Abstract
The purpose of this study was to establish microscopic normal in the middle ear of the cat while concurrently characterizing gross and microscopic lesions reflecting spontaneous otitis media. Both ears from 50 cats were examined grossly and processed for histologic examination of the external, middle, and internal ear on a single slide. Gross lesions of the middle ear were present in 14 of 100 (14%) and included turbid fluid, frank pus, hemorrhage, and fibrous thickening of the auricular mucoperiosteum. Histologically, 48 of 100 (48%) ears had evidence of ongoing or previous inflammatory middle ear disease, including proteinaceous fluid; vascular ectasia; expansion of the auricular mucoperiosteum by neutrophils, lymphocytes, and macrophages; cholesterol clefts; hemorrhage; fibrin; granulation tissue; membranous pseudo-glands; fibrosis; proliferation and/or osteolysis of the tympanic and septum bullae. Histologic lesions were identified in 34 of 100 ears (34%) lacking gross evidence of disease. Ears were classified histologically as either normal (52/100 [52%]) or diseased (48/100 [48%]). Diseased ears were further classified as mild to moderate (37/100 [37%]) or severely (11/100 [11%]) affected. Internal ear involvement was present in 11 of 100 (11%) ears. Histologic evidence of middle ear disease in cats is far greater than gross lesions or clinical literature suggests; further investigation and correlation of clinical and histologic disease are warranted. With minimal additional preparation, diagnostic specimens may be readily prepared and evaluated for this integral sensing organ.
Ear disease is a common presenting complaint in veterinary medicine. Many of the perpetuating factors of otitis externa, such as bacterial and yeast organisms, conformation, or environmental complications, more commonly affect dogs, 5 and as a result, publications documenting microbial and histologic features of otitis externa and otitis media in dogs are more common. Similar accounts in cats are rare, with few clearly documenting microbial or histologic features of naturally occurring otitis media. 6,18
In 1957, Lawson 6 first described otitis media in the cat as a specific clinical entity, characterized as an inflammatory middle ear disease typically associated with clinical neurologic disturbances, including hind limb gait abnormalities, head tilt, and inability to maintain balance. Gross or radiographic evidence of purulent material and bony changes to the tympanic bulla were confirmed in many but not all clinically affected cats. To further determine the general frequency of middle ear disease, Lawson completed a survey of 66 stray cats and found that 6 of 66 (9.1%) had gross evidence of middle ear disease, including variably inspissated purulent to bloody effusion and/or frank hemorrhage. No further microscopic characterization was completed. In contrast to Lawson’s study, in 2009, Schlicksup et al 18 reported that 59 of 3442 (1.7%) necropsied cats had gross evidence of nonneoplastic ear disease. Although their conclusion was that nonneoplastic disease of the feline middle ears occurs relatively rarely, this study excluded all cats with medical records that lacked clinical information related to the aural, respiratory, or nervous systems, and histologic examination was limited to a few cases. The wide discrepancy in the resulting reported disease prevalences could be explained by (1) a fundamental difference in the cat populations examined, (2) modern diagnostic techniques and therapeutics that have dramatically reduced the number of cats that present for necropsy with otitis media, or (3) inherent limitations in one or both of these previous study methods.
Investigation of middle ear disease in domestic animals is hindered by the seemingly protracted and involved specimen preparation, requirements associated with difficult tissues (bony and membranous interfaces), and an apparent knowledge void in the veterinary literature. A lack of investigation into these structures is at least partially responsible for the general acceptance that middle ear disease is uncommon in the cat. In addition, if determining prevalence of disease is reliant solely on clinical expression of disease, it is likely otitis media is significantly underestimated since only 10% of cats with gross evidence of middle ear disease may have specific clinical signs. 18
The purpose of this study is to establish the spectrum of histologic normal within the feline middle ear, to describe the characteristics of naturally occurring gross and histologic inflammatory lesions of the feline middle ear, and to report a newly established, step-by-step tissue technique to allow for efficient and rapid collection and processing resulting in consistent, repeatable results for routine use in diagnostic practice.
Materials and Methods
Animals
Fifty cats accessioned through the Oklahoma Animal Disease Diagnostic Laboratory, Stillwater, Oklahoma, between December 2010 and September 2011 were included. Six submittals during this time period included only heads: 3 for rabies examination and 3 from stray animals with limited or no accompanying history.
Gross Examination
After completion of routine necropsy, the head was routinely disarticulated from the carcass at the atlanto-occipital joint. Bone cutters were used to remove the calvaria and the brain removed using blunt dissection. From the ventral aspect, the musculature overlying the ventral surface of the tympanic bullae was bluntly dissected to expose the bone. Using bone cutters, the ventral floor of the tympanic bulla was removed bilaterally, revealing the auricular mucoperiosteum lining the tympanic bulla. The auricular mucoperiosteum was evaluated for gross changes, including increased thickness, cloudiness, or hemorrhage, and then perforated to allow for gross examination of the tympanic cavity.
Tissue Processing
To facilitate more rapid processing, a section of skull containing the left and right petrous portions of the temporal bones was isolated. Detailed procedures for dissecting the skull for ear examination have been previously described. 12 In brief, with the brain removed, a cut was extended through the floor of the skull just caudal to the optic nerves and extending bilaterally through the lateral most edges of the zygomatic arches (Fig. 1). Alternatively, after removing the brain, bone cutters were used to make bilateral cuts through the temporomandibular joints, lateral to the tympanic bullae and external ear canal, and the floor of the skull was fractured just caudal to the optic nerves. Either method produced a single piece of ventral skull containing portions of the left and right squamous and petrous portions of the temporal bone. With the ventral bullae previously opened to facilitate examination and fixation, the bone was placed in 10% buffered formalin for a minimum of 48 hours. Following fixation, ears were decalcified for 7 to 14 days by immersion in formic acid decalcifying solution (Surgipath decalcifier I; Leica Biosystems Richmond, Richmond, IL).
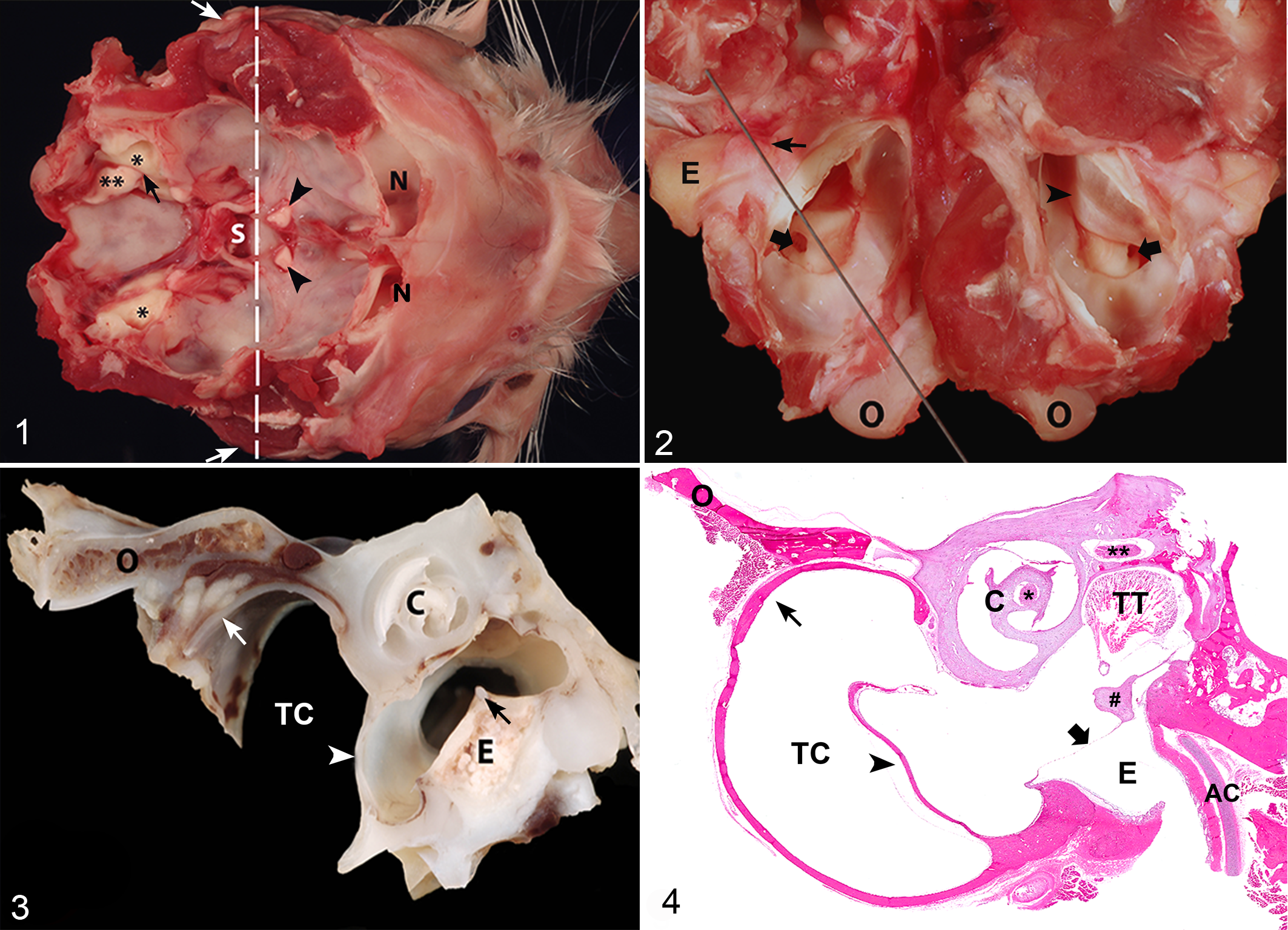
Following fixation and decalcification, ears were trimmed to facilitate examination of the external, middle, and internal ear in a single tissue section. Using the opening in the ventral bulla, a sharp microtome blade was positioned at the junction of proximal and middle third of the occipital condyle, extending a millimeter or two medial to the round window and finishing midway through the bony opening of the external acoustic meatus (Fig. 2). A second cut, parallel and 2- to 3 mm medial to the first, resulted in a section (Fig. 3) appropriate for further processing. After trimming, tissues were dehydrated, embedded in paraffin wax, cut in 4-μm sections, and stained with hematoxylin and eosin (HE). HE was applied per standard operating procedures with the exception of doubling the exposure time to Harris hematoxylin.
