Abstract
The low-affinity nerve growth factor receptor p75NTR is a major neurotrophin receptor involved in manifold and pleiotropic functions in the developing and adult central nervous system (CNS). Although known for decades, its entire functions are far from being fully elucidated. Depending on the complex interactions with other receptors and on the cellular context, p75NTR is capable of performing contradictory tasks such as mediating cell death as well as cell survival. In parallel, as a prototype marker for certain differentiation stages of Schwann cells and related CNS aldynoglial cells, p75NTR has recently gained increasing notice as a marker for cells with proposed regenerative potential in CNS diseases, such as demyelinating disease and traumatic CNS injury. Besides its pivotal role as a marker for transplantation candidate cells, recent studies in canine neuroinflammatory CNS conditions also highlight a spontaneous endogenous occurrence of p75NTR-positive glia, which potentially play a role in Schwann cell–mediated CNS remyelination. The aim of the present communication is to review the pleiotropic functions of p75NTR in the CNS with a special emphasis on its role as an immunohistochemical marker in neuropathology. Following a brief illustration of the expression of p75NTR in neurogenesis and in developed neuronal populations, the implications of p75NTR expression in astrocytes, oligodendrocytes, and microglia are addressed. A special focus is put on the role of p75NTR as a cell marker for specific differentiation stages of Schwann cells and a regeneration-promoting CNS population, collectively referred to as aldynoglia.
Neurotrophins are capable of enhancing survival, proliferation, and differentiation of neuroepithelial cells but may in parallel induce apoptosis and cell death, respectively. 4,35,163 The term neurotrophins encompasses various molecules, including nerve growth factor (NGF), 122,123 brain-derived neurotrophic factor (BDNF), 9,120 neurotrophin (NT)–3, 84,128 NT-4, 79,91 and their respective precursors (proNGF, proBDNF, proNT-3). 14,119,170
These neurotrophins exert their effects by binding to their respective cell surface receptors. Each of the receptors belonging to the tyrosine receptor kinase (Trk) family (TrkA, TrkB, and TrkC) binds specific neurotrophins with high affinity, 8,23,35,109,116,176 whereas the p75 neurotrophin receptor (p75NTR) is able to bind all aforementioned neurotrophins, although with lower affinity (Fig. 1). 23,35,45,147 Since its first description in the 1970s and 1980s, p75NTR has been the object of numerous studies seeking to elucidate its functions and its role as a cell marker. Indeed, this receptor has been shown to fulfill contradictory tasks in central nervous system (CNS) regeneration and degeneration, depending on the cellular context and its interaction with other receptors. While substantial progress has been made in understanding the underlying mechanisms, the ambivalent functions of p75NTR are not fully understood yet.
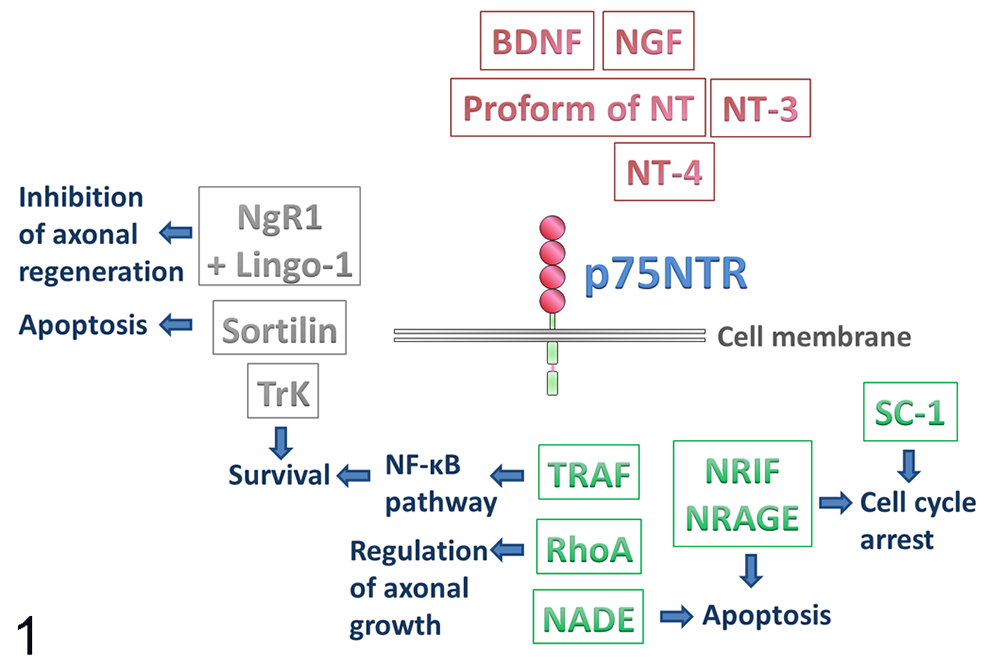
Schematic representation of the various interactors of nerve growth factor receptor p75 (p75NTR). The extracellular domain binds neurotrophins as well as their respective proforms (red). Interaction with other receptors (gray) influences the outcome of p75NTR activation. As the intracellular domain lacks catalytic activity, the signaling of p75NTR is partially carried out by interactors of the intracellular domain (green), which also accounts for the different effects of p75NTR stimulation. BDNF, brain-derived neurotrophic factor; Lingo-1, immunoglobulin-like domain-containing nogo receptor interacting protein 1; NADE, p75NTR-associated cell death executor; NGF, nerve growth factor; NgR1, Nogo-66 receptor 1; NRAGE, neurotrophin receptor-interacting melanoma antigen homolog; NRIF, neurotrophin receptor interacting factor; NT, neurotrophin; RhoA, ras homolog gene family, member A; SC-1, Schwann cell factor 1; TRAF, TNF receptor–associated factor; TrK, tyrosine receptor kinase.
p75NTR does not signal through traditional pathways as its cytoplasmic domain is lacking catalytic activity. 29,125 Hence, signaling of this receptor is partially carried out by interacting proteins, which are recruited by or associated with the receptor in response to neurotrophins. 83,163 Several such interactors of the intracellular domain of p75NTR have in fact been identified so far, all of them mediating differing biological functions (Fig. 1). For instance, p75NTR-associated cell death executor (NADE), 141 neurotrophin receptor interacting factor 1 and 2 (NRIF1 and 2), 17,33 and neurotrophin receptor–interacting melanoma antigen homolog (NRAGE) 167 promote p75NTR-dependent apoptosis. NRIF, NRAGE, and Schwann cell factor 1 37 additionally promote cell-cycle arrest via p75NTR. 83 The p75NTR-induced activation of the nuclear factor-κB pathway is regulated by members of the tumor necrosis factor (TNF) receptor–associated factor family. 213 Further, interaction between p75NTR and Ras homolog gene family member A (RhoA) is reported to regulate axonal growth. 212
Conversely, interaction with other receptors also strongly influences the outcome of p75NTR activation (Fig. 1). While p75NTR positively influences Trk-mediated survival and trophic effects, 83,147 its interaction with sortilin leads to induction of apoptosis subsequent to binding of proNGF. 146 p75NTR also forms a receptor complex with Nogo-66 receptor 1 and leucine-rich repeat and immunoglobulin-like domain-containing nogo receptor interacting protein 1. 132,201 Binding of certain myelin components (CNS-derived myelin-based growth inhibitors), such as myelin-associated glycoprotein, by this receptor complex has been shown to inhibit axonal regeneration. 132,201,211
In addition, the intracellular domain of p75NTR can be released through receptor intermembrane proteolysis, thus being enabled to act as a signaling molecule itself. 26,100,102
Besides binding neurotrophins, p75NTR is also able to bind the glycoprotein of rabies virus. 71,195 Although not crucial for the infection itself, interaction with p75NTR leads to enhanced internalization and retrograde axonal transport of the virus in vitro as compared with p75NTR-independent transport. 71,195
Moreover, although with contradictory results, p75NTR is also implicated in the pathogenesis of Alzheimer’s disease. While binding of amyloid β and amyloid precursor protein by p75NTR has been associated with neuronal damage, the extracellular domain of this receptor, released via ectodomain shedding, seems to play a neuroprotective role by inhibiting amyloid β deposition. 54,210,219,223
Binding of PrP106-126, a synthetic fragment of the prion protein, to p75NTR induces apoptosis in neuroblastoma cell cultures. 6,46 Further underscoring a potential role of p75NTR in transmissible spongiform encephalopathies, there is increased expression of p75NTR in bovine spongiform encephalopathy–inoculated transgenic mice correlating with lesion distribution, prion protein deposition, and gliosis. 130
Recent studies demonstrate a role of p75NTR in pathological changes of the CNS, including inflammatory diseases of the canine CNS. Further emphasizing the importance of this molecule, p75NTR also serves as a marker for Schwann cells in clinical transplantation studies for CNS conditions such as demyelinating disease and spinal cord injury. This article summarizes the role of p75NTR in the CNS with a special emphasis on its role as a cell marker in veterinary neuropathology.
