Abstract
The histologic classification of canine perivascular wall tumors (PWTs) is controversial. Many PWTs are still classified as hemangiopericytomas (HEPs), and the distinction from peripheral nerve sheath tumors (PNSTs) is still under debate. A recent histologic classification of canine soft tissue sarcomas included most histologic types of PWT but omitted those that were termed undifferentiated. Twelve cases of undifferentiated canine PWTs were evaluated by transmission electron microscopy. The ultrastructural findings supported a perivascular wall origin for all cases with 4 categories of differentiation: myopericytic (n = 4), myofibroblastic (n = 1), fibroblastic (n = 2), and undifferentiated (n = 5). A PNST was considered unlikely in each case based on immunohistochemical expression of desmin and/or the lack of typical ultrastructural features, such as basal lamina. Electron microscopy was pivotal for the subclassification of canine PWTs, and the results support the hypothesis that canine PWTs represent a continuum paralleling the phenotypic plasticity of vascular mural cells. The hypothesis that a subgroup of PWTs could arise from a pluripotent mesenchymal perivascular wall cell was also considered and may explain the diverse differentiation of canine PWTs.
Perivascular wall tumors (PWTs) include nonendothelial vascular neoplasms arising from structural or supportive cells of the vessel wall. 31 Whereas humans PWTs are considered a heterogeneous group of tumors with variable differentiation, 13 many canine tumors of this sort are still diagnosed as hemangiopericytoma (HEP) based on histologic features, including perivascular whorls, and variable immunohistochemical expression of desmin, pan-actin, S100, or CD34. 16 In addition, when extensive immunohistochemistry cannot be performed and neoplastic cells express variable or discordant antigens, the more general morphologic term of “canine spindle cell tumor with whorling pattern” has been recommended. 18 In fact, the whorling pattern, once regarded as distinctive of canine HEP, 16 is considered nonspecific and has been identified in numerous tumors, including human myopericytoma and angioleiomyoma. 31 Currently, the World Health Organization classification of human tumors of soft tissues recommends the term myopericytoma—first introduced by Requena and Sangueza 27 —for pericytic tumors with perivascular myoid cell or myopericytic differentiation. 22 Myopericytomas usually follow a benign clinical course with occasional recurrence; however, although rare, malignant behavior has been reported for deep-seated lesions. 23
The canine HEP shares most of its morphologic and biologic features with human myopericytomas.2,17,31 Accordingly, Avallone et al 2 suggested that canine PWTs are a mixed group of tumors, including HEP, angioleiomyoma, myopericytoma, angiomyofibroblastoma, and angiofibroma. 2 For the specific subclassification of PWTs, characterization by cytology, histology, and immunohistochemistry has been recommended. 2 However, in some cases, this approach is insufficient for the lack of typical histologic patterns or a specific tumoral phenotype. Such cases were categorized as undifferentiated PWT. 2 In addition, much controversy and debate still pertains to the differential diagnosis between PWTs and peripheral nerve sheath tumors (PNSTs). 14
The current study was designed to assess the utility of transmission electron microcopy for the ultrastructural characterization and diagnosis of canine PWTs with indeterminate differentiation.
Materials and Methods
Case Material
From 79 previously evaluated PWTs, 2 12 cases that could not be classified based on morphologic and immunohistochemical criteria were selected for the study. The signalment of the 12 dogs and the site and size of each tumor are listed in Table 1. In each case, preoperative cytologic examination of a fine-needle aspirate had indicated spindle cell sarcoma, so a portion of the tumor had been fixed in formalin, a portion snap-frozen, and multiple samples obtained for electron microscopy.
Signalment and Tumor Location and Size in 12 Canine Perivascular Wall Tumors of Uncertain Classification.
