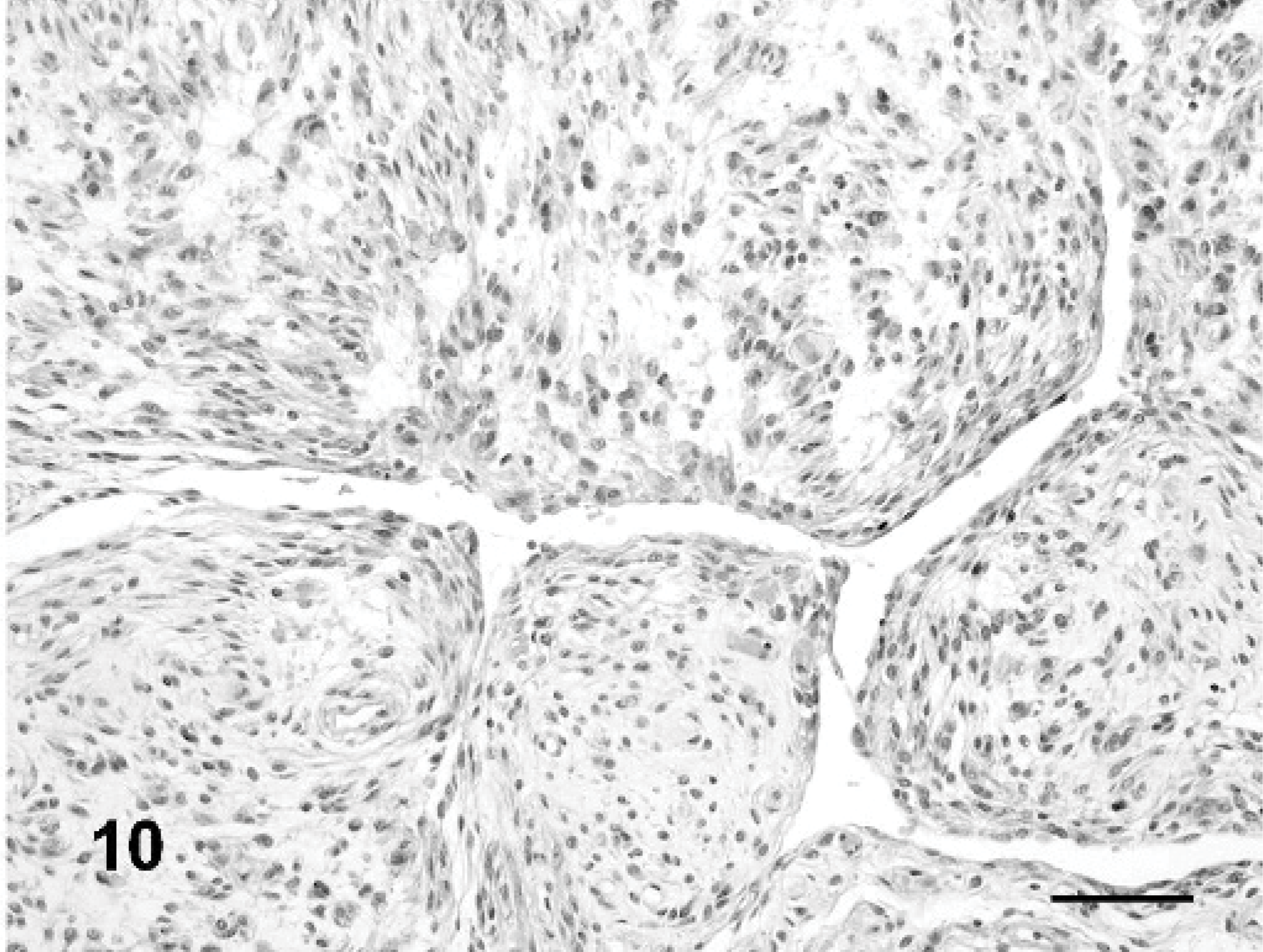
Hemangiopericytoma, dog No. 1. Staghorn and myxoid growth patterns. HE stain. Bar = 70 μm.
Immunohistology
Immunohistochemical and immunofluorescence results are listed inTable 2. Cytokeratin pool (AE1/AE3), CD18, CD31, Factor VIII, glial fibrillar acidic protein (GFAP), melan A, myoglobin, neuron specific enolase (NSE), and S100 were negative in all dogs with PWT. The clone Qbend/10 did not react with the vascular endothelium and was considered noncrossreactive with dogs. CD34 (clone 1H6) stained the endothelium of normal capillaries in frozen sections and identified positive neoplastic cells in 15 of 20 dogs. All dogs were intensely vimentin positive. Most of the dogs had an intermediate phenotype showing variable expression of muscular markers and CMG-3G5.
