Abstract
Dogs with mucopolysaccharidosis (MPS) IIIA were bred within an experimental colony. As part of characterizing them as a model for testing therapeutic strategies for the analogous disease of children, a pathologic study was undertaken. By histology, there were variably stained storage cytosomes within neurons, including many that stained for gangliosides. On ultrastructure examination, these cytosomes contained either moderately dense granular material, tentatively interpreted as precipitated glycosaminoglycan; a variety of multilaminar bodies, interpreted as being associated with secondary accumulation of gangliosides; or a mixture of both types. In the liver, storage vesicles also contained excess glycogen as a secondary storage product. In various tissues, there were large foamy macrophages. In the brain, many of these were in juxtaposition with neurons, and, on ultrastructure examination, they contained storage cytosomes similar to those in neurons. However, the neuron in association with such a macrophage frequently showed little such material.
Keywords
Mucopolysaccharidosis (MPS) IIIA is an inherited lysosomal storage disease of children caused by deficient activity of lysosomal heparan N-sulfatase. 17 It is 1 of 4 types of MPSIII (A–D) that lead to the Sanfilippo syndrome, each one resulting from a deficiency of a catalytic enzyme particular to sequential catabolism of heparan sulfate. The syndrome is characterized by progressive neurodegeneration from 3 to 6 years of age and leads to mental retardation; sleep disturbances; and behavioral problems, such as hyperactivity, temper tantrums, destructive behavior, and physical aggression. Other signs include severe diarrhea, speech impairment, hearing loss, early onset of puberty, and seizures. Most patients die in their late teens or early twenties, by which time there is moderate cortical atrophy of the brain.
MPSIIIA has also been described in Wirehaired Dachshund dogs, 8, 12 Huntaway dogs, 11, 28 and mice. 6, 16 In the 2 canine breeds, the mutations are different. A deletion at 737–739 leads to a loss of threonine at position 246 in Dachshunds, 1 whereas an insertion of adenosine at 708–709 occurs in Huntaways, which leads to a frame shift and results in chain termination at position 228. 28 Clinical signs are essentially those of cerebellar disease, with onset from 18 months of age. 11 To date, because no specific therapy is available for MPSIIIA, models are needed to test evolving therapeutic methodologies, both specific and ancillary. This pathologic study of MPSIIIA disease in Huntaway dogs was undertaken as a basis to their use in such experiments.
Materials and Methods
Experimental animals
The dogs with MPSIIIA were bred in a colony established for the purpose, with a diagnosis of genotype by a deoxyribonucleic acid/polymerase chain reaction test on blood. 28 This pathologic study was based on 2 dogs (29 and 39 months of age), with additional information from 3 others (21, 29, 35 months of age) that were used in the course of acute experiments on therapy. Controls were a normal Huntaway-type dog and a heterozygous male, a litter mate of one of the affected dogs. Dogs were euthanatized with sodium pentobarbitone and subjected to necropsy.
Histopathology
Tissues were fixed in 4% buffered paraformaldehyde and processed into paraffin. Sections from serial coronal blocks of brain and from mid cervical, thoracic, and lumbar spinal cord were stained with hematoxylin and eosin (HE); Sudan black; toluidine blue; Luxol fast blue; periodic acid–Schiff (PAS), on its own and in combination with Luxol fast blue. Selected unstained sections of brain were examined by fluorescent microscopy (440-nm excitation filter and 530-nm barrier filter). Sections from other body tissues were stained with HE and PAS methods.
Electron microscopy
Selected areas of brain cerebral cortex, hippocampus and cerebellum, liver, and kidney were fixed in 4% paraformaldehyde and 2% glutaraldehyde with secondary fixation in 1% osmium tetroxide and processed into epoxy resin. “Thick” sections were stained with toluidine blue for light microscopy, and “thin” sections were processed with the lead citrate/uranyl acetate method for electron microscopy.
Golgi studies
Golgi studies were carried out by a previously published methodology. 30
Immunostaining
Anti-GM2 ganglioside mouse antibodies were generously provided by Dr. P. Livingston (Memorial Sloan Kettering, New York, NY) and by Progenics Pharmaceuticals, Inc. (Tarrytown, NY). Anti-GM3 ganglioside mouse antibody was generously provided by Dr. S. Hakomori (Pacific Northwest Research Laboratory, Seattle, WA). Biotinylated goat anti-mouse immunoglobulin (Ig) M and biotinylated goat anti-mouse IgG were purchased from Vector Laboratories (Burlingame, CA).
Immunocytochemical procedures were carried out as previously reported. 16, 31 Briefly, hemicoronal blocks of brain were sectioned on a Leica 9000S vibratome to generate free-floating sections (20–30 μm) in a cooled chamber that contained a phosphate buffer. These were incubated overnight with primary antibody, followed by biotinylated secondary antiserum and the ABC Elite complex (Vector Laboratories). Visualization of labeled cells was accomplished by using a diaminobenzidine kit (Vector Laboratories). Some sections were counterstained with Nissl, or were stained with Nissl alone, to assess cell distribution.
Histochemistry
Filipin was obtained from Sigma-Aldrich (St. Louis, MO). For visualization of unesterified cholesterol, vibratome sections were incubated in a quenching solution of 1.5 mg/ml glycine in phosphate buffered saline solution and then were probed with 0.5 mg/ml filipin diluted in 0.1% dimethyl sulfoxide or dimethyl sulfoxide alone for 10 minutes, washed, and mounted on slides with Prolong antifade reagent (Molecular Probes, Eugene, OR). 16
Animal welfare
All procedures were approved by the Animal Ethics Committee of Massey University and conformed to “the code of ethical conduct for the use of live animals for teaching and research” as approved under the New Zealand “Animal Welfare Act 1999.”
Results
Gross pathology
With the exception of 2 dogs affected with MPSIIIA and with myxomatous thickening of the tricuspid heart valve, gross pathologic lesions were confined to the central nervous system (CNS). There was mild-to-moderate hydrocephalus in 2 affected dogs (1 treated and 1 not treated) and in the control heterozygous animal. The cerebellum was considered slightly atrophic in affected dogs at 21 and 29 months of age and moderately so in the 35-month-old animals. Despite this, there was no obvious difference, or trend, in brain weights or brain–to–body-weight ratios between 5 affected dogs and 2 controls (data not shown).
Histopathology
In the CNS of affected dogs, storage material within neurons was difficult to see in HE-stained paraffin sections, except for that in large motor neurons in the ventral horns and brain-stem nuclei, where it appeared as a pale, granular, slightly eosinophilic accumulation that distended much of the neuron. This material stained intensely with the PAS stain. Elsewhere, storage material stained variably with this method, ranging from negligible to intense, with most in an intermediary range. Storage bodies were best stained and defined by the Luxol fast blue stain. In sections stained by combined Luxol fast blue and PAS methods, Luxol fast blue tended to dominate and mask PAS +ve material. However, in some neurons, it stained only with the PAS stain; in others, there were both Luxol fast blue and PAS +ve red granules; and, in a few, an intermediate staining by a mixture of the 2 colors. Some storage material stained lightly with Sudan black and was autofluorescent in unstained paraffin sections. In epoxy-resin toluidine-blue sections, storage material stained as either variably light- or dark-blue rounded bodies, sometimes with one or the other type predominating in any 1 neuron. Purkinje cells contained both very pale- and dark-blue inclusions, the 2 types being more or less compartmentized within the cell (Fig. 1). Many neurons, particularly in the cerebral cortex, did not appear to contain storage material of any description.
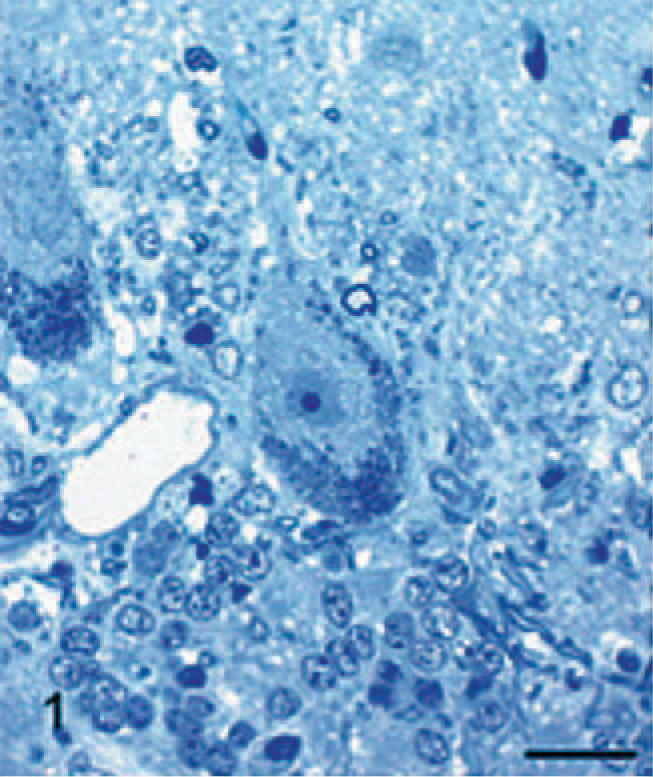
Cerebellum; 29-month-old dog with MPSIIIA. Purkinje cell containing numerous dark staining storage bodies at the periphery of the cell and clear or light-staining storage cytosomes in the perikaryon region. Epoxy resin, toluidine blue. Bar = 25 μm.