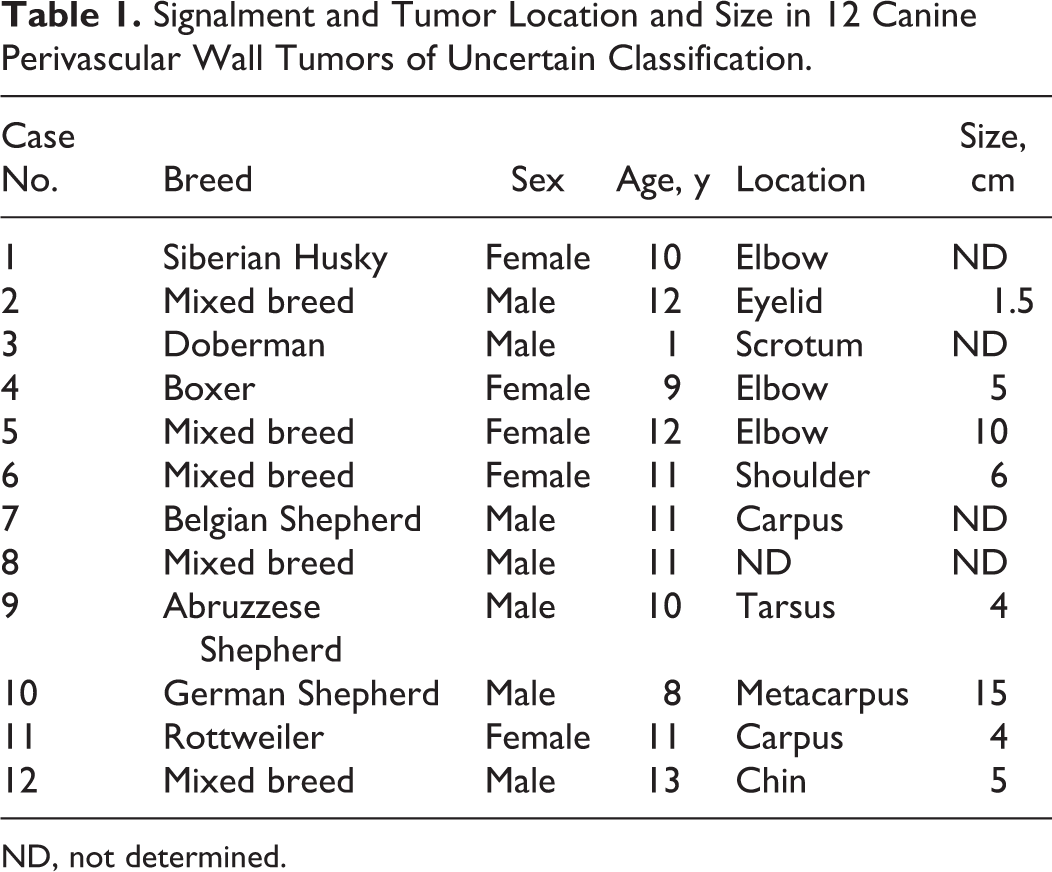
ND, not determined.
Histology and Immunohistochemistry
Samples for histologic evaluation were fixed in 10% neutral buffered formalin and routinely processed for histology. On hematoxylin-eosin (HE)–stained sections, morphologic features and vascular growth patterns were evaluated.2,20,27 For immunohistochemistry, cryosections of 5 μm were cut, air dried, fixed in acetone (3 minutes), and stored at –70°C. Frozen sections were stained with a streptavidin-biotin peroxidase complex method using a panel of antibodies, as previously described. 2 Primary antibodies, sources, and dilutions are listed in Table 2. Negative controls consisted of substitution of specific antibodies with isotype-matched irrelevant monoclonal antibodies or omission of the primary antibody. Frozen sections of canine dermis and striated muscle were used as positive controls and to check for the normal distribution of antigens.
Primary Antibodies for Immunohistochemistry of Canine Perivascular Wall Tumors of Uncertain Classification.

Transmission Electron Microscopy
Part of each biopsy specimen was fixed in 2.5% glutaraldehyde (pH 7.2) for 3 hours and postfixed in 1% OsO4 in 0.1 M cacodylate buffer for 60 minutes. The samples were subsequently dehydrated through an ethanol series, rinsed in propylene oxide, and embedded in epoxy resin (Durcupan; ACM Fluka, Sigma-Aldrich, St Louis, Missouri). One-micron sections were stained with toluidine blue and examined under the light microscope. Ultra-thin sections (70 nm) were prepared from selected areas, and grids were contrasted with uranyl acetate and lead citrate. Five different areas of each tumor were processed for electron microscopy, and at