Abstract
An 8-year-old, male neutered Irish Setter was presented with a 2-week history of pain upon opening the mouth and chronic otitis externa. Computed tomography examination revealed destruction of the left tympanic bulla with a soft-tissue density within the remains of the tympanic cavity. Cytology of aspirates collected from tissue adjacent to the tympanic bulla revealed suppurative inflammation and bacteria. A ventral bulla osteotomy was performed. Histopathology of granulation tissue within the remains of the tympanic cavity was diagnostic of cholesterol granuloma.
Cholesterol granuloma (CG) is a granulomatous lesion containing cholesterol crystals. 4 In humans, CG frequently develops in the middle ear or mastoid region. 3 In the middle ear, CG is usually associated with chronic otitis media; however, it may also be associated with adenomatous tumors and endolymphatic sac tumors in this location. 4, 13 Cholesterol granulomas have also been reported in the petrous apex, maxillary sinus, frontal sinus, orbit, breast, tunica vaginalis, peritoneum, brain, and kidney of humans. 4, 7, 9 In domestic animals, there are reports of CG in the brain of the horse, 8 the middle ear, the maxilla and brain of the dog, 2, 10, 12 and the uterus and brain of the cat. 5, 14 This report describes a canine case of CG that is associated with otitis media and destruction of the tympanic bulla.
An 8-year-old, 36.6-kg, male neutered Irish Setter was referred with a 2-week history of lethargy, anorexia, difficulty chewing, and pain upon opening the mouth. Additionally, there had been a 2-year history of intermittent otitis externa on the left. Physical examinations by the referring veterinarian in the 2 weeks prior to referral revealed pain and swelling in the left submandibular region, pain upon opening the mouth, and brown exudate in the left horizontal ear canal. A mixed population of bacterial cocci and rods were present on cytologic examination of the exudate. Treatment with oral meloxicam and cephalexin resulted in some clinical improvement, and the dog was referred for further evaluation.
On examination at the University of Melbourne Veterinary Clinic and Hospital, there was discomfort upon opening the mouth; however, pain, swelling, atrophy, and asymmetry were not detected upon palpation of the head and masticatory muscles. The left horizontal external ear canal was narrowed and inflamed; a moderate volume of brown exudate in the canal prevented visualization of the tympanic membrane. The rectal temperature was normal, and the remainder of the physical examination was unremarkable. At this time, differential diagnoses for the pain upon opening the mouth included temporomandibular joint disease, masticatory myositis, otitis media, migrating foreign body, and retrobulbar disease.
Results of a preanesthetic blood analysis were normal, and the dog was anesthetized for further diagnostic tests. Electromyography was performed on the temporalis and masseter muscles; an absence of spontaneous electrical activity excluded masticatory myositis.
A computed tomographic examination of the cranium showed destruction of the left tympanic bulla and replacement by material that was isodense to the brain; the material within the remains of the bulla did not contrast enhance with intravenous iohexol administration (Omnipaque, Amersham Health, North Ryde, N.S.W., Australia). There was swelling and contrast enhancement of the soft tissues immediately lateral and ventral to the remains of the bulla.
Ultrasound-guided, percutaneous, fine-needle aspirates were collected from the tissue adjacent to the remains of the left tympanic bulla. On cytology, large numbers of nucleated cells were present, with a prominent background precipitate of protein and red blood cells; the nucleated cells comprised approximately 95% degenerate and nondegenerate neutrophils and 5% activated macrophages. There were large numbers of intracellular and extracellular cocci, occasional fibroblasts, and occasional sheets of atypical epithelial cells that exhibited some anisocytosis and anisokaryosis. The origin of the epithelial cells was unclear from the cytologic examination, and individual cell features included round or ovoid nuclei, clumped nuclear chromatin, prominent nucleoli, and a variable volume of basophilic cytoplasm; occasional binucleate epithelial cells were also present. The cytology was consistent with suppurative inflammation and epithelial cell dysplasia. It was thought that the epithelial cells probably originated from the middle ear mucosa; the possibility of neoplasia of the middle ear was not excluded at this time.
