Abstract
An 8-year-old, male neutered Siamese cat was presented with Horner syndrome and right head tilt. A soft tissue mass was observed in the right tympanic cavity, and bulla osteotomy was performed. Tissue samples retrieved from the tympanic cavity were sent for histology, and a middle ear fluid swab was sent for bacterial culture and sensitivity. Histologic diagnosis was of otitis media associated with cholesterol granuloma (CG). Bacterial culture yielded
Cholesterol granuloma (CG) is a histologic term used to describe a granulomatous lesion with numerous cholesterol clefts surrounded by fibrous reaction.4,16,21 In human beings, CG is found in many organs and is often observed in temporal bone and middle ear.4,16,21 In veterinary medicine, spontaneous CG is most commonly reported in the choroid plexus of older horses.
15
It has been described in maxillary sinus,
17
middle ear,2,5,20 and brain
23
of dogs, in choroid plexus of fat-tailed dwarf lemurs (
Formalin-fixed samples of a middle ear mass from a cat were received at the Tifton Veterinary Diagnostic and Investigational laboratory (University of Georgia, Tifton, Georgia). The samples were from an 8-year-old, male neutered Siamese cat that presented with unilateral Horner syndrome signs and a right head tilt. Horner syndrome signs included miosis, ptosis of upper eyelid, protrusion of third eyelid, and enophthalmos. Clinically, a soft tissue mass was observed through the right tympanic membrane. Pupillary light reflexes appeared normal, and fluorescein staining was negative. Radiographs of the head revealed increased opacity in both tympanic bullae. Right tympanic bulla osteotomy was performed. Samples of a thick, tannish fluid were swabbed from the distal compartment of the tympanic bulla and submitted for aerobic and anaerobic bacterial cultures and sensitivity. The rostral and distal compartments of the tympanic cavity were curetted, and samples of the soft tissue mass retrieved from the tympanic cavity were fixed in 10% formalin and submitted for histology.
