Abstract
vHypertrophic cardiomyopathy (HCM) is the most commonly diagnosed cardiac disease in cats. The complex pathophysiology of HCM is still far from clear, but myocardial remodeling is a key process, and cardiomyocyte disarray, interstitial fibrosis, leukocyte infiltration, and vascular dysplasia are described histopathologic features. The present study systematically investigated the pathological processes in HCM, with the aim to shed more light on its pathogenesis. Hearts from 18 HCM cases and 18 cats without cardiac disease (controls) were examined, using light and transmission electron microscopy, immunohistochemistry, and morphometric approaches to identify and quantify the morphological changes. Reverse transcription–quantitative polymerase chain reaction was applied to provide additional mechanistic data on remodeling processes. In HCM, the left and right ventricular free wall and septal myocardium exhibited a significantly reduced overall cellularity, accompanied by a significant increase in interstitial Iba1-positive cells with macrophage morphology. In addition, the myocardium of almost half of the diseased hearts exhibited areas where cardiomyocytes were replaced by cell-rich fibrous tissue with abundant small and medium-sized vessels. HCM hearts also showed significantly higher transcription levels for several inflammatory and profibrotic mediators. Our findings suggest that HCM is the consequence of cardiac remodeling processes that are the result of cardiomyocyte damage and to which macrophages contribute by maintaining an inflammatory and profibrotic environment.
Keywords
Feline hypertrophic cardiomyopathy (HCM) is defined by an unexplained thickening of the left ventricular wall without dilation of the chambers and in absence of any other cardiac or noncardiac disease that itself is capable of causing hypertrophy of the heart. 28,29,31 HCM is the most commonly diagnosed cardiac disease in cats. 8,29,31 It is a heterogeneous disease, in both its clinical presentation and progression. 1 A retrospective study has shown a familial predisposition in Persian, American Shorthair, Maine Coon, and Ragdoll cats. 8 In Maine Coon and Ragdolls, HCM has been linked to mutations in a sarcomeric gene. 23,24 Epidemiological studies have identified a sex predisposition for male cats and a mean age of 6 years at the time of diagnosis. 1,28,29 Affected cats can present with signs of congestive heart failure (CHF) or arterial thromboembolism, or they can suffer sudden death, although many never exhibit any clinical signs of cardiac disease and die of noncardiac conditions. 4,28,32
To the authors’ knowledge, the first study on feline primary myocardial disease and CHF was published in 1970; this and subsequent papers reported a range of pathological features for HCM. 2,3,11,17,20 Macroscopically, feline HCM is characterized by a concentric symmetrical (generalized) or asymmetrical (focal) thickening of the interventricular septum, left ventricular free wall, and papillary muscles, often with dilation of the left atrium. 32 Cardiomyocyte hypertrophy and disarray, a variable degree of interstitial fibrosis, interstitial inflammatory cell aggregates, and dysplasia of vessel walls have been described most frequently as histological features of HCM. 3,4,17,20 More recent studies attempted to further characterize and quantify these features; their results are, at least in part, controversial. 3,4,16,17,20,29 In a study on familial HCM in domestic shorthair cats, one end-stage case with multifocal extensive areas of myocardial fibrosis accompanied by a mononuclear cell infiltration was reported; the changes were interpreted as myocardial infarcts. 4 Studies using a quantitative approach partially confirmed a diffuse or multifocal increase in interstitial fibrosis but questioned cardiomyocyte hypertrophy and myofiber disarray as consistent features of HCM; they did not find evidence of fiber branching or significant differences in diameter and length of cardiomyocytes. 3,16,17 The inflammatory component in HCM has so far been characterized based on the cell morphology (hematoxylin-eosin [HE] stain) and limited cytochemical stains (Leder stain for neutrophils) and appears to comprise lymphocytes, plasma cells, neutrophils, and macrophages. 4,17 An ultrastructural study of the myocardium of cats with HCM identified degenerative changes, including mitochondrial damage, of cardiomyocytes and abundant extracellular matrix deposition in the interstitium. 6
With age, the human heart undergoes various functional and morphological changes, including a thickening of the left ventricular free wall (LV), gradual decline of the number of cardiomyocytes accompanied by an increase of collagen, fibrotic changes in the valves, and vascular remodeling with progressive intimal thickening. 9 Similarly striking processes have not been reported in the myocardium of old cats without cardiac disease. However, we have previously shown that a range of genes for inflammatory and remodeling mediators are actively transcribed in the normal feline myocardium, with a shift from a pro-inflammatory state in young age that is more pronounced in male cats to a pro-fibrotic state with increasing age. 10 Considering these results and the plethora of partly contradictory findings in the myocardium of cats with HCM, we hypothesized that feline HCM is associated with progressive remodeling processes to which age contributes. Therefore, the aim of the present study was to systematically assess the morphological changes and their underlying mechanisms in HCM to gain a better understanding of the complex pathophysiology of this disease.
