Abstract
American trypanosomiasis is caused by the zoonotic protozoa Trypanosoma cruzi and primarily results in heart disease. Organisms also infect the central nervous system (CNS). The Texas A&M University veterinary teaching hospital archive was searched for dogs with CNS disease with intralesional protozoal amastigotes. This study summarizes 4 cases of dogs with disseminated trypanosomiasis and CNS involvement confirmed by quantitative polymerase chain reaction (qPCR) with T. cruzi primers. Clinical signs included lethargy, respiratory distress, tetraparesis, and seizures. Central nervous system lesions included meningeal congestion (1/4), necrosis with hemorrhage in the spinal cord gray and white matter (2/4), and histiocytic meningoencephalitis (4/4), and meningomyelitis (2/4) with intralesional and intracellular protozoal. Genotyping identified 1 case of T. cruzi discrete typing unit (DTU) TcI and 2 cases as TcIV, both are common variants in the United States. Trypanosomiasis should be considered a differential diagnosis for dogs with CNS signs in T. cruzi-endemic areas.
Trypanosoma cruzi, a zoonotic protozoan parasite, is the causative agent for American trypanosomiasis or Chagas disease. Trypanosomiasis is a significant public health concern with over 6 to 7 million people infected.12,20 T. cruzi is enzootic in the Americas and infects a wide range of both wild and domestic species.13,14 This parasite is known for its tropism for the heart, especially the right ventricular free wall.15,22 Infection can lead to a variety of clinical signs such as lethargy, coughing, and collapse due to dilated cardiomyopathy, arrhythmias, and atrioventricular blocks. 15
Trypanosomiasis is primarily transmitted by vectors. Humans and animals are infected when insects in the Reduviidae family, colloquially known as kissing bugs, deposit infectious trypomastigotes from their urine or feces onto the skin during a blood meal. 6 In animals, oral transmission is considered to be an important mode of T. cruzi transmission through the consumption of infected triatomine insects or their feces. 2 Infectious trypomastigotes circulate until they locate and invade susceptible host cells and form pseudocysts containing amastigotes. 6
T. cruzi can be subclassified into 7 discrete typing units (DTUs) TcI to TcVI and Tcbat, based on genetic analysis. 3 Different DTUs have varied geographic distributions, which can be used to predict disease distribution and host preferences. Surveys of triatome insects in the United States have found DTU TcI and TcIV subtypes.3,10 DTU TcIV is often found in dogs with cardiac disease but has also been reported in a horse with neurologic signs and humans with cardiac and neurologic disease.4,7,9,18,19
While the hallmark of T. cruzi infection is cardiac disease, the 4 cases herein presented demonstrated clinical signs of primary central nervous system (CNS) disease. Little is reported about T. cruzi tropism for the CNS; therefore, we describe the CNS lesion in dogs and evaluated the associated DTU to determine if the DTU correlated with CNS tropism.
A search of the Texas A&M University College of Veterinary Medicine and Biomedical Sciences pathology database from 1995 to 2021 was performed using keywords: trypanosome, Chagas, and trypanosomiasis. Reports were evaluated for clinical presentation, gross lesions, and histologic findings. Cases with CNS involvement were selected for inclusion. This search revealed 78 reports of which 4 met the inclusion criteria based on CNS involvement. All available information regarding signalment, clinical presentation, diagnostic tests, and pathology results were collected.
Case 1 was a 3-month-old, male Boxer dog that presented for ascites, respiratory distress, and acute collapse. Case 2 was a 2-year-old, castrated male Mastiff dog that presented for fever, hemiparesis, and tetraparesis. Despite aggressive treatment, clinical signs in case 2 progressed to intermittent extensor rigidity, hyper-reflexic hindlimbs, and grand mal seizures. Case 3 was a 7-month-old, female Mastiff dog that presented for pitting edema, lethargy, cachexia, and fever. Case 4 was a 5-year-old, castrated male American Pit Bull Terrier that presented for respiratory distress and neurologic signs including right hindlimb paresis that rapidly progressed to paralysis with urinary and fecal incontinence. Analysis of cisternal cerebrospinal fluid (CSF) was only performed in case 2, in which it revealed an increased total protein concentration of 128 mg/dL, Refractive Index (RI): < 30, and a mild, mixed cell pleocytosis with a total nucleated cell count of 7 cells/µL (RI: < 5). The leukocyte differential consisted of 8 large mononuclear cells, 3 small lymphocytes, 2 eosinophils, and 1 non-degenerate segmented neutrophil; however, no infectious organisms were seen.