A drawback in the interpretation of immunohistochemical data regarding p75NTR expression in veterinary neuropathology is represented by the fact that different antibodies of variable clonalities against p75NTR, either directed against the extra- or the intracellular domain of the receptor, are used in various studies. Hence, and also because of the use of different immunohistochemical protocols in different laboratories, these data are not necessarily entirely comparable; however, general data upon p75NTR expression in the CNS can be expected to correlate throughout these studies. For all figures in this report that display immunohistochemistry for p75NTR, we have applied antibodies directed against the extracellular domain of the receptor (murine tissue: polyclonal p75NTR antibody, Merck Millipore AB 1554; canine, feline, and ferret tissue: monoclonal p75NTR-antibody, ATCC, clone HB8737). Furthermore, the following antibodies have been tested for immunohistochemistry in canine and murine samples and specifically reacted with p75NTR in our lab: polyclonal p75NTR antibody (extracellular domain; Novus Biologicals NBP1-25971) and polyclonal p75NTR antibody (intracellular domain; Alomone labs ANT-011).
Expression of p75NTR in Neurons During Development, Adulthood, and CNS Injury
In neurons, p75NTR is expressed at certain developmental stages in various regions of the brain and spinal cord. 51,61,207 For instance, neurons in dorsal root ganglia (DRG), spinal cord motor neurons, brain stem neurons, cholinergic basal forebrain neurons, Purkinje cells, trigeminal ganglion neurons, and neurons in the caudate putamen express p75NTR at specific stages of their development. 51,61,127,137,207 Still, after reaching adulthood, p75NTR expression becomes significantly down-regulated in most neurons. 88,163 Illustrating its functions in neuroaxonal development, knockout mouse models demonstrate that p75NTR plays a regulatory role in developmental axon growth. 16 Complete deletion of p75NTR in knockout mice leads to a significantly reduced number of DRG neurons as well as a decreased thickness of sciatic nerves 198 but also causes a long-lasting increase in the number of basal forebrain cholinergic neurons, thus seemingly modulating their survival. 144 These findings show that proper functioning of p75NTR is mandatory for normal CNS development.
In adulthood, expression of p75NTR is retained in certain anatomical localizations (Figs. 2 and 3), such as cholinergic basal forebrain neurons in various species, including humans, mice, dogs, monkeys, ferrets, and sheep. 7,52,82,90,127,162 Similarly, spinal cord motor neurons and supraspinal neurons are p75NTR immunoreactive at low levels during adulthood in rodents. 11,51 Adult canine DRG neurons express both p75NTR mRNA and protein in situ (Supplemental Figs. S1 and S2), but not in vitro. 65 Contrary, up-regulation of p75NTR is observed in adult rat DRG neurons in vitro. 221 This discrepancy might be attributed to the different species, culturing protocols, or detection methods used. 65 Moreover, expression of p75NTR is also retained in cerebellar Purkinje cells under normal conditions in rats. 110,154,200
Physiological expression of nerve growth factor receptor p75 (p75NTR) in normal nervous system of representative domestic animals.
Despite its restriction to certain neuronal populations and its general down-regulation after reaching adulthood, neuronal p75NTR expression is interestingly up-regulated during various pathological conditions. 45,80,160,164 For instance, experimentally induced focal cerebral ischemia in rats results in a transient overexpression of p75NTR in striatal cholinergic neurons, which are markedly resistant to ischemic cell death. 112 However, whether p75NTR increases this resistance or is involved in the induction of apoptosis in these cells remains to be elucidated. 112
Likewise, axotomy via internal capsule lesion leads to an increase of p75NTR expression in corticospinal neurons in the murine brain. 67 Co-expression of TrkC and p75NTR is necessary to induce neuronal cell death in this model. 67
Further, hypo-osmolar stress increases p75NTR expression in murine primary neurons in vitro. 159 In rat cerebellar Purkinje cells in proximity of the lesion induced by unilateral needle puncture, up-regulation of p75NTR expression is demonstrated via immunohistology when compared with the contralateral side and to controls. 131 Likewise, p75NTR immunoreactivity in spinal cord motor neurons is increased during disease progress in a transgenic mouse model of amyotrophic lateral sclerosis. 40 The majority of these neurons does not co-express caspase-3, a marker for apoptosis, so p75NTR might also be involved in the cellular stress response apart from inducing apoptosis. 40
Following peripheral nerve crush or transection, strong expression of p75NTR appears in spinal cord and facial motor neurons in the rat, paralleling the regeneration of the respective axons. 51,166 Hence, the receptor might also play a role in axonal regeneration.
Chronic administration of an anti-p75NTR antibody substantially reduces loss of cholinergic neurons in the rat forebrain following application of kainic acid. 149 Thus, Oh et al 149 concluded that increased p75NTR expression in these neurons contributes to their death.