Semiquantitative Scoring of Ear Lesions
The following structures of the ear were thoroughly examined histologically: temporal bone, including petrous and squamous portions, tympanic membrane, tympanic cavity, malleus, incus, stapes, tensor tympani or stapedius muscles, facial (CN VII) and vestibulocochlear (CN VIII) nerves, and the auricular mucoperiosteum (Fig. 4).
Descriptive analysis of ear lesions led to the identification of 4 major categories of microscopic changes: (1) vascular, including hemorrhage and edema; (2) inflammatory infiltrates, particularly neutrophils, macrophages, lymphocytes, plasma cells, and fibrin; (3) thickening/polypoid proliferation of membranous structures, including formation of pseudo-glandular structures; and (4) thickening, irregular erosion, and resorption of bone. Structures of the external ear and internal ear were also examined.
Quantitative Scoring of Ear Lesions
The thickness of bony segments of the tympanic bulla and septum bulla, as well as associated overlying auricular mucoperiosteum, was measured using an in-eyepiece micrometer at 200× magnification.
Statistical Analysis
All statistical analyses were conducted with SAS version 9.2 (SAS Institute, Cary, NC). The effects of sex and disease severity on the width of the bulla bone were investigated with analysis of variance methods. The comparisons of categorical variables, such as disease severity (high vs low) and respiratory ailment (yes vs no), were made with the use of contingency tables and the Fisher exact test. Statistical significance was set at the .05 level.
Results
During the study period, cats ranging from <1 week to >17 years of age were submitted for rabies or necropsy examination. There were 13 castrated males, 11 spayed females, 16 sexually intact males, 8 sexually intact females, and 2 unspecified. Forty-three cats were domestic shorthairs, 2 were domestic longhairs, 1 was not specified, and there was 1 each of the following breeds: Bengal, Norwegian Forest Cat, Siamese, and Devon Rex.
Clinical signs associated with upper respiratory tract disease were cited as the cause of euthanasia in 4 cats. An additional 6 cats euthanized due to poor prognosis or insufficient funds presented with a variety of clinical signs attributable to upper respiratory tract disease, including “difficulty breathing” associated with markedly increased mucus production, labored breathing, extension of the head and neck, increased respiratory rate and effort, dyspnea, lethargy, anorexia, serous ocular discharge, wheezing, and purulent discharge from eyes and nares. In total, 4 cats were euthanized due to clinical signs associated with upper respiratory tract disease, 35 cats were euthanized because of poor prognosis or insufficient funds, 3 were euthanized as rabies suspects, 6 died, and mode of death was not recorded in 2 cases.
Necropsy diagnoses included trauma (13 cats), cytauxzoonosis (7 cats), neoplasia (2 lymphosarcoma, 2 meningioma, 1 pancreatic ductular carcinoma, 1 biliary carcinoma, 1 undetermined, and 1 both meningioma and biliary adenocarcinoma), pneumonia and sepsis (3 cats), for rabies diagnostics (3 cats), chronic renal disease (2 cats), urinary obstruction (2 cats), and 1 each for upper respiratory tract infection, feline leukemia virus (FeLV)–associated disease, feline panleukopenia, ethylene glycol toxicosis, inanition, feline infectious peritonitis (FIP), hypertrophic cardiomyopathy (HCM), diabetes mellitus, bacterial peritonitis, and anesthesia-associated death. Necropsy diagnoses were undetermined in 3 cases.
Gross Examination
Of the 100 total ears examined, 14 of 100 (14%) ears were grossly affected, representing 8 cats with 1 (2 cats) or both (6 cats) ears affected. Fluid or suppurative material was identified in the tympanic bullae in 7 of these 8 cats. Bilateral (3 cats) or unilateral (1 cat) suppurative otitis media was most common and characterized by abundant, white-yellow to slightly green, variably mucoid, soft exudate (Fig. 5). In the lone cat with unilateral suppurative exudation, the other ear contained small amounts of clear to cloudy fluid. Bilateral (3 cats) or unilateral (1 cat) hemorrhagic middle ear disease occurred in 4 cats and was variably characterized by mild petechial hemorrhage, red-tinged suppurative exudate, or frank blood. Five of 100 (5%) ears were grossly classified as having chronic disease typified by bilateral lysis of the septum bulla (2 cats), bilateral (1 cat) or unilateral (1 cat) tympanic bulla mucoperiosteal thickening, or unilateral thickening of the ventral bulla (1 cat).
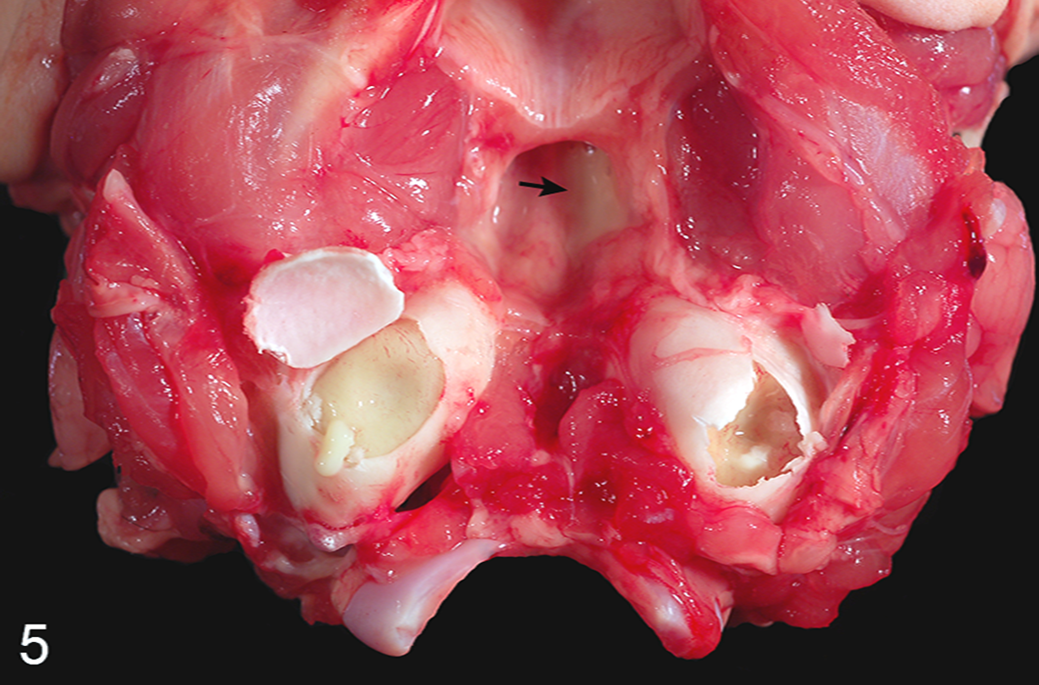
Fresh skull, ventral aspect; cat. The ventral bone of the tympanic bullae has been opened bilaterally. The tympanic cavities are filled with soft, yellow-tan exudate. Similar exudate is present in the nasopharynx (arrow) due to extension through the left auditory (eustachian) tube.
Microscopic Examination
Histologic Characteristics of Normal Ears
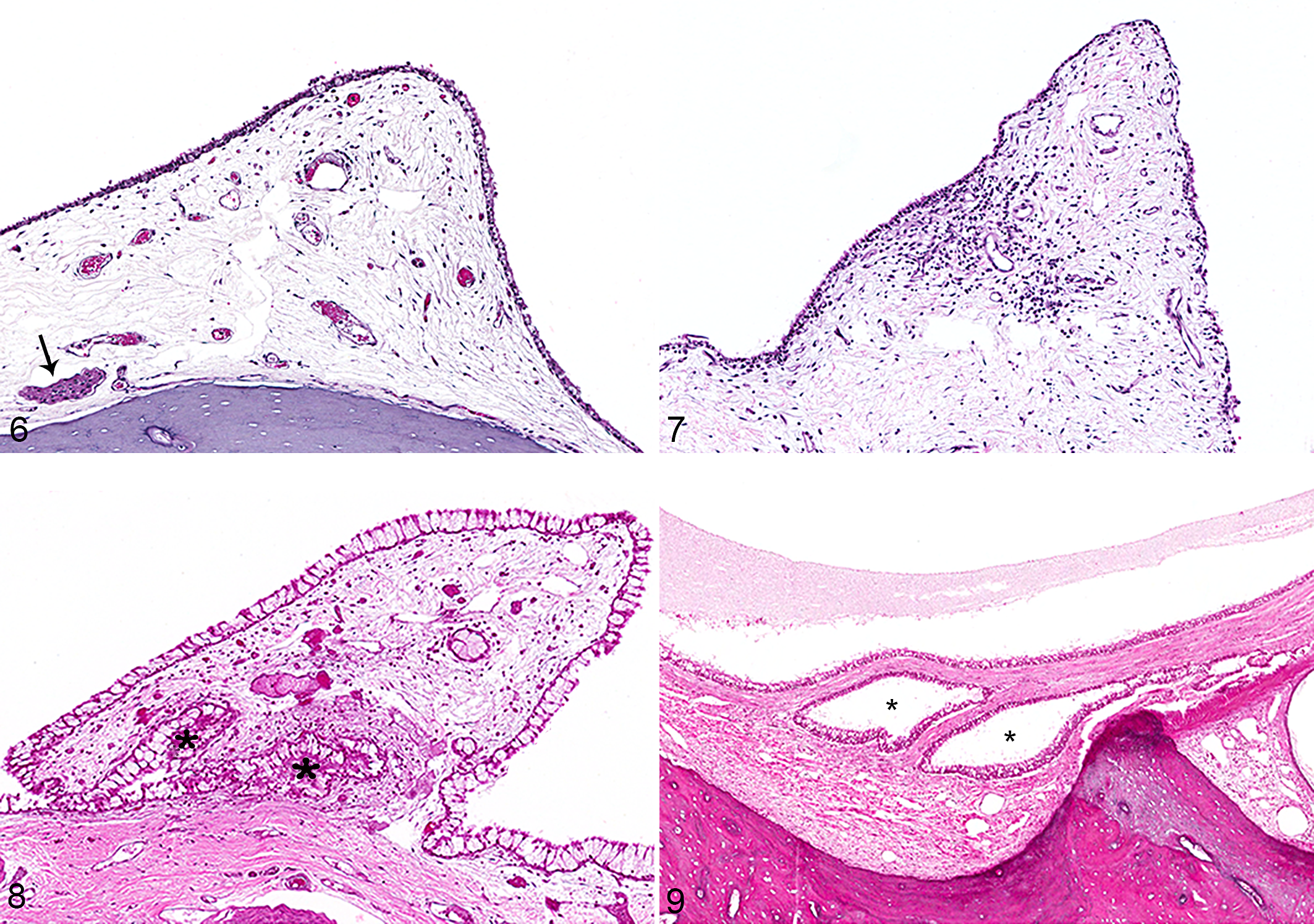
Changes were considered within the limits of normal if they were limited to vascular changes of edema within the auricular mucoperiosteum with or without mild transudation into the tympanic cavity. Of the 100 total ears examined, 52 of 100 (52%) ears were histologically normal, representing 29 cats with 1 (6 cats) or both (23 cats) ears normal. Within histologically normal ears, the most common findings included minimal expansion of auricular mucoperiosteum by clear edema fluid and mildly ectatic vessels or lymphatics in 15 of 52 (28.8%), minimal proteinaceous homogeneous to flocculent material within the tympanic cavity in 15 of 52 (28%), and minimal infiltration by 25 or fewer lymphocytes per 400× field within the auricular mucoperiosteum overlying the petrous portion of the temporal bone in 11 of 52 (21.1%) ears (Fig. 6). Lifting of the auricular mucoperiosteum from bony surfaces was quite common and considered a trimming artifact.