least 50 cells were evaluated per area. Electron micrographs were taken using a Zeiss EM900 electron microscope (Carl Zeiss, Oberkochen, West Germany). Unless otherwise stated, all chemicals were purchased from Electron Microscopy Sciences (Fort Washington, Pennsylvania).
Results
Histology and Immunohistochemistry
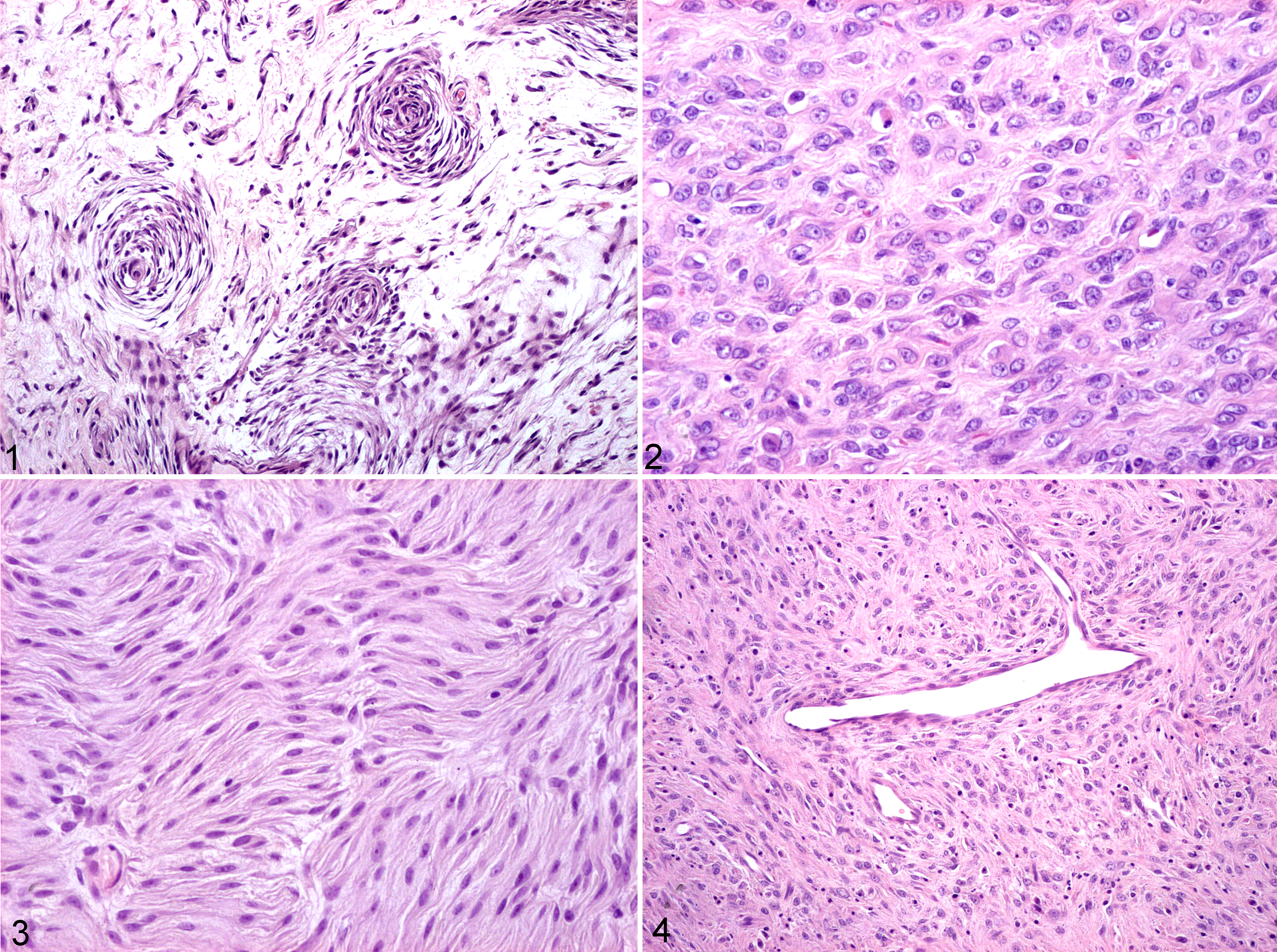
The histologic features and immunophenotype of the 12 PWTs are summarized in Table 3. Cases Nos. 1 to 10 had perivascular whorls (Fig. 1) (10/10), interlacing bundles (5/10), myxoid areas (6/10), solid (4/10) or storiform (3/10) patterns, and occasional staghorn vessels (1/10). In all cases, the neoplastic cells expressed vimentin. Smoothelin and glial fibrillary acidic protein were never expressed; S100 protein expression was observed in <5% of neoplastic cells from a single case (dog No. 12). Seven of 10 cases had desmin expression and lack of α–smooth muscle actin (α-SMA), consistent with asynchronous muscular differentiation. The remaining 3 tumors (dog Nos. 8–10) had absent or minimal muscular differentiation. In dog Nos. 11 and 12, cytologic features were typical of PWT, 2 but a perivascular growth pattern was absent (dog No. 11) or insufficient for a PWT diagnosis (dog No. 12). In dog No. 11, histologically characterized by solid areas (Fig. 2) and interlacing bundles (Fig. 3), α-SMA was expressed in less than 25% of the cells; calponin and caldesmon were not detected. In dog No. 12, the cells diffusely expressed calponin, α-SMA, and desmin and had a solid growth pattern with occasional staghorn vessels (Fig. 4).
Histologic, Immunohistochemical, and Ultrastructural Features of 12 Canine Perivascular Wall Tumors of Uncertain Classification.