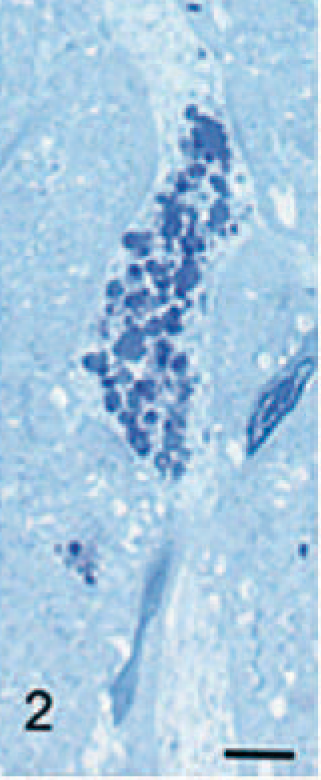
There was degeneration and loss of Purkinje cells, which was most severe in the older dogs. In surviving Purkinje cells, storage granules were sometimes noted extending into and distending their dendrites, which were often enlarged (Fig. 2). In the 2 oldest dogs, there was mild-to-moderate loss of granule cells of the cerebellum. Occasional spheroids were noted in cerebellar white matter, and other small eosinophilic spheroids occurred in gray matter, including nuclei of the brain stem.
Immunocytochemical staining for GM2 ganglioside in the cerebral cortex showed widespread staining of neurons and glia (Fig. 3a). Most prominently stained were large pyramidal neurons of layers III and V, with large numbers of stained storage cytosomes scattered throughout the cell body and extending into apical and basilar dendrites (Fig. 3b). GM3-immunostaining had a similar distribution in the cortex but was generally less prominent (Fig. 4a, b). On occasions, GM3-negative-stained neurons were in close association with cells interpreted as glial cells, which stained strongly for this ganglioside (Fig. 5). In addition to storage of gangliosides, filipin histochemistry showed the accumulation of large amounts of unesterified cholesterol in cell bodies and dendrites, particularly of large pyramidal neurons (Fig. 6).
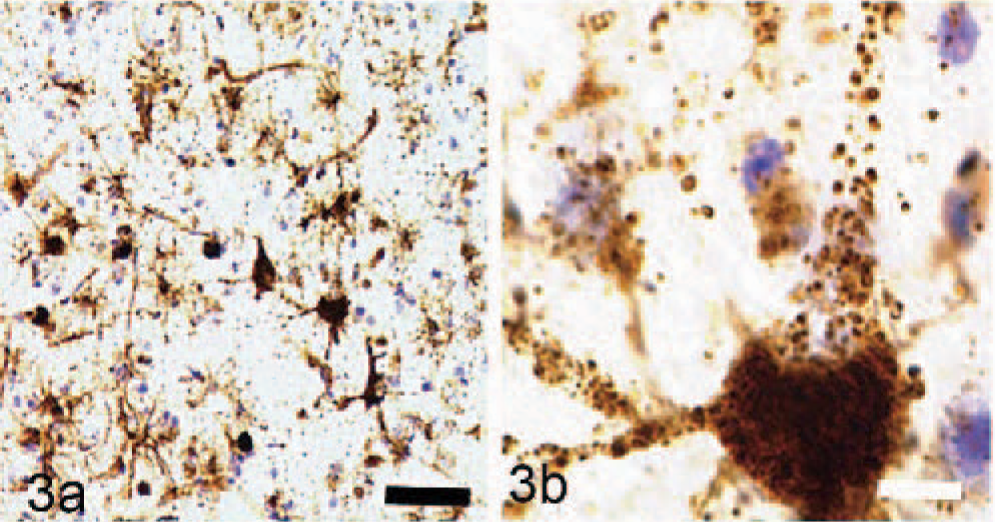
Immunocytochemical and histochemical studies of storage material from the cerebral cortex of dogs with MPSIIIA; GM2 ganglioside immunoreactivity of neurons, astrocytes and macrophages. Nissl counterstain Bar = 100 μm.
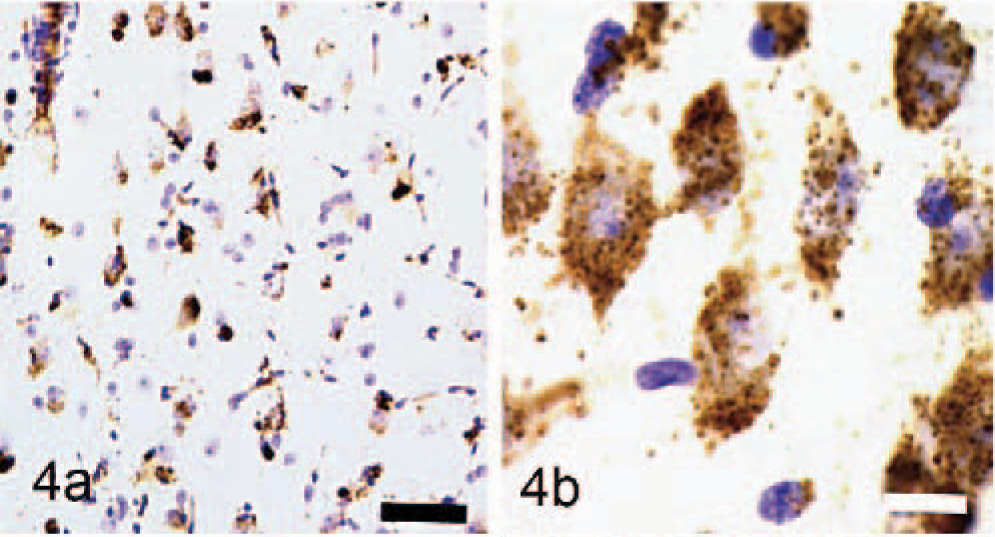
GM3 ganglioside immunoreactivity of neurons, astrocytes and macrophages. Nissl counterstain Bar = 100 μm.
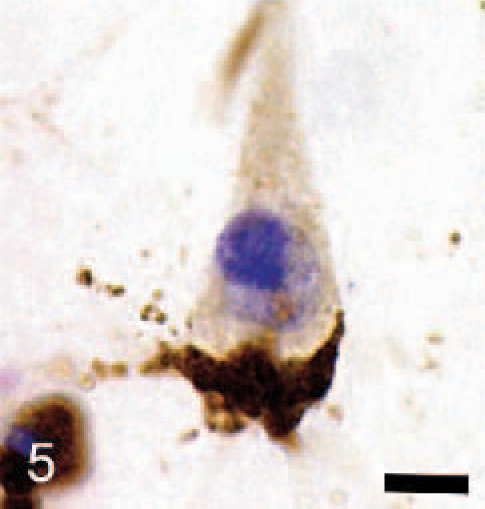
Cerebral cortex; dog with MPSIIIA. GM3 ganglioside immunoreactivity of a satellite glial cell shows extensive storage of GM3 ganglioside compared with that in the juxtaposed neuron. Nissl counterstain Bar = 10 μm.

Cerebral cortex; dog with MPSIIIA. Histochemical staining of unesterified cholesterol in a pyramidal cell is shown by diffuse blue staining. No counterstain Bar = 50 μm.
In the cerebellum, GM2-ganglioside antibody staining occurred mainly in smaller cells and cell profiles of the molecular layer, to some extent in surviving Purkinje cells, and in Bergmann glia. GM3-ganglioside staining was more prominent in neuronal somata and apical dendrites of Purkinje cells and Golgi neurons. Filipin staining also showed presence of unesterified cholesterol in surviving Purkinje cell bodies and apical dendrites. Golgi impregnated neurons in the cerebral cortex showed normal dendrites and dendritic spines. Large pyramidal cells of layers III and V sometimes exhibited enlarged axon hillocks, and, in rare instances, these were large enough to be termed meganeurites. 21 These latter occasionally exhibited short neuritic processes that resembled ectopic dendrites.
Protoplasmic astrocytes contained granular material that stained moderately with Sudan black but poorly with the PAS and Luxol fast blue stains in paraffin sections but lightly with toluidine blue in resin sections. There was mild proliferation of Bergmann glia, whose fibers sometimes appeared hypertrophied. Slight Wallerian-type degeneration occurred in cerebellar white matter and was most severe in the oldest dogs. Autonomic ganglion cells in the small intestine were minimally vacuolated, but supportive cells with fibroblast-like characteristics were highly vacuolated. The various layers of the retina were present in normal proportions. Ganglion cells contained granular storage material, and pigment epithelial cells were lightly vacuolated.
The most generalized histologic lesion was as foamy macrophages, whose structures were best appreciated in toluidine-blue-stained epoxy-resin–embedded sections. In the brain and the spinal cord, they appeared as satellite cells to neurons (Fig. 7), free in the neuropil, in perivascular spaces (Fig. 8), and in the meninges. Those macrophages adjacent to neurons and in the neuropil contained varying amounts of coarse granular material that stained moderately with PAS, Sudan black, and variably with Luxol fast blue in paraffin; and lightly with toluidine blue in epoxy resin sections. This material was also autofluorescent in unstained paraffin sections. Similar macrophages with granules were sometimes noted immediately adjacent, as well as within, the periarterial spaces; however, most macrophages within these spaces were characterized by clear vacuoles or, less commonly, a hyaline material persisted, which sometimes had a yellow-brown tinge. Macrophages with granular storage material were particularly numerous in and adjacent to the depleted Purkinje cell layer of the cerebellum. In the meninges, they were variably vacuolated, often with granules that tended to be larger and stain more intensely with the PAS method than in similar cells within the brain. These were found both in the subpial space and in the leptomeninges.
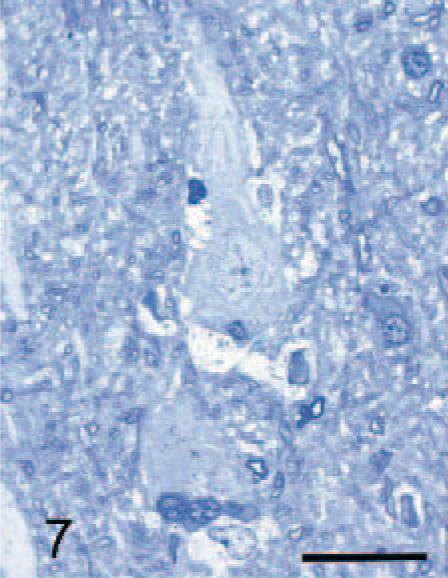
Cerebral cortex; dog with MPSIIIA. Pyramidal neuron from cerebral cortex of 29-month-old dog with MPSIIIA surrounded by foamy macrophages in close juxtaposition to the neuron. Epoxy resin, toluidine blue. Bar = 25 μm.