After sciatic nerve injury, rat neurons of the ipsilateral DRG show decreased p75NTR expression, while no change in neuronal p75NTR reactivity is present on the contralateral side. 222 The decrease in p75NTR expression is attributed to the damage to the peripheral sensory axons and the consecutive disruption of regulatory signals from the periphery. 222
In humans, cortical neurons rarely express p75NTR at an advanced age (>50 years) but do so frequently in brains affected by Alzheimer’s disease. 139 Under these circumstances, p75NTR expression is interpreted as a compensatory reaction aiming to sustain neuronal integrity. 139
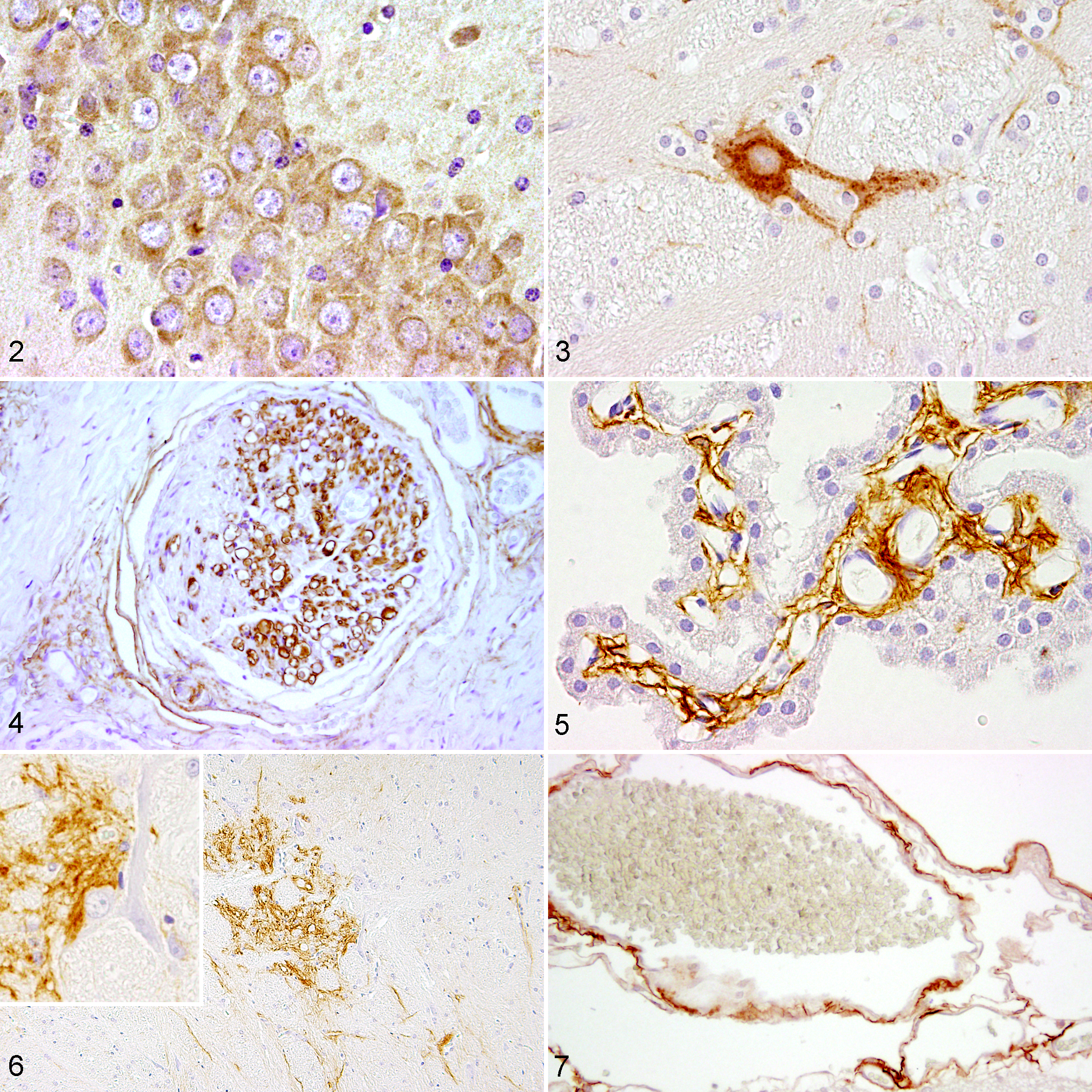
Using immunohistochemistry, p75NTR-positive Schwann cells and perineurial cells are demonstrated in the normal peripheral nerves of adult dogs (Fig. 4). In the healthy canine brain, p75NTR immunopositivity is observed in meningeal and ependymal fibroblasts as well as perineurally in the mesencephalic nucleus of the trigeminal nerve (Figs. 5 and 6). In cats and ferrets, p75NTR is also present within meningeal fibroblasts (Fig. 7). Besides single p75NTR-positive neurons (Fig. 3), ependymal fibroblasts also show immunoreactivity for p75NTR in ferrets.
p75NTR as a Marker for Neuronal Progenitor Cells
Neurogenesis is the active process of forming new neurons from stem or progenitor cells. 12,134 It has long been believed that neurogenesis exclusively occurs during development of the CNS, while adult neurons represent a fixed population that does not regenerate. 12,78,111 However, nowadays, there are several lines of evidence that neurogenesis in fact continues throughout adult life. 2,63,103 A limited number of neural stem cells remains in the subventricular zone (SVZ) and in the subgranular zone of the dentate gyrus (SGZ) in adulthood. 12,73,199 These cells are capable of generating neurons and glial cells in adults. 12
Within the SGZ of mice, p75NTR-positive cells partially express either neuronal nuclear antigen (NeuN) or glial fibrillary acidic protein (GFAP), suggesting neuronal and astrocytic differentiation, respectively. 18 In p75NTR-knockout mice, the number of bromodeoxyuridine (BrdU)–positive cells in the dentate gyrus is significantly reduced, suggesting that p75NTR promotes proliferation of progenitor cells within the SGZ. 18
In postnatal and adult rats, a small number of cells in the SVZ shows an immunohistological reaction for both p75NTR and nestin, the latter being a prototype marker for neural precursors. 215 However, in postnatal and adult rats, p75NTR-positive cells account for less than 1% of the total number of cells in the SVZ. 215 In adult individuals, the p75NTR-positive population encompasses all of the neurosphere-producing precursor cells, whereas in newborn rats, the precursor cells can be either p75NTR-positive or -negative. 215 Nevertheless, interestingly, only p75NTR-positive cells possess neurogenic potential in both newborn and adult rats in vitro. 215 Demonstrated via immunohistology, some p75NTR-positive cells in the SVZ of adult male rats co-express nestin, whereas most of these cells co-express Ki-67, a marker for proliferating cells. 70 Similarly, p75NTR-expressing cells are positive for BrdU in the murine SGZ, suggesting that these cells are proliferating. 18 Likewise, adjacent to the SVZ, expression of p75NTR is up-regulated in oligodendroglial progenitors in response to cuprizone-induced demyelination. 153
Thus, in the adult brain, these p75NTR-positive cells appear to show primarily neurogenic properties under physiological conditions, although they may give rise to glial progenitors in pathological events. 41
Consequently, p75NTR may be used as a cell marker for neural progenitor cells with a primary neurogenic potential in the SVZ.
The Role of p75NTR in Glial Cells
Microglia
Myeloid cells in the CNS, a heterogeneous group of innate immune cells, encompass microglia as well as perivascular, meningeal, and choroid plexus CNS macrophages. 155 Microglia are the only parenchymal CNS myeloid cells and originate from the yolk sac and probably the fetal liver. 69,155 Interestingly, recent studies indicate that the macrophages found at CNS interfaces also derive from the same sources. 155 In addition, microglia as well as perivascular and meningeal CNS macrophages are long-lived and do not receive input from the blood or bone marrow in adulthood. 155
Following their widespread migration during development, microglia are found scattered throughout all regions of the CNS. 107 Under normal conditions, these cells are in a resting state characterized by a ramified cell morphology. 107 However, alterations of their environment, such as ischemia or inflammation, lead to rapid microglial activation. 107,113 Microglia play a pivotal role in phagocytosis of cellular debris, 107,113 control of neuronal activity, 15 and regulation of the number of functional synapses. 99 They also are a source of neurotrophins 50,81 and at the same time act as a target for these molecules. 217 Using Western blot, Nakajima et al 142 reported expression of p75NTR alongside Trk receptors in cultured microglia of neonatal rats, while in a different study, p75NTR expression was detected neither in murine microglial cell lines nor in primary microglia from neonatal rats. 217 This discrepancy might be due to differences in the process of culturing the cells. Using immunohistology and in situ hybridization, p75NTR-positive microglia/macrophages are demonstrated in long-term organotypic murine brain slice cultures. 177 Binding of NGF by p75NTR negatively influences the induction of major histocompatibility class II in microglia in rat slice cultures, which is demonstrated by the reversal of the effect through antibody-mediated receptor blockage. 145 Therefore, microglial p75NTR expression may play a role in immunomodulation.
Despite these lines of evidence from in vitro studies, there are surprisingly few reports concerning microglial p75NTR expression in vivo. For instance, microglial up-regulation of p75NTR is observed in multiple sclerosis (MS) plaques in humans compared with control white matter and interpreted as part of the stress response of these cells. 48 In contrast, there is neither microglial expression of p75NTR in the normal human CNS nor in the MS-affected CNS in a second study. 168 This disparity might be related to the different immunohistological antibodies used in the studies. 168
Further studies, especially with emphasis on its expression in vivo, are needed to examine the role of p75NTR in microglia.
Astrocytes
Astrocytes represent the largest proportion of glial cells in the mammalian CNS. 3 Astrocytes produce and secret neurotrophins and proNGF, 47,62,194 especially after CNS injury. 133 Not only functioning as a source of neurotrophins, they may also act as a target for these mediators. 41 In the normal adult rodent brain, p75NTR expression in astrocytes is limited and, using immunohistology, primarily found in astrocytes of the glia limitans. 165 In vitro, primary cultures of rat type I astrocytes express low levels of p75NTR, while treatment with NGF causes these cells to significantly up-regulate mRNA expression of p75NTR. 87 Following global transient cerebral ischemia in rats, up-regulation of both p75NTR and TrkA in the majority of astrocytes in the cornu ammonis 1 area of the hippocampus is demonstrated using immunohistology and double immunofluorescence. 148 Within lesions induced either by quinolinic acid or by 3-nitropropionic acid, a dose-dependent up-regulation of p75NTR in astrocytic progenitor cells, but not in mature astrocytes, of the rat striatum is shown via immunohistology and double immunofluorescence. 80 However, the consequence of this up-regulation of p75NTR in astrocytes remains elusive.