Materials and Methods
Animals and Tissue Sampling
The study was conducted on the hearts of 18 cats with HCM and 18 control cats (ie, cats that had died or were euthanized with diseases not involving the heart). The group of HCM cats (Supplemental Table S1) comprised 1 female, 2 male, and 15 male neutered cats, with a mean age of 7.6 ± 3.1 years (mean ± standard deviation) and a mean body weight of 5.6 ± 1.8 kg. The control group (Supplemental Table S2) comprised 1 female, 11 female neutered, 2 male, and 4 male neutered cats, with a mean age of 7.7 ± 4.8 years and a mean body weight of 3.9 ± 1.1 kg.
All HCM cats underwent a full postmortem examination with owners’ consent. Two-thirds (12/18) had been clinically confirmed and had undergone echocardiography carried out by a specialist in veterinary cardiology. The remaining 6 cases had been submitted for necropsy due to sudden death and clinical suspicion of cardiac disease. All 6 cats showed marked left ventricular hypertrophy and left atrial dilation consistent with previously reported gross features of HCM, 11 in the absence of gross and histological changes suggestive of systemic disease. The majority of the control animals (12/18) had also undergone a full postmortem examination, the remaining third (6/18) were only available for a partial necropsy, and the diagnosis relied on the clinical examination and the gross assessment of the heart by the cardiologist.
Samples for histology were collected from the right ventricular free wall (RV), LV, and interventricular septum (IVS) and fixed in 10% neutral-buffered formalin. To obtain the samples, the apex of the heart (lower half) was separated by a cross-section, and a sample from each of the mid LV, IVS, and RV towards the heart base was obtained. One cross-sectional slice of the removed apex was processed for histology after fixation, in addition to longitudinal sections of the LV, IVS, and RV. To ensure optimal complementarity, samples for quantitative polymerase chain reaction (qPCR) were collected next to these areas in cats whose hearts had been removed within 1 h after death (11 HCM and 10 control animals) (Table 1). The latter samples from each cardiac location were placed in RNAlater (Ambion, Life Technologies, Paisley, UK) and frozen at –80°C for subsequent RNA extraction. 10 Of the 11 HCM cats examined by reverse transcription—qPCR (RT-qPCR), 10 were male neutered and 1 was male intact. The cats had a mean age of 8.5 ± 3.2 years and a mean body weight of 5.3 ± 1.8 kg. The 10 control cats examined by RT-qPCR were female neutered animals, except 1 male and 1 male neutered. Their mean age was 10.0 ± 5.1 years, and the mean weight was 4.1 ± 1.3 kg; both were similar to those of the HCM cats (age: P = .215, weight: P = .115).
Antibodies and Immunohistological Methods.
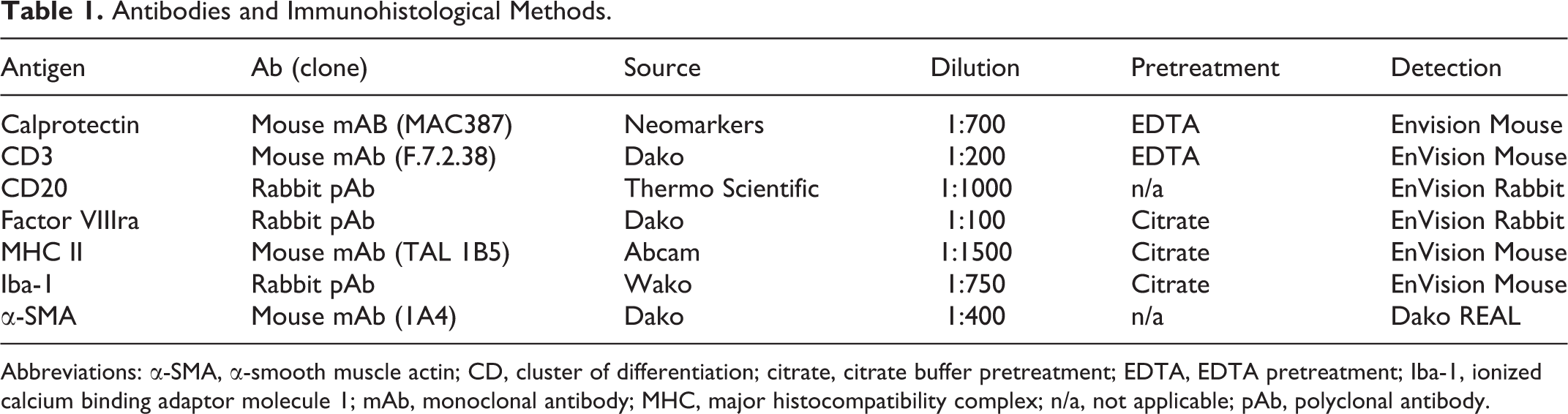
Abbreviations: α-SMA, α-smooth muscle actin; CD, cluster of differentiation; citrate, citrate buffer pretreatment; EDTA, EDTA pretreatment; Iba-1, ionized calcium binding adaptor molecule 1; mAb, monoclonal antibody; MHC, major histocompatibility complex; n/a, not applicable; pAb, polyclonal antibody.