Diagnosis
A specific diagnosis was established in 17 of 20 cases (Table 2). In the three remaining cases a precise diagnosis was not achieved due to uncharacteristic immunophenotype and the absence of a perivascular growth pattern despite the typical cytologic findings.
HEP was diagnosed in 2 dogs. One tumor (dog No. 1) was located in the perineal area (Fig. 2) of a female, spayed, 11-year-old Irish setter and had a prevalent myxoid and staghorn growth pattern (Fig. 10) associated with abundant capillaries but no perivascular whorling. This HEP was characterized by expression of CD34, pan-actin, α-smooth muscle actin, calponin, CMG-3G5 (Fig. 11), cytoplasmic laminin and lack of reactivity with markers specific for well-differentiated muscle. The second HEP (dog No. 2) was located in the digit of the hind limb of a female, 10-year-old German shepherd crossbreed. This HEP had a staghorn and myxoid growth pattern with expression of pan-actin, α-smooth muscle actin and calponin.
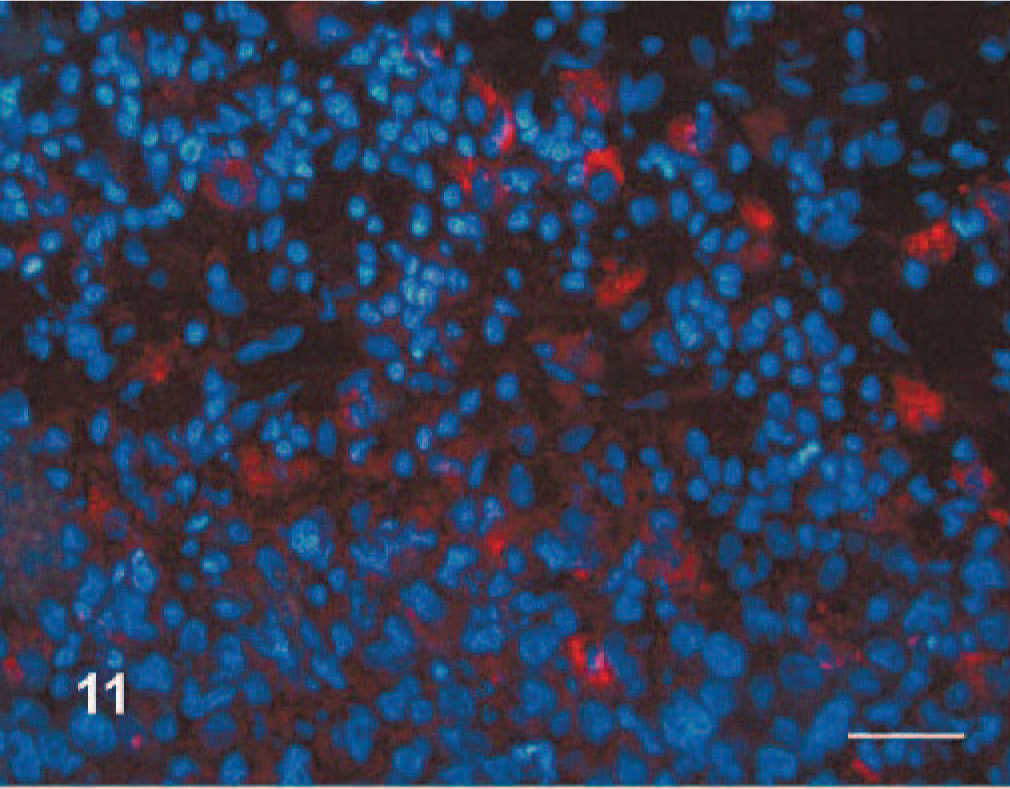
Hemangiopericytoma, dog No. 1. Multifocal cytoplasmic expression (red) of the cell membrane related ganglioside 3G5 in a hemangiopericytoma. Immunofluorescence for rhodanine labeled 3G5-mAb and DNA fluorochrome-Hoechst 33342 counterstain. Bar = 50 μm.