Even though treatment of rat hippocampal astrocytes with NGF in primary cultures causes a significant reduction in the cell number, this effect does not seem to be mediated through apoptosis via p75NTR. 42 Rather, activation of p75NTR by NGF appears to reduce proliferation of astrocytes, leading to the conclusion that this interaction may possibly restrict glial scar formation in vivo. 42
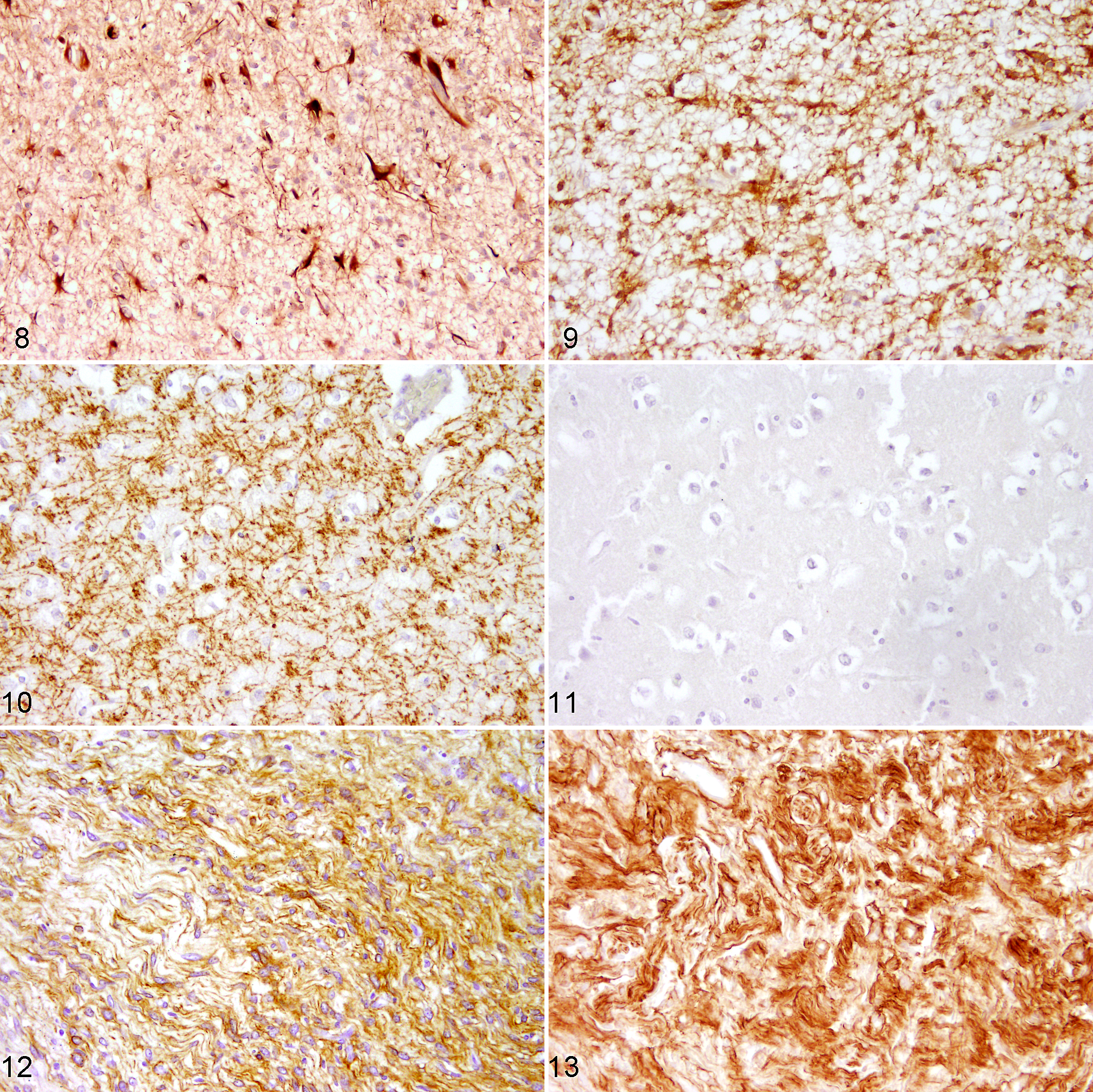
In a recent study, using tissue microarrays to compare different markers in several canine CNS neoplasms, p75NTR turned out to be useful in discriminating between oligodendroglioma and astrocytoma as it is expressed only in the latter (Figs. 8–11). 178 In addition, p75NTR is not expressed in canine anaplastic astrocytoma, thus possibly being an indicator of a higher differentiation of the neoplasm. 178
Nerve growth factor receptor p75 (p75NTR) as tumor cell marker.
Oligodendrocytes
As the counterpart of Schwann cells in the peripheral nervous system (PNS), oligodendrocytes are the myelinating cell in the CNS. 24 At birth, only a few regions in the brain are myelinated, and the myelination process continues until it is completed, which may take up to 30 years in humans. 53 Development, proliferation, migration, and differentiation into myelinating oligodendrocytes depend on various regulatory factors and are in part controlled by NGF, BDNF, and NT-3. 10,38,136,208 However, these effects seem to be predominantly mediated by Trk receptors. 38,49,101,114 Using immunohistology, p75NTR is not detected in oligodendrocytes in the normal adult rodent CNS. 39,143 Similarly, it is expressed by only few oligodendrocytes in the healthy adult human brain. 48
In different pathological CNS changes, animal experiments, and cell culture experiments, oligodendrocytes up-regulate p75NTR expression, however. In cultured mature oligodendrocytes obtained from postnatal rat cerebral cortex, treatment with NGF causes p75NTR-dependent cell death. 32 Following spinal cord injury in mice, p75NTR-mediated apoptosis of oligodendrocytes is induced by proNGF, 14 most probably by simultaneous binding of sortilin and p75NTR. 147 Oral administration of LM11A-31, a small molecule designed to block proNGF-p75NTR binding, promotes myelin sparing and functional recovery after spinal cord injury in mice due to a >50% increase in the number of surviving oligodendrocytes and myelinated axons. 192 Therefore, p75NTR indeed seems to play a role in oligodendrocyte apoptosis in rats and mice.
In contrary, treatment with NGF does not increase the apoptotic rate in cultured adult human oligodendrocytes, suggesting that p75NTR may fulfill other functions than apoptosis in human oligodendrocytes. 115
In MS white matter plaques, up-regulation of p75NTR mRNA and protein is demonstrated in oligodendrocytes. 48 However, only a part of these p75NTR-expressing oligodendrocytes simultaneously reacts positively with terminal deoxynucleotidyl transferase-mediated dUTP nick end-labeling (TUNEL). 48 Furthermore, a subpopulation of nerve-glial antigen 2–positive oligodendrocyte progenitor cells with an elongated shape expresses p75NTR in chronic MS lesions but not in the normal adult human brain. 34 This population is not associated with TUNEL positivity. 34 Taken together, these studies suggest that oligodendrocyte apoptosis in MS lesions is not primarily mediated via p75NTR signaling. This view is supported by an experiment designed to study the role of p75NTR in the fate of oligodendrocytes in a cuprizone-induced demyelination model in p75NTR-knockout versus C57BL/6 wild-type mice. 39 Despite transient expression of p75NTR in oligodendrocytes in wild-type mice, neither the degree of remyelination nor the number of surviving oligodendrocytes differs between wild-type and knockout mice, 39 substantiating the hypothesis that p75NTR is not necessarily associated with oligodendrocyte death.