Histology, Immunohistochemistry, and Fluorescence Examinations
After formalin fixation, myocardial samples were trimmed and embedded in paraffin wax, and consecutive sections (3–5 µm) were prepared and routinely stained with HE and van Gieson stain for the demonstration of collagen deposition, or they were subjected to immunohistochemical and fluorescence staining. Immunohistochemistry was performed using a Dako autostainer (Dako, Glostrup, Denmark) or the Discovery XT autostainer (Ventana Medical System, Inc, Tucson, Arizona, USA) and was applied to detect CD18 and MHC II (leukocytes), CD3 (T cells), CD20 (B cells), Iba-1 (macrophages) and calprotectin (recently blood-derived macrophages), factor VIII–related antigen (Factor VIII) (endothelial cells), and α–smooth muscle actin (α-SMA) (smooth muscle cells). Antibodies and the respective antigen retrieval and detection methods are listed in Table 1. Briefly, after deparaffinization, antigen retrieval was performed for all antigens except for α-SMA, by incubation of the slides with citrate buffer (pH 6) at 98°C for 10 minutes or EDTA buffer (pH 9) at 98°C for 20 minutes. Endogenous peroxidase was blocked by incubation with hydrogen peroxide solution for 10 minutes. Slides were incubated with equine serum for 30 minutes at room temperature followed by the primary antibodies and matching secondary antibodies; different detection kits were then applied (Table 1). Sections were washed with phosphate buffered saline between each incubation step (pH 8). Finally, sections were counterstained with hematoxylin for 40 seconds and mounted. Consecutive sections incubated with an isotype-matched irrelevant antibody or without the primary antibody served as negative controls. A further section underwent fluorescence staining to mark cell nuclei, using the ProLong Diamond Antifade Mountant with DAPI (4’, 6-diamidino-2-phenylindole; Invitrogen, Carlsbad, California, USA). Briefly, after deparaffinization and rehydration, sections were incubated with DAPI for 24 hours at room temperature in the dark.
Transmission Electron Microscopy (TEM)
Additional myocardial samples from 2 HCM cases (Cases 1.8 and 1.9) (Supplemental Table S1) were fixed for 24 hours in 5% glutaraldehyde, buffered in 0.2 M cacodylic acid buffer, pH 7.3, for TEM, then trimmed and routinely embedded in epoxy resin. Toluidine blue–stained semithin sections (1.5 μm) were prepared to select areas of interest for the preparation of ultrathin sections (75 nm) that were contrasted with lead citrate and uranyl acetate and viewed with a Philips CM10, operating with a Gatan Orius Sc1000 digital camera (Gatan Microscopical Suite, Digital Micrograph).
Morphometric Analyses
A morphometric approach was taken to quantify the overall cellularity of the myocardium, the interstitial cellularity, as well as the amount of interstitial collagen and the interstitial space in HCM in comparison to the myocardium of control cats.
Slides stained for Iba-1 and α-SMA with the van Gieson stain and for DAPI fluorescence were scanned using a digital slide scanner (NanoZoomer-XR C12000; Hamamatsu, Hamamatsu City, Japan) and evaluated with the computer program VIS (Visiopharm Integrator System, Version 5.0.4. 1382; Visiopharm, Hoersholm, Denmark). For all quantitative approaches, 20 regions of interest (ROIs) with a size of 2.37 mm2 (the area of a high power field with one ocular of 22 mm field of view) were randomly selected in the LV, RV, and the IVS of all hearts. The van Gieson stain served to assess the percentage of space occupied by collagen, the percentage of interstitial empty space (ie, cell- and fiber-free space), and the percentage of space occupied by cardiomyocytes (ie, contractile tissue). A threshold classification allowed recognition of fibrous tissue (bright pink), cardiomyocytes (yellow), and interstitial empty space (empty) in each ROI, and the results were expressed as percentage of assessed tissue per ROI.