Myopericytomas (dog Nos. 3–8) were characterized by pericapillary whorling (Fig. 8) as a major growth pattern. Placentoid pattern was present in 3 myopericytomas (Fig. 9). Myxoid, storiform, and solid areas were frequent. Calponin (Fig. 12) and pan-actin were expressed in all myopericytomas. Desmin was observed in 4 of 6 dogs (Fig. 13a) and myosin was always negative (Fig. 13b). Cytoplasmic positive staining for laminin was identified in 3 of 6 dogs. CMG-3G5 was present only in dog No. 8. This case was characterized by an intermediate phenotype among myopericytoma and HEP and had a prevalence of myxoid growth with capillary whorling.
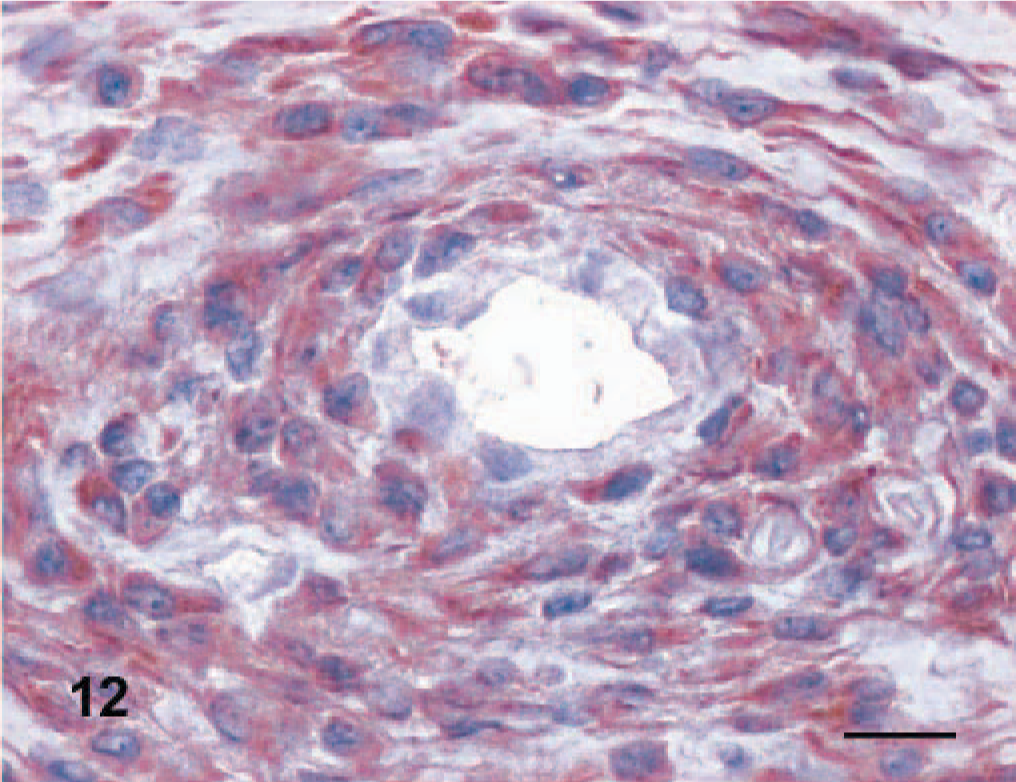
Myopericytoma, dog No. 3. Intense and diffuse cytoplasmic expression of calponin by perivascularly arranged neoplastic cells. Anti-calponin immunoperoxidase stain, AEC chromogen, hematoxylin counterstain. Bar = 30 μm.