The formalin-fixed specimen received consisted of multiple, small fragments, from 3 mm × 2 mm × 2 mm to 12 mm × 5 mm × 3 mm, of a crumbly gray to yellow tissue. For histology, formalin-fixed samples were trimmed, embedded in paraffin, sectioned at 5-µm thickness, and stained with hematoxylin and eosin. Multiple fragments of the mass from tympanic bulla were examined histologically. All sections examined had large numbers of cholesterol clefts surrounded and separated by epithelioid macrophages, neutrophils, fibrin, red blood cells, very few lymphocytes and plasma cells, a few hemosiderin-laden macrophages, and reactive fibroblasts (Fig. 1). Areas with predominance of necrotic debris and degenerated neutrophils were also observed among cholesterol clefts (Fig. 2). The granulomatous reaction with cholesterol clefts was encircled by fibrous tissue. A few gland-like structures lined by cuboid epithelium were seen within the granulation tissue and the collagen-rich connective tissue (Fig. 3) at the margins of the sections. The histologic diagnosis was of chronic otitis media with CG formation. Bacterial culture yielded heavy growth of
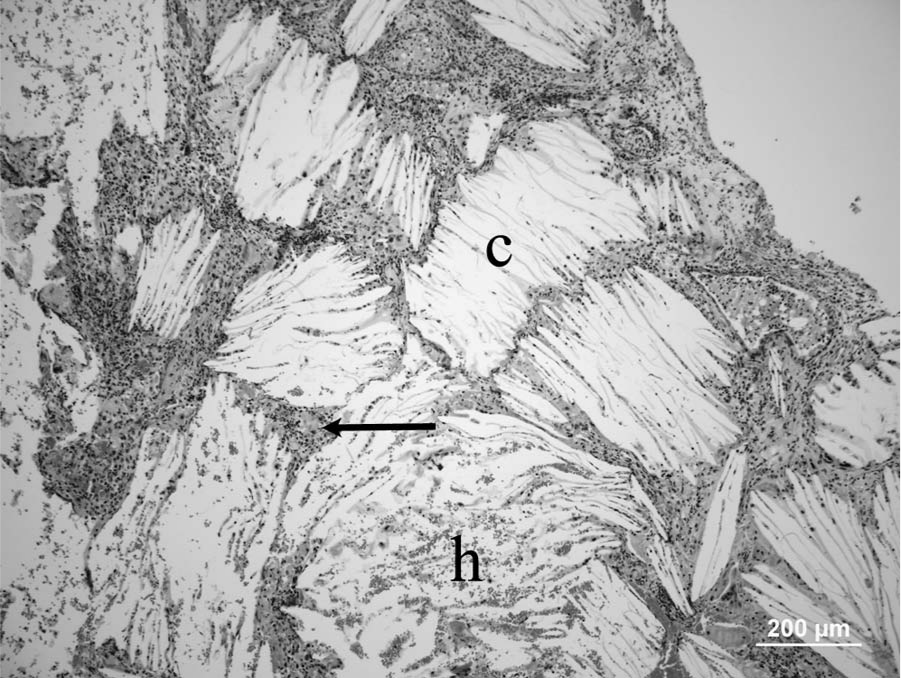
Micrograph; middle ear cholesterol granuloma; cat. Note multiple typical acicular clefts that represent cholesterol crystals (c) separated and surrounded by inflammatory cells (arrow) and areas of hemorrhage (h). Hematoxylin and eosin. Bar = 200 µm.
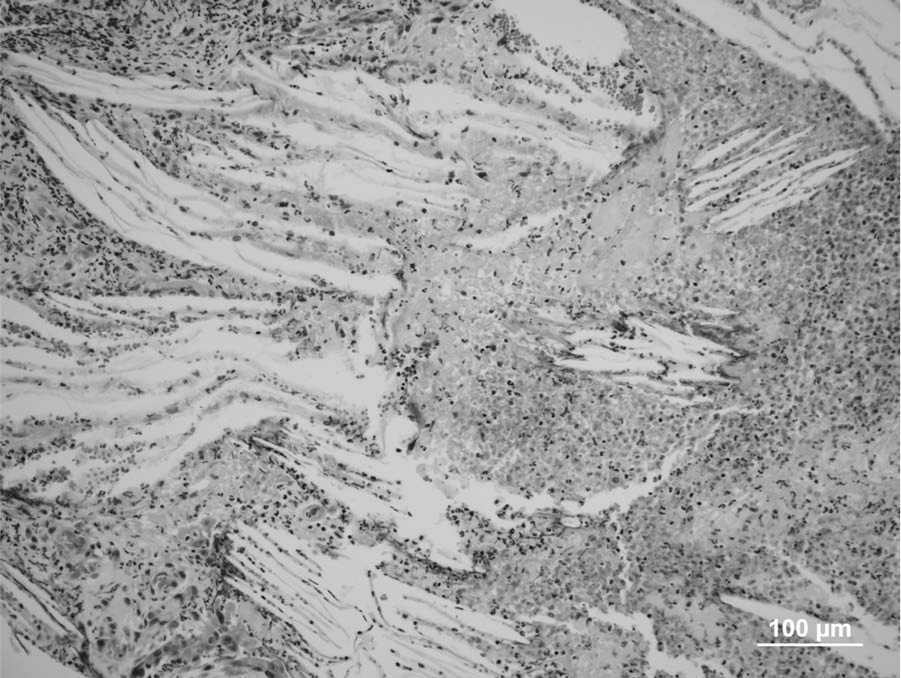
Micrograph; middle ear cholesterol granuloma; cat. Note extensive areas of cell debris, fibrin, and viable and degenerated neutrophils among cholesterol clefts. Hematoxylin and eosin. Bar = 100 µm.
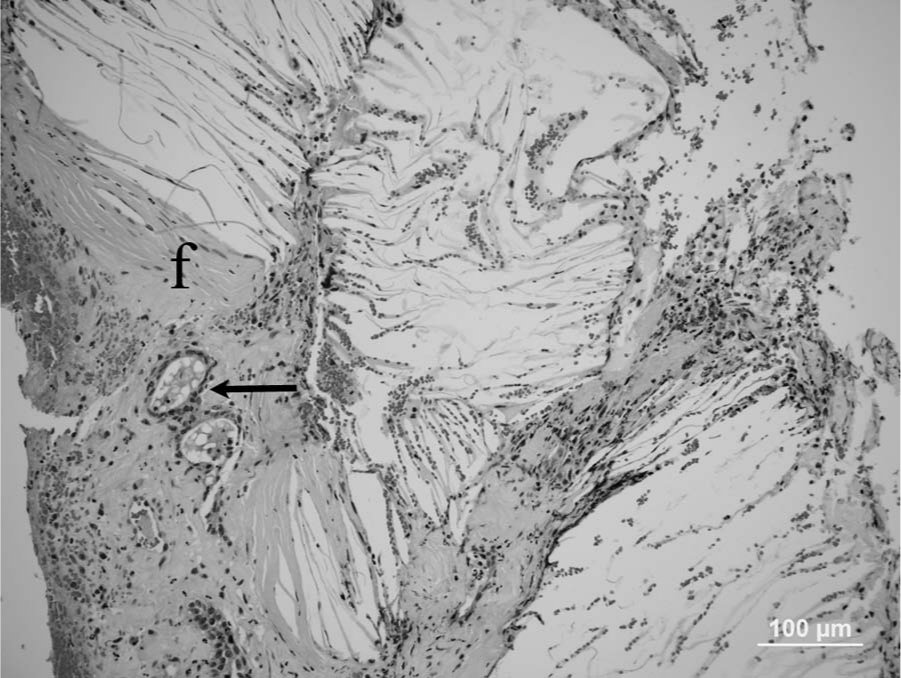
Micrograph; middle ear cholesterol granuloma; cat. Intense fibrosis (f) and a few gland-like structures (arrow) are observed along the periphery of the cholesterol granuloma. Hematoxylin and eosin. Bar = 100 µm.
Immediate postoperative ear treatment consisted of ear drops containing orbifloxacin, mometasone furoate monohydrate, and posaconazole a for 2 days. Based on culture and sensitivity, a 30-day treatment with amoxicillin and clavulanic acid b was started. Twenty-one days after the initial tympanic bulla osteotomy, the cat was clinically evaluated. Minor improvement of initial clinical signs was noted. The cat still had a very obvious right head tilt and all signs of Horner syndrome initially observed. The cat did not show any desire to jump up onto higher objects; however, he was not showing signs of pain and was able to maneuver around his environment very well. He was eating and drinking normally. At 35 days postoperative, the cat showed very little of the Horner syndrome signs initially observed but still had an obvious right head tilt. At 44 days postoperative, the cat showed no signs of Horner syndrome with any other clinical signs noted except the right head tilt that possibly will persist for the remaining of the cat’s life.
In human beings, CG of the middle ear system is a benign expansile lesion that occurs in the middle ear or mastoid region and petrous apex.3-5,9,21 Middle ear CG has been associated with chronic middle ear diseases including otitis media, middle ear adenomatous tumors, cholesteatoma, and endolymphatic sac tumors.3,4 Cholesterol granuloma is a histological term used for the description of a tissue response to cholesterol crystals, and it is also used as a clinical diagnosis. 9 In human beings, CG of the middle ear is a frequently encountered complication of otitis media and has been found in as many as 12–20% of temporal bones with chronic otitis media. 21 Very few cases of CG involving middle ear have been described in veterinary medicine. In dogs, CG has been reported in the middle ear, 2 has been associated with otitis media and destruction of tympanic bulla, 5 and as a long-term complication of total ear canal ablation. 20
In cats, cholesterol crystals are a common finding within lesions of endogenous lipid pneumonia, 12 and cholesterol crystals are also a common finding in meningiomas. 13 Typical CG has been described in the uterus of a female cat with cystic endometrial hyperplasia-pyometra complex. 25 Two intracranial CGs were reported in cats6,19 that presumably originated from meninges and compressed adjacent brain.6,19 A choroid plexus CG was described in a cat 8 that had the typical gross and histological appearance of the CG of the choroid plexus described in older horses. 15 Cholesterol granuloma is also known as cholesteatoma in horses 15 ; however, the term “ear cholesteatoma” in human medical literature is used to describe a benign keratinizing squamous cell cyst, 4 and the term “CG” is used to describe a granulomatous lesion containing cholesterol crystals that occur in a wide setting of middle ear diseases. 4 Aural cholesteatoma or aural keratinizing cyst has also been described in middle ears of dogs. 24 In human beings and dogs, CG and ear cholesteatoma may coexist.21,24
Cholesterol granuloma of the middle ear has been experimentally induced in several species of animal including cats,7,10 chinchilla (
The pathogenesis of the formation of middle ear system CG is still controversial; however, it is believed that 3 factors play an important role in its development: hemorrhage, impaired clearance or drainage, and obstruction of air exchange or ventilation.9,21 Obstruction at any level along the middle ear may result in the interruption of clearance and ventilation. Various factors including remaining mesenchyme, polyps, scar formation, cholesteatoma, inflammatory effusions, and congenital epidermoid cysts can be expected to cause obstruction. 16 Bleeding may result from pressure changes within air cells, trauma, acute hemorrhagic otitis, mucosal inflammation, and chronic hemorrhagic inflammatory effusion.16,21 Compared with middle ear and mastoid CGs, petrous apex CG is considered a distinct entity by some authors. 11 Based on experimental models, it is believed that petrous apex CG is the result of blockage of the normal aeration of the petrous apex air cells. The obstruction leads to absorption of air and the development of negative pressure within the air cells, causing mucosal engorgement and hemorrhage and granuloma formation. 3 The cholesterol crystals that result from the red cell degradation incite a typical foreign body reaction leading to further hemorrhage and inflammation. 3 Cholesterol granuloma seems to promote its own growth in a vicious cycle by becoming a source of subsequent hemorrhage and obstruction. 16 A new pathogenesis for petrous apex CG speculates that petrous apex CG is due to sustained hemorrhage from exposed bone marrow elements within petrous apex cells with development deficiency of the cortical bone that normally separates the air cell system from the marrow compartment. 11
Cholesterol granuloma formation was experimentally induced in the tympanic bullae of cats,7,10 and has been experimentally induced in cats through eustachian tube obstruction leading to middle ear effusion with evidence of organization of the inflammatory effusion within 3 months. Organized effusion in the presence of inflammation and mucosal epithelial lining breaks was considered a potential source for granulation tissue formation and CG. 10 In another similar experiment, tympanic membrane perforation followed by eustachian tube obstruction was induced in cats. Massive reaction of the mucosal periosteum with granulation and polypoid tissue formation and CG, with the classic characteristics as described in human beings, were also observed within 3 months. 7
In human beings, the choice of treatment of CG depends on the location, the presence of any complications, the associated disease, the status of the patient, and his or her hearing. The treatment for cases with CG in the middle ear cleft is usually conservative, with drainage and permanent aeration often very useful. For the treatment of CG of the petrous apex, a variety of surgical procedures have been described.4,21
In conclusion, middle ear CG as observed in human beings may occur in the middle ear of cats with otitis media. The lesion described in the cat from the present report was similar to not only the CG described in human beings but the CG experimentally induced in cats in animal models to study the disease in human beings.7,10 Postmortem examination of the middle ear should be routinely incorporated into necropsy techniques in cats. A previous article describes step-by-step methods for collection and preparation of dog and cat ears for histologic examination. 18
Footnotes
Acknowledgements
The authors would like to thank the staff from the Tifton Veterinary Diagnostic and Investigational Laboratory (Tifton, Georgia) for assistance in tissue processing.
a.
Posatex Otic Suspension, Merck Animal Health, Summit, NJ.
b.
Clavamox, Pfizer Animal Health, New York, NY.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