CNS gross and histologic findings are summarized in Table 1. Gross CNS lesions were variable and ranged from no lesions (Cases 1 and 3) to meningeal congestion with unilateral cerebral swelling (Case 2), and multifocal areas of dark red to brown discoloration in the cervical spinal cord gray matter (Case 2) and white matter (Case 4). Case 4 exhibited spondylosis deformans from L2 to S1 and had multifocal superficial abrasions along the dorsal aspect of the right hindlimb secondary to hind limb paresis.
Gross and histologic lesions in the CNS associated with T. cruzi infection.

Hematoxylin and eosin (H&E) stained sections from each case were reviewed for histopathologic changes within the CNS. Within the cerebrum, all cases had scattered intracellular and extracellular 2 to 4 µm, round to oval protozoal amastigotes with distinct nuclei and parallel, rod-shaped kinetoplasts accompanied by multifocal infiltrates of a mixed inflammatory population characterized by histiocytes and fewer lymphocytes and plasma cells in the gray and white matter as well as the meninges (Fig. 1a, b). Virchow-Robbins spaces were expanded by aggregates of lymphocytes and plasma cells. In 2 cases (3 and 4), the inflammation extended into the brainstem (Fig. 1c). Cases 2 to 4 had multifocal infiltrates of predominantly macrophages with fewer lymphocytes and plasma cells in the gray and white matter with extracellular, intraneuronal, and intrahistiocytic amastigotes (Fig. 1d). The spinal cord was not examined for Cases 1 and 3; however, spinal cord was available for examination in Cases 2 and 4. Within the cervical spinal cord, the gray and white matter had multifocal areas of histiocytic infiltration with fewer neutrophils with numerous intraneuronal, intrahistiocytic, and extracellular amastigotes (Fig. 1e).
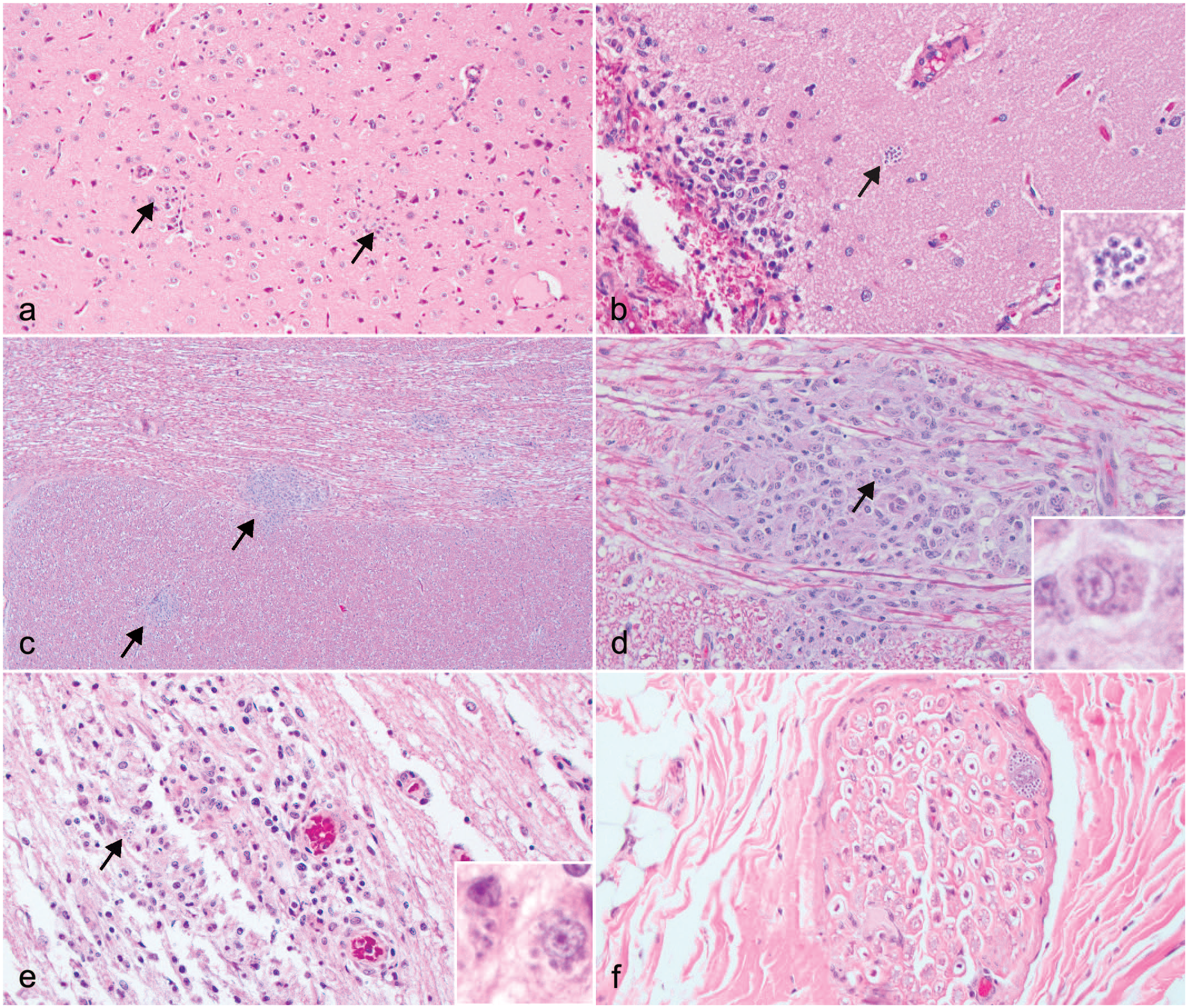
(a) Multifocal, nonsuppurative encephalitis (arrows). Cerebrum, Case 1. HE. (b) Multifocal to coalescing, lymphohistiocytic meningoencephalitis with extracellular protozoal amastigotes (arrow). Inset, high magnification of extracellular amastigotes. Cerebrum, Case 1. HE. (c) Multifocal, histiocytic encephalitis (arrows). Brainstem, Case 2. HE. (d) Multifocal to coalescing, histiocytic encephalitis. Amastigotes are within macrophages (arrow). Inset, high magnification of intracellular amastigotes. Brain stem, Case 2. HE. (e) Multifocal to coalescing, lymphohistiocytic myelitis with intralesional protozoal amastigotes (arrow). Amastigotes are within macrophages (inset). Spinal cord, Case 4. HE. (f) Focus of protozoa within a nerve. Peripheral nerve, Case 3. HE, hematoxilyn and eosin.