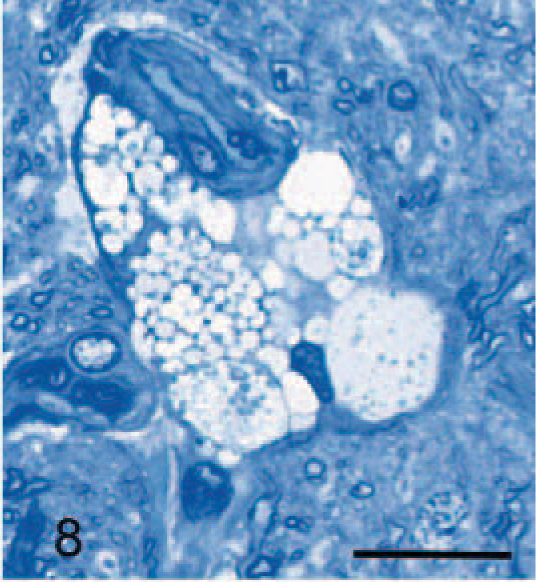
Cerebral cortex; dog with MPSIIIA. Large vacuolated macrophages within the periarterial space of the cerebrum in the same dog as in Fig. 7. Epoxy resin, toluidine blue. Bar = 20 μm.
Accumulations of vacuolated macrophages were commonly found in the sinusoids of the spleen and lymph nodes, and were particularly dense in the medulla of lymph nodes that drain the intestine. They were also common between smooth-muscle fibers and along fascial planes in the intestinal tract. Similar cells were found between cardiac muscle fibers, in the interstitial tissue of the renal pelvis, in a subepithelial position in the bladder, and, to a lesser extent, in other tissues. In resin-embedded sections of the liver, Kupffer cells were highly vacuolated.
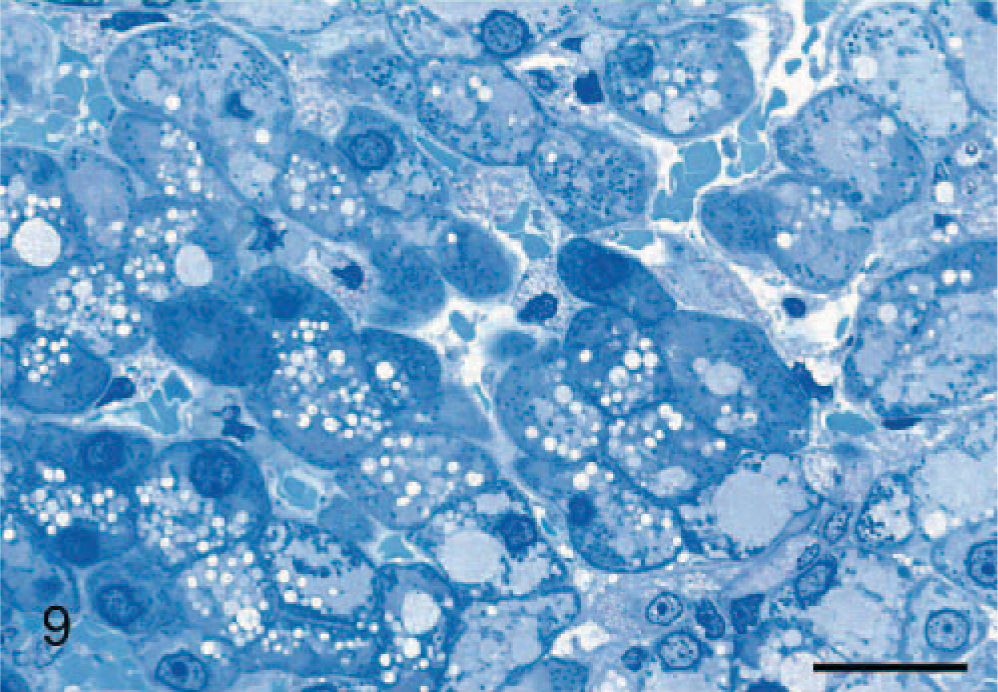
In the liver, hepatocytes were severely vacuolated, leading to their enlargement and encroachment on sinusoids. In resin-embedded sections, some vacuoles were clear and contained no stained material, whereas other cytosomes contained some opaque hyaline material (Fig. 9). There was a range in size of vacuoles/cytosomes, which collectively filled much of the cytoplasm. Mild vacuolation was observed in bile-ductule and bile-duct epithelium. In the pancreas, slight vacuolation of acinar cells occurred, compared with a more severe vacuolation of intralobular-duct epithelium. Vacuolation occurred in epithelial cells of convoluted tubules and collecting ducts of the kidney, being most severe in the distal tubules. Although vacuoles often appeared empty, other similar cytosomes contained slightly opaque hyaline material that stained very palely with the PAS method in paraffin sections and by toluidine blue in epoxy-resin sections (Fig. 10).
Liver; 29-month-old dog with MPSIIIA. Hepatocytes contain small clear or lightly staining vacuoles, contrasting with larger, moderately staining, round, opaque inclusions. The small dark-staining granules are interpreted as mitochondria. Epoxy resin, toluidine blue. Bar = 15 μm.
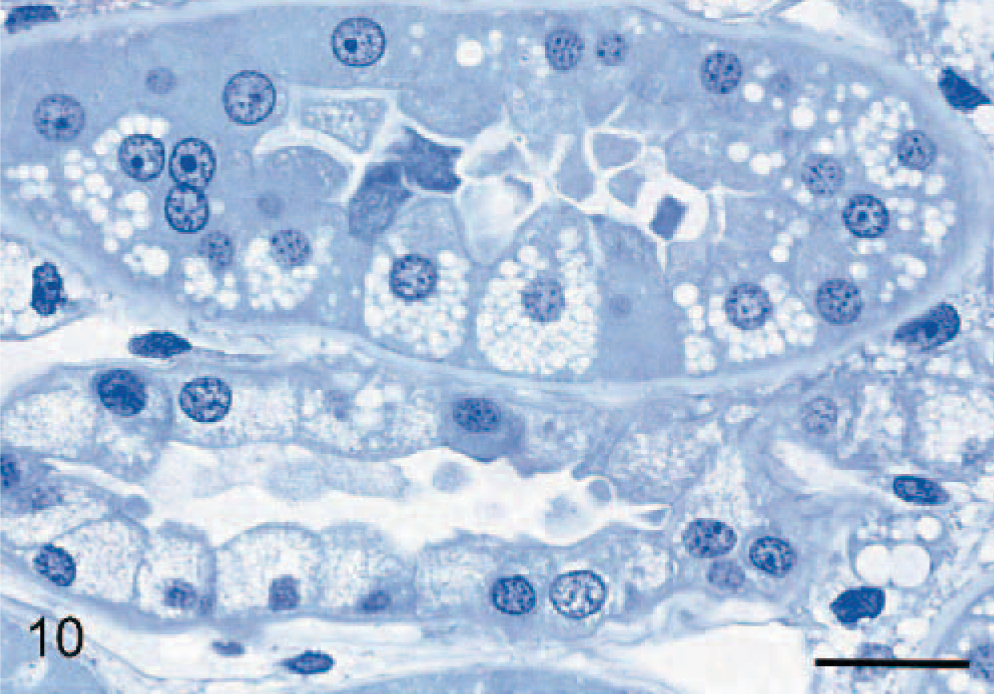
Kidney; 29-month-old dog with MPSIIIA. There is severe vacuolation of proximal (upper) and distal (lower) convoluted tubules that appear to contain very lightly staining material. Epoxy resin, toluidine blue. Bar = 20 μm.
There was severe vacuolation of thyroid follicular cells, and, in various tissues, there was a fine vacuolation of fibrocytes. The enlarged tricuspid valves in 2 affected dogs were composed of loose myxomatous connective tissue separated by bands of mature collagenous tissue.