Sections stained for Iba-1 were used to quantify the number of macrophages in the interstitium. A threshold classification allowed recognition of positive (intense brown staining of the entire cytoplasm) and negative cells in each ROI, and the results were expressed as the absolute number of positive nuclei per ROI. In a post-processing step, very small nuclei (nuclear area < 5 μm2) were excluded from counting in order to avoid falsely classifying areas of increased background staining as nuclei. Nuclei that occurred in groups were often mistakenly classified as only one nucleus by the program and were therefore excluded from automated counting (nuclear area > 120 μm2). Sections stained for α-SMA were used to determine the number of small- to medium-sized vessels (identified as α-SMA-positive vascular rings). Sections stained with DAPI fluorescence served to assess the number of nuclei and thereby the overall cellularity of the myocardium. The intensity of the DAPI channel in NDP.view was set at 150% with a γ-value of 1.5 for all scans. A median unsharp filter (x = 55, y = 55) was added to the blue color channel (RGB-B) as a pre-processing step to remove background variation. With this method, the nuclei of all cell types were counted (cardiomyocytes, interstitial cells, vascular endothelial cells, and smooth muscle cells) and expressed as the number per ROI.
RT-qPCR for Relevant Remodeling Mediators
For the amplification of the cytokines interleukin (IL)–1, IL-2, IL-4, IL-6, IL-8, and IL-18; tumor necrosis factor (TNF)–α; transforming growth factor (TGF)–ß; interferon (IFN)–γ; the matrix metalloproteinases (MMP)–2, -3, and -13; and the tissue inhibitors of matrix metalloproteinases (TIMP)–1, -2, and -3, published primer pairs and protocols were applied. 10
Statistical Analysis
Data from morphometric measurements and RT-qPCR were entered into spreadsheets, and statistical analysis was performed using a commercially available software package (IBM SPSS Statistics 21, Portsmouth, UK). Basic descriptive statistics (mean, median, variance, standard deviation, interquartile range, and confidence interval) were calculated for the variables recorded. Distribution of data was analyzed applying graphical Q-Q plots, Kolmogorov Smirnov, and Shapiro-Wilk analyses. Data from morphometric measurements and RT-qPCR results were not normally distributed and log-transformed to improve normality and the model assumptions necessary for parametric analysis. Results between groups and regions were subsequently compared using 1-way analysis of variance with Tukey post hoc test and using unpaired t-tests. Scatter plots and linear regression analysis was used to investigate the relationship between data from morphometric measurements and age. No correction for multiple comparisons was obtained, and results are displayed as mean and standard deviation. Statistical significance was defined as P < .05.
Results
Study Population
While both study groups were of similar age, HCM cats had significantly greater body weights than control cats (5.6 kg vs 3.9 kg; P = .001). The 3 HCM cats that had a left atrial thrombus (1.7, 1.10, 1.11) were older than the 15 HCM cats without an atrial thrombus (10 ± 2.6 years vs 7.1 ± 2.9 years; mean ± SD).
The Composition of the Feline Myocardium Is Influenced by Age
The unaltered myocardium (RV, LV, IVS) of the control cats was morphometrically assessed for cellularity, number of interstitial vessels (α-SMA+) and macrophages (Iba-1+), percentage of area consisting of cardiomyocytes (ie, contractile tissue), and interstitium (Tables 2 –4). There were no regional differences in these parameters among sections of RV, IVS, and LV (Table 3). However, age of the cats was correlated with several parameters (Table 4). Specifically, compared to younger cats, older cats had a reduced overall cellularity (DAPI), proportion of contractile tissue (cardiomyocytes), number of small vessels (α-SMA), macrophages (Iba-1) in the interstitium, and a reduced amount of interstitial collagen (van Gieson stain) when they suffered from HCM (Table 4).