Histologically, all cases had pseudocysts with amastigotes in the heart accompanied by lymphohistiocytic myocarditis. Less commonly, amastigotes were in the tongue (Case 1), spleen (Cases 2 and 3), and bone marrow (Case 2). Additional pseudocysts were found in the peripheral nerve of skeletal muscles (Case 3; Fig. 1f) and within various skeletal muscles with lymphohistiocytic myositis (Cases 1 and 4).
Other findings included interstitial pneumonia with pulmonary fibrosis (2/4), interstitial nephritis (1/4), lymphoid follicular hyperplasia with sinus histiocytosis (1/4), enteritis (1/4), pancreatitis (1/4), pituitary adenitis (1/4), cystitis and vaginitis (1/4), and dermatitis (1/4). All cases also had hepatic vacuolar changes that varied from glycogen to lipid type.
To confirm the diagnosis of trypanosomiasis, DNA was extracted from 50 µm curls of formalin-fixed, paraffin-embedded CNS tissue with documented intralesional protozoa (Cases 1-3) using the QiAMP Formalin-Fixed Paraffin-Embedded (FFPE) advanced kit (Qiagen, Valencia, CA), following the manufacturer’s instructions. Fresh tissue was available for case 4, and DNA was extracted using Omega E.Z.N.A tissue DNA kit from Omega Bio-Tek (Norcross, GA) following the manufacturer’s instructions. Extracted DNA was tested for T. cruzi infection by amplification of repetitive nuclear satellite DNA (Cruzi1/Cruzi2/Cruzi3) as previously described.5,11 This PCR method has demonstrated sensitivity and specificity for T. cruzi compared to other PCR applications. 23 T. cruzi DNA extracted from isolate Sylvio X10 CL4 (ATCC 50800, American Type Culture Collection [ATCC]) was used as the positive control; negative controls included absence of template DNA and DNA-free water. 5 Samples were considered positive if the Ct value was below 35. All 4 cases were found to be positive with low Ct values (Case 1 = 27.53; Case 2 = 21.50; Case 3 = 30.04; Case 4 = 23.73) confirming infection with T. cruzi.