Transmission electron microscopy
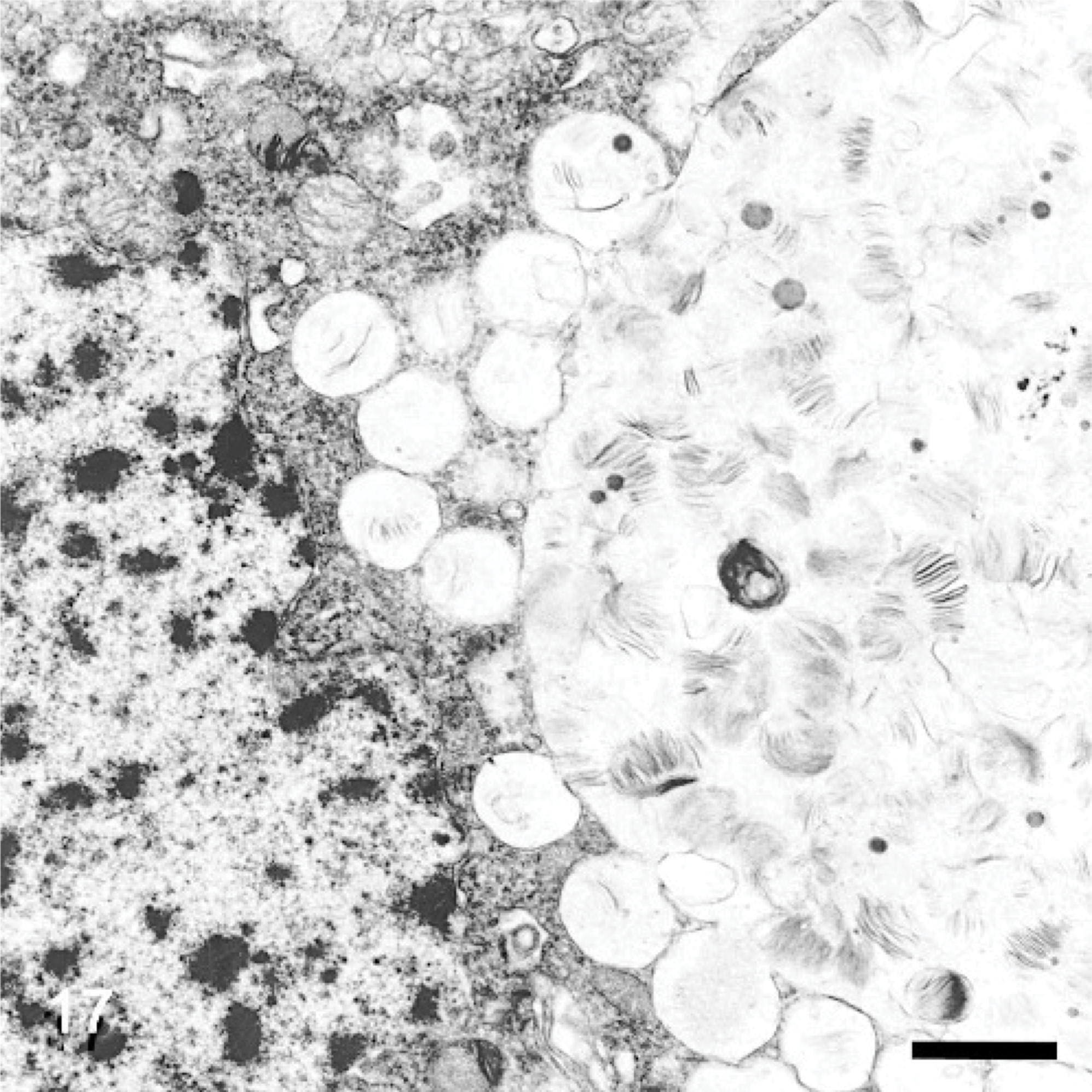
The ultrastructure of intraneuronal storage material was heterogeneous and appeared as membrane-bound vesicles that contained moderately dense granular material; similar organelles that contained the same material plus straight multilaminar arrays or electron dense globules; organelles with only straight multilaminar arrays (“zebra bodies”); organelles with multilaminar circular profiles; or very dense bodies, often with some underlying evidence of circular multilaminar profiles (Figs. 11– 16,). Clear vesicles, which sometimes contained wispy multilamellar arrays or light amorphous material, occurred but were uncommon (Fig. 17). In some neurons, one particular type of storage material, either granular or of a multilaminar nature predominated; in others, a mixture occurred. The storage material in Purkinje cells was mainly of the granular and multilaminar circular profile forms, which coincided with the pale (clear) and densely staining organelles, respectively, as noted by light microscopy (Fig. 1). Apart from storage bodies, some hypertrophied Purkinje cell dendrites contained stacks of smooth endoplasmic reticulum (Fig. 18). Storage bodies were also observed in large dendrites of pyramidal cells in the cerebral cortex. The vacuolated macrophages observed by light microscopy contained membrane-bound vesicles compatible with the structure of secondary lysosomes. In the CNS, a proportion of these macrophages also contained varying amounts of amorphous, granular, or multilaminar material (Fig. 19). Macrophages in the neuropil contained similar material, whereas, most of those in the periarterial spaces contained no such stained material within vesicles. However, in some of these perivascular macrophages, there was a light electron-dense precipitate and small electron dense globules.
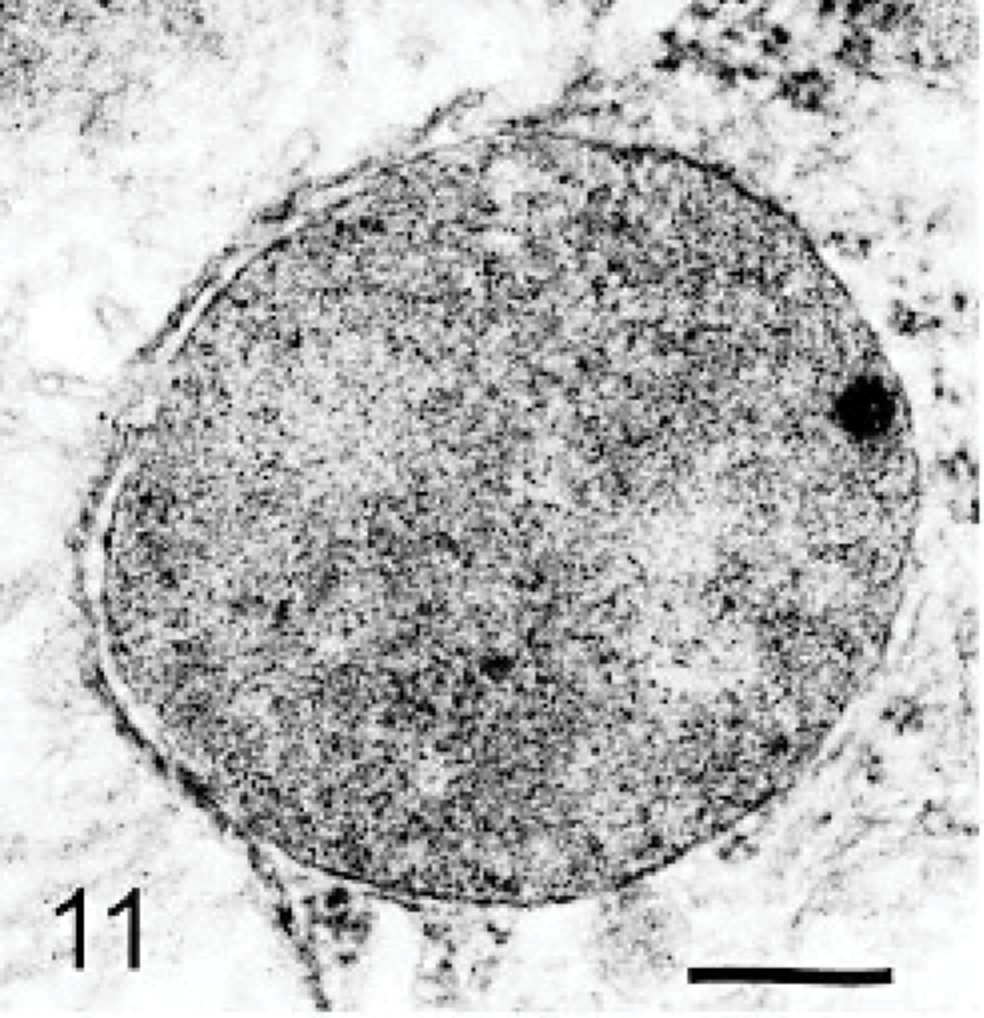
Brain; dog with MPSIIIA. A neuron containing a moderately electron-dense granular storage body. Lead citrate and uranyl acetate. Bar = 0.025 μm.
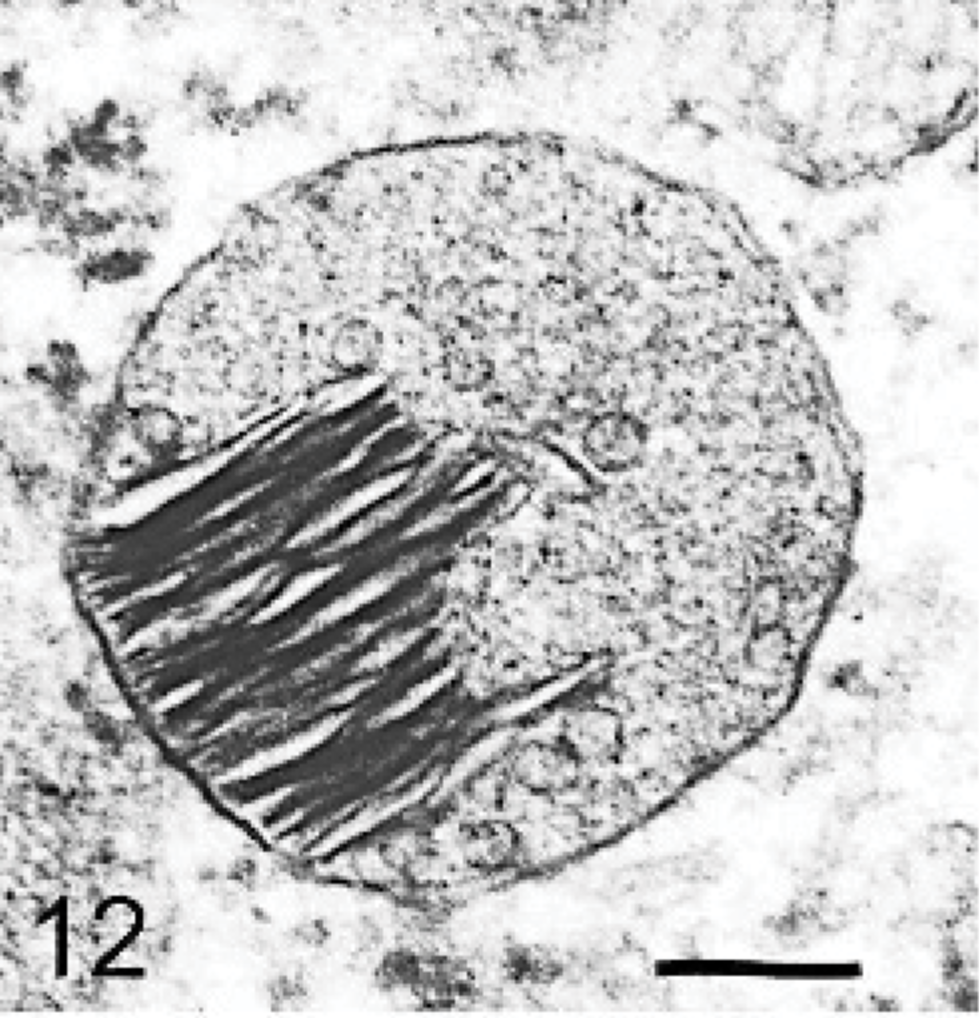
Brain; dog with MPSIIIA. A neuron containing a moderately dense granular storage body with electron-dense laminae. Lead citrate and uranyl acetate. Bar = 0.025 μm.