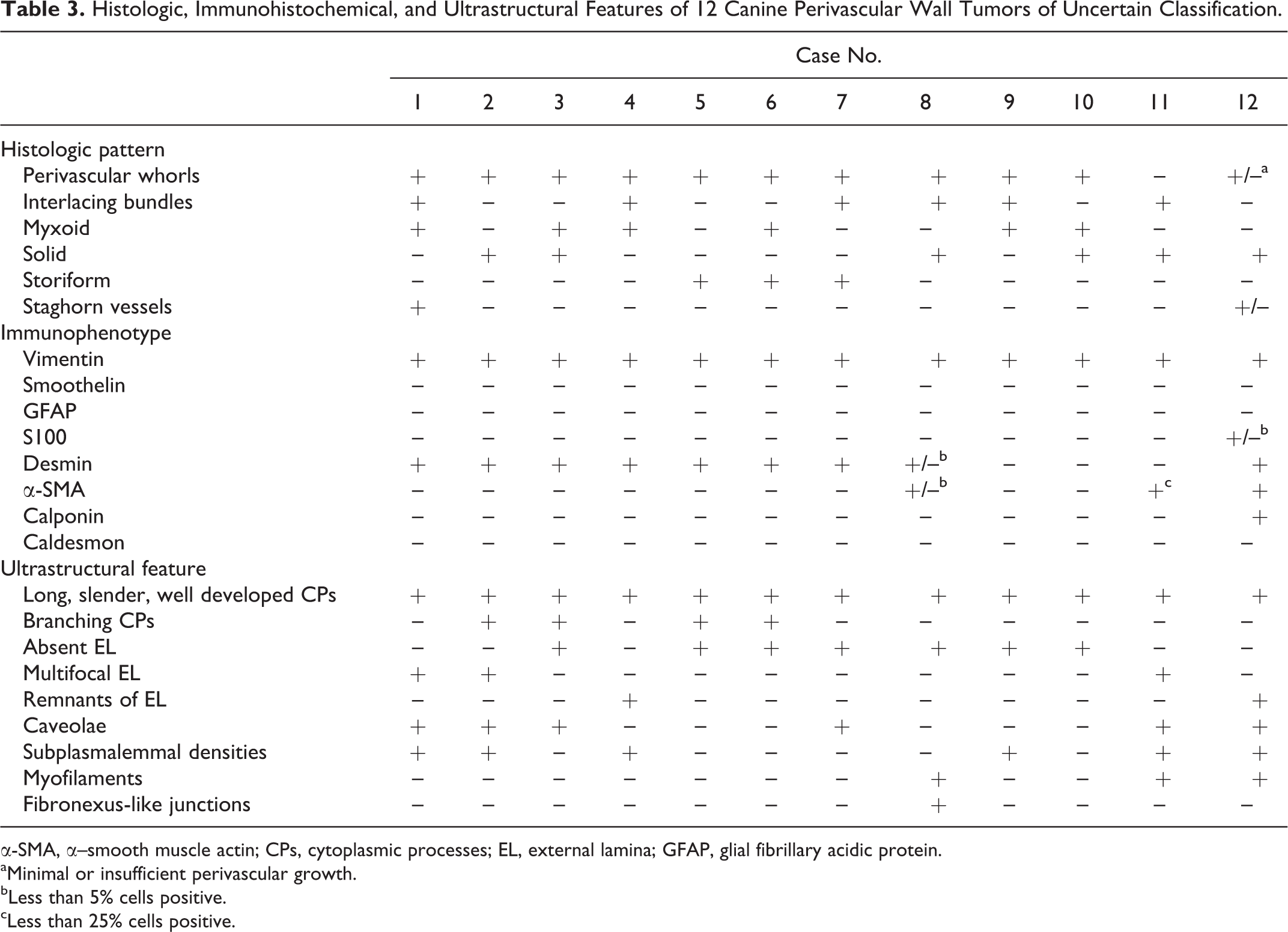
α-SMA, α–smooth muscle actin; CPs, cytoplasmic processes; EL, external lamina; GFAP, glial fibrillary acidic protein.
aMinimal or insufficient perivascular growth.
bLess than 5% cells positive.
cLess than 25% cells positive.
Electron Microscopy
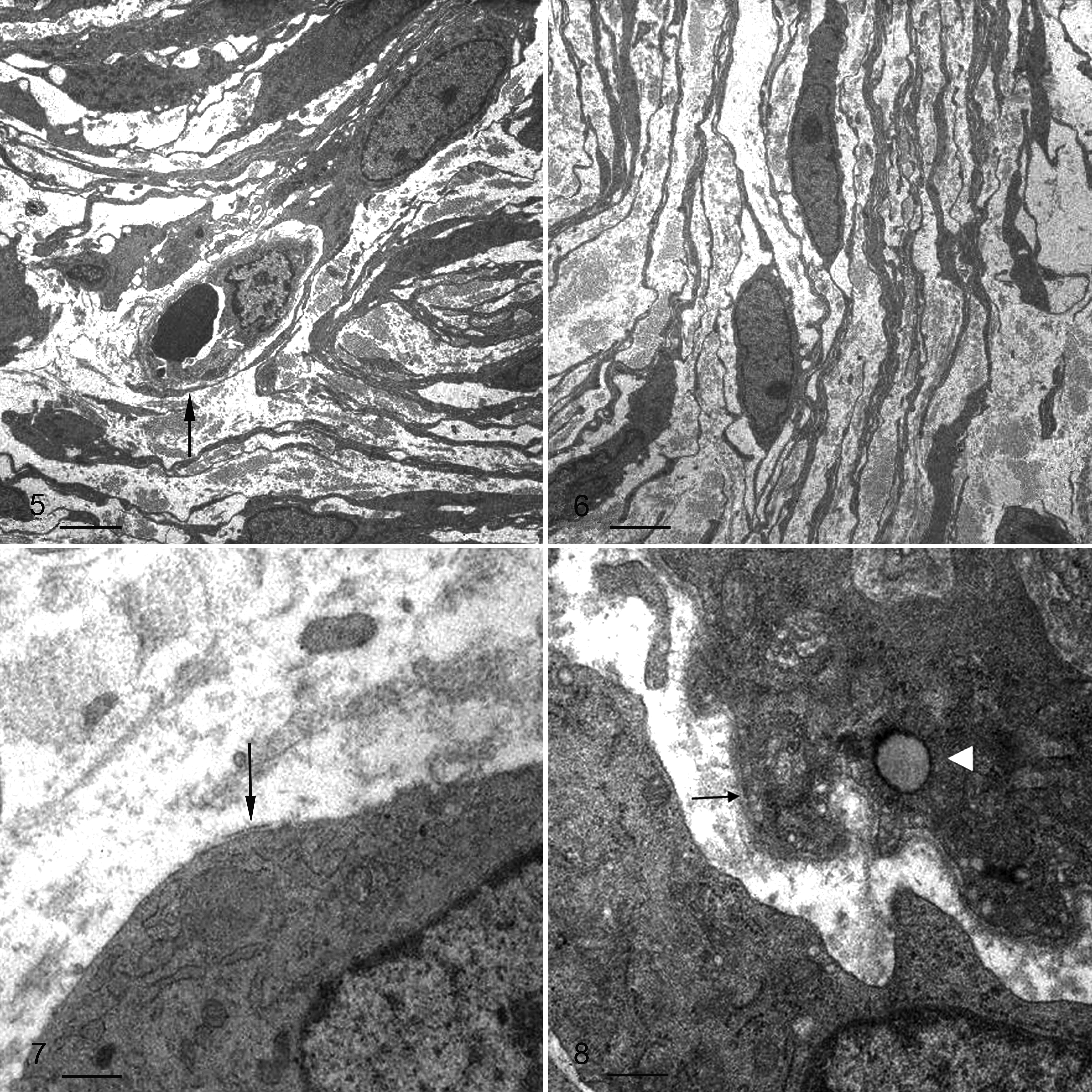
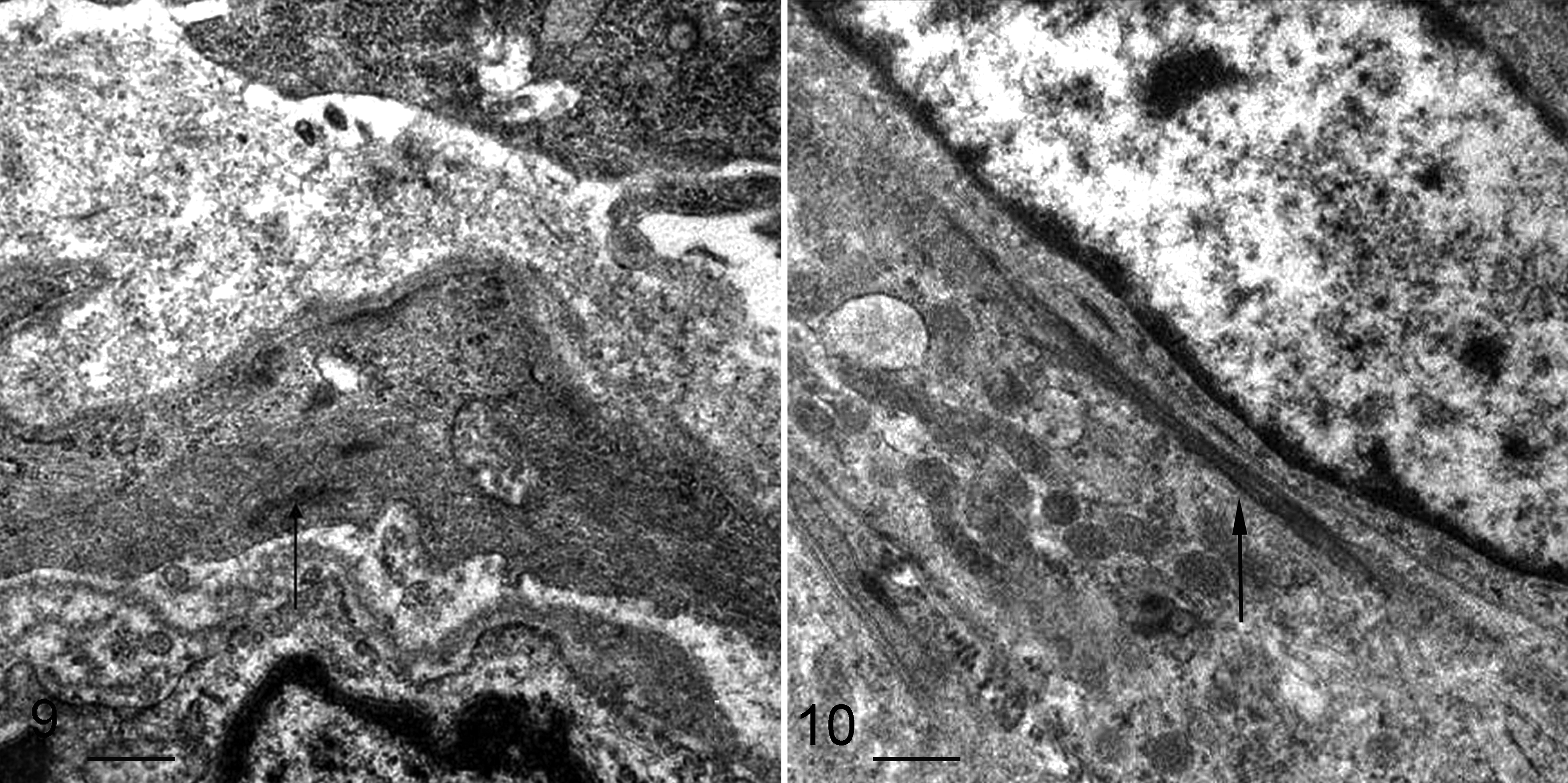
Ultrastructural findings are summarized in Table 3. In semithin sections, the distinctive feature for all cases was a perivascular whorling arrangement, apparent even in dog Nos. 11 and 12, in which it was not detected histologically. The association between neoplastic cells and vessel walls (eg, cytoplasmic processes encircling the vessels, neoplastic cells radiating from tunica media) was always documented. Multifocal myxoid matrix and highly branching cytoplasmic processes were also observed. In all cases, the neoplastic cells were spindle shaped (occasionally polygonal), with oval nuclei, slightly to moderately indented nuclear envelope, dispersed chromatin, and fibrillogranular nucleoli. Cytoplasmic processes, some (4/12) with branching (Fig. 5), were long, slender, and well developed in all 12 cases (Fig. 6). Intercellular junctions were absent. The external lamina was lacking (7/12), was discontinuous and poorly developed (3/12), or consisted of rare focal remnants (2/12) (Fig. 7). Lamina appeared amorphous, consisting of homogeneous, finely textured, and moderately dense material closely abutting plasma membranes (Fig. 8). Rare plasmalemmal caveolae (Fig. 8) and multifocal subplasmalemmal densities were observed in 6 of 12 cases. Occasional myofilaments (fine actin-sized filaments with focal densities) were present (3/12) (Fig. 9). Neoplastic