All T. cruzi positive samples were f evaluated by PCR to assess the nuclear spliced leader intergenic region (SL-IR) for typing of T. cruzi. Twenty microliter reactions were performed using the QIAGEN Multiplex PCR kit (Qiagen) with previously described modifications. 10 Positive controls include DNA from TcI (ATCC 50800), T. cruzi-infected Triatoma sanguisuga from Texas (DTU TcIV), and T. cruzi Y-strain (ATCC 50832, DTU TcII). PCR for Leishmania spp. was performed as previously described. 16
Genotyping was performed on all cases using undiluted and 1:10 dilution samples. Case 1 was consistent with T. cruzi DTU I, and cases 3 and 4 were consistent with DTU IV. Unfortunately, the DTU from case 2 could not be determined. DTU determination on case 2 presumably failed due to DNA degradation from prolonged formalin-fixation. 9
Central nervous system disease is an uncommon clinical manifestation of T. cruzi and was not initially considered a differential at the time of necropsy. 23 The diagnosis was made primarily based on the histologic presence of amastigotes with a perinuclear kinetoplast, which narrowed the differentials to Trypanosoma cruzi or Leishmania sp. The orientation of the kinetoplast is a key histologic feature used to differentiate between T. cruzi and L. infantum. Trypanosomes are characterized by a parallel kinetoplast whereas the kinetoplast is perpendicular in Leishmania spp. 1 Although the kinetoplast can be used to differentiate between the 2 genera, H&E stained sections are less sensitive than transmission electron microscopy (TEM) for evaluating kinetoplast orientation. TEM was performed on cases 2 and 3 at the time of initial diagnosis, which revealed a parallel kinetoplast consistent with T. cruzi. Immunohistochemistry (IHC) can also be performed to highlight the organisms; however, IHC has lower specificity due to the potential cross reaction between surface antigens of the 2 organisms. 19
Polymerase chain reaction is both sensitive and specific for diagnostic purposes.8,10,11,23 qPCR was positive for all cases, and SL-IR typing identified DTU TcI (case 1) and TcIV (case 3,4). qPCR for Leishmania spp. was negative for all cases. Interestingly, an experimental infection in Swiss mice comparing the tissue tropism of TcII and TcIV found that TcIV appeared to have greater tropism for the CNS. 17 Due to the limited typing results for the cases presented herein, we are unable to confirm that DTU TcIV is associated with CNS tropism.
Types I, II, IV, V, VI have all been found within the CNS of non-human mammals. 24 A recent literature review indicated amastigotes were primarily within the encephalon when present. 24 Generally, lymphohistiocytic inflammation and amastigotes in the myocardium is expected with T. cruzi. In cases with disseminated disease that involves the CNS, the histologic findings include inflammatory aggregates of lymphocytes and macrophages, often with perivascular orientation in the brain and spinal cord and intralesional free and intracellular amastigotes. 21 Chagas disease in humans can localize to the brain and spinal cord, and immunocompromised patients are at an increased risk. Headaches, fever, and acute meningoencephalitis are noted in these patients. 7 Central nervous system dissemination also has been noted in a horse with chronic ataxia and lameness, which had protozoal amastigotes within the spinal cord but not the heart. 4 Similarly, Case 4 had only 1 cyst in the heart, while most lesions were localized to the brain and spinal cord.
Types I, II, and III have been associated with domestic cycles between domestic animals and humans while types I, II, V, and VI have been more closely associated with the sylvatic cycle between wildlife and the vector, though most of this testing focuses on Central and South America. 3 Types I and IV are the only subtypes reported in Texas at this time. 10
Overall, the 4 cases evaluated exhibit CNS dissemination of T. cruzi, with varying degrees of inflammation in other organs. As some of the cases presented with only neurologic signs, T. cruzi should be a differential for CNS disease in animals in endemic areas.
Footnotes
Acknowledgements
The authors would like to acknowledge the Texas A&M histology lab for their excellent work on these cases, and the owners of the animals for allowing further study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