Overall Cellular and Matrix Composition of the Myocardium in 18 Control Cats and 18 Cats With Hypertrophic Cardiomyopathy (HCM).a
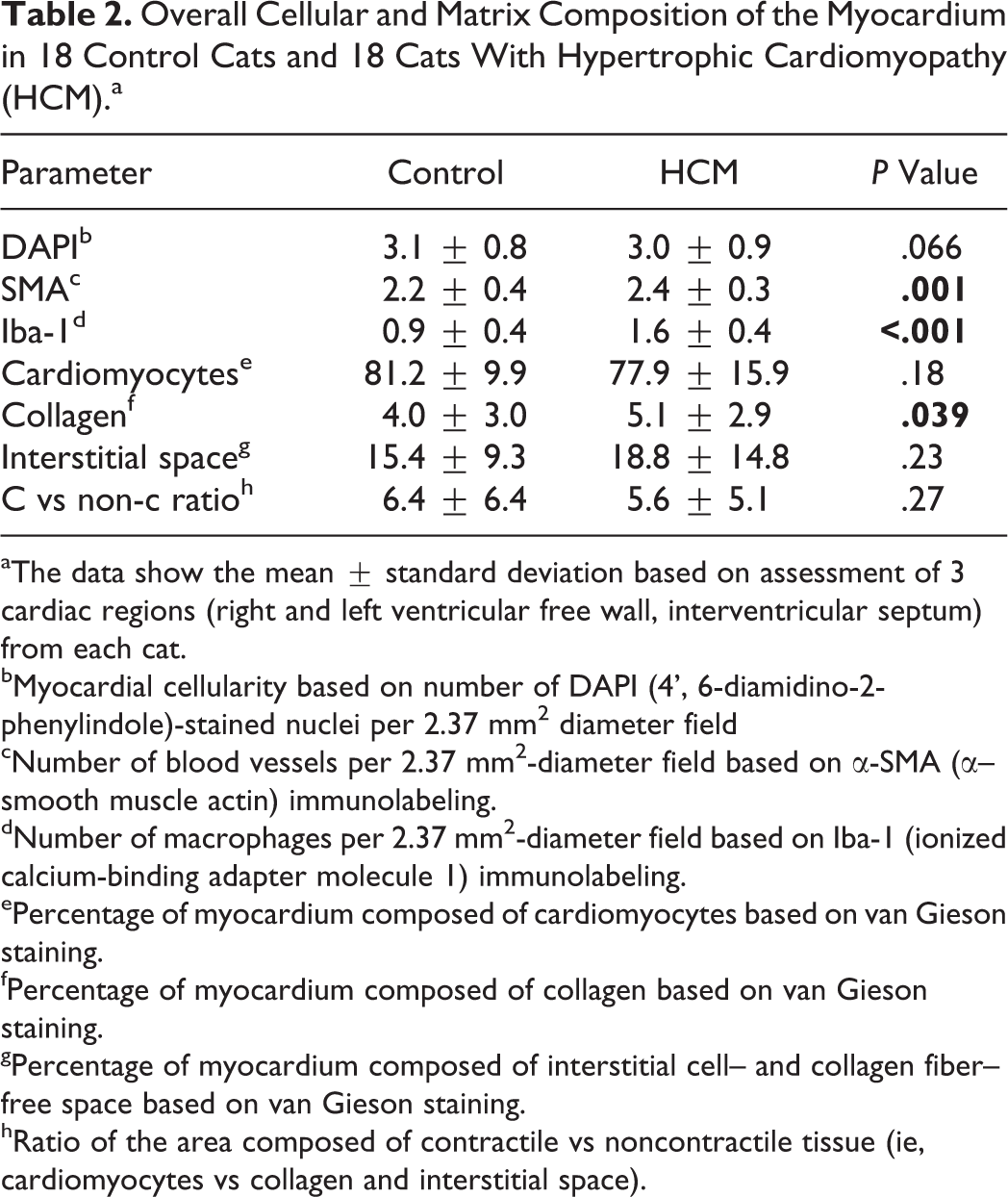
aThe data show the mean ± standard deviation based on assessment of 3 cardiac regions (right and left ventricular free wall, interventricular septum) from each cat.
bMyocardial cellularity based on number of DAPI (4’, 6-diamidino-2-phenylindole)-stained nuclei per 2.37 mm2 diameter field
cNumber of blood vessels per 2.37 mm2-diameter field based on α-SMA (α–smooth muscle actin) immunolabeling.
dNumber of macrophages per 2.37 mm2-diameter field based on Iba-1 (ionized calcium-binding adapter molecule 1) immunolabeling.
ePercentage of myocardium composed of cardiomyocytes based on van Gieson staining.
fPercentage of myocardium composed of collagen based on van Gieson staining.
gPercentage of myocardium composed of interstitial cell– and collagen fiber–free space based on van Gieson staining.
hRatio of the area composed of contractile vs noncontractile tissue (ie, cardiomyocytes vs collagen and interstitial space).