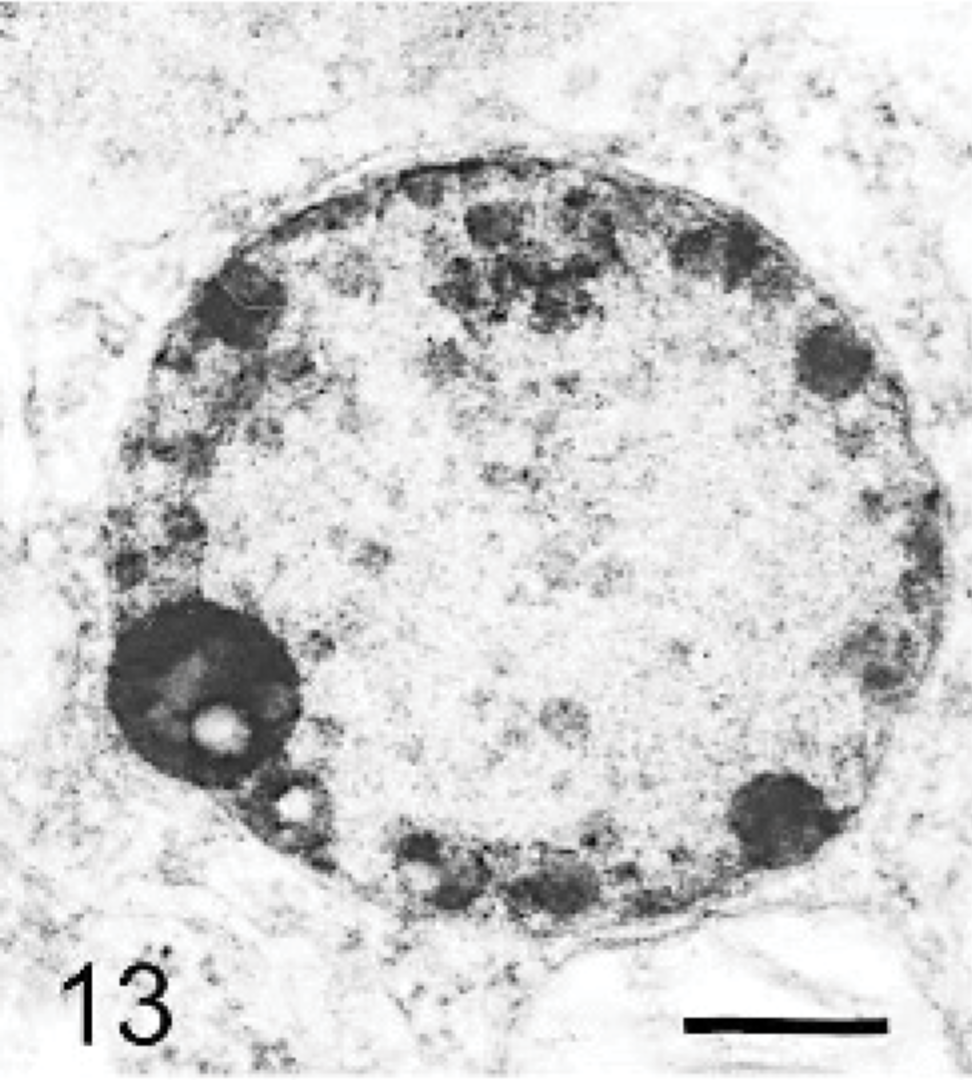
Brain; dog with MPSIIIA. A neuron containing a moderately dense granular storage body with electron-dense globular material. Lead citrate and uranyl acetate. Bar = 0.025 μm.
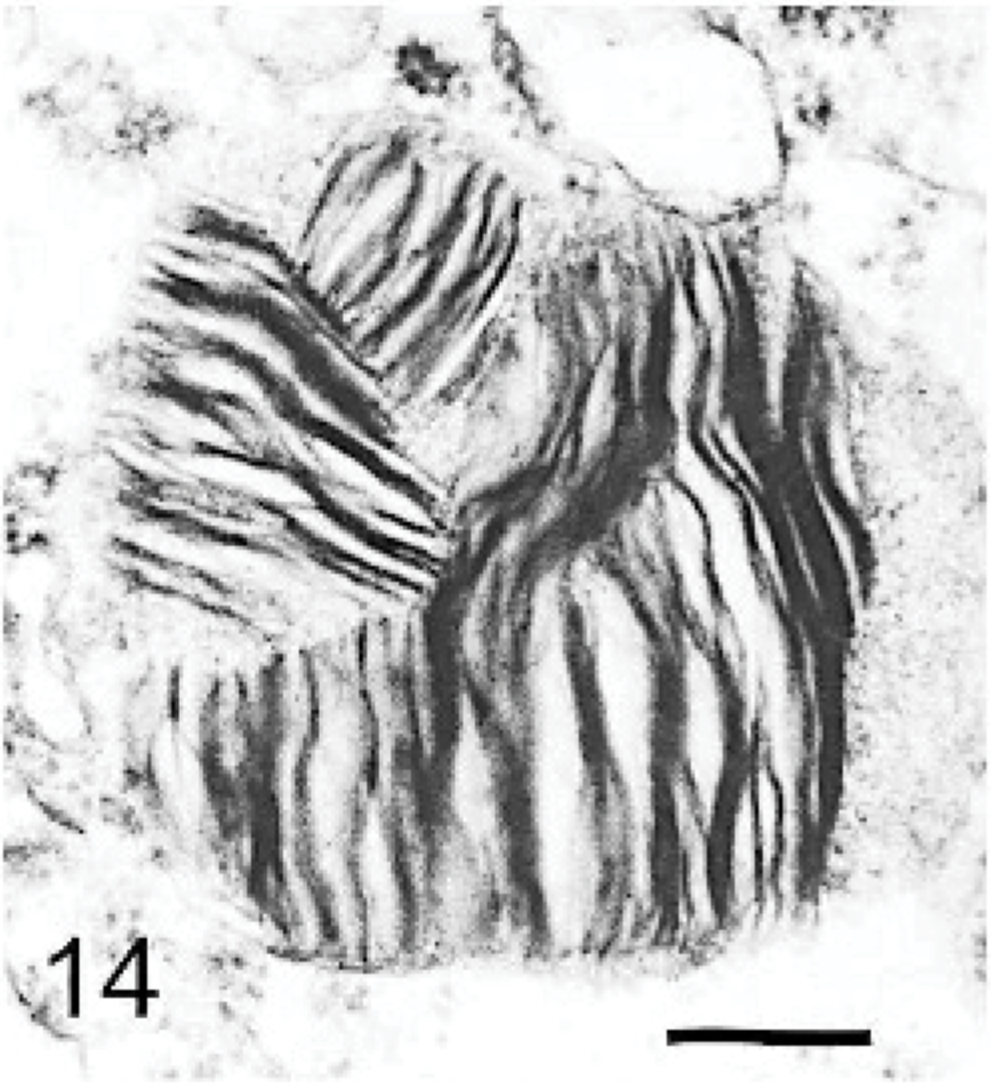
Brain; dog with MPSIIIA. A neuron containing a storage body with multilaminar material (“zebra body”). Lead citrate and uranyl acetate. Bar = 0.033 μm.
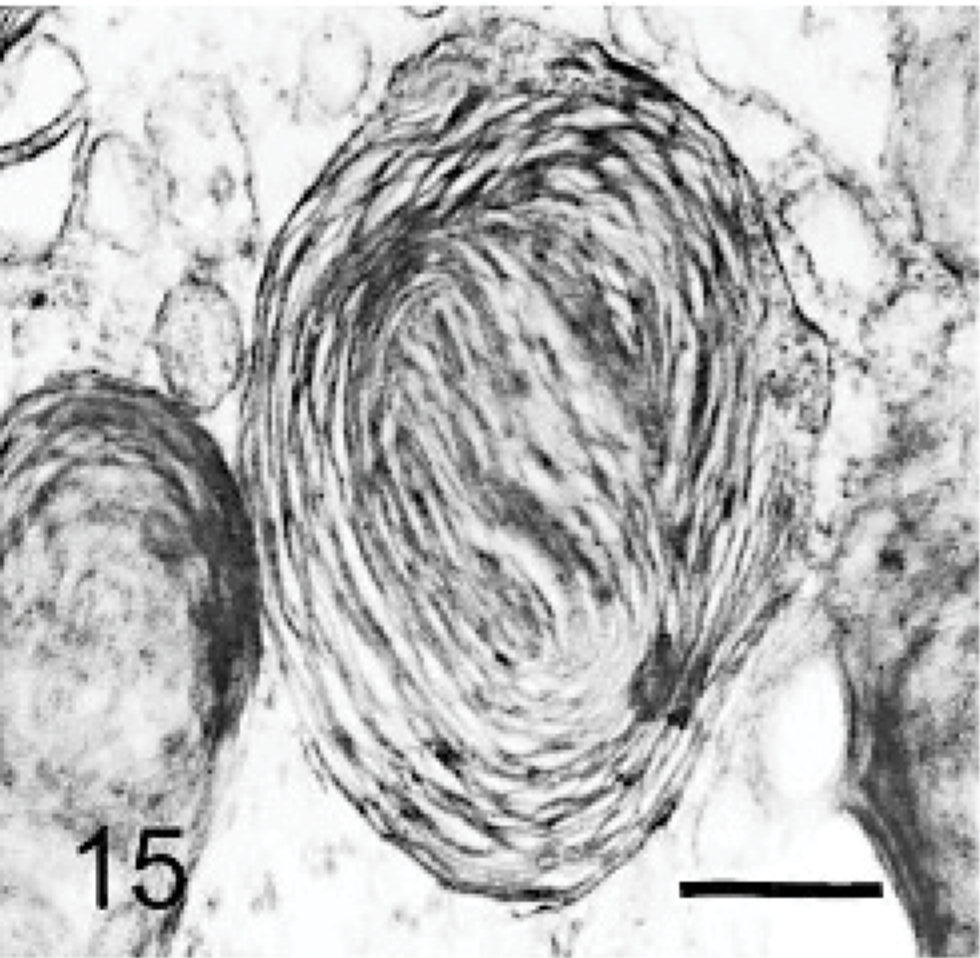
Brain; dog with MPSIIIA. A neuron containing a storage body made of multilaminar material, with circular profiles. Lead citrate and uranyl acetate. Bar = 0.033 μm.