cells contained intermediate filaments (vimentin bundles), numerous free ribosomes, prominent rough endoplasmic reticulum (11/12) with dilated stacks (7/12), and few (2/12) to numerous (10/12) mitochondria (ring shaped in 5/12). The cytoplasm contained occasional glycogen aggregates (3/12), lipid droplets (4/12), or myelin figures (1/12). Neoplastic cells of dog No. 8 had poorly developed fibronexus-like junctions, consisting of longitudinal fibrils projecting from the cell surface into the extracellular space (Fig. 10).
Definitive Diagnosis
The combination of immunohistochemical and ultrastructural features confirmed the diagnosis of PWT in all cases and identified variable degrees of tumor cell differentiation. A diagnosis of PNST was ruled out in each case based on the immunohistochemical expression of actin and/or desmin (dog Nos. 1, 2, 8, 11, 12) and the absence of typical PNST ultrastructural features (dog Nos. 3–7, 9, 10)—namely, a well-developed, continuous, or discontinuous basal lamina; rudimentary cell junctions; broad and occasionally entangled cytoplasmic processes with an onion skin arrangement; or long-spacing collagen or Luse bodies (collagen fibrils with periodicity markedly greater than the 64-nm periodicity of the common or native collagen).
Four categories of neoplastic differentiation were identified by combined immunohistochemistry and electron microscopy results: (1) poorly differentiated myopericytomas and myopericytoma-like tumors, (2) fibroblast-like PWTs, (3) poorly differentiated angiomyofibroblastomas, and (4) undifferentiated PWTs. The first category was characterized by variable myopericytic differentiation. In poorly differentiated myopericytomas (dog Nos. 1, 2), perivascular