The following day, a ventral bulla osteotomy was performed. At surgery, exudate and granulation tissues were visualized in the remains of the left tympanic cavity; there was no mass or discrete granuloma. Remaining fragments of tympanic bulla were removed, 5 fragments of granulation tissue, up to 6 mm in width, were excised and submitted for histopathology and bacterial culture, and the region was lavaged with saline. Surgical excision of the granulation tissue appeared to be complete. A temporary surgical drain was placed, and the tissues were closed routinely. Postoperatively, the dog was treated with subcutaneous amoxicillin-clavulanic acid and meloxicam; oral amoxicillin-clavulanic acid administration was continued for 4 weeks. A coagulase-negative Staphylococcus species that was sensitive to amoxicillin-clavulanic acid was cultured from the tissue samples.
Biopsy samples were fixed in 10% buffered formalin, routinely processed, embedded in paraffin, sectioned at 5 μm, and stained with HE. On microscopy, the tissue was predominantly of dense, hyaline collagen with intersecting vascularized bands. Irregular areas of ossification of connective tissue showed some remodeling to osteonal bone. Polyps were present that were partially covered by flattened, simple squamous epithelia (Fig. 1). There were numerous acicular clefts, typical of cholesterol crystals, surrounded by fibrous tissue and infiltrates of mononuclear inflammatory cells (Fig. 2). The inflammatory cells were mostly macrophages, some of which had pigmented cytoplasm. Lower numbers of lymphocytes and plasma cells and occasional neutrophils were also present. There was no evidence of neoplasia. The histopathology was diagnostic of CG. 4
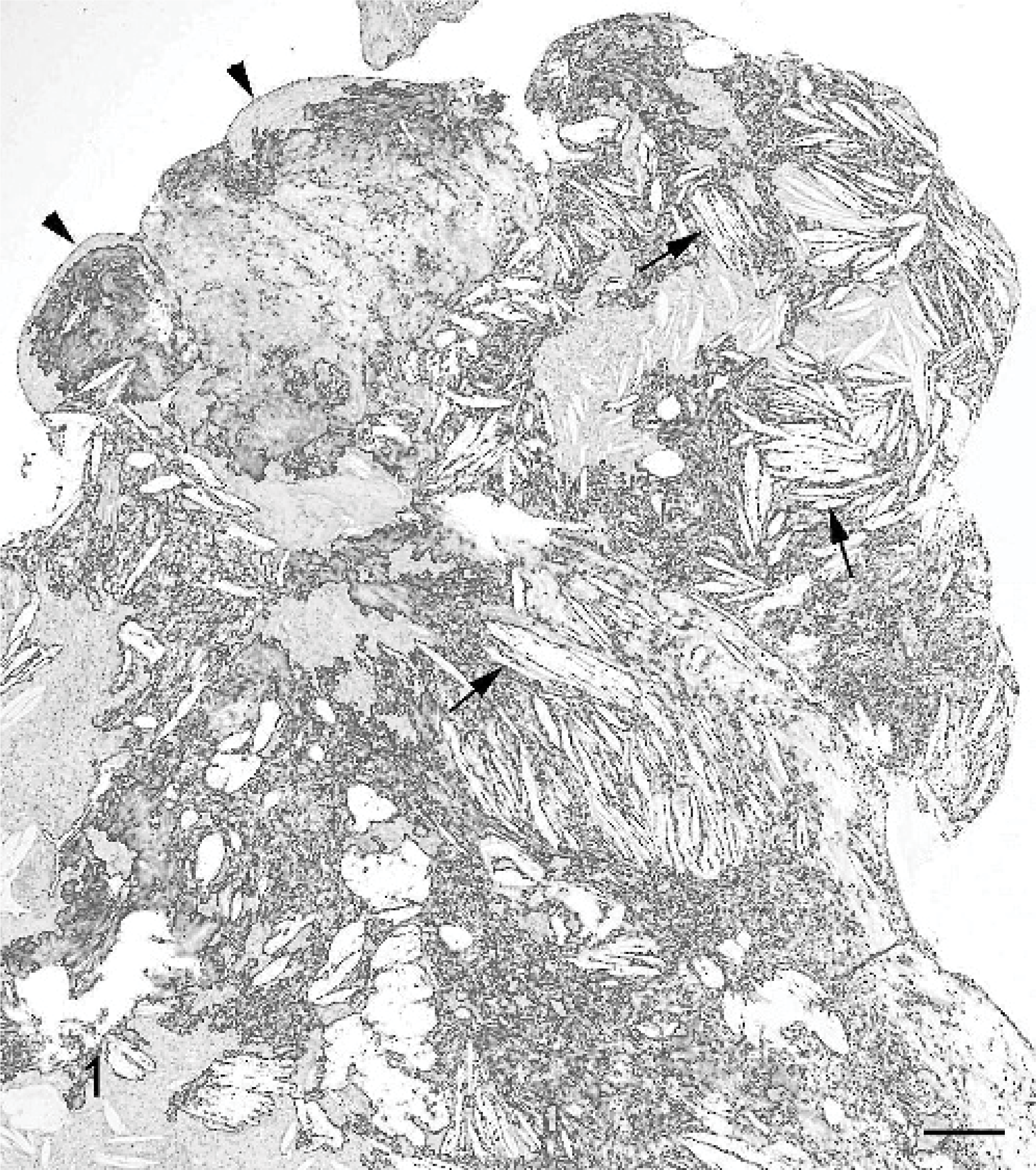
Middle ear cholesterol granuloma; dog. Note the multiple acicular clefts that are typical of cholesterol crystals (arrows) and the formation of a polyp covered by a flattened simple squamous epithelium (arrow heads). HE. Bar = 200 μm.
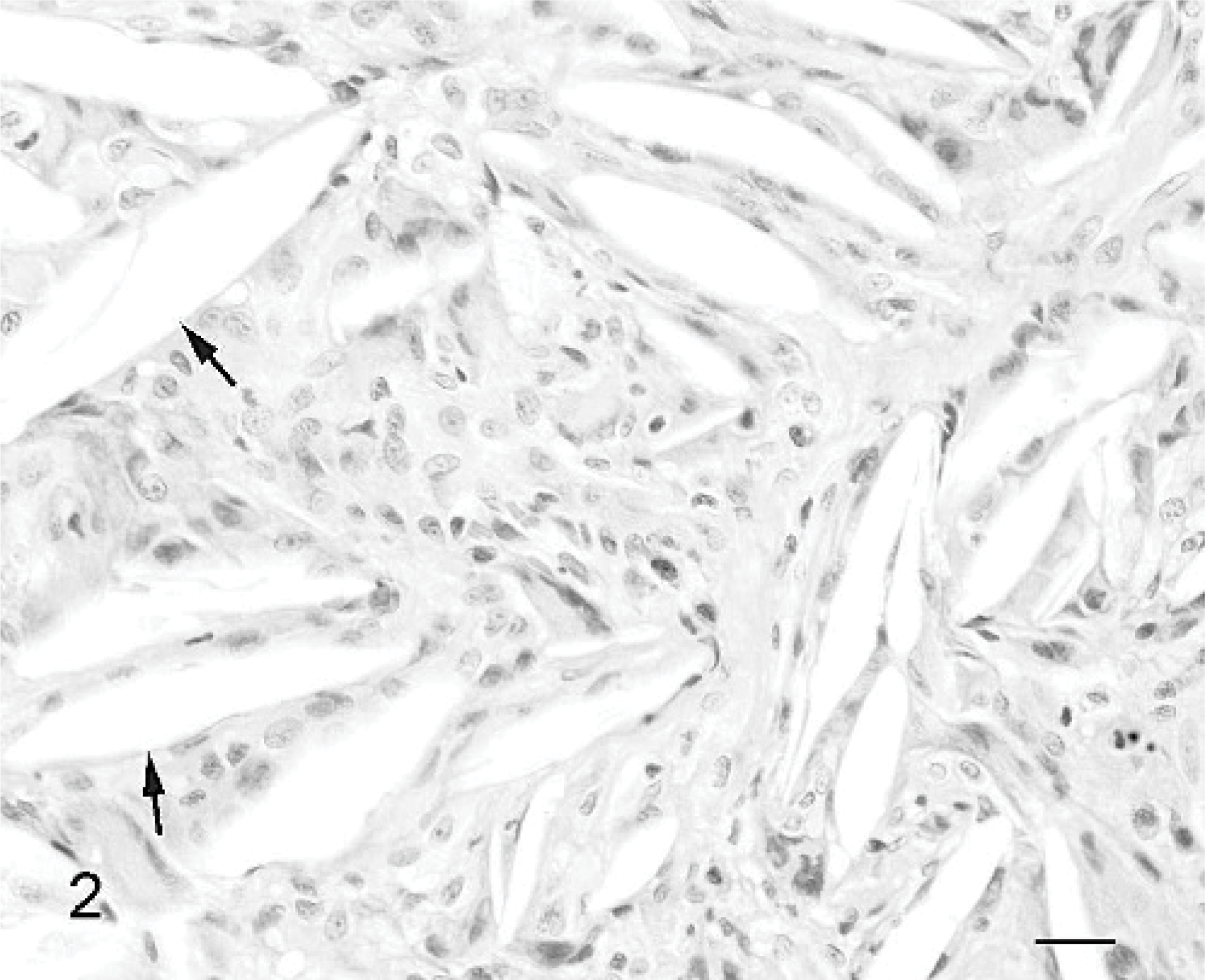
Middle ear cholesterol granuloma; dog. Note the acicular (cholesterol) clefts (arrows) surrounded by mononuclear inflammatory cells. HE. Bar = 20 μm.
The dog's clinical signs resolved with therapy and, at the time of this writing, 12 months after initial presentation, the dog was reported to be normal.
Cholesterol granulomas are benign expansive lesions that, in humans, are most commonly found in the middle ear or mastoid region. 3 The necessary factors for the development of CG in the middle ear and other normally aerated bony cavities are thought to be hemorrhage, interference with drainage, and obstruction of ventilation. 3, 4, 13 Possible causes of the initial hemorrhage include mucosal inflammation, trauma, and pressure changes within the air cells. 3, 7, 13 Cholesterol, originating from erythrocyte membranes and possibly mucosa or transudate, precipitates forming crystals which, stimulate a granulomatous reaction and the development of CG. 3, 11
Ventilatory obstruction is postulated to have a significant role in the pathogenesis of CG in some cases. Mucosal swelling or other lesions initially obstruct the pneumatic pathways to the air cells. Trapped gas resorption then results in negative pressure within the air cells, which is thought to cause transudative mucosal hemorrhage. 3 This theory has been supported by experiments in the squirrel monkey, in which obstruction of the pharyngeal orifice of the eustachian tube caused the development and continued growth of CG in the mastoid region. 6 The pathogenesis of petrous apex CG may differ from that of middle ear or mastoid CG. Based on the evaluation of high-resolution computed tomographic images in humans, it is hypothesised that a developmental deficiency of the cortical bone that normally separates the air cell system from the marrow compartment predisposes to the development of CG in the petrous apex. 7 Hemorrhage from the highly vascular exposed marrow is able to coagulate within the mucosal air cell lining and occlude outflow pathways leading to CG development. 7
In the early stages of development, the lesions of CG may only have the gross appearance of mucosal thickening. 3 The mature granulomas are often brown or yellow and may be round, ovoid, or irregular; in advanced cases the granulomas can cause osteitis and erosion of adjacent bone. 3, 4, 13 The histologic appearance of CG is diagnostic. Microscopically, the lesions are composed of a large number of acicular clefts, typical of cholesterol crystals, surrounded and engulfed by multinucleated giant cells and embedded in fibrous granulation tissue. 4 Additional findings may include areas of old and recent hemorrhage with numerous macrophages containing hemosiderin. 4
The destruction of the tympanic bulla, evident on computed tomography, initially raised the suspicion of neoplasia in this case. Lysis of the tympanic bulla is commonly associated with neoplasia of the middle ear in the dog; however, it may occur in severe cases of otitis media. 1 In humans erosion of adjacent ossicles and bone may be seen on computed tomography in advanced cases of middle ear CG. 4 The previously reported canine case of middle ear CG was an Irish Setter without evidence of concurrent bacterial otitis or lysis of the tympanic bulla. 2
In conclusion, CG can occur in conjunction with otitis media in the dog and may be associated with lysis of the tympanic bulla.