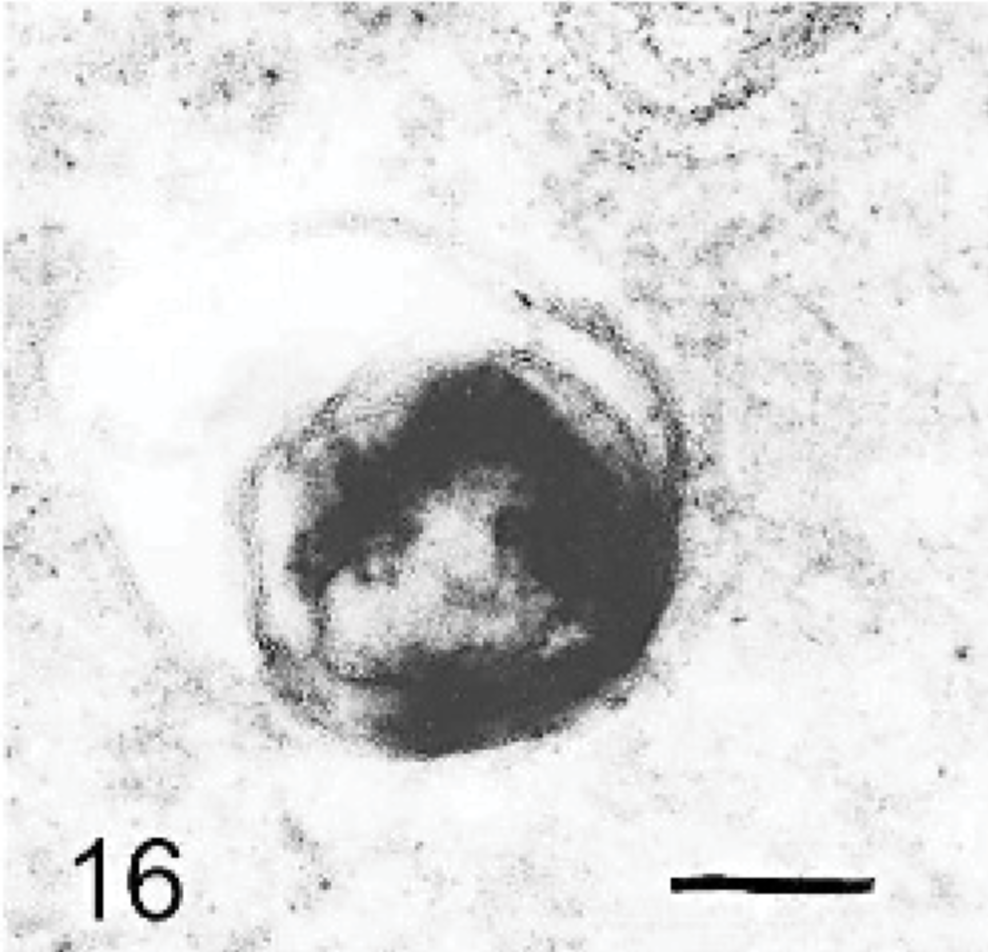
Brain; dog with MPSIIIA. A neuron containing a very electron-dense storage body, with some underlying membranous material. Lead citrate and uranyl acetate. Bar = 0.015 μm.
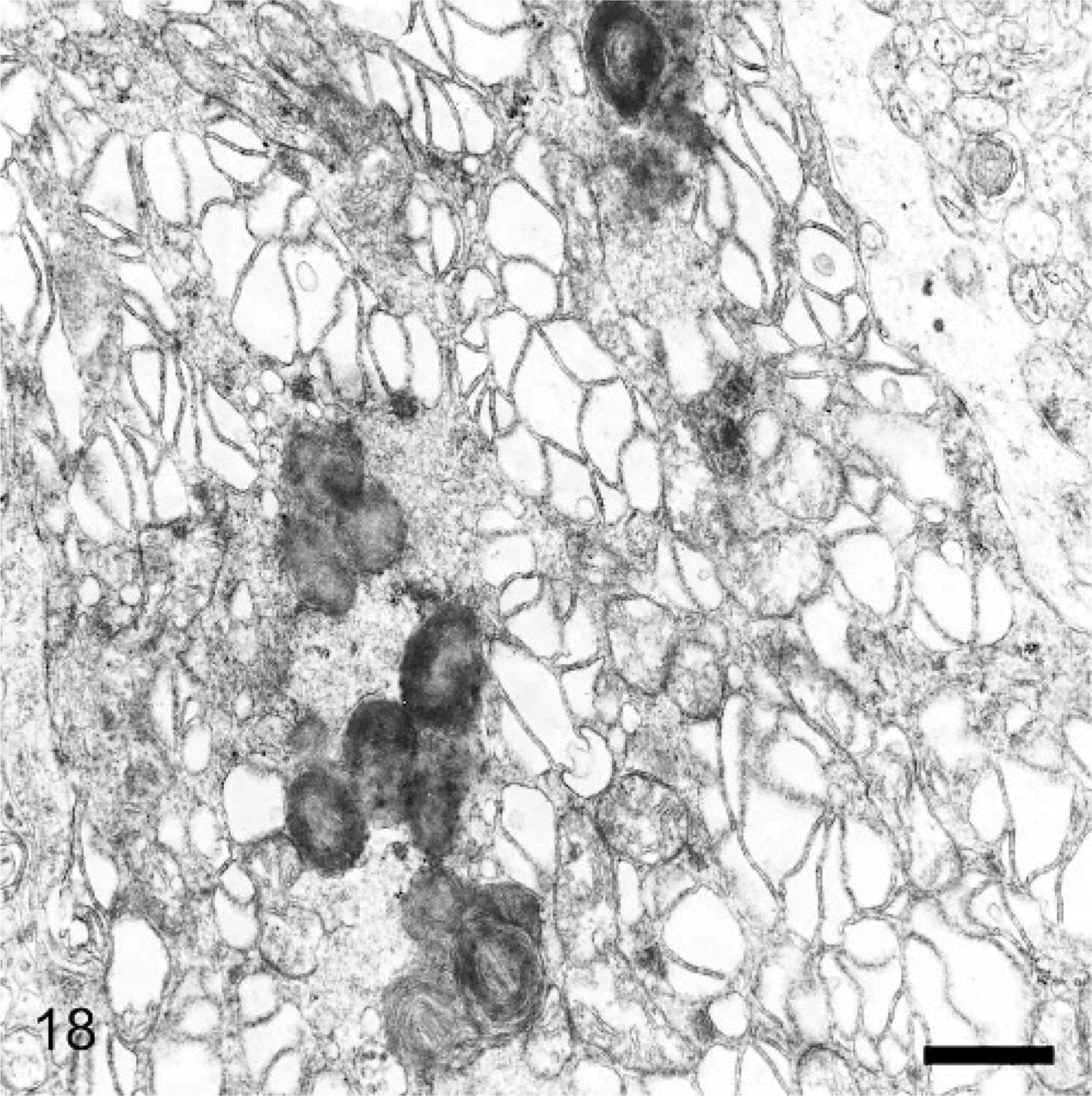
Brain; 29-month-old dog with MPSIIIA. A Purkinje cell dendrite containing both electron-dense storage cytosomes and sacs of dilated endoplasmic reticulum. Lead citrate and uranyl acetate. Bar = 0.1 μm.