whorls, asynchronous muscular marker expression, and ultrastructural early myoid differentiation (poorly developed external lamina, rare subplasmalemmal caveolae and densities) were evident, although these cases lacked typical features of normally developed myopericytes (smooth muscle actin expression and intracytoplasmic myofilaments). Neoplastic cells of myopericytoma-like tumors (dog Nos. 11, 12) had morphology and immunoreactivity partially overlapping HEP but expressed early smooth muscle differentiation markers.
Fibroblast-like PWTs (dog Nos. 9, 10) were characterized by perivascular whorls and absence of myoid differentiation by immunohistochemistry or ultrastructure.
One case classified as poorly differentiated angiomyofibroblastoma (dog No. 8) was characterized by perivascular whorls, weak expression of muscular markers, and fibronexus-like junctions.
Typical features of the last category (undifferentiated PWTs) (dog Nos. 3–7) were perivascular whorls and asynchronous muscular marker expression but absence of specific ultrastructural features of myoid differentiation (rare or absent external lamina, caveolae and subplasmalemmal densities, absence of myofilaments).
Discussion
In this study, the ultrastructural spectrum of canine PWTs with uncertain classification was evaluated to identify typical features that may aid in their characterization. The combination of immunohistochemistry and ultrastructure supported a perivascular origin of all tumors. The variation in degree of differentiation led to the identification of 4 categories: poorly differentiated myopericytomas and myopericytoma-like tumors, fibroblast-like PWTs, poorly differentiated angiomyofibroblastomas, and undifferentiated PWTs.