When only normal ears were compared, the width of the tympanic bullae bone of cats did not correlate to sexual status (mean [SE]; male, 298.8 [26.2] μm; female, 248.5 [19.7] μm; P = .13), although male cats did trend toward thicker bullae.
Histologic Characteristics of Diseased Ears
Mild to moderately affected ears
Mildly to moderately affected ears exhibited 2 or more categories of change, most commonly vascular as well as inflammatory changes. Thirty-seven of the 100 ears examined (37%) were considered histologically mildly to moderately affected. A total of 22 cats had 1 (7 cats) or both (15 cats) ears affected. Ears were characterized by the presence of variable vascular changes, including mild to moderate proteinaceous fluid within the tympanic cavity, mild vascular and lymphatic ectasia, and expansion of auricular mucoperiosteum by fluid or hemorrhage. Other changes included infiltration of auricular mucoperiosteum by more than 25 inflammatory cells per 400× field (Fig. 7), polypoid membranous proliferation and goblet cell metaplasia (Fig. 8), formations of epithelial pseudo-glands (Fig. 9), lymphoid follicular development, and fibrosis of the auricular mucoperiosteum. In 3 of 100 examined ears (3%), perineuritis of the facial nerve was present. Facial neuritis was not identified in any ear.
Severely affected ears
Severely affected ears had active inflammatory infiltrates accompanied by proliferation of the auricular mucoperiosteum and/or evidence of current or previous bony proliferation, lysis, or remodeling. Eleven of the 100 ears examined (11%) were considered histologically severely affected. A total of 6 cats had 1 (1 cat) or both (5 cats) ears affected. Severely affected ears were characterized by filling and often obliteration of the tympanic cavity by inflammatory exudate (10/11 [90.1%]), consisting predominantly of degenerate neutrophils (11/11 [100%]) with variable numbers of macrophages (7/11 [63.6%]). Inflammation was largely confined to the dorsolateral compartment in 3 of 11 (27.3%) ears. Bony lysis of the septum bulla (3/11 [27.3%]) and irregular lysis (8/11 [72.7%]) or proliferation (5/11 [45.5%]) of auditory ossicles or bone of the tympanic bulla was also observed. Membranous changes were typically associated with marked inflammation and included expansion of the auricular mucoperiosteum by granulation tissue (10/11 [90.1%]), hemorrhage frequently associated with cholesterol cleft formation (5/11 [45.5%]), formation of pseudo-glandular structures (9/11 [81.8%]), and erosion (8/11 [72.7%]) or effacement (6/11 [54.5%]) of the auricular mucoperiosteum epithelium. In severely affected ears, the tympanic membrane was typically involved (10/11 [90.1%]), with frequent extension into the muscle and tendon of the tensor tympani (5/11 [45.5%]). Rupture of the tympanic membrane was not observed.
The normal tympanic bulla is uniform in width (mean [SE], 279.0 [17.9] μm), without a marrow cavity. A significant difference in bulla width was noted between animals with no to moderate disease (mean [SE], 288.2 [15.6] μm) and those with severe disease (mean [SE], 398.2 [52.1] μm; P = .02). The occurrence of severe disease in the ears associated with cats presenting with signs of respiratory disease was 7 of 22 (31.8%), while only 4 of 78 (5.2%) ears of cats without clinical respiratory signs had severe disease, a difference that is statistically significant (P < .01). Conversely, the relationship of a neurological disorder did not correlate with severe ear disease. Severe disease was present in 2 of 18 (11.1%) ears in cats presenting with neurologic signs, while 9 of 82 (11.0%) ears of cats without a neurological disorder exhibited severe disease (P = 1.0). When combining respiratory and neurological signs (having either), 9 of 40 (22.5%) ears had severe disease, while only 2 of 60 (3.3%) ears with neither respiratory nor neurologic signs had severe disease (P < .01).
Histologic Characteristics of Chronic Otitis Media
Chronically affected ears had 1 or more of the following: active inflammatory infiltrates composed predominantly of macrophages or lymphocytes, lymphoid nodules, regionally extensive fibrosis of the auricular mucoperiosteum, and/or evidence of current or previous bony proliferation, lysis, or remodeling. Twenty-two of the 100 ears examined (22%) were classified as chronically affected. A total of 14 cats had 1 (6 cats) or both (8 cats) ears affected. Ten of the 22 chronically affected ears (45.5%) were also severely affected. The remaining 12 chronically affected ears (54.5%) were considered mildly to moderately affected as characterized by resolution of active inflammation and the presence of lymphoid follicles embedded within dense fibrosis of the auricular mucoperiosteum (1 ear) or mild residual mononuclear inflammation and fibrosis of the auricular mucopersioteum.
Histologic Characteristics of Internal Ear Involvement
Twenty-four of 100 ears (24%) had histologic changes in the internal ear. Six of the 24 ears (25%) were considered histologically normal, and internal ear changes were limited to minimal to mild eosinophilic finely flocculent material. Eleven of 24 ears (46%) were considered histologically mildly to moderately affected, and internal ear findings included eosinophilic fluid in 8 of 11 (73%) and minimal to mild hemorrhage in 9 of 11 (82%). Seven of 24 ears (29%) were histologically severely affected, and internal ear changes included eosinophilic fluid in 7 of 7 ears (100%) and infiltration of inflammatory cells in 6 of 7 (86%). Inflammatory cells were limited to the cochlea (2 ears), the vestibulocochlear nerve (2 ears), or both locations (2 ears). Internal ear changes were considered minimal to mild for all ears except for 2 in which there was marked infiltrates of macrophages into the vestibulocochlear nerve. Vestibulochoclear neuritis was present in 4 of 100 (4%) ears.
The presence of internal ear findings was significantly correlated with a grade of severe otitis media, present in 7 of 24 (30%) severely affected ears, while only 4 of 77 (5.2%) ears without internal ear changes were severely affected (P < .01).
Discussion
Understanding and characterizing pathologic changes in the middle ear requires a firm foundation in the normal gross and histologic anatomy. The external ear is composed of the auricle (pinna) and external acoustic meatus (ear canal), which terminates medially at the tympanic membrane. The middle ear is composed of the tympanic cavity, an air-filled space located medial to the tympanic membrane. In the cat, this cavity is nearly completely separated into dorsolateral (epitympanic) and ventromedial compartments by the septum bulla, a thin shelf of bone that extends from the lateral aspect of the tympanic bulla medially to appose the petrous portion of the temporal bone. A narrow slit between this bony septum and the petrous portion of the temporal bone allows communication between the 2 compartments. In the dog, the septum bulla forms an irregular ridge of sharp bony spicules with incomplete delineation of the 2 compartments. This anatomic variation is important, since otic pathology may be entirely limited to the dorsolateral compartment in the cat. In the present study population, significant inflammatory changes were limited to the dorsolateral compartment in 3 of 100 (3%) ears.
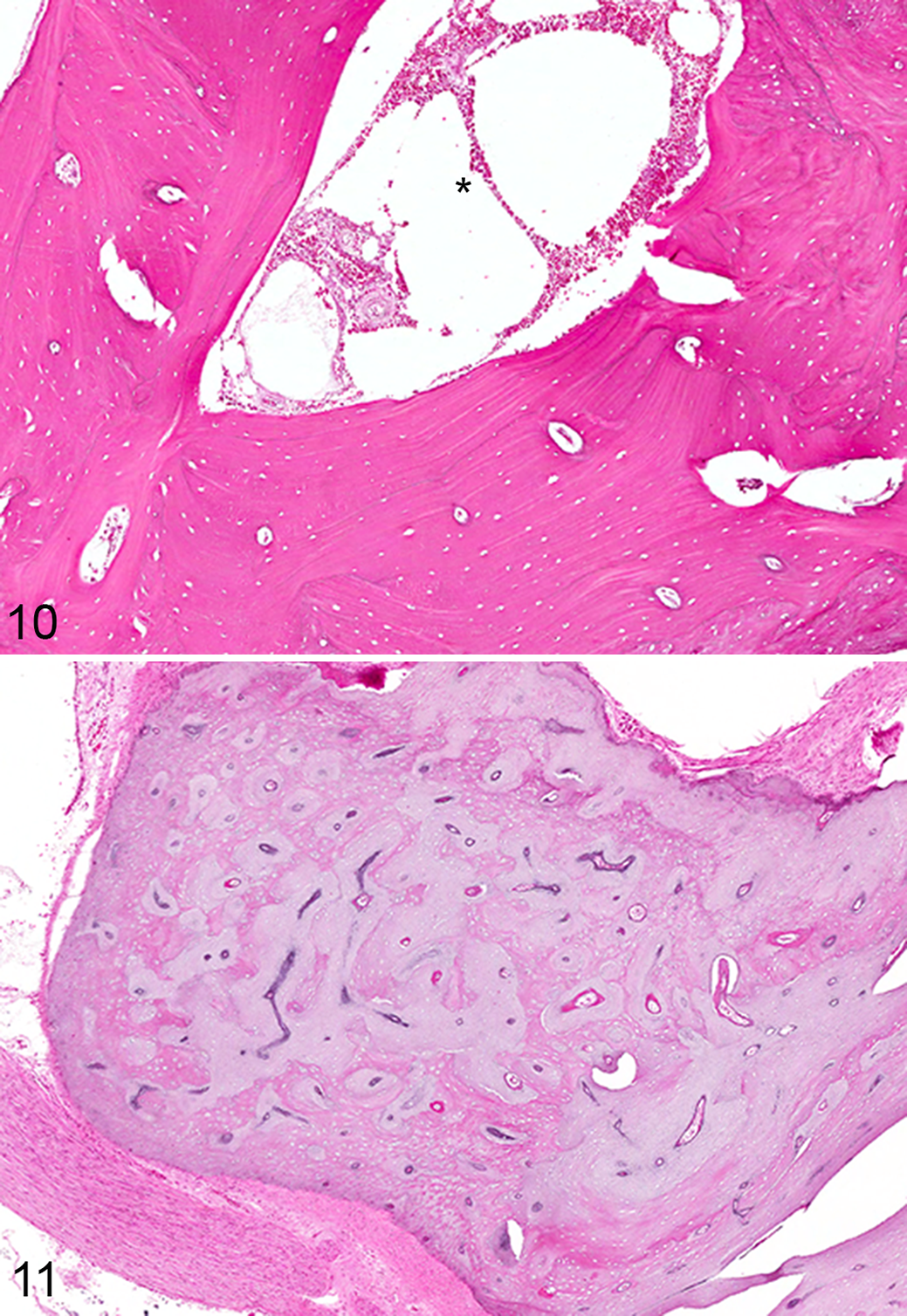
The tympanic cavity is nearly circumferentially encased in a bony shell composed dorsally of the squamous portion of the temporal bone, medially by the petrous portion of the temporal bone, and ventrolaterally by the tympanic bulla. Regionally, the bones encasing the tympanic bulla are developmentally distinct. The squamous portion of the temporal bone is lamellar to osteonal bone with a prominent marrow cavity (Fig. 10), while those of the tympanic bulla and bulla septum are similar but without a prominent marrow cavity, unless markedly thickened. The petrous temporal bone, grossly distinguished by its distinctly yellow hue, is histologically slightly pale and basophilic, reflecting its composition as calcified cartilaginous matrix interwoven with lesser lamellar bone (Fig. 11). 20 The petrous temporal bone lacks a marrow cavity. The 3 auditory ossicles—the malleus, incus, and stapes—are of similar composition to the petrous portion of the temporal bone.
Forming the lateral most border of the middle ear, the tympanic membrane (Fig. 12) is grossly a thin, semitranslucent, gray-white membrane spanning the width of the fibrocartilaginous rings forming the entrance to the external ear canal. Other than pathologic rupture, gross changes are not easily identified and are frequently overlooked. Microscopically, the tympanic membrane is composed of 3 layers: an outer layer of keratinizing squamous epithelium continuous with the keratinizing squamous epithelium lining the external ear canal; a middle, extremely thin, variably vascularized layer of fibrous connective tissue; and an inner, low cuboidal to nonkeratinizing squamous epithelium. 13 Microscopically, these layers are easily identified within the dorsal pars flaccida (Fig. 13) but much less distinct in the ventral pars tensa, which comprised most of the tympanic membrane in routine sections (Fig. 14).
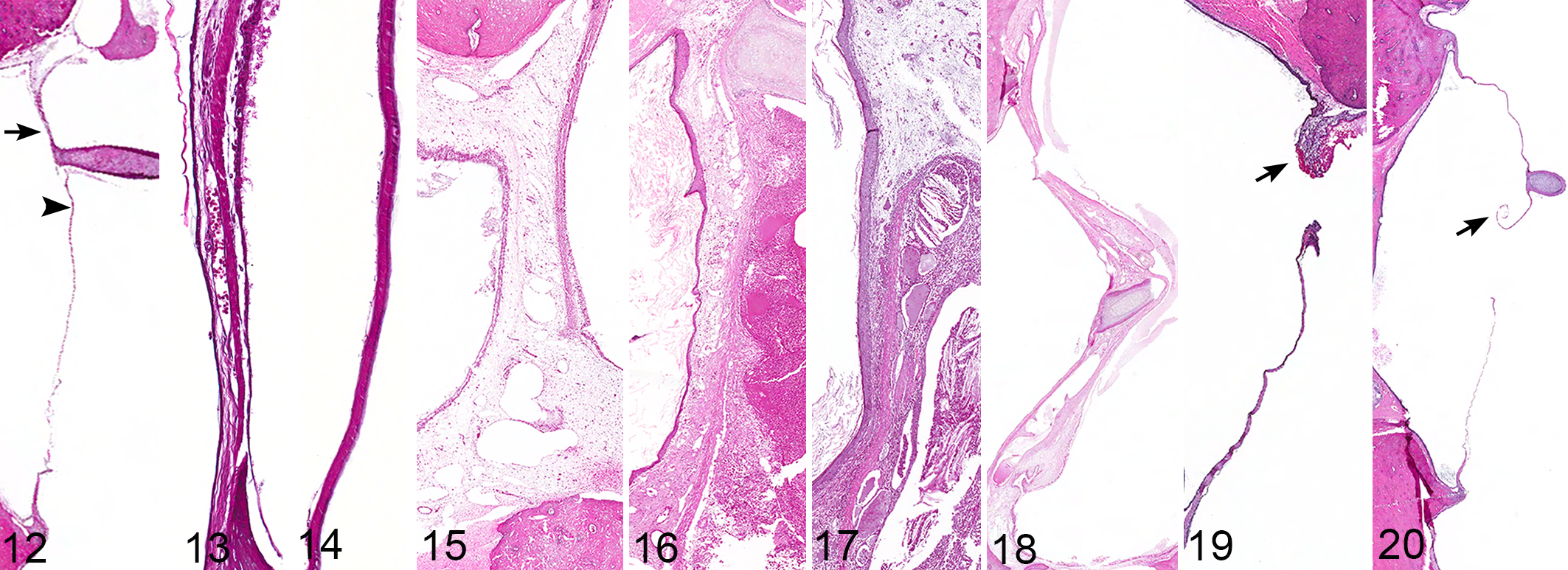
In the cat, inflammation of the tympanic membrane, myringitis, is typically reflective of otitis media and is stereotypical in nature. Early changes include expansion by edema and ectatic lymphatics (Fig. 15) and progresses to include, with increased severity, hemorrhage, inflammation (Fig. 16), granulation (Fig. 17) or mature fibrous connective tissue (Fig. 18), vascular ectasia, formation of cholesterol clefts, and bony remodeling of the manubrium of the malleus. These changes are typically similar to, and continuous with, inflammatory changes within the rest of the auricular mucoperiosteum of the middle ear. Although tympanosclerosis, or formation of hyaline plaques, is commonly reported in humans with chronic myringitis, 11 this lesion was not observed within the study population. In only 1 case was pathologic rupture of the membrane confirmed histologically. In this case, the animal had a history of head trauma, and traumatic rupture was confirmed via locally extensive hemorrhage and edema within the affected portion of the membrane (Fig. 19). Traumatic or inflammatory rupture of the tympanic membrane should be differentiated from the much more common artifactual rupture (Fig. 20) that is a frequent complication of trimming and processing.
Rupture of the tympanic membrane secondary to inflammatory changes appears to be an infrequent event in the cat and was present in none of the examined ears of this study or in any of the ears in Lawson’s initial report. 6 In the dog, otitis media is frequently associated with severe otitis externa. 5 Perforation of the tympanic membrane is seen in upward of 25% of cases of chronic otitis externa 3,16 and may be positively associated with grass awns or the presence of rod-shaped bacteria in the external ear canal. 16 With their upright pinna and relatively open, hairless ear canals, primary bacterial and/or foreign body otitis externa is a much less common complication in the cat. 5 Similarly, despite the severity of some cases of otitis media, extensive involvement of the tympanum did not correlate with extension of disease into the external ear canal. This finding mirrors otitis media in humans, in whom tympanic membrane perforation is inexorably associated with otitis media despite its occurrence in a minority of cases. 9 In cats, as in humans, otitis media may commonly occur without otitis externa, 5 and thus external ear examination alone is unlikely to be a sensitive tool for the diagnosis of otitis media.
Spanning from the tympanic membrane, across the air-filled space of the tympanic cavity and articulating at the oval window, are the 3 auditory ossicles: the malleus, the incus, and the stapes. The auditory ossicles are responsible for the physical transmission of sound converting vibrations of the tympanic membrane to the round window, where it will continue on as fluid waves within the perilymph and endolymph of the internal ear. The manubrium (arm) of the malleus is embedded within the tympanic membrane and is the most common section of ossicle examined in routine sections of the ear employing this study’s technique. The malleus articulates with the incus, which articulates with the stapes via synovial joints. 13 The stapes articulates as a syndesmosis within the oval window of the petrous portion of the temporal bone, forming a tight and effective barrier while allowing precise transmission of tympanic movement to the internal ear. The middle ear ossicles develop from cartilage with a single center of ossification. Persistence of cartilage in each of the ossicles is normal and distinguishes them from other long bones. 20 Histologic changes to these bones are typically reflective of the severity of the surrounding inflammatory infiltrate, with lysis or irregular proliferation and remodeling associated with suppurative otitis media being the most common.
Within the routine middle ear sections of this study, portions of the stapedius muscle and tendon and tensor tympani muscle and tendon are frequently encountered, while that of the tensor veli palatine muscle, which controls the opening of the auditory tube, is not typically present. Both the stapedius and tensor tympani muscles are composed of a central tendon with muscle fibers radiating in what is described as “feather shaped” (Fig. 21). The tensor tympani contains prominent adipose tissue, which may provide the cochlea insulation against the electrical effects of contraction. 20 Inflammatory changes of the muscles of the middle ear were uncommon (7/100 [7%] ears) and were reflective, both in severity and composition, of the middle ear disease (Fig. 22). Minor intramuscular hemorrhage was present in 4 ears, associated with inflammation in 2.
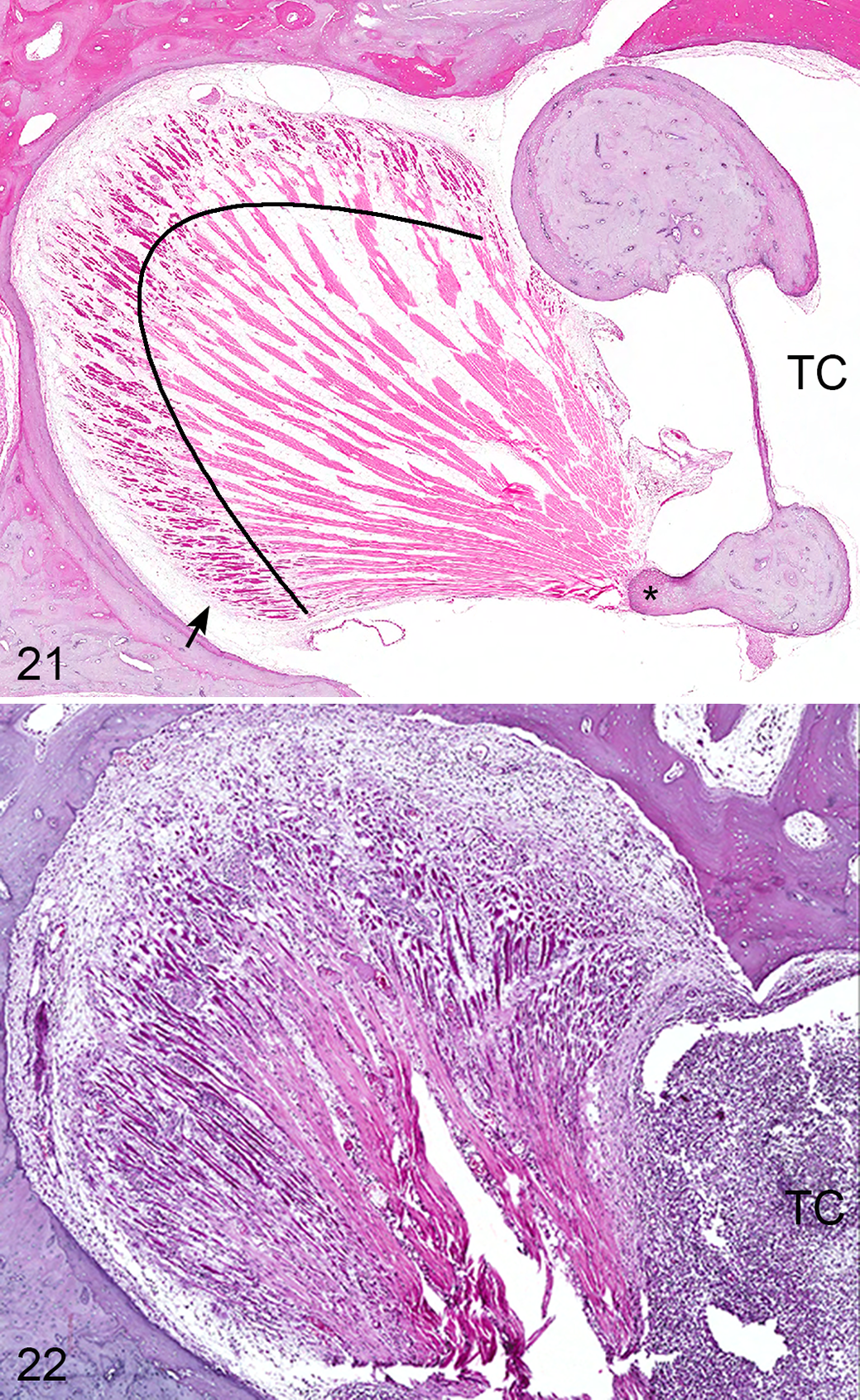
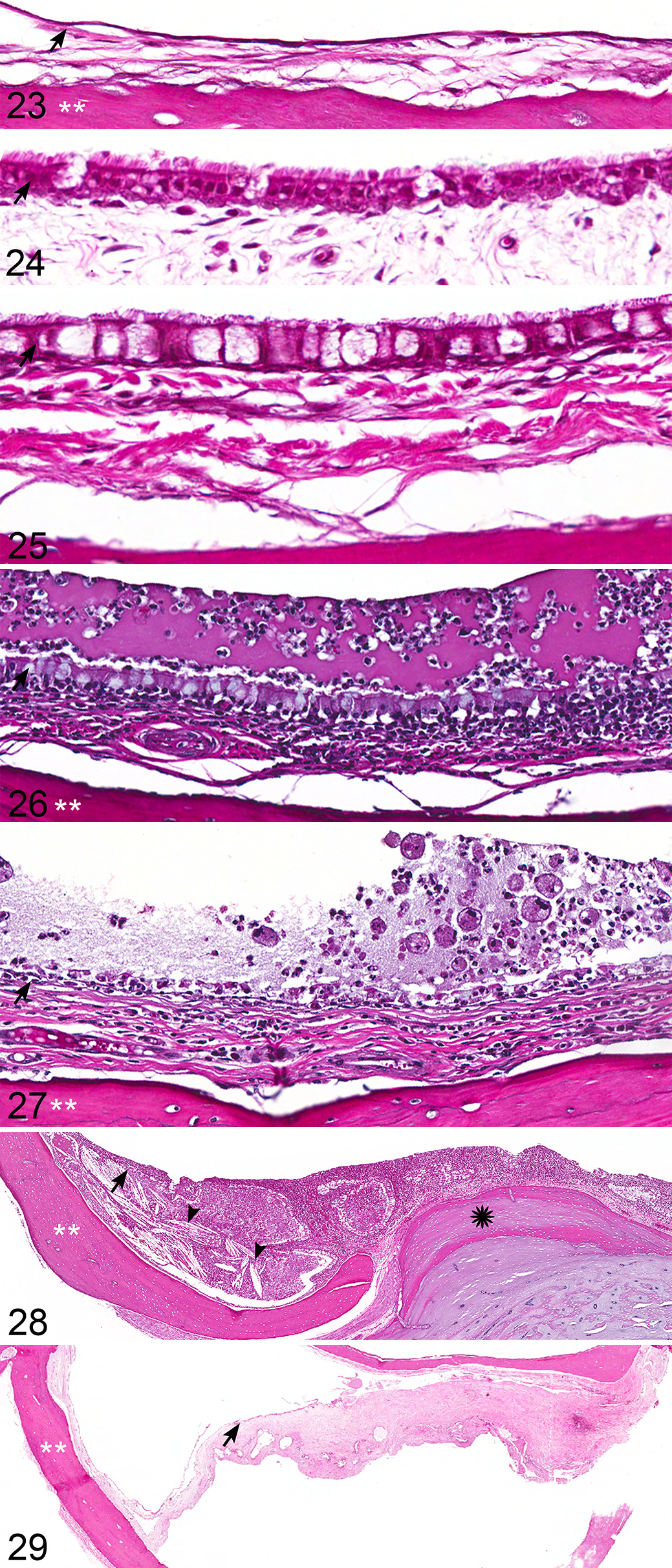
The tympanic cavity is lined by a modified mucosal membrane composed of a single layer of epithelial cells on a basement membrane; a regionally, variably prominent propria-submucosa; and a loose layer of fibrous connective tissue much thinner but otherwise synonymous with the periosteum of other bones. The direct apposition of epithelia and submucosa with periosteal connective tissues is unique to the ear, and we use the term auricular mucoperiosteum to demarcate this unique mucosal membrane. The composition of epithelial cells within the auricular mucoperiosteum varies significantly by location and is typically simple, nonkeratinizing squamous (Fig. 23) to low cuboidal when lining the ventromedial compartment and auditory ossicles; low cuboidal to columnar, variably ciliated when lining the medial aspect of the tympanic membrane (Fig. 24) and overlying the petrous portion of the temporal bone; and tall columnar to variably pseudostratified and ciliated, analogous to respiratory epithelium, lining the dorsolateral compartment and continuous with the auditory (eustachian) tube connection with the nasopharynx. Intermixed with the respiratory epithelium of the dorsolateral compartment are variable numbers of goblet cells and low numbers of nonciliated basilar cells (Fig. 25). The origin of these anatomic differences has recently been described to be the result of a dual origin of the epithelium lining the middle ear. 19 Within regions of the ear closely associated with the auditory (eustachian) tube, the epithelium is of endodermal origin. These typically ciliated cells facilitate the clearance of material from the middle ear through the auditory tube. Regions of epithelium that are simple and nonciliated are of neural crest origin. 19
In the cat, otitis media is thought to occur primarily as a sequela of upper respiratory tract disease, often as a direct consequence of bacteria from the nasopharynx traveling via the auditory (eustachian) tube into the tympanic bulla. In addition, removal of debris from the middle ear relies on passage through the auditory tube, and tubal dysfunction is thought to be a necessary precursor for the development of otitis media. 13 The importance of the auditory tube to middle ear health is the foundation of the tubal ligation method used in the induction of experimental models of otitis media. In the dog, but not in the cat, extension through the tympanic membrane is also considered a common route of infection. Less common routes of infection in both cats and dogs include extension via the temporohyoid joint, direct extension via erosion of the tympanic bullae, or migration along vascular or neural pathways. 13
As with any other tissue, the middle ear reacts to injury with a stereotypical inflammatory response that, regardless of injurious agent, tends to progress though a continuum common to otitis media in both animals and humans. 15 This continuum includes an early vascular and exudative phase that, with persistence, leads to a proliferative phase and eventual resolution with fibrosis. Changes in the middle ear cavity were classified into 4 major categories: vascular dynamic (fluid), inflammatory, membranous, and bony.
Normal to minimally affected ears typically had minimal homogeneous to faintly flocculent proteinaceous fluid within the tympanic cavity with mild to moderate expansion of the auricular mucoperiosteum by ectatic lymphatics and vessels, as well as minimal edema fluid. Low numbers of lymphocytes and/or plasma cells were often present within the auricular mucoperiosteum, particularly those of the dorsolateral compartment overlying the petrous portion of the temporal bone. In a normal ear, these cells should number less than 25 in any 400× field. More significant expansion of the membranes by protein-rich edema fluid was typically accompanied by inflammatory infiltrates and fibrin exudation and variably accompanied by hemorrhage. In all instances of acute exudative otitis media, the bulk of the inflammatory infiltrate is composed of neutrophils (Fig. 26), which may be localized, initially accumulate within the dorsolateral compartment, or, in the most severe cases, entirely fill the tympanic cavity. Neutrophils are typically accompanied by lesser numbers of macrophages, lymphocytes, and plasma cells, which may increase in number with chronicity (Fig. 27). Regardless, neutrophils remain present in any active case of otitis media. Frequently accompanying marked suppuration is abundant granulation tissue (Fig. 28), typically expanding the auricular mucoperiosteum and variable fibrosis (Fig. 29), which may occur early in the disease process. With chronicity, lymphocytes and plasma cells may become the predominant leukocyte and, with resolution, may remain as multifocal lymphoid nodules (follicles). Without other evidence of active inflammation, the presence of lymphoid follicles is interpreted to represent a low-grade, chronic, and resolving otitis media.
In the early human otic literature, under the condition of chronic spontaneous otitis media, and in nearly all animal models of the disease, the presence of glandular structures within the auricular mucoperiosteum is reported. Typically, these have been considered simple tubular mucous glands 10 that may arise as a metaplastic response 14 to inflammatory ear disease. While cystic structures were commonly present in the membranes of diseased ears in the current study, their origin as metaplastic glands is doubtful. With any significant inflammation of the auricular mucoperiosteum, polypoid proliferation of the membranes is an early and common change, and folding with entrapment of the overlying epithelium was not uncommonly observed (Fig. 8). The resulting pseudo-glandular structures typically remain long after resolution (Fig. 9) of the inflammation and may, with fibrosis, be considered a positive indicator of previous inflammatory disease. Except in the region immediately adjacent to the exit of the auditory (eustachian) tube within the epitympanic recess, glands or gland-like structures are not present in the normal auricular mucoperiosteum of the middle ear, and their presence is always considered abnormal. Other membranous changes included attenuation of the overlying epithelial cells with frequent loss of cilia, vacuolation and degeneration of epithelial cells, and increased goblet cell density. As noted in experimentally induced disease, acute otitis media is associated with an increased total area of the middle ear with respiratory-like epithelium, with increased goblet cells in regions of epithelium where goblets calls are not typically observed. 2
Acicular (cholesterol) clefts are a common feature of severe, exudative otitis media and are typically associated with sites of previous hemorrhage. In otitis media, cholesterol clefts are quite common and, in the current study, were present both within the inflammatory exudate of the tympanic cavity and more commonly within the auricular mucoperiosteum, often in association with the previously described pseudo-glandular structures. Their presence, while distinctive, should be considered a common, diagnostic feature of severe otitis media, and their presence alone should not be diagnostic of a cholesterol granuloma, a rare condition in which cholesterol clefts induce a granulomatous foreign body–type inflammatory mass causing secondary, often pressure-related damage. Despite the frequent occurrence of cholesterol clefts, cholesterol granulomas were not identified in the current study; however, a recent report in a cat with Horner syndrome suggests that this entity may be an uncommon complication of otitis media in the cat. 4
Bony changes indicative of chronic ear disease were common in the examined ears and are most easily recognized in the tympanic bulla, septum bulla, and the petrous portion of the temporal bone. The normal tympanic bulla is uniform in width, smooth, and without a marrow cavity (Fig. 30), and changes are readily identified as proliferative or erosive, characterized by marked, regular to irregular expansion; irregular erosion (Fig. 31); development of a marrow cavity (Fig. 32); and occasionally irregular bony spicule development (Fig. 33). Thickening of the bulla bone is significantly associated with severe otitis media, and as such, any thickening of bulla, often evident even on cursory gross examination, should warrant further histologic investigation. Proliferative changes of the septum bulla (Fig. 34) were occasionally accompanied by complete bony lysis (Fig. 35). Although the petrous portion of the temporal bone is a calcified cartilagenous matrix with minimal lamellar bone, deposition of proliferative lamellar bone is typical. Lysis of the petrous portion of the temporal bone was not observed.
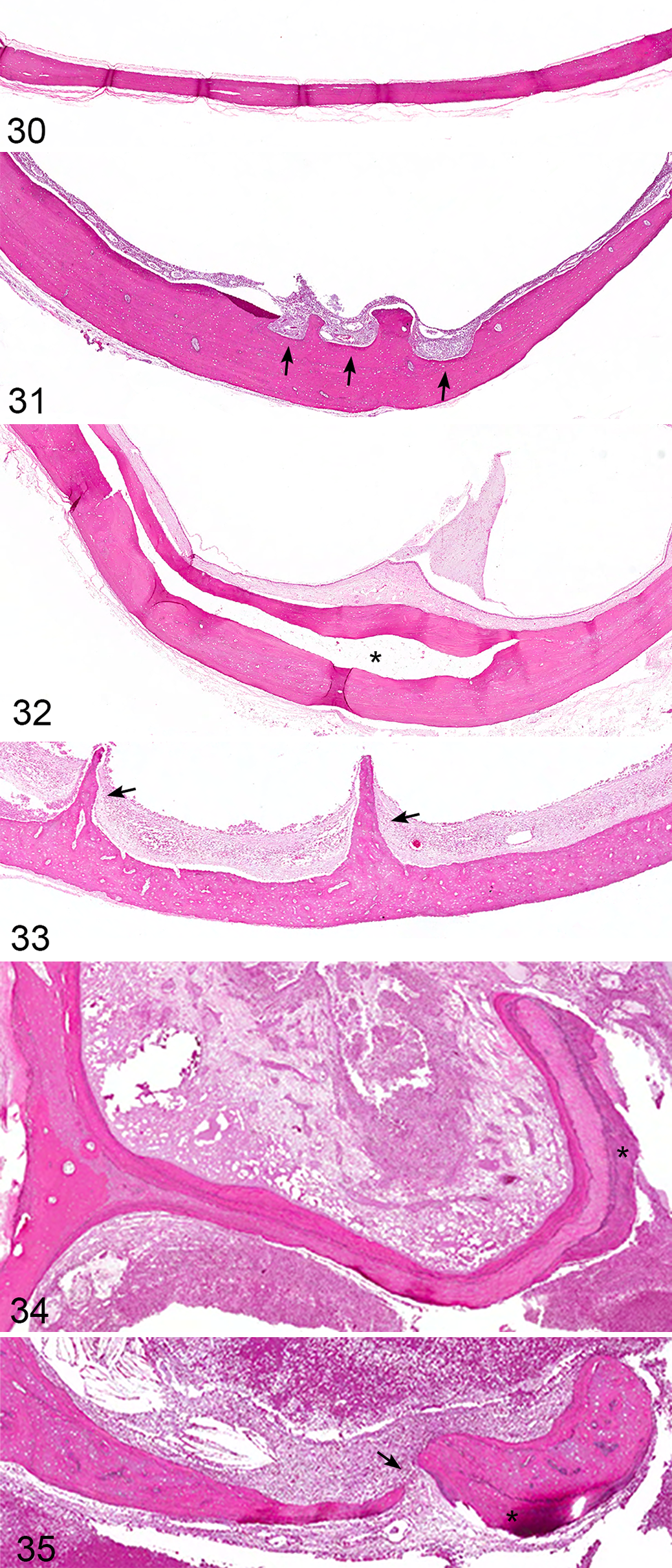
Forming the dorsomedial most border of the middle ear is the internal ear, encased within the petrous portion of the temporal bone. The internal ear is composed of a bony labyrinth, a membranous labyrinth, and the vestibulocochlear nerve (CN VIII) traversing the internal acoustic meatus from the brainstem, along with the facial nerve (CN VII). The internal ear directly communicates with the middle ear at both the round and oval windows (Figs. 36–41). The oval window, containing the circumferential syndesmosis of the stapes with the petrous temporal bone, is generally considered impermeable to extension of inflammation from the middle ear. 14 In humans, osteosclerosis near and variably involving this syndesmosis is common. 10 Neither extension of inflammation through the oval window nor osteosclerosis was identified in any of the 28 sections of ear in which the oval window was present.
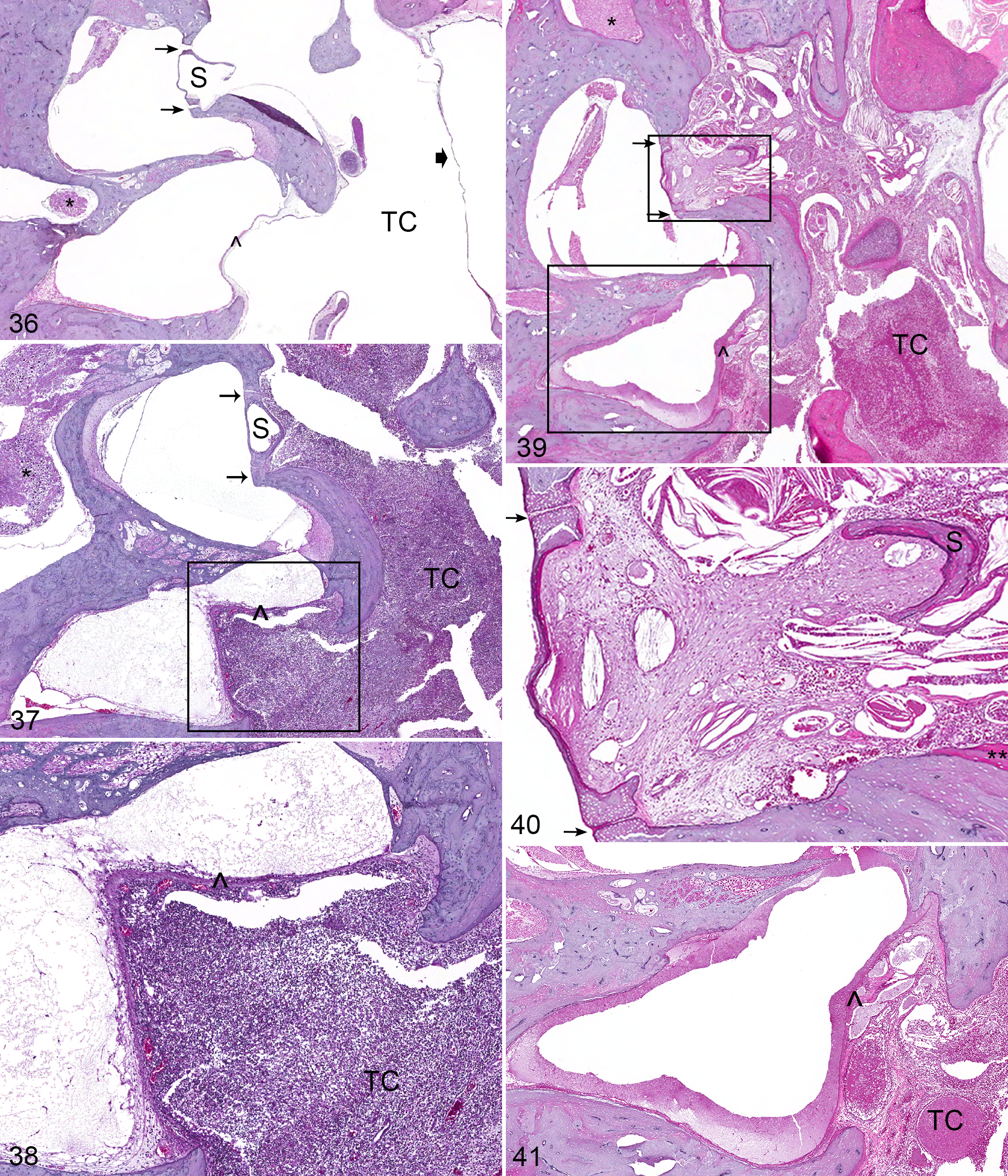
In experimental feline models of otitis media, the permeability of the membrane of the round window is increased to a variety of macromolecules, including tritiated albumin, horseradish peroxidase, and some bacterial toxins, 14,17 during the acute phase of the disease (Fig. 38); despite this, morphologic evidence of otitis interna remains relatively uncommon. In response to chronic otitis media, the membranes of the oval (Fig. 40) and round windows (Fig. 41) exhibit the stereotypical inflammatory changes of the auricular mucoperiosteum, including expansion by granulation tissue and inflammatory cells. Some studies suggest that the reduced permeability of the round window in chronic otitis is associated with physical obstruction of the membrane by the mat of fibrin, proteinaceous material, and granulation tissue that routinely develops in these inflammatory conditions. 15,17
Detailed examination of the internal ear was not the intent of this study, and as such, abundant processing artifact was common in the examined ears. Slow, gentle decalcification with EDTA solutions and plastic embedding is required for best preservation of membranous and epithelial structures. 8 Since this is not practical for the diagnostic pathologist, changes to the internal structures noted in this study must be interpreted in light of these limitations.
In routine preparations of the ear, portions of the vestibule and cochlea are most commonly present. In sections of the vestibule, the fine membranes of the utricle and saccule may be observed, while in the cochlea, the scala vestibuli, cochlear duct (scala media), and scala tympani should be easily identified, as are the structures of the mechanosensory portion of the organ of Corti, including the hair cells that lie on the basilar membrane and the overlying tectorial membrane. Although identification of these structures is fairly uncomplicated, artifactual damage makes determination of significant pathologic change beyond the scope of the routine diagnostic section. Increased proteinaceous fluid (Figs. 38, 41), inflammatory cells, or hemorrhage are typical lesions that can be identified with some confidence on routine diagnostic sections.
Within routine sections of ears, branches of the trigeminal nerve (CN V), facial nerve (CN VII), and vestibulocochlear nerve (CN VIII) are present; however, only sections of facial and vestibulocochlear nerves are easily and routinely identified. Dysfunction of both the vestibulocochlear (CN VIII) and the facial nerve (CN VII) is clinically associated with middle ear disease, and vestibular signs associated with vestibulocochlear neuritis may be the presenting complaint in cases of otitis media and interna. Both nerves enter the petrous portion of the temporal bone via the internal acoustic meatus (Fig. 42); the vestibulocochlear nerve courses into the cochlea and semicircular canals, while the facial nerve transverses the facial canal. Within the region of the stapedius muscle, the bone of the facial canal is incomplete (dehiscence of the facial canal), 1,7 particularly within the region of the round window (Fig. 43), allowing direct communication between the tympanic cavity and the epineural connective tissues (Fig. 44). Despite this anatomic exposure, facial neuritis is rarely reported and was not present in any of the 12 study sections that captured this portion of the facial canal, although facial perineuritis was present in 3 of 12 (25%) ears (Fig. 45). Despite this lack of histologic evidence to support facial neuritis as the primary neurologic manifestation of otitis media, it is important to note that only 12 of 100 (12%) routine sections examined had the facial canal at the level of the dehiscence. Although it would be expected that significant facial neuritis might be observed in the more proximal sections present in every ear examined, localized facial neuritis could not be ruled out in cases where the dehiscence of the facial canal was not observed. Since the clinical diagnosis of facial neuritis can be quite difficult in the cat, it is likely that facial neuritis is underreported both clinically and histologically, and sections targeting the facial nerve might be a further avenue of study.
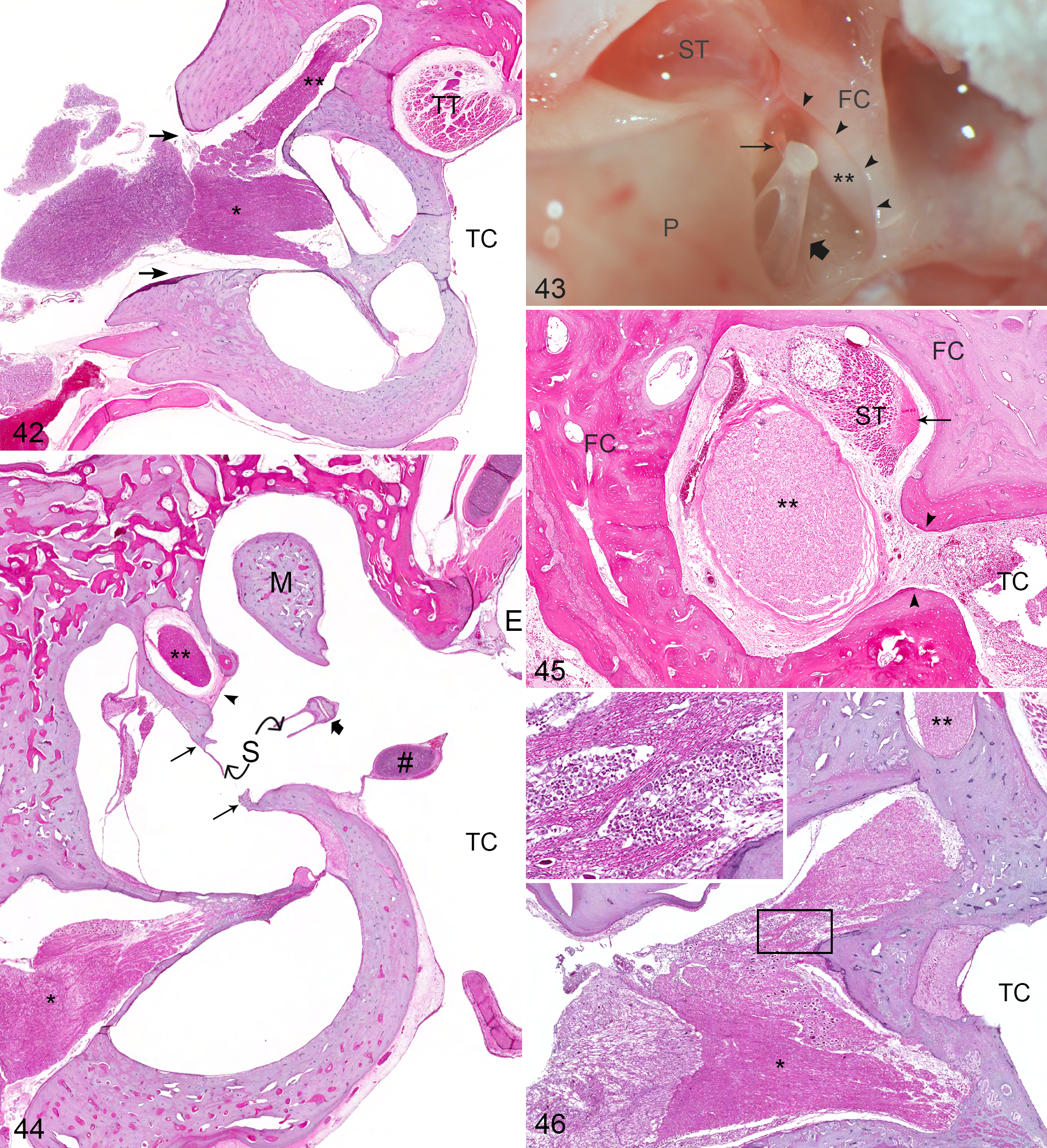
In the current study, vestibulocochlear neuritis was present in 4 of 100 (4%) ears (Fig. 46); most significantly, bilateral vestibulocochlear neuritis was present in a cat that presented with neurologic abnormalities originally attributed to rabies virus infection. Otitis interna was not present in either ear. Although it seems most probable that vestibulocochlear neuritis is a sequela to otitis interna, which in itself is most commonly a sequela of otitis media, primary hematogenous or nervous routes of infection cannot be entirely ruled out in these cases. In the above case, the brain was unavailable for examination, and extension of meningitis via the vestibulocochlear nerve is considered a distinct possibility. The relative frequency of vestibular cochlear neuritis in cases of otitis media lacking significant otitis interna is not well understood, and this anatomic paradox merits further study.
In the current study, 24 of 100 (24%) ears had histologic findings in the internal ear. In all 6 otherwise histologically normal ears, the histologic finding was limited to small amounts of flocculent eosinophilic material. While this material is easily recognizable, its likely significance is limited. In humans, similar material is associated with formalin fixation or increased death to fixation interval, 8 and its presence in an otherwise normal ear is considered an inevitable artifact of the current study method. However, inflammatory changes of the internal ear were significantly associated with severe otitis media. Inflammation of the cochlea and vestibulochlear nerve was limited to severely affected ears, and the presence in both the cochlea and vestibulocochlear nerve in 2 ears supports the currently accepted theory that otitis interna is an extension of otitis media that may, in some cases, result in vestibulocochlear or facial neuritis. More specific or definitive conclusions are likely premature and not warranted without the use of processing and fixation techniques specific to the delicate structures of the internal ear.
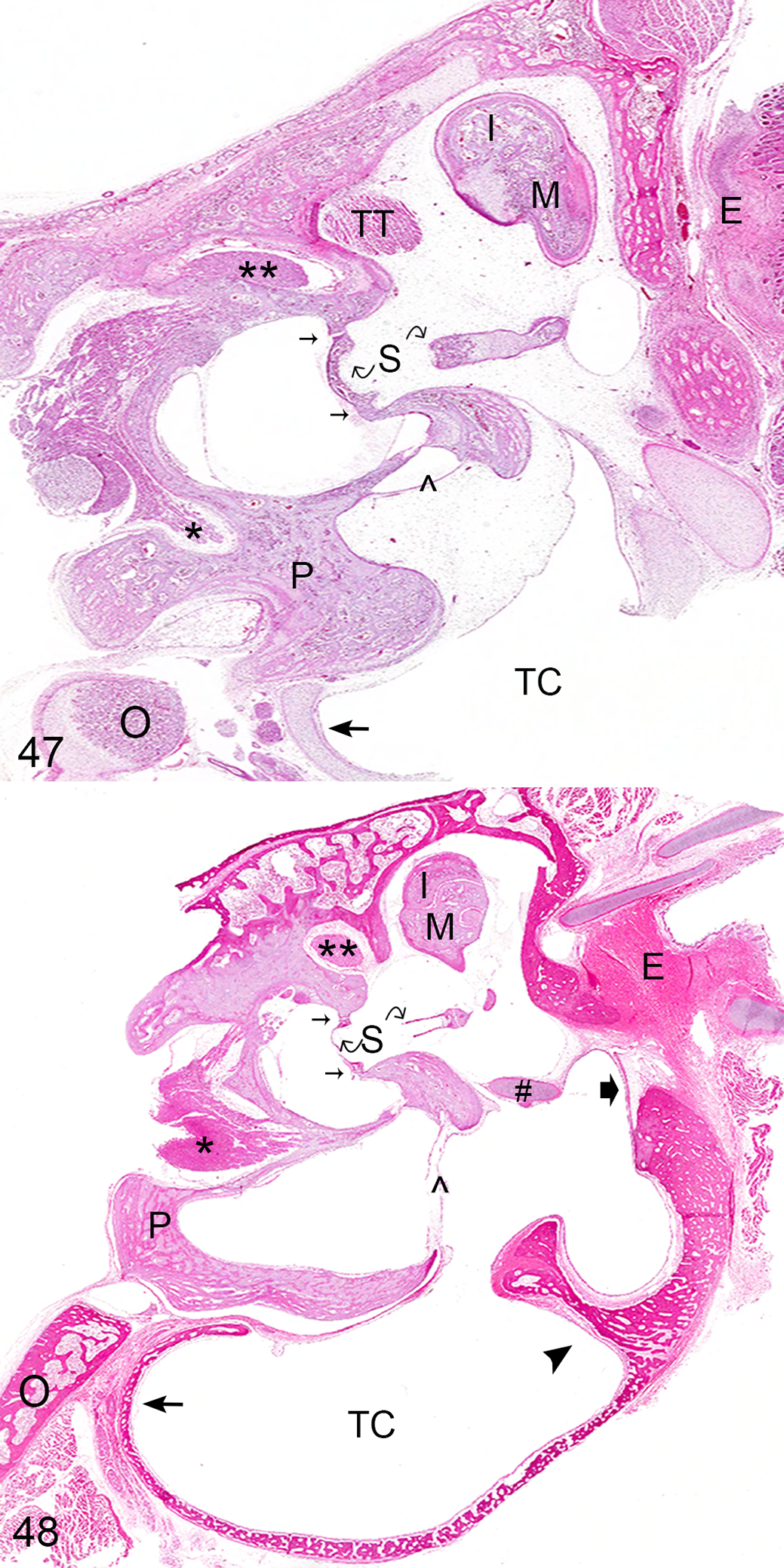
Two significant age variants warrant discussion. In kittens up to 4 weeks of age, the middle ear may be filled with a finely basophilic, paucicellular, mesenchymal matrix that appears to expand the auricular mucoperiosteum of the neonatal ear (Fig. 47). This finding is a normal feature of the development of the middle ear and should not be mistaken as a proliferative or inflammatory change. This mesenchymal matrix is thought to be material released from rupture of the first pharyngeal pouch during development of the middle ear. 19 During postnatal maturation of the ear, this mesenchyme “cavitates,” or retracts, leaving an epithelial-lined air-filled space. In addition, in skeletally immature cats, the tympanic bone is loosely woven and lacks significant compact lamellar bone until maturity (Fig. 48).
Results of the present study indicate that both gross and histologic evidence of middle ear disease in the cat is more frequent than the current literature suggests. In the current study, gross evidence of disease was present in 14 of 100 (14%) examined ears. In addition, histologic evidence of current or prior ear disease was present in 48 of 100 (48%) examined ears and considered severe in 11 of 100 (11%). In 34 of 100 (34%) ears, histologic evidence of current or prior otitis media, including expansion of the auricular mucoperiosteum by leukocytes, cholesterol clefts, hemorrhage, fibrin, and granulation tissue containing membranous pseudo-glands, was present in ears that were grossly normal, indicating the importance of histologic examination to accurately define the prevalence of otitis media in the cat, as well as the significant likelihood of underestimating disease prevalence if gross examination is the sole method of identifying disease. In addition, although otitis media is typically considered a unilateral disease, 5 bilateral gross and histologic disease was predominant in the current study.
The significant association between severe ear disease and clinical signs of respiratory disease is important to the diagnostician in which young and/or immunocompromised animals are frequently presented for examination. Equally important, given the significant association between severe otitis media and clinical signs of upper respiratory tract disease, investigation for otitis media may be warranted in any cat with a previous history of respiratory disease. Although this is a preliminary study, with no specific correlation of histologic changes to functional evaluation of hearing and overall health status, this study suggests disease is not as uncommon as previously reported and may be a significant contributor to morbidity in the cat population. In addition, this study indicates that it is not improved health care for pets that has decreased the frequency of ear disease but rather the lack of significantly detailed investigation.
While this study clearly indicates that both gross and histologic evidence of disease is more prevalent than previously described, it must be noted that the current study cohort included both stray and owned animals; as such, the role of veterinary care, environmental factors such as indoor or outdoor housing, frequency of exposure to disease, and underlying immune status on disease prevalence could not be evaluated. Using a representative sample of animals received in a state diagnostic laboratory, the current study establishes the spectrum of morphologic normal and diseased feline middle ears, irrespective of clinical signs. While this is interpreted by the authors to reflect disease prevalence in the general population of cats in the greater Oklahoma state region, the correlation between histologic findings and clinical signs is limited by this element of the study design.
Finally, although many different techniques to examine the ear have been employed in the course of experimental investigation, this study’s sectioning technique is the first to offer an easily reproducible procedure that allows the routine evaluation of most of the important structures of the ear on a single slide, while minimizing additional effort and prolonged tissue processing. This technique readily allows evaluation of many structures while still allowing for easy modification should examination of other anatomic sites be required. As special sectioning, fixation, and processing techniques are required to accurately evaluate histologic lesions of the internal ear, the currently described method is not recommended for those with particular interest in the membranous labyrinth.
The intent of this study was to catalog the spectrum of inflammatory disease affecting the middle ear. In addition, a novel, reproducible method of examination of all compartments of the ear in a single section was an unexpected tangible benefit of the study. In aggregate, it is hoped that this study will serve as both a core resource and impetus for pathologists, clinicians, and scientific investigators to begin the process of definitive correlation of clinical and histological disease.
Footnotes
Acknowledgements
We acknowledge the technicians of the histopathology laboratory at the Oklahoma Animal Disease Diagnostic Lab, particularly Ms Darlene Minor.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This research was supported by the “Roger J. Panciera Pathology Educational Fund,” Oklahoma State University Foundation, Stillwater, Oklahoma.