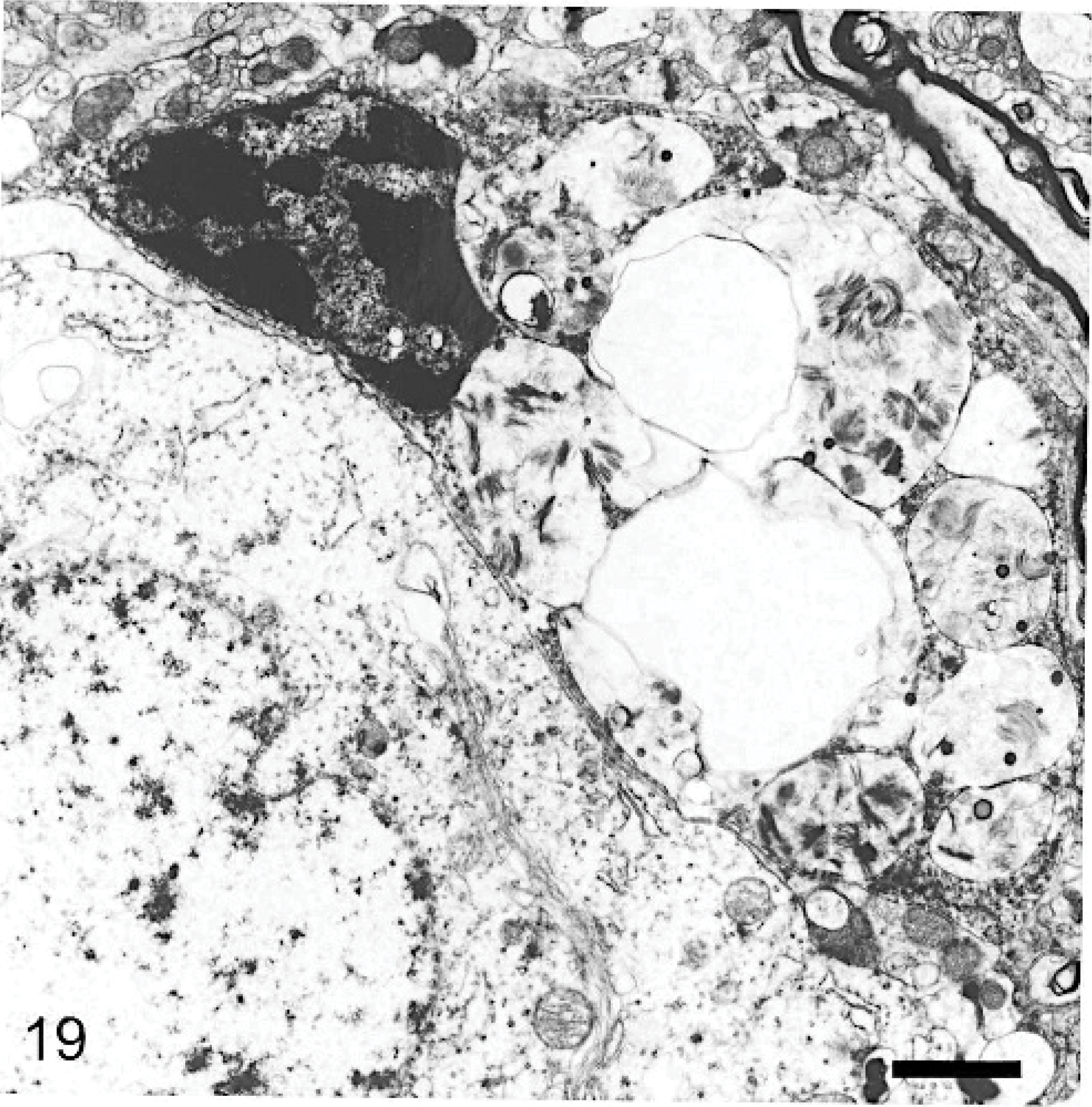
Cerebral cortex; 29-month-old dog with MPSIIIA. A neuron (bottom left) is without storage bodies or vesicles, in contrast to the adjacent macrophage (top right) containing empty vesicles and vesicles containing electron-dense laminar material. The cell membranes of both cells are in close juxtaposition. Lead citrate and uranyl acetate. Bar = 0.2 μm.
Many macrophages noted by light microscopy to be in proximity to neurons had their plasma membrane in close association with that of the respective neuron over a considerable distance. They were usually highly distended with storage-type vesicles and storage material typical of the types found in neurons (Fig. 19). In many such neuron/macrophage juxtapositions, however, the adjacent neuron contained no, or very little, storage material. In one association, the neuron contained vesicles that contained a wispy laminar material identical to that found in the adjacent macrophage (Fig. 17). Protoplasmic astrocytes appeared slightly swollen and contained varying numbers of storage bodies, most commonly in the form of “zebra bodies.” There was an increase in glial fibrils, this being particularly noted in hypertrophied Bergmann glia. Wallerian-type degeneration of axons was occasionally noted.
In hepatocytes in the liver, smaller vesicles were essentially empty of electron-dense material, but larger ones frequently contained varying amounts of electron-dense material in the form of rosettes that resembled glycogen (Fig. 20). This material was particularly extensive in those cytosomes where the stored material was noted as opaque by light microscopy of epoxy-resin sections (Fig. 9). Some smaller vesicles contained a small amount of multilamellar material. In most hepatocytes, storage material had displaced rough endoplasmic reticulum and mitochondria to be either adjacent to the nucleus or to the periphery of the cell.
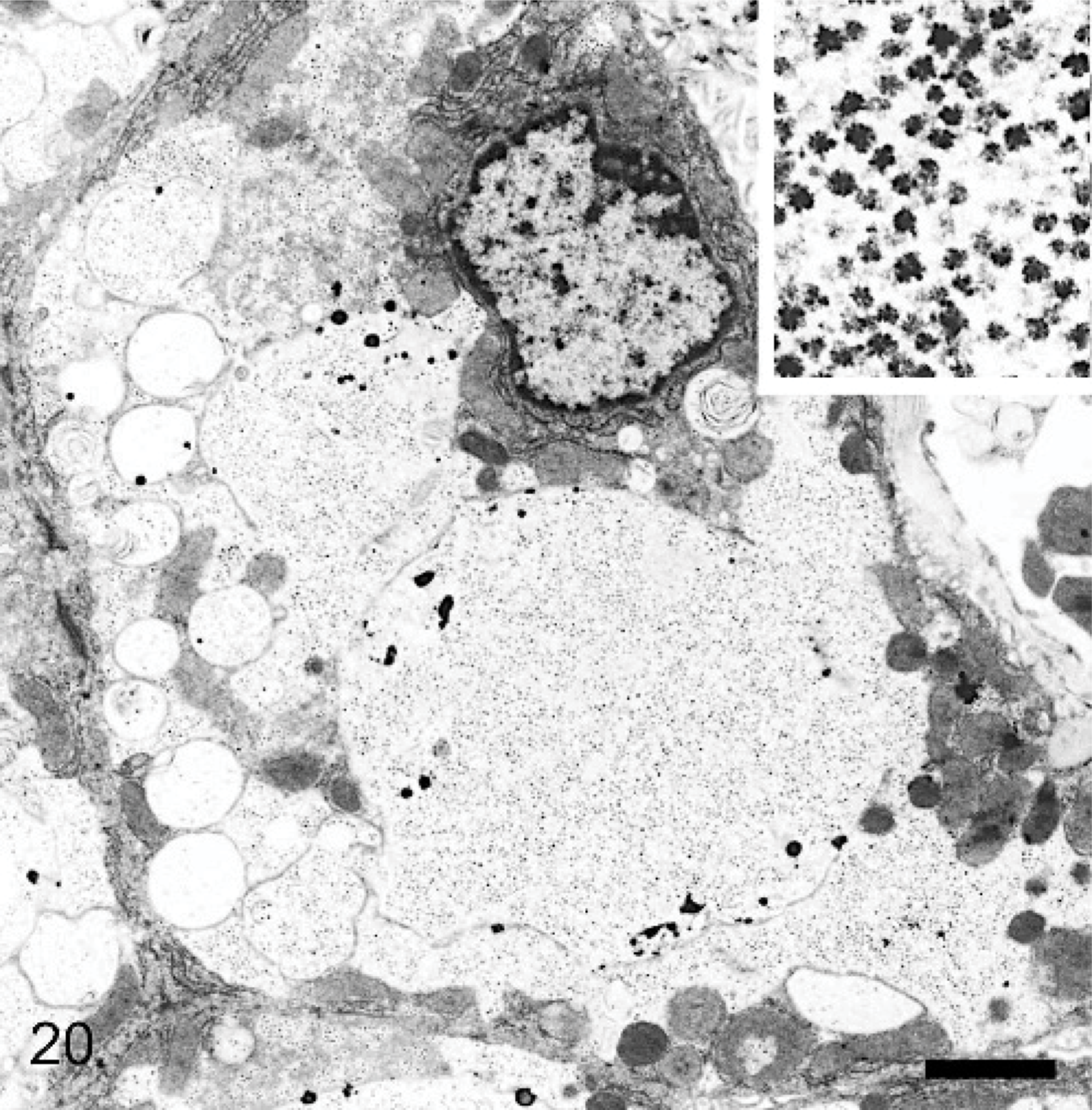
Liver; 29-month-old dog with MPSIIIA. Hepatocyte cytoplasm is distended, with some empty vesicles and some larger ones containing granular material. Bar = 0.07 μm. The inset, at higher magnification, shows that the granular material is made of electron-dense rosettes resembling glycogen granules. Lead citrate and uranyl acetate. Bar = 0.025 μm.
Renal-tubule cells contained numerous vesicles with light-to-moderate flocculated material. The thyroid-gland follicular cells contained many membrane-bound vesicles with light floccular material that was similar morphologically to those normally found in the degradation of thyroglobulin. These vesicles were considered more numerous in affected dogs and of more uniform light density.
Discussion
Hydrocephalus, present in 2 dogs affected with MPSIIIA, was an inconstant feature in the present study and also occurred in an unaffected heterozygous dog used as a negative control. Thus, it may not be directly related to MPSIIIA. Despite cerebellar atrophy in affected dogs, overall brain weights and brain weights relative to body weight (data not shown) showed considerable individual variation and no relation to disease status.
The loss of Purkinje cells and other abnormalities of the cerebellum are consistent with neurologic signs in MPSIIIA in Huntaway dogs of ataxia, hypermetria, and a wide-based stance.