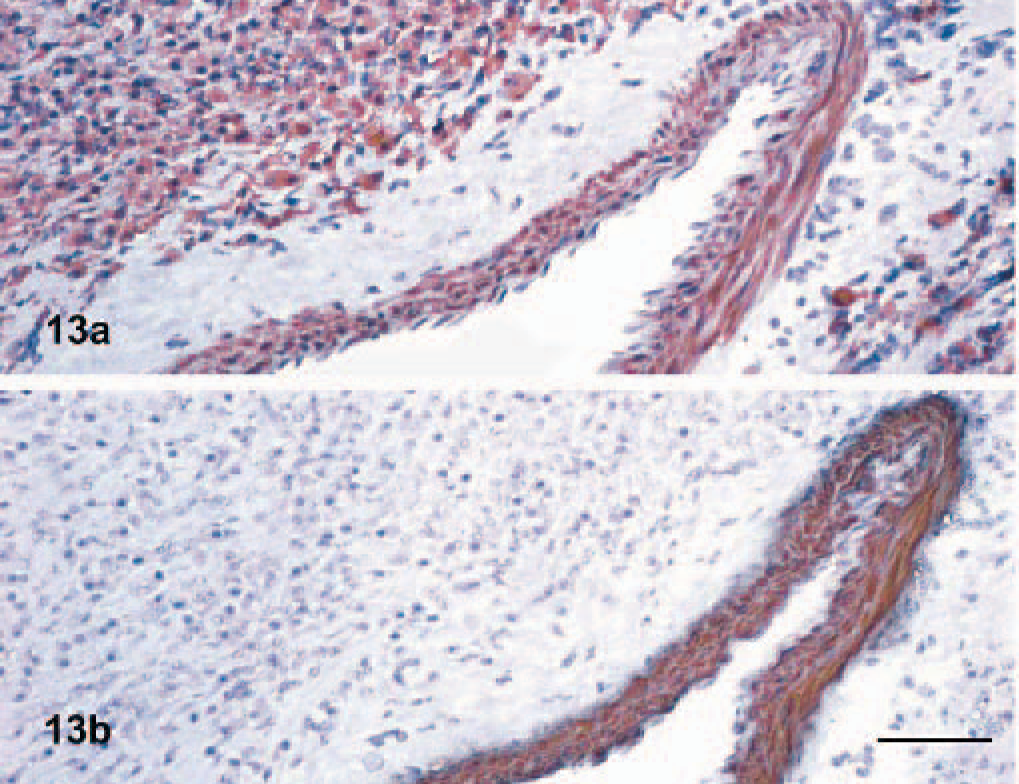
Myopericytoma, dog No. 7. Positivity to desmin (13a) and negativity to myosin (13b) of neoplastic cells in a myopericytoma. A normal vessel (internal positive control) not involved in the tumor is present. The tunica media of the vessels expresses desmin and myosin. Anti-desmin (13a) and anti-myosin (13b) immunoperoxidase stain, AEC chromogen, hematoxylin counterstain. Bar = 100 μm.
Vascular myomas (dog Nos. 9–15) were characterized by parallel to interlacing bundles of spindle cells, often originating from tunica media of large (Fig. 7a) to medium-sized vessels (Fig. 7b). Additional minor growth patterns were perivascular whorling and placentoid. Smoothelin expression was detected in 5 of 20 cases and 6 of 20 cases were heavy caldesmon positive (Fig. 14). These dogs (dog Nos. 9–15) also expressed desmin, actin, and calponin. All had a mature muscular phenotype that was considered diagnostic for this group of tumors. The diagnosis of angioleiomyosarcoma was based on cytologic atypia and increased mitotic rate and was confirmed by the malignant behavior.
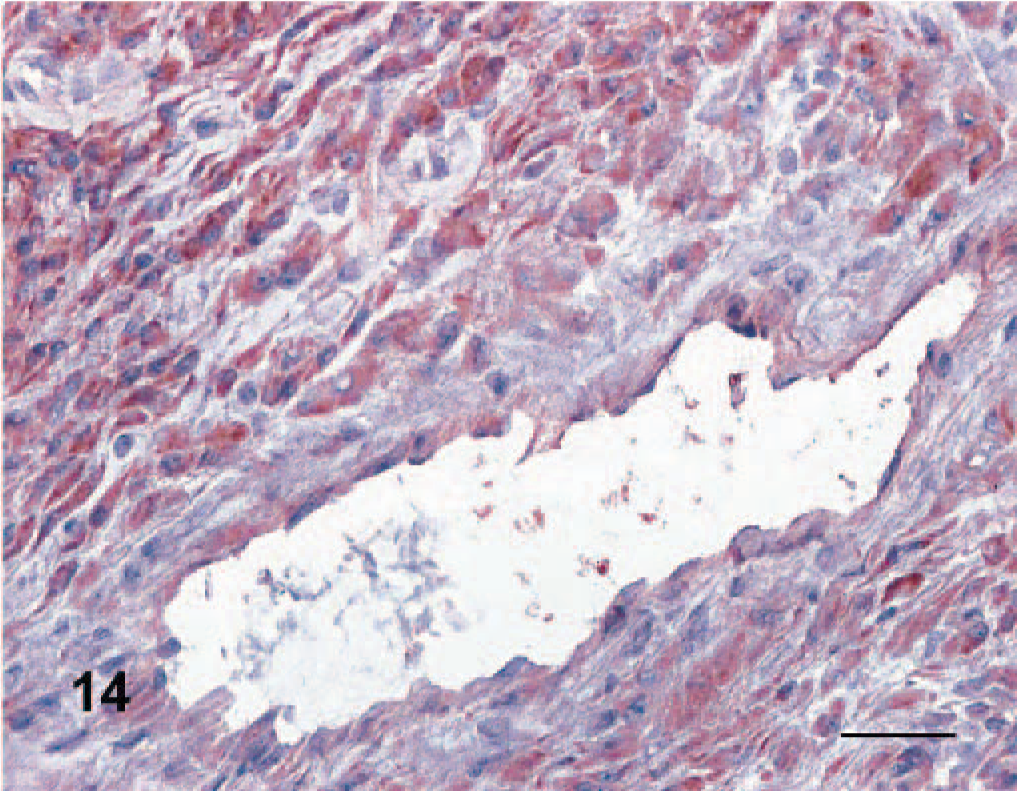
Angioleiomyoma, dog No. 13. Neoplastic cells are characterized by intense caldesmon expression. Anti-caldesmon immunoperoxidase stain, AEC chromogen, hematoxylin counterstain. Bar = 50 μm.
Two dogs (dog Nos. 17 and 18) expressed only vimentin. Dog No. 17 was diagnosed as angiofibroma based on the presence of perivascular whorling. In dog No. 16, adventitial whorling was associated with an intermediate muscular phenotype and was considered indicative of an adventitial myofibroblastic origin.
The differential diagnosis of canine PWT related to the most useful markers and histologic vascular pattern is summarized inTable 3. Based on immunohistochemical staining, a phenotypic continuum from HEP to adventitial tumors was evidenced in 16 dogs (dog Nos. 1–16). Contractile protein expression (Table 3) was minimal in the HEP; the amount increased in the group of myopericytomas, was maximal in the myomas, and was progressively reduced among the adventitial neoplasms recapitulating the phenotypic expression of these proteins in the normal vascular wall cell types.
Summary of useful marker expression and vascular patterns in the differential diagnosis of canine PWTs; + or +/− indicates the most helpful markers and histologic patterns for the differential diagnosis of each single category of Perivascular Wall Tumor.∗

HEP = hemangiopericytoma; MPC = myopericytoma; ALM = angioleiomyoma; ALMS = angioleiomyosarcoma; ADV = adventitial tumor; AGF = angiofibroma; ∗CMG-3G5 = cell membrane ganglioside 3G5.
Followup
In all cases an excisional biopsy was performed. Followup was collected from 14 of 20 dogs (dog Nos. 1–5, 7, 8, 11–15, 17, 19). In 6 dogs tumors recurred (dog Nos. 1, 2, 5, 11, 14, and 15). Of these, 2 were more than 10 cm in size (dog Nos. 14 and 1), and all 6 had infiltration of surgical margins by the neoplastic cells. In 4 dogs, the tumor recurred once. Of these, 2 were angioleiomyosarcomas (dog Nos. 14 and 15), 1 was a myopericytoma (dog No. 5), and 1 was a HEP (dog No. 2). One angioleiomyoma (dog No. 11) recurred twice, but no reemergence was recorded 30 months after the third surgery. One HEP (dog No. 1) recurred 3 times. Tissue invasion and recurrence were the cause of death or euthanasia in 4 dogs: the 2 with HEP (dog Nos. 1 and 2) and the 2 with angioleiomyosarcoma (dog Nos. 14 and 15). Four dogs (dog Nos. 8, 11, 13, 19) were alive and free of disease with a followup of 12 to 24 months. Five dogs (dog Nos. 3, 4, 7, 12, 17) died of causes other than the tumor, but there was no recurrence of the PWT and there was a disease-free interval of 1.5 to 3 years. Margins of excision evaluated by histology did not contain neoplastic cells in 4 dogs (dog Nos. 3, 4, 7, 12, 17). Neoplastic cells extended to the surgical margins in 1 dog (No. 4).
Discussion
The simultaneous assessment of cytology, histology, and phenotype allowed the diagnosis of a specific PWT in 85% of the dogs evaluated. In accordance with morphologic and immunohistochemical criteria applied, 6, 24, 68 the most common canine PWTs were vascular myomas followed by myopericytomas; only 2 cases of canine HEP were diagnosed. Despite a “typical” cytology, a definitive diagnosis of PWT could not be obtained in 3 dogs because of the lack of a specific vascular growth model and a nondiagnostic phenotype. This might be attributable to a lack of differentiation of the neoplastic cells owing to downregulation of lineage-specific proteins that are necessary to allow the immunohistochemical identification of the cell of origin. 38 The loss of contractile proteins has been reported in gastric leiomyosarcomas in humans. 28, 58 A second possibility could be the derivation of the tumors from pluripotential subendothelial cells that are able to differentiate into several mesenchymal cell lineages. 24, 50
Cytologic evaluation did not allow distinction among the different types of canine PWTs, but it was highly indicative of a perivascular origin of the tumors investigated. Formerly these tumors were cytologically diagnosed as HEP. 9 The combination of moderate to high cellularity, cohesion of spindle cells, presence of capillaries, and multinucleation should be considered typical of canine PWT as a group as the coexistence of these cytologic features is unique and has not been observed in other types of canine cutaneous spindle cell tumors. 62 The presurgical cytologic screening was crucial for distinguishing PWT from spindle cell melanoma, angiosarcoma, histiocytic sarcoma, and other canine cutaneous tumors that have been reported in a cytologic survey. 22 Moreover, the pivotal role of cytology in the selection of our cases was confirmed by the immunohistochemical results that were characterized by S100 and GFAP negativity of all the cases selected. Thus, cytology was discriminating to differentially diagnose PWTs and peripheral nerve sheath tumors (PNSTs) and to allow us to exclude PNSTs from this study.
The combination of histologic pattern analysis and immunohistochemistry was essential for the differential diagnosis of canine PWT. Immunohistochemistry was useful for characterizing PWT and for differential diagnosis PWT from synovial sarcoma. The effectiveness of immunohistochemistry was attributable to the application of a large panel of antibodies on frozen sections and included a group of muscle markers also used in the study of human PWT. 6, 19, 24, 42, 65, 66, 68 The diagnosis of vascular myomas was supported by the identification of a mature muscular phenotype defined by smoothelin and/or h-Caldesmon (h-CD) positivity. This expression paralleled that described in humans. 40, 66 The h-CD is a calcium-dependent protein that regulates contraction 57 and is expressed by visceral and vascular mature smooth muscle cells, but it is absent in myofibroblasts and pericytes. 19, 40, 66 Smoothelin is a cytoskeletal protein that has an auxiliary role in contraction. 65 Both h-CD and smoothelin are expressed in the late phases of muscular cell embryologic development and identify well-differentiated, mature, smooth muscle cells. 19, 65, 67 Therefore, the identification of either one of these molecules seems useful in the differential diagnosis of canine PWT. 19, 40, 66 Negativity to myosin characterized most cases and may indicate a precapillary to capillary origin of most canine PWT.
Of note, only 1 canine angioleiomyosarcoma was myosin positive, although myosin expression was consistently observed in the wall of the normal large vessels present in the sections. Smooth muscle myosin is generally absent in subintimal contractile cells of the precapillary and capillary beds, 3 while it is expressed with increasing intensity especially in the postcapillary compartment. 33 Interestingly, perivascular cells of the peripheral precapillary and capillary beds are the cells that bear higher blood pressures 55 and that might be the cell type involved in the development of PWT as a consequence of continuous mechanical stress. Pan-actin and α-smooth muscle actin were widely expressed but were lost in some angioleiomyoma/sarcomas. This loss may derive from discordant contractile filament production, and not a lack of differentiation, as the maturity of the neoplastic cells was demonstrated by the expression of h-CD and/or smoothelin. 40, 57, 65
The main criteria useful for diagnosing canine myopericytoma in this case series were the presence of perivascular whorling in association with pan-actin, calponin, and/or desmin expression. These findings paralleled what has been described in humans. 24, 37, 45, 68 The combination of muscular markers identified in myopericytomas is considered indicative of an intermediate muscular phenotype. 19, 65, 66 Desmin is an intermediate filament of skeletal (Z zone), cardiac, and smooth muscle cells (dense bodies). 61 Thus, desmin is not specific for smooth muscle, but its expression increases with smooth-muscle cell differentiation. 61 Desmin expression has been reported in subendothelial smooth muscle cells and myopericytes of precapillary and postcapillary vascular beds 3, 46 and is absent in pericytes. 3 Desmin was expressed in canine angioleiomyomas/sarcomas and variably in myopericytomas, but it was negative in the 2 canine HEP. This paralleled the expression pattern reported in human PWT. 68 Noteworthy, desmin is considered the marker of choice to differentiate myopericytoma from HEP in humans. 