An influence of the lesion type (spinal cord injury and demyelination, respectively) on p75NTR-mediated cell death has been proposed, possibly explaining the variations in the above-mentioned studies. 41
Schwann Cells and Aldynoglia: Glial Cells With Regenerative Capacity
p75NTR immunohistochemistry has been demonstrated as a useful tool to distinguish peripheral nerve sheath tumors (PNST) from other spindeloid neoplasms (eg, perivascular wall tumors) in dogs (Fig. 12). 36,184 The p75NTR immunoreactivity in PNST probably indicates an undifferentiated Schwann cell and/or neural crest origin of the neoplastic cells. 184 p75NTR expression in feline PNST is also reported (Fig. 13) but seems to be less constantly observed. 129
Moreover, because of their regenerative potential, both Schwann cells and olfactory ensheathing cells (OEC) have recently gained importance in research as promising candidates for the transplantation into the demyelinated or otherwise injured CNS. In fact, beneficial effects following transplantation of these glial cells into lesions are reported by numerous studies using rodent models as well as by studies conducted in the dog, although positive (ie, regenerative) effects still need to be demonstrated in humans.* Similarly, transplantation of canine OEC and Schwann cells into a rat model of spinal cord injury resulted in an improvement of the locomotor function. 157 Hence, additional studies in translational animal species, such as the dog, would be valuable.
Schwann cells are the principal glia found in the PNS. They are involved in many important physiological and pathological aspects of the PNS such as providing trophic support for neurons, conduction of nerve impulses along axons, development and regeneration of nerves, production of extracellular matrix, modulation of neuromuscular synaptic activity, and presentation of antigens. 5,28,30,98,182,183 However, their prototype function is to myelinate peripheral axons.
Schwann cells derive from the neural crest, and their development undergoes 3 different stages: Schwann cell precursor cells, immature Schwann cells, and mature Schwann cells can be distinguished, while the latter are further divided into myelinating and nonmyelinating Schwann cells. 97,135 Several immunohistological markers exist that are useful in identifying the different stages (Fig. 14). 135 Both immature Schwann cells and nonmyelinating mature Schwann cells express p75NTR. 98 Yet, interestingly, myelinating Schwann cells are not immunoreactive for p75NTR (Fig. 14). 98
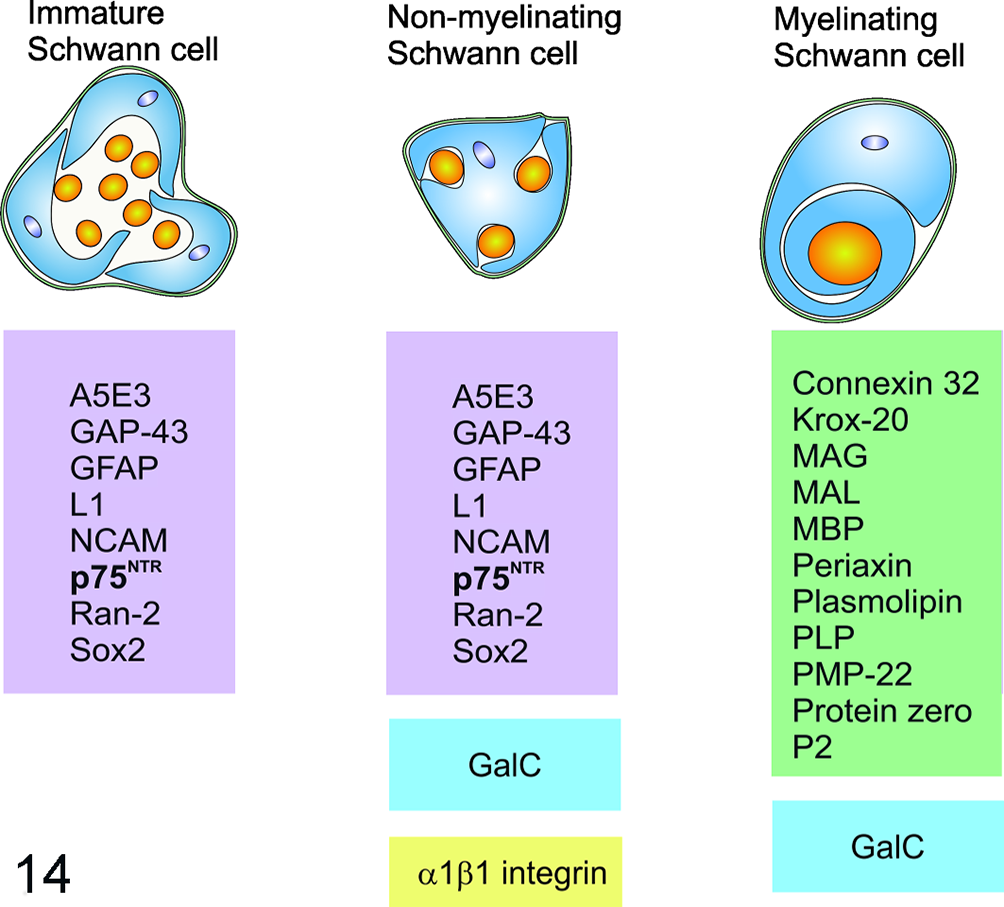
Antigen expression in different Schwann cell stages (schematic representation). Markers in the violet box are expressed in immature and nonmyelinating Schwann cells but not in myelinating Schwann cells. GalC (blue) is demonstrated in both myelinating and nonmyelinating Schwann cells. α1β1 integrin (yellow) and markers in the green box are specific for nonmyelinating and myelinating Schwann cells, respectively. GalC, galactocerebrosid; GAP-43, growth-associated protein 43; GFAP, glial fibrillary acidic protein; Krox-20, early growth response protein 2 (EGR2); MAG, myelin-associated protein; MAL, myelin and lymphocyte protein; MBP, myelin basic protein; NCAM, neural cell adhesion molecule; p75NTR, nerve growth factor receptor; PLP, proteolipid protein; PMP-22, peripheral myelin protein 22; P2, peripheral myelin protein 2 (PMP2); Ran-2, rat neural antigen-2. Modified from Mirsky et al and Lempp et al. 121,135
Nevertheless, p75NTR is widely expressed in Schwann cells during development and up-regulated after peripheral nerve injury as well as during regeneration processes. 41,88,105,187,214 p75NTR seems to regulate Schwann cell migration as a reduced migration rate is observed in knockout mice. 16 Although p75NTR generally plays a crucial role in apoptosis, the numbers of apoptotic Schwann cells in control and p75NTR-deficient mice during normal development are similar. 185 Thus, p75NTR does not seem to play a major role in apoptosis of Schwann cells during development.
However, adult p75NTR-knockout mice exhibit thinner myelin sheaths in the sciatic nerve than adult wild-type mice. 175 Following sciatic nerve crush injury, remyelination occurs in both mouse types, but histological analysis reveals that the number of myelinated axons and the thickness of myelin sheaths are reduced in mutant mice compared with wild-type mice. 175 These results indicate a crucial role of p75NTR in myelination. Still, the exact mechanism how p75NTR influences myelination remains enigmatic.
Nerve grafts harvested from either p75NTR-knockout or wild-type mice, transplanted into the sciatic nerve injury site of nude mice, lead to impaired motor recovery in p75NTR-knockout Schwann cell grafted mice in comparison with wild-type Schwann cell grafted animals. However, this reduced motor recovery rate is thought to be at least in part caused by impaired axonal growth. 193
Demonstrating the dual role of p75NTR in terms of either promoting regeneration or cell death, the death of Schwann cells in the distal nerve segment following sciatic nerve axotomy is mediated by p75NTR in postnatal rodents. 152 This process is enhanced by application of NGF in wild-type but not in p75NTR-knockout mice. 152
As Schwann cells also express p75NTR in vitro, this receptor is employed in the purification of Schwann cell cultures. 22,180
Aldynoglia (from the greek αλδατνω, “to make grow”) are a form of specialized glia encompassing OEC, tanycytes, pituicytes, and Müller glia. 76,206 These subpopulations of macroglia express some of the immunological markers of non-myelinating Schwann cells, including p75NTR, GFAP, and O4. 76,206
OEC are unique glial cells of the olfactory system that ensheath, guide, and stimulate growth of axonal processes of olfactory neurons that enter the CNS. 181 As in other cell types, rat OEC express p75NTR during early development and the expression decreases to reach the lowest level in adulthood. 72 In situ, p75NTR is expressed in only a small population of OEC located in the olfactory nerve layer of the olfactory bulb in neonatal rats. 55
Cultured OEC show a similar morphology and share several markers with cultured Schwann cells. 205 Most OEC in primary cell suspensions of rat and canine nasal mucosa do not express p75NTR, while the receptor is up-regulated during OEC culturing (Figs. 15 and 16). 22,25,150,203,224 Up-regulation is apparent only in those rat OEC displaying O4-positive axonal fragments on their surface. 203 This implies intimate contact with olfactory receptor neurons in situ and suggests that axonal signaling down-regulates p75NTR expression in vivo. 25,203 In general, p75NTR is considered a prototype marker of OEC in vitro, although CNPase is deemed a more reliable marker for rat OEC. 25,150 Interestingly, canine but not rat OEC share several characteristics with primate OEC in vitro (eg, growth without stimulation by mitogens and stable expression of p75NTR in long-term culture). 190,204 Further, expression of p75NTR remains stable even after immortalization of cultured canine OEC. 191 Hence, with an emphasis on transplantation approaches in humans, dogs might serve as an important translational animal model for studies focusing on OEC.
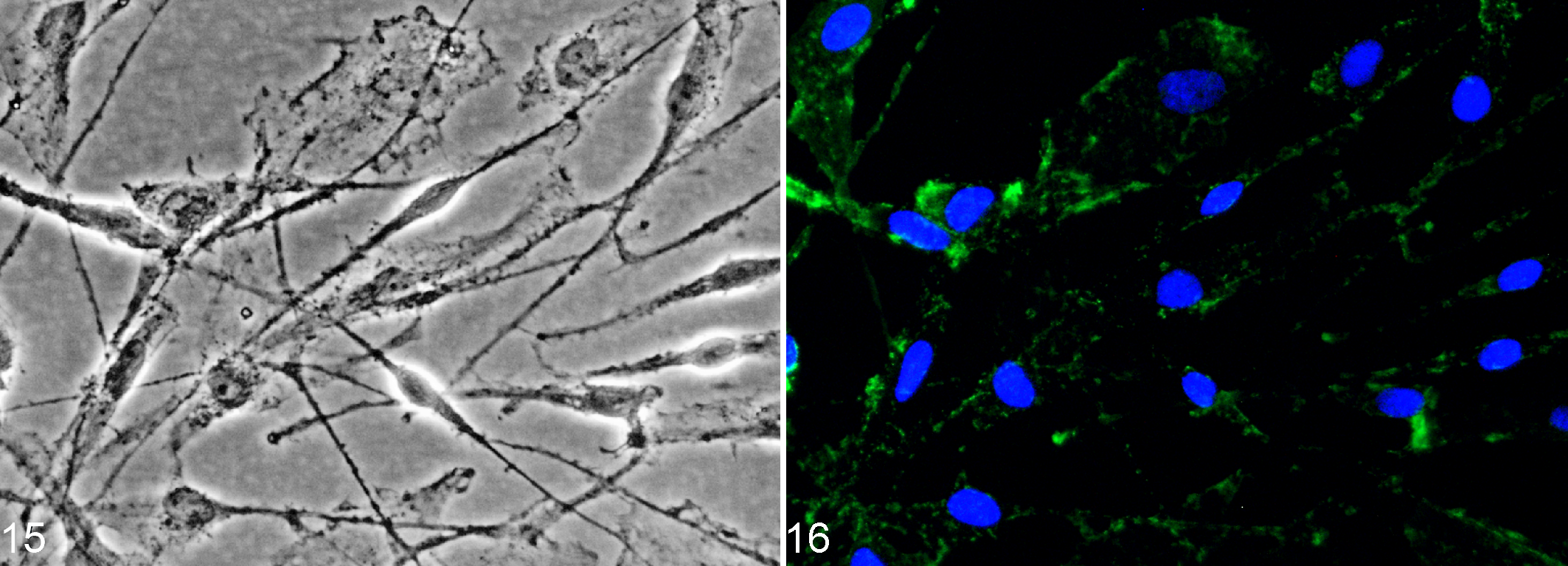
Canine olfactory ensheathing cells (OEC) express nerve growth factor receptor p75 (p75NTR) in vitro.
Tanycytes line the third ventricle, 74,206 whereas pituicytes are located in the neurohypophysis. 64 Both cell types are capable of ensheathing axons 64,76 and grow under similar conditions as OEC when extracted from the adult rodent brain. 76 In co-culture with neurons, these macroglia seem to envelope neurites and evolve into a myelinating phenotype. 76 There is strong evidence that at least a portion of tanycytes situated in the ependymal layer of the third ventricle may represent neural progenitor cells. 161,209 Further studies are needed to elucidate the function of p75NTR in tanycytes as well as pituicytes.
Within the retina of both primates and rodents, expression of 75NTR by Müller glial cells, another aldynoglia subtype, has been demonstrated. 85,86,169 p75NTR is indirectly involved in the death of p75NTR-negative retinal ganglion cells: proNGF forms complexes with the p75NTR and sortilin receptors of Müller glial cells, thus leading to robust production of TNF in these glial cells followed by TNF-dependent death of retinal ganglion cells. 118
Remarkably, p75NTR-expressing glial cells, which share properties with Schwann cells and aldynoglia, spontaneously occur within organotypical slice cultures of the murine cerebrum and brainstem as well as of the canine olfactory bulb. 89,177 These Schwann cell-like glia have synonymously been called aldynoglial Schwann-like glia, CNS Schwann cells, and Schwann cell–like brain glia. 89,151,188,196 Further, these cells are detected in mixed cell cultures derived from adult canine brain and represent the predominating cell population after about 11 weeks. 151 In these cultures, the Schwann cell–like glia are characterized by the expression of p75NTR as well as their bi- to tripolar, spindle-shaped morphology. 151
Interestingly, in cell cultures originating from adult dogs, OEC and Schwann cell–like glia completely lack differences in their global transcriptome when compared with each other 196 Moreover, cultured OEC and Schwann cell–like glia share a highly similar transcriptome with peripheral Schwann cells, which is in line with observations of a comparable increase in p75NTR expression after ∼2 to 7 days in vitro in all of these cell types. 22,55,151 Hence, OEC and Schwann cell–like glia, both being highly related to peripheral Schwann cells, seem to represent nearly identical cell types, at least in vitro, whose major difference appears to be made up by their respective location. Yet few transcriptomic differences between peripheral Schwann cells and their CNS counterparts have been revealed in this study. In line with this, voltage-gated potassium (K+) channels display KD-type K+ currents in canine cultured peripheral Schwann cells, whereas Schwann cell–like glia exhibit both KD- and KA-type K+ currents, as demonstrated via patch clamping. 104 This suggests that CNS Schwann cell–like glia, although transcriptionally related to their peripheral counterpart, do indeed possess unique functional properties. Further, expression of A2B5 and O4 is significantly higher in Schwann cell–like glia than in peripheral Schwann cells. 104 Contrary, GFAP-expression is significantly higher in peripheral Schwann cells compared with Schwann cell-like glia. 104 It needs to be emphasized that most of the aforementioned data with regard to Schwann cell–like glia within the CNS are based on in vitro studies, thus not allowing a conclusive summary of the relatedness of peripheral Schwann cells and aldynoglia in vivo. Nevertheless, as myelination is the key functional hallmark of Schwann cells within the PNS, it appears not unlikely that CNS-endogenous Schwann cell–like glia may fulfill similar functional tasks. In fact, several recent lines of evidence indicate that Schwann cells might play a so far underestimated role in spontaneous CNS regeneration, which will be detailed within the next paragraph.
p75NTR: A Marker for Schwann Cells With Remyelinating Potential in the CNS?
Schwann Cell Remyelination: An Underestimated Regenerative Process in CNS Disease?
Remyelination is a process of regenerating the myelin sheath of demyelinated axons, thus restoring saltatory conduction. 95,172 Many experimental demyelination models show that remyelination is effectively achieved by oligodendrocyte progenitor cells (OPC), which differentiate into myelinating oligodendrocytes following demyelination within the CNS. 31,58,124 The new myelin sheath is thinner but still ensures functional recovery. 57 In MS, remyelinated regions are referred to as shadow plaques because of the paler staining of the thinner myelin sheath. 44,126 Remyelination in MS is insufficient, however, and oligodendrocytes fail to remyelinate naked axons. 44,57
Interestingly, remyelination in the CNS is additionally mediated by Schwann cells under certain conditions. Schwann cell remyelination is observed in several experimental animal models of demyelination, including injection of 6-aminonicotinamide or lysolecithin into rat spinal cord, Theiler’s murine encephalomyelitis in mice, chronic experimental allergic encephalomyelitis in guinea pigs, as well as in spinal cord lesions of humans affected by MS. 20,21,66,92,93,156,174 Moreover, Schwann cell remyelination also ensues following spinal cord injury in rodents as well as humans. 13,77,220 In addition, an aberrant proliferation of Schwann cells and nerve fibers, termed Schwannosis, has been reported in chronic human spinal cord injury. 27
As p75NTR is expressed in Schwann cells, except the myelinating stage, this marker could be useful in demonstrating Schwann cells associated with demyelinated lesions. 105,121,179 On the other hand, the presence of myelinating Schwann cells can be demonstrated using myelin protein zero (P0) and periaxin as markers. 10 5,121,135,179
Whether remyelination is initiated by either oligodendrocytes or Schwann cells significantly depends on the presence of activated astrocytes. 138 Remyelination by Schwann cells in the CNS occurs predominantly in regions deficient of astrocytes, while oligodendrocytes need the presence of astrocytes to remyelinate (Fig. 17). 19,21,56,58,59,171,186
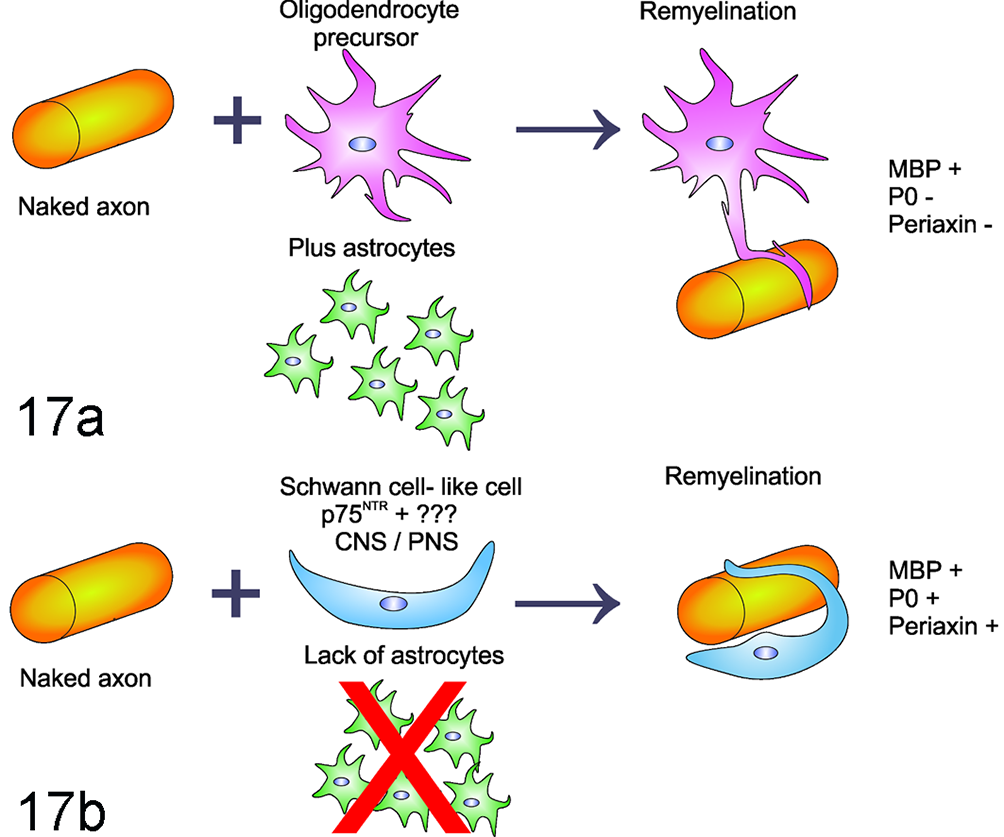
Proposed theory of oligodendrocyte versus Schwann cell remyelination. (a) In demyelinated lesions where astrocytes are present, oligodendrocyte progenitor cells mature into myelinating oligodendrocytes, which can remyelinate naked axons. Among other myelin components, the oligodendroglial myelin sheaths are positive for myelin basic protein (MBP). (b) Absence of astrocyte involvement favors Schwann cell–mediated remyelination. Schwann cell remyelination differs from oligodendrocyte remyelination in terms of antigen expression: myelin components produced by Schwann cells include myelin protein zero (P0) and to a lesser extent MBP. Further, myelinating Schwann cells express periaxin. CNS, central nervous system; PNS, peripheral nervous system.
In ethidium bromide–induced lesions of the rat spinal cord, the extent of Schwann cell remyelination remains similar at 4, 6.5, and 24 weeks post injectionem. 68 Therefore, Schwann cell remyelination seems to be a stable process that is not replaced by oligodendrocyte remyelination, 68 although replacement of transient Schwann cells by permanent oligodendrocyte remyelination has been demonstrated in one study. 94
Based on the observation of significant Schwann cell–mediated remyelination in the CNS, a logical aim is to identify the source of these cells. For years, the main thesis was that a significant number of peripheral Schwann cells (eg, derived from the spinal nerve root), infiltrate the lesion when the integrity of the astrocytic glia limitans is disrupted. 21,59,158 However, the ability of endogenous CNS neural and glial precursor cells to give rise to Schwann cells in vitro and after transplantation into the demyelinated spinal cord has recently challenged this hypothesis. 1,43,106,140 Further studies revealed that a CNS subpopulation of neural and glial precursor cells can be intrinsically programmed to differentiate into the Schwann cell lineage (Fig. 17). 106,140,216
Evidence for Spontaneous Emergence of p75NTR-Positive Glia With Schwann Cell Characteristics in Neuroinflammatory Conditions of the Dog
Canine Distemper Virus Infection
Previous studies have shown that both canine Schwann cells and OEC are susceptible to infection with canine distemper virus (CDV) in vitro. 189,196 In canine mixed brain cell cultures, Schwann cell–like glia are the first to be infected by CDV and also represent most infected cells in vitro. 151 In contrast, no CDV infection is detectable in these cells in vivo. 89 As mentioned above, spontaneously arising Schwann cell–like glia in organotypical slice cultures of the murine and canine CNS express p75NTR (Fig. 18). 89,177
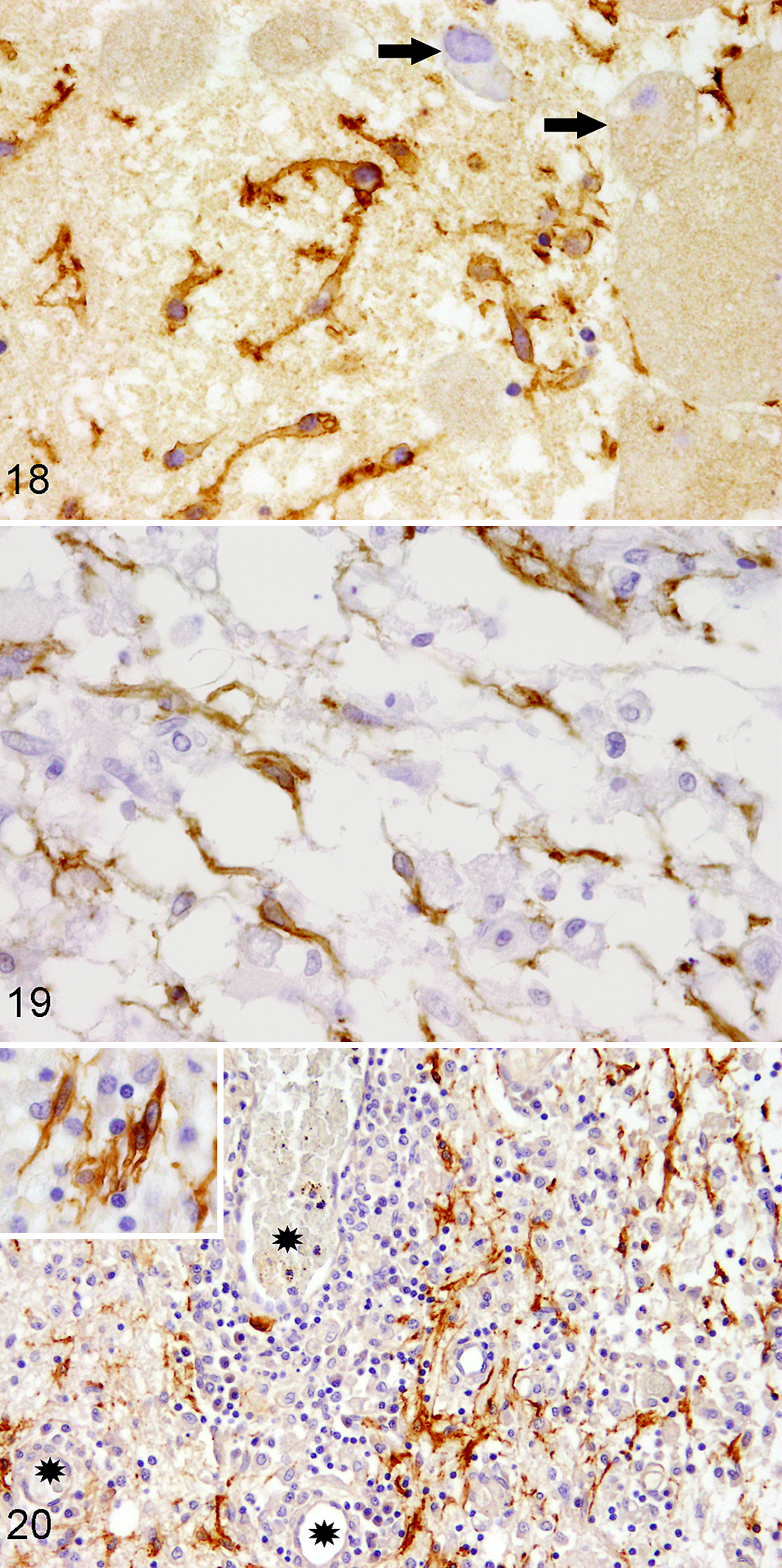
Nerve growth factor receptor p75 (p75NTR) expression in the lesioned central nervous system.
Intriguingly, spontaneous occurrence of bi- to multipolar, p75NTR-immunopositive Schwann cell–like glia are similarly demonstrated in the demyelinated white matter but also in early lesions preceding demyelination in dogs naturally infected with CDV (Fig. 19). 89,179,206 As the increase in p75NTR-expressing Schwann cell–like glia parallels the increase in axonal damage, the latter has been proposed to be a triggering mechanism for the generation of Schwann cell–like glia in the CNS. 89,121 However, in distemper leukoencephalitis, a very low proportion of lesions is positive for periaxin, a marker for myelinating Schwann cells. 179 In fact, only 2 out of 121 distemper leukoencephalitis lesions (1.65%) showed evidence of periaxin-positive myelinating Schwann cells, indicating that the vast majority of p75NTR-positive Schwann cell–like glia are arrested in a premyelinating stage. 179 In addition, no co-expression of p75NTR and sex determining region Y-box 2 (Sox2), a maker for immature, nonmyelinating and dedifferentiated Schwann cells, is present in distemper leukoencephalitis. 179
Canine Granulomatous Meningoencephalitis (GME) and Other Lympho-histiocytic Meningoencephalitides of Unknown Cause
In dogs with GME and other lympho-histiocytic meningoencephalitides of unknown cause, p75NTR-positive Schwann cell–like glia are observed in more than half of the cases, mainly in close vicinity to blood vessels and predominantly within the brain stem (Fig. 20). 105 Interestingly, the emergence of these cells in these primarily nondemyelinating canine encephalitides is not affected by the presence of astrocytes. 105 Contrary to distemper leukoencephalitis, periaxin-positive myelinating Schwann cells were present in 6% and 13% of lesioned areas of the cerebral and cerebellar white matter, respectively. 105 Even 37% of the brain stem lesions displayed periaxin positivity. 105
In GME and other lympho-histiocytic meningoencephalitides of unknown cause, p75NTR-positive Schwann cell–like cells co-express Sox2, which is found in immature, nonmyelinating and dedifferentiated Schwann cells, but not growth-associated protein 43, GFAP, and early growth response protein 2 (EGR2/KROX-20) as markers for different Schwann cells stages (see Fig. 14) or platelet-derived growth factor receptor alpha as a marker for OPC. 105 These results, together with the fact that co-localization of p75NTR and Sox2 is not observed in the control sciatic nerve but only in the injured peripheral nerve, might indicate that these Schwann cell–like glia are indeed dedifferentiated Schwann cells. 105 Interestingly, Sox2/p75NTR-double positive cells are strongly associated with periaxin-positive lesions, suggesting that they may differentiate into remyelinating Schwann cells. 105
Schwann cell remyelination in GME and other lympho-histiocytic meningoencephalitides of unknown cause might be carried out by dedifferentiated peripheral p75NTR-expressing Schwann cells invading the CNS, which redifferentiate within the lesions. 105 Moreover, a macrophage-dominated inflammatory milieu has been proposed to represent an adequate environment for Schwann cell plasticity within the injured CNS. 105
Taken together, p75NTR-positive cells seem to differ in their phenotype in distemper leukoencephalitis and GME/lympho-histiocytic meningoencephalitides of unknown cause. This could explain the variation in the amount of Schwann cell remyelination observed in the 2 disease entities. Moreover, regional differences might contribute to differences in effective Schwann cell remyelination in these disease entities. However, the exact reasons for this discrepancy remain unclear.
Concluding Remarks: p75NTR as a Cell Marker in the Normal and Diseased CNS
p75NTR is widely expressed during development in the CNS in both neurons and various glial cells. During adulthood, most of these cells down-regulate p75NTR expression, but it appears that these cells retain the ability to up-regulate p75NTR expression during neuropathological conditions. Yet its most interesting feature is that p75NTR serves as a marker for Schwann cells of peripheral and central origin and aldynoglia in lesions of the CNS. Hence, p75NTR is also of clinical significance, as these glial cell types are considered promising candidates with intrinsic regenerative properties and for transplantation into the lesioned CNS, respectively. Moreover, promotion of the spontaneous occurrence of p75NTR-positive cells in CNS pathologies might represent a target for future studies.
Supplemental Material
Supplemental Material, DS1_VET_10.1177_0300985818781930 - p75 Neurotrophin Receptor: A Double-Edged Sword in Pathology and Regeneration of the Central Nervous System
Supplemental Material, DS1_VET_10.1177_0300985818781930 for p75 Neurotrophin Receptor: A Double-Edged Sword in Pathology and Regeneration of the Central Nervous System by Kathrin Becker, Armend Cana, Wolfgang Baumgärtner, and Ingo Spitzbarth in Veterinary Pathology
Footnotes
Acknowledgements
We thank Dr I. Gerhauser, PhD, Annika Lehmbecker, PhD, and Yimin Wang, PhD, for providing unpublished photographs.
Authors Contribution
Kathrin Becker and Armend Cana contributed equally to this work and should be considered as co-first authors.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: A. Cana was supported by a scholarship from the European Union. This study was supported in part by the Niedersachsen-Research Network on Neuroinfectiology (N-RENNT) of the Ministry of Science and Culture of Lower Saxony, Germany.
Supplementary material for this article is available online.
Notes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