11
We postulate that the stacks of smooth endoplasmic reticulum noted in electron micrographs of hypertrophied Purkinje cell dendrites, reflected a response involving calcium ion (Ca2+) regulation. This organelle is normally present in dendrites as thin cisterns beneath the plasmalemma and is associated with Ca2+ regulation. Cells have a considerable ability to buffer Ca2+ through Ca2+ binding proteins, such as parvalbumin and calbindin, and uptake into smooth endoplasmic reticulum and mitochondria. Under conditions of anoxia or intraventricular injection of
In human MPSIIIA, cognitive and behavioral changes are prominent early clinical features of disease. These changes in neurologic function may be associated with the occurrence of meganeurites with ectopic dendrites that lead to aberrant synaptic function. Such ectopic dendrites occur in the gangliosidoses and other disorders in which gangliosides accumulate 21 but are an infrequent finding in the dogs with MPSIIIA, which may explain the lack of cerebrocortical dysfunction in this species.
Families of heparan sulfate proteoglycans (HSPG) occur on the surface of cells and play a major function in modulating the actions of extracellular protein ligands. 5, 27 The glycosaminoglycan moiety is covalently bound to transmembrane proteins (core linked) known as syndecans or to glypicans linked to glycosylphosphoninositide-anchored proteins within the cell membrane. The heparan sulfate helps immobilize and regulate the turnover of ligands that bind to receptors on the cell surface at calveolae and clathrin-coated pits. These are internalized and sorted in endosomes, with the return of HSPGs and receptor apparatus to the plasma membrane. Alternatively, they may be degraded in the endosomal/lysosomal system by heparanase and other specific enzymes for the sequential degradation of the glycosaminoglycans released from proteoglycans. 17, 27 Core-linked and glycosylphosphoninositol-anchored proteoglycans may have different kinetic parameters for their endocytosis and passage through the endosomal/lysosomal systems. 27 HSPGs may also be shed from the cell in an orderly fashion close to the cell surface. Such soluble fragments of HSPG are biologically active and may inhibit binding of ligands to receptors.
Storage lysosomes were heterogeneous, ranging from clear vacuoles, such as those found in macrophages and reflecting vesicles from which soluble glycosaminoglycans may have been leached during preparation of tissues, to discrete round cytoplasmic inclusions of variable density with toluidine-blue staining in epoxy-resin sections and PAS in paraffin sections. The variable staining with Luxol fast blue/PAS stains is further evidence of heterogeneity of storage material. In the brain, the light-stained bodies corresponded in general to those described ultrastructurally as of a granular moderately dense type; the darker stained to the more electron dense multilaminar inclusions. It is postulated that the former contained heparan sulfate concentrated in a gel but precipitated on tissue dehydration. Electron-dense globules, sometimes noted within them (Fig. 13), may reflect products of nonenzymic cross-linking of glycosaminoglycan (advanced glycation products).
The multilaminar membranous bodies have been shown to contain gangliosides that accumulate as secondary products in the mucopolysaccharidoses. 16, 24 Their presence in this canine disorder was also confirmed by immunostaining (Figs. 3a, b; 4a, b; 5). GM2 and GM3 likely derive from GM1 ganglioside, an integral lipid component of the neurolemma, which is believed to be a constituent of membrane rafts and calveolae. 29 It is degraded in the endosomal/lysosomal system by a series of hydrolases aided by a specific activator protein. 15 It has been shown that glycosaminoglycans reversibly inhibit numerous lysosomal enzymes, most strongly below pH 4.5. 2 Although it is possible that excess heparan sulfate disturbs the critical conditions for ganglioside catabolism, which results in ganglioside accumulation as a secondary lesion, why this results in sequestration of gangliosides in largely separate vesicle populations remains unknown. 16 In some neurons, these multilaminar bodies predominated to such an extent that there was little evidence of primary storage product. The varied membranous ultrastructure of storage bodies is likely a reflection of the major type of ganglioside present.
Ganglioside oligosaccharides stain by the PAS method, 7 and, in at least some cells, e.g., Purkinje cells, where multilaminar bodies were common, the bodies stained well by this method. Heparan sulfate has the propensity to stain positively, but, in practice, results can be very variable. 4 The reason for the accumulation of unesterified cholesterol is less clear; however, it routinely accompanies the storage of gangliosides in a variety of storage disorders. 16, 21– 23 This may reflect the fact that gangliosides and cholesterol are colocalized at the neuronal surface in specialized microdomains known a lipid rafts or calveolae. Secondary defects in endosomal/lysosomal disease may thus be responsible for the accumulation of both ganglioside and cholesterol. 21
The occurrence of foamy macrophages in a wide variety of tissues was a generalized lesion that probably reflects uptake of heparan sulfate from tissue spaces or blood. These cells are presumed to contain soluble glycosaminoglycan leached during the preparation of sections, although, in some, it may still be seen as a slightly opaque hyaline material. Foamy macrophages in the CNS contained storage material ultrastructurally similar to that in neurons, some of which may have derived from dead neurons. However, many of these macrophages were in close juxtaposition to neurons, a feature noted also in other reports, where they were referred to as satellite cells. 6 The near absence of storage material in many of the associated neurons (Figs. 5,7,17,19) and the many other similar observations made during the course of the study, provide morphologic evidence that support a hypothesis that a transfer of storage cytosomes occurs between neurons and macrophages. Alternatively, endocytosed ligand/receptor domains and associated HSPGs could be recycled from endosomes to be phagocytosed at the cell surface. This was particularly suggested inFig. 17. The large clear vesicles within some macrophages are compatible with endocytosis of HSPGs released by proteolysis near the cell surface, because similar large clear vesicles were seldom seen within neurons.
The distribution of macrophages with complexes of storage material within the CNS implies that, having obtained storage material from neurons or the surface of neurons, macrophages traverse the neuropil toward the perivascular spaces where they may be held up briefly before entering the spaces and draining to the meninges. Within the periarterial spaces, they contrasted with other macrophages that contained mainly clear vesicles that suggested these latter were resident cells taking up soluble heparan sulfate circulating in tissue fluid from the neuropil to the perivascular spaces. The occasional yellow-brown coloration of this nongranular material, as seen by light microscopy, and its apparent precipitation with occurrence of electron dense globules, suggests polymerization through nonenzymic cross-linking.
The severe vesiculation of renal tubules and collecting-duct cells indicates significant circulating levels of blood heparan sulfate, which is excreted through the glomerular tuft to be reabsorbed in the tubules and ducts, rather than glycosaminoglycan derived in situ from faulty catabolism. It is noteworthy that similar excretion of mannose-rich oligosaccharides occurs through the glomerular tuft in bovine α-mannosidosis, as shown by a ratio >2 : 1 in prerenal and postrenal blood. 13
The accumulation of storage material in hepatocytes is also likely to reflect an ability to take up circulating heparan sulfate from plasma in addition to that generated intracellularly in hepatic lysosomes. The liver has a great capacity to adsorb metabolites from circulating blood and to metabolize and channel them into a wide variety of metabolic pathways. The inability to degrade heparan sulfate would preclude its sugars from entering the general metabolic pool. Hepatocytes contained many large storage lysosomes, and, on electron microscopy, these contained rosettes that were similar in appearance to glycogen. It is concluded that this material is glycogen, secondarily accumulated because of interference in its catabolism by excess heparan sulfate, which may reversibly inhibit lysosomal enzymes, including α-glucosidase. 2 Enlargement and distortion of heart valves in 2 affected dogs are probably disease related, as similar lesions commonly occur in the mucopolysaccharidoses, including MPSIIIA.
Accumulation of substrate within secondary lysosomes as a consequence of deficient activity of a hydrolase is an easy concept to understand. However, this may be an oversimplification, because large amounts of glycosaminoglycan are lost in the urine. This suggests that considerable amounts of this substrate arise from compromised digestion of HSPGs shed from the surface of cells 5, 27 or that there may be an increase in the amount of extracellular glycosaminoglycan to be cleared because of perturbation of the endosome/lysosomal system, resulting in excess heparan sulfate or HSPGs being returned to the cell surface. Either explanation may explain the widespread occurrence of vesiculated macrophages in the CNS and elsewhere. Material not immediately cleared by this means may give rise to elevated levels of HSPG or heparan sulfate in tissue fluids, blood, and eventually urine. An increase in heparan sulfate within the neuropil may have subtle changes on the binding and uptake of ligands, on the growth and structure of dendrites, or on synapses. Such changes in the interstitial milieu of the brain are perhaps signalled by the swelling and mild fibrous metaplasia of astrocytes, cells intimately associated with homeostasis of the ionic environment and pH of the extracellular fluid in the brain.
There is a growing interest in the relationship between the activation of glial cells and neuronal death in lysosomal storage diseases, 10, 14, 19, 25 and, at least in mouse Sandhoff disease (a form of GM2 gangliosidosis), this may be mediated by autoantibodies to GM2 and GA2 gangliosides. 26 In another study, it was noted that microglial activation, which preceded acute neurodegeneration in murine Sandhoff disease, was suppressed by bone-marrow transplantation. 20 In mouse models of MPSI and MPSIIIB, in which similar lesions have been investigated, there is upregulation of messenger ribonucleic acid transcripts related to macrophage function. 18 The authors questioned whether this increase results in an exacerbation of the disease process and suggested that, at the very least, microglia should be considered a target cell in therapeutic strategies. The apparent intimate relationship between macrophages and neurons in dogs with MPSIIIA is thus of particular interest beyond the implication of transfer of storage material from neuron to macrophage.
MPSIIIA in dogs, with signs related mainly to the cerebellum, varies clinically from the more severe and varied syndrome in children, but, the underlying biochemical pathogenesis of the disorder and the way this may react to therapy are likely to be similar. The less severe and varied clinical manifestation of this disorder in dogs may facilitate the use of this animal as a model for therapeutic trials.
Footnotes
Acknowledgements
We wish to thank Mrs. P. Davey and Mr. A. Hicks for assistance in preparing paraffin and epoxy resin sections for light and electron microscopy. D. Hopcroft also helped with cutting sections for electron microscopy and printed the electron micrographs.