Composition of the Myocardium in Different Cardiac Regions of Cats With and Without Hypertrophic Cardiomyopathy (HCM).a
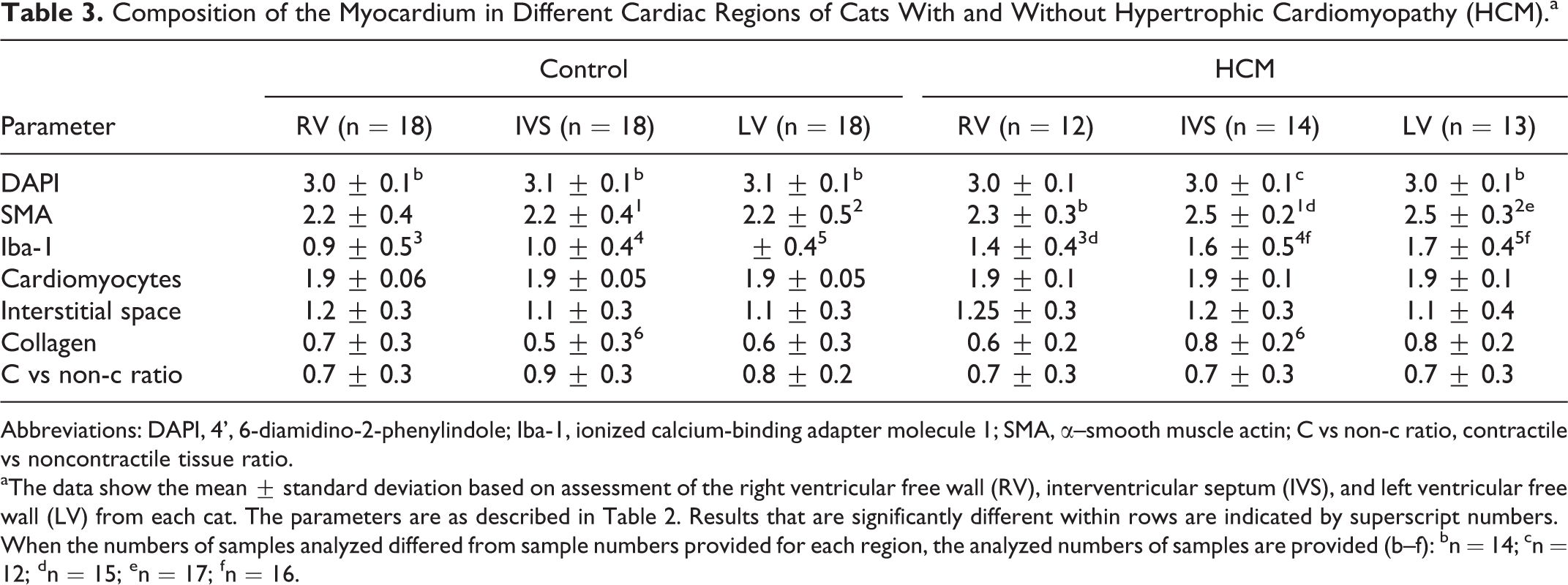
Abbreviations: DAPI, 4’, 6-diamidino-2-phenylindole; Iba-1, ionized calcium-binding adapter molecule 1; SMA, α–smooth muscle actin; C vs non-c ratio, contractile vs noncontractile tissue ratio.
aThe data show the mean ± standard deviation based on assessment of the right ventricular free wall (RV), interventricular septum (IVS), and left ventricular free wall (LV) from each cat. The parameters are as described in Table 2. Results that are significantly different within rows are indicated by superscript numbers.
When the numbers of samples analyzed differed from sample numbers provided for each region, the analyzed numbers of samples are provided (b–f): bn = 14; cn = 12; dn = 15; en = 17; fn = 16.