21, 68 In contrast to what has been previously reported in veterinary medicine, 2, 41, 48 desmin should be consistently negative to diagnose canine HEP. One myopericytoma showed unusual features with the prevalence of a myxoid growth associated with CMG-3G5 expression. This tumor had morphology and reactivity partially overlapping with HEP, but it also expressed desmin, which is considered typical of myopericytomas. 68 This myopericytoma might derive from an intermediate pericytic-myopericytic cell type localized most likely in the precapillary bed. 46 The 2 HEP had growth and staining patterns that closely resembled the human counterpart. 68 CD34 and vimentin are the most frequent markers expressed by HEP in humans 6, 44, 49, 54, 68 and are associated with low-intensity pan actin and α-smooth muscle actin expression. 6, 31, 68 However, CD34 cannot be considered specific for HEP as CD34-negative HEP have been reported in humans 12 and the molecule is also expressed by other types of soft tissue sarcomas. 16 Regrettably, no data regarding calponin, h-CD and CMG-3G5 reactivity of human HEP are available to compare with our cases. Unfortunately, based on our findings and on the reported expression of CMG-3G5 by human melanocytes, 15 the expression of this antigen may be considered helpful, but not diagnostic, for canine HEP. Therefore, based on morphology, the diagnosis of canine HEP should be limited to those tumors that do not bear whorling growth and are characterized by prominent myxoid to staghorn aspects in association with variable expression to actin, CD34, and CMG-3G5, but negative to desmin and all mature muscular markers. The distinction of HEP and myopericytoma is important, because HEP is a malignant tumor while myopericytoma is generally benign in humans. 6, 31, 42, 54, 68 In our case series, 5 myopericytomas were benign and 1 myopericytoma recurred, but the second surgery was curative. On the contrary, the 2 HEP had a locally aggressive behavior with repeated recurrences that led to euthanasia of both dogs. Thus, the biologic behavior of canine myopericytomas and HEP in this case series paralleled what has been described in humans. 5, 31, 42, 54, 68
One angiofibroma was characterized by predominant pericapillary whorling associated with negativity to muscular markers, closely resembling the human counterpart. 52 Calponin is a proteinregulating contraction 59, 60 that was included in our panel as an indicator of a mature smooth muscle cell, similar to that of humans. 19 However, a wide expression was identified in canine PWT, and calponin was useful only to differentiate the angiofibroma from other PWT. An angiomyofibroblastoma was diagnosed on the basis of the prevalence of adventitial whorling in conjunction with an intermediate muscular immunophenotype and paralleled previous cases described. 37, 47 However, the specific origin from myofibroblasts needs ultrastructural confirmation by demonstration of fibronexus and stress fibers. 16
Overall reactivity of canine PWT evidenced a phenotypic muscular continuum with intermediate phenotypes between HEP, myopericytomas, and angioleiomyomas that recapitulated the normal progressive variation in amount and type of contractile proteins distributed along the peripheral vascular beds. 46 Our findings paralleled the observation that some human PWT also derive from intermediate differentiation cell stages between pericytes, myopericytes, and smooth muscle cells. 24 To partially substantiate this hypothesis, a subgroup of canine PWT were characterized by pericapillary myopericytoma-like growth as a minor pattern associated with a mature muscular phenotype characterized by h-CD, desmin, and smoothelin positivity. In humans, these PWT tumors are defined as myopericytoma-like perivascular myoma 45 and seem to have a subendothelial origin from a cell intermediate among myopericytes and smooth muscle cells of the metarteriolar vascular compartment. 46 Additionally, a myopericytoma with simultaneous expression of desmin and CMG-3G5 could have been derived from a transitional cell type among pericytes and myopericytes.
A potential diagnostic algorithm that could be applied to the differential diagnosis of canine PWT is proposed inFig. 15. As proposed by this algorithm, cytology and histology are essential, and immunohistochemistry helped diagnose PWT. A specific immunohistochemical panel should include antibodies recognizing h-CD, smoothelin, desmin, myosin, calponin, and CMG-3G5. All markers used in this report were also tested on formalin-fixed canine skin and striated muscle with and without antigen retrieval, but the majority reacted only with fresh frozen specimens. The antibodies of our panel that might be applied to formalin-fixed, paraffin-embedded tissue sections were limited to vimentin, cytokeratin, factor VIII, CD31, GFAP, MBP, S100, melan A, and the muscular markers α-sma, pan-actin, myosin and desmin. The CD34 clone Qbend10, which was previously reported to work on formalin-fixed specimens, 41 did not identify canine endothelial cells in frozen sections and was excluded from our panel. This technical problem has been previously reported. 20 On the contrary, CD34 (clone 1H6) stained canine endothelial cells and was expressed by most PWT and by the epithelioid angiosarcoma. Thus, CD34 is widely expressed in PWT and was not useful to distinguish among these tumors.
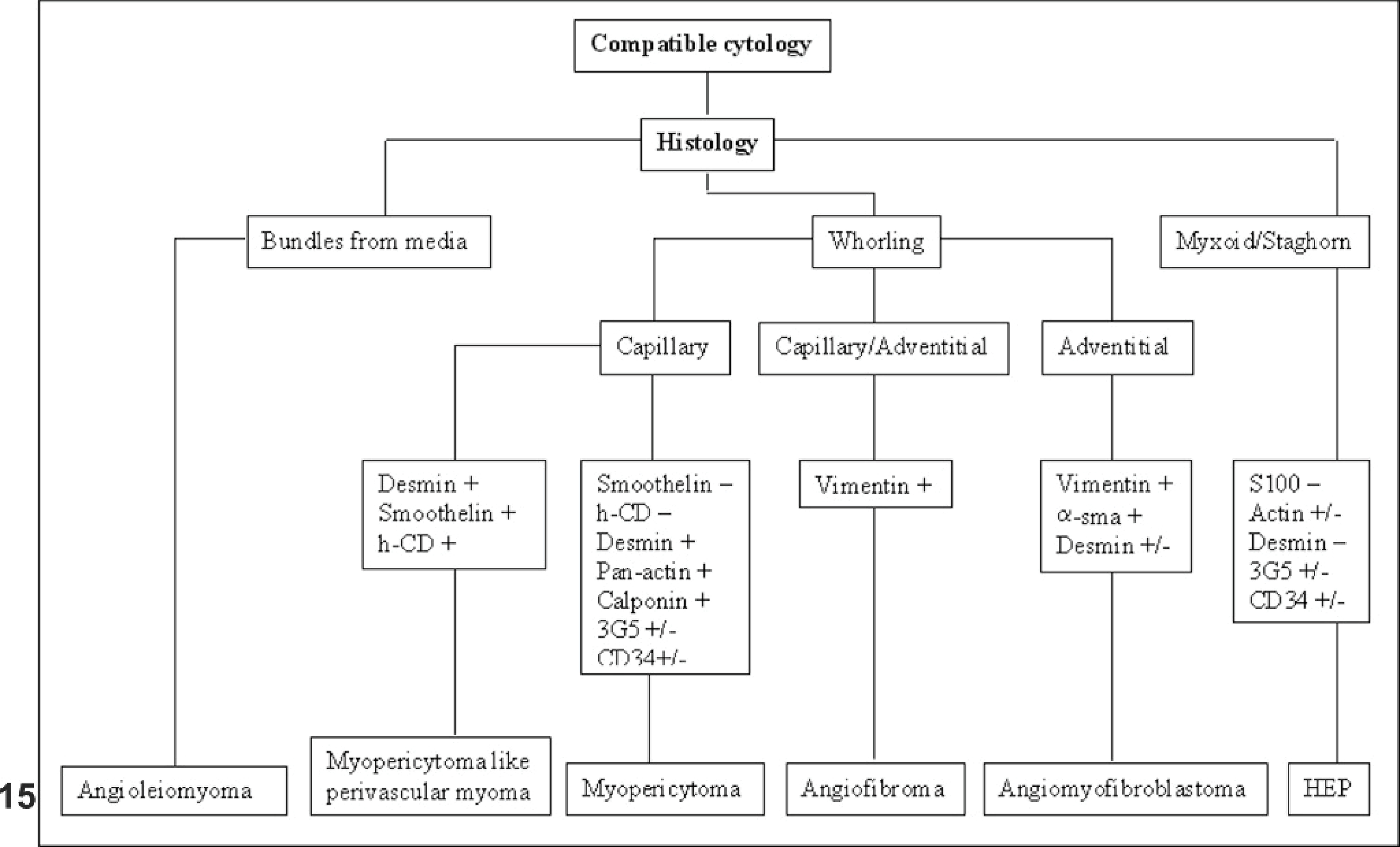
Diagnostic algorithm for the differential diagnosis of canine Perivascular Wall Tumor.
The main implications of this study are that, as reported in humans, 21 HEP is less frequent than previously believed and that canine PWT represents a mixed group of distinct biologic entities comprising hemangiopericytomas, angioleiomyomas, myopericytomas, and most likely angiomyofibroblastomas and angiofibromas. The diagnosis of canine PWT should be considered a multistep process in which cytology, histology, and immunohistochemistry may be necessary, but not always sufficient, to differentially diagnose these tumors.