The first PWT category, poorly differentiated myopericytoma and myopericytoma-like tumor, is based on intermediate features between HEP and myopericytoma.2,16,22 Both cases of myopericytoma-like tumors were useful to demonstrate that, although the typical whorling pattern could be missed by histology, myopericytoma can be still diagnosed by combining immunohistochemical and ultrastructural data. External lamina, myofilaments, subplasmalemmal caveolae and densities, and actin immunoreactivity were all useful features to rule out pericytic and/or myofibroblastic differentiation. Interestingly, the cytological features typical of PWT 2 in these tumors were more diagnostically useful than the histologic pattern. Tumor cells with early myoid differentiation can derive from a poorly differentiated myopericyte, in which the transcription of lineage-specific proteins is downregulated 21 or from a transitional cell between pericyte and myopericyte. 24 The current findings support the premise that canine PWTs represent a phenotypic continuum paralleling the progression from pericytes to myopericytes and smooth muscle cells in the vascular wall. 2 Hence, PWTs may actually originate from perivascular cells other than pericytes, as traditionally accepted in veterinary oncology, and therefore the new nomenclature of PWT should be preferred to the old HEP.
The second category of tumors identified in this study was the fibroblast-like PWTs. These tumors are generally diagnosed as HEP and have cells arranged in whorls that express vimentin and are negative for desmin and actin. 21 However, the lack of external lamina and caveolae, the classic ultrastructural features of neoplastic pericytes, 32 justified the diagnosis of fibroblast-like PWT. The likely cell of origin of fibroblast-like PWTs is the perivascular fibroblast, a cell lacking myoid differentiation. Perivascular fibroblasts do not have contractile filaments or basal lamina but have elaborate long, extremely thin cytoplasmic processes, 25 as demonstrated in these cases by electron microscopy. Perivascular fibroblasts are considered the cell population, second to pericytes, that is most likely to have mesenchymal stem cell potential. 5
A single case of angiomyofibroblastoma, representing the third category, was identified in this study. Its neoplastic cells were arranged around blood vessels and had myofibroblastic differentiation. 11 Electron microscopy is the only reliable technique to distinguish myofibroblasts from smooth muscle cells, a distinction that is often impossible by morphology or immunohistochemistry, 28 as for our case. The morphologic marker considered specific for myofibroblasts is the fibronexus junction, 11 although attenuated versions of these structures have been seen in aortic smooth muscle cells, 6 during in vitro transdifferentiation into myofibroblasts, 10 and in human endothelium in vivo. 12
The last group of tumors identified was the undifferentiated PWT. These cases could derive from pluripotent mesenchymal perivascular cells. 1 The ultrastructure of these cells has not been well characterized; however, the features of our cases overlap with those of the vascular wall resident progenitor cells, described by Pacilli and Pasquinelli, 25 that form the so-called vasculogenic niche and can differentiate into smooth muscle cells, pericytes, or fibroblasts. 25 The hypothesis that a subgroup of PWTs could arise from a pluripotent perivascular cell would explain the diverse differentiation of PWTs in this and previous studies 2 and is in agreement with our observations, as well as those of Requena et al 26 and Granter et al. 17
Peripheral nerve sheath tumors and PWTs may have overlapping microscopic features. 18 Occasional concentric whorls can be found in PNST, 18 although this pattern is not as extensive as that in PWTs. 16 On the other hand, the perivascular arrangement of tumor cells in HEP is not always detectable, because the proportion of neoplastic cells and vascular channels may vary. 8 Moreover, S100 protein is considered to be a sensitive but nonspecific marker of PNSTs both in human and veterinary medicine.14,29 Indeed, S100 expression in HEP 14 and lack of S100 expression in human and canine PNST14,19 have been reported. For a differential diagnosis, muscular marker expression can be useful since PNSTs do not express desmin, 18 which is commonly present in PWTs. 2 Although immunoreactivity for α-SMA has been described in human PNSTs, 7 this seems unusual and has never been reported for canine PNSTs. 14 The “simple” phenotype of undifferentiated and fibroblast-like PWTs, which lack the expression of muscular and nervous markers, in conjunction with ultrastructural features of branching cytoplasmic processes, pinocytotic vesicles, and subplasmalemmal densities, which are shared by PWT and PNST, 9 complicates the diagnosis. However, PNSTs have well-developed, continuous, or discontinuous basal lamina and rudimentary cell junctions,9,30 which were not present in the cases of this study. Moreover, benign neoplastic perineurial cells are orderly arranged, with parallel layers of thin, elongated cytoplasmic processes in an onion skin fashion. 15 The main diagnostic problem with undifferentiated PWTs is their distinction from low-grade, usually poorly differentiated, malignant PNSTs. Basal lamina is an important diagnostic feature of malignant PNSTs but may be scant and focal in the highly cellular and less differentiated form. 4 Ultrastructural features deemed useful for the diagnosis of malignant PNST include broad, occasionally entangled, cytoplasmic processes partially invested by basal lamina, rudimentary cell junctions, and long-spacing collagen or Luse bodies.3,9 Furthermore, definitive evidence of nerve fiber involvement may be needed for the distinction of PNSTs, 18 whereas perivascular orientation is characteristic of PWTs. On the basis of the above observations, the authors suggest that neoplastic cells arranged in a proven perivascular growth pattern, with round to oval (not serpentine or fusiform) nuclei, an immunohistochemically and ultrastructurally undifferentiated phenotype without rudimentary cell junctions, Luse bodies, or nerve fiber involvement, should be considered undifferentiated perivascular cells and not Schwann or perineurial cells.
In conclusion, ultrastructural evaluation supported the hypothesis 2 that the indiscriminately applied term of canine HEP is nonspecific, and PWTs—a more suitable term in these cases—represent a group of tumors that may undertake several partially overlapping lines of differentiation (Fig. 11). This overlap gives rise to a “gray zone” where the specific diagnosis of a PWT subtype is difficult to achieve without additional techniques because of the high plasticity of the cell populations of origin. In summary, a small subgroup of PWTs with equivocal morphology at the light microscopic level and lack of specific immunohistochemical or molecular markers requires ancillary ultrastructural investigation for an accurate diagnosis.
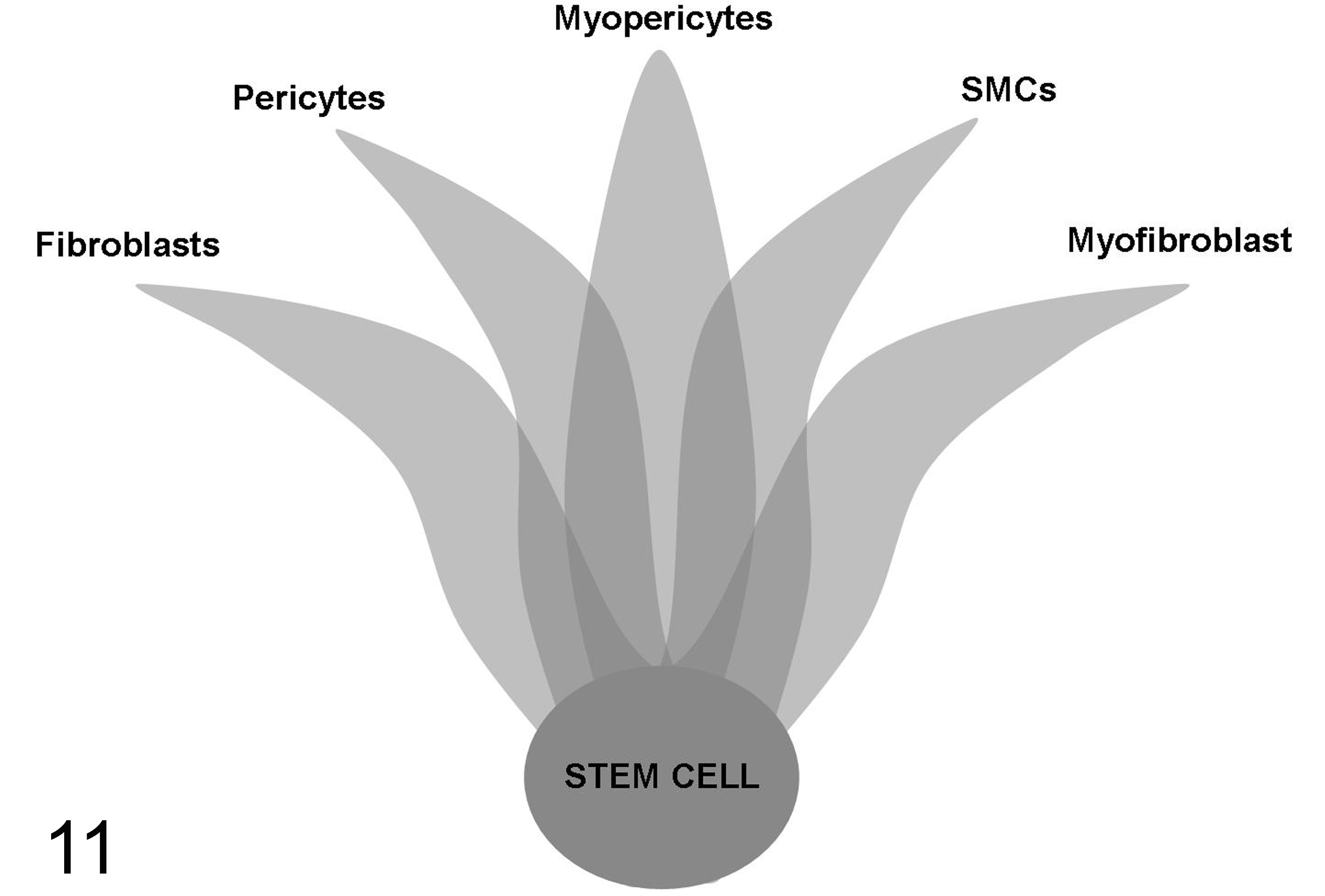
Scheme depicting the lines of differentiation of perivascular stem cells. Perivascular wall tumors could derive from undifferentiated stem cells, perivascular fibroblasts, pericytes, myopericytes, smooth muscle cells (SMCs), myofibroblasts, or transitional cells with overlapping features, represented by the dark gray zone.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