Correlation of Parameters of Myocardial Composition With Age in Control Cats (n = 18) and Cats With HCM (n = 18), Based on the Available Data for All 3 Cardiac Regions.a
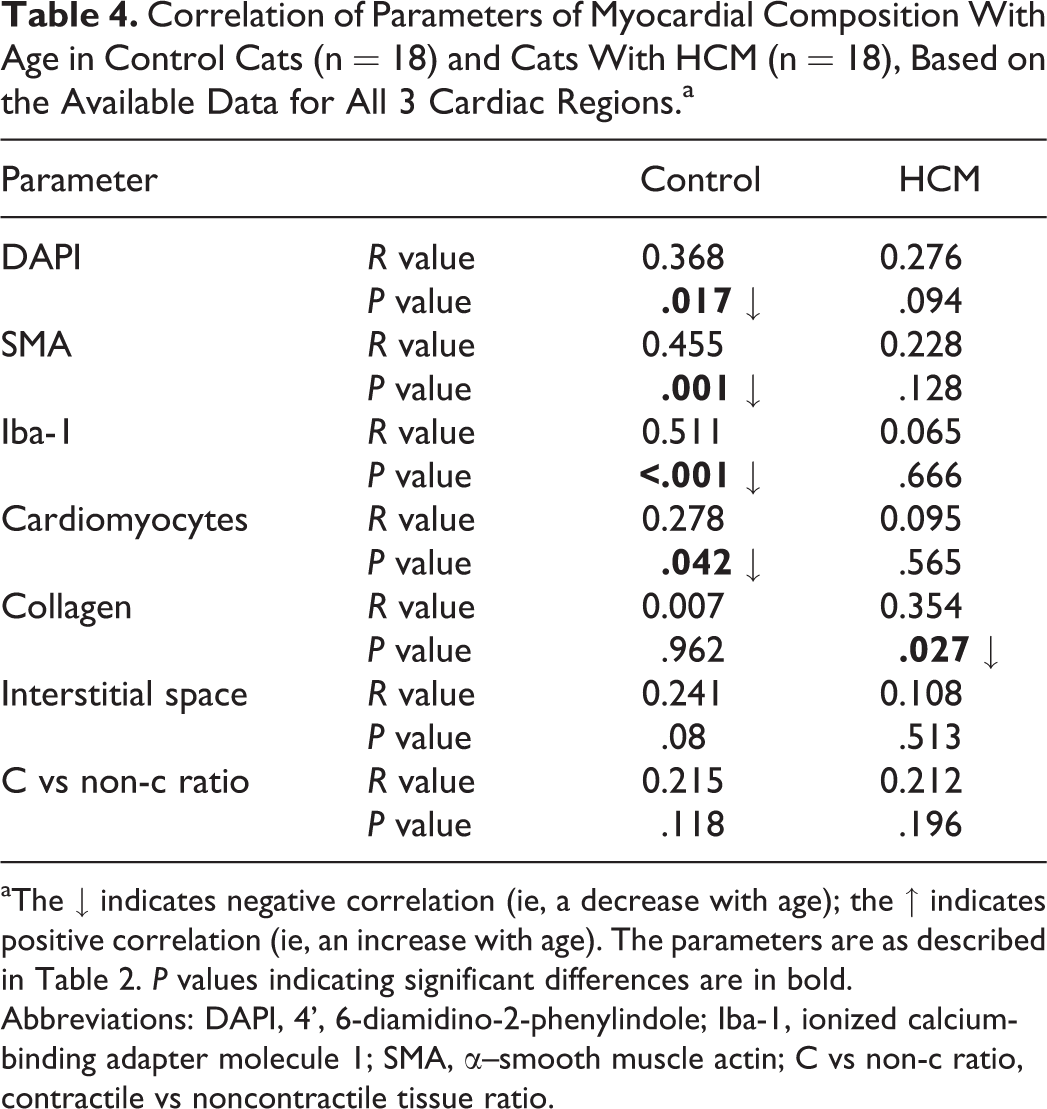
aThe ↓ indicates negative correlation (ie, a decrease with age); the ↑ indicates positive correlation (ie, an increase with age). The parameters are as described in Table 2. P values indicating significant differences are in bold.
Abbreviations: DAPI, 4’, 6-diamidino-2-phenylindole; Iba-1, ionized calcium-binding adapter molecule 1; SMA, α–smooth muscle actin; C vs non-c ratio, contractile vs noncontractile tissue ratio.
HCM Is Associated With an Increase in Interstitial Macrophages and Small- to Medium-Sized Vessels and a Regional Increase in Interstitial Collagen
All hearts were screened for histological features described in HCM, that is, cardiomyocyte disarray and degeneration, leukocyte infiltration, and interstitial fibrosis. 3,4,12,17,20 Some or all of these features were observed in most HCM cases (15/18; 83%) (Supplemental Table S3). There was patchy to disseminated mild multifocal loss and/or degeneration of cardiomyocytes, the latter represented by loss of striations and occasional intracytoplasmic microvacuolization (Fig. 1), and variably intense, slight to mild multifocal interstitial collagen deposition (Figs. 2, 3) together with the presence of spindle-shaped cells (fibroblasts) (Fig. 2) that were occasionally α-SMA positive (myofibroblasts).
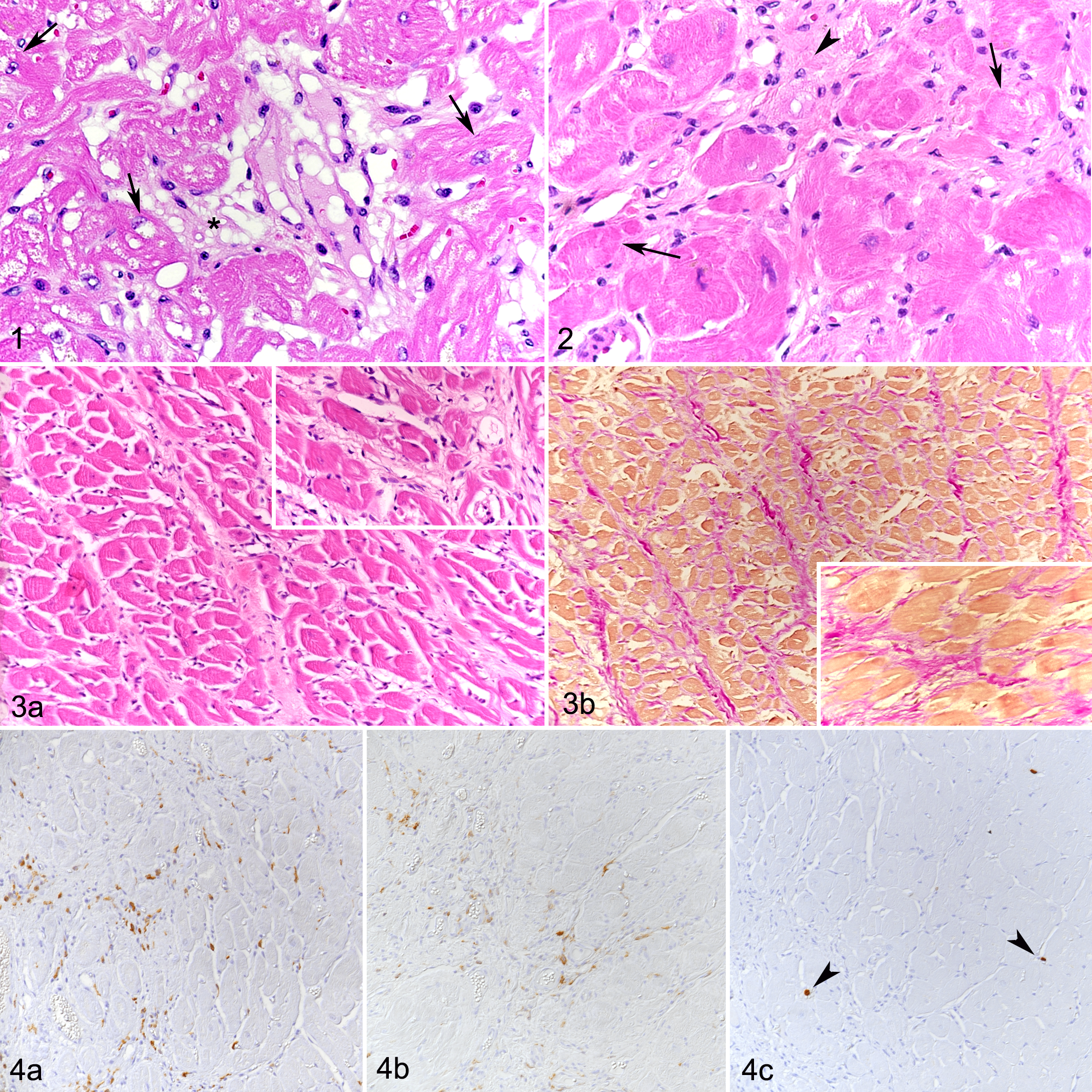
Hypertrophic cardiomyopathy (HCM), left ventricular myocardium, cat.
In addition, all HCM cases exhibited evidence of mild diffuse expansion of the interstitium (Fig. 1), occasional interstitial accumulations of mature adipocytes (fatty infiltration), and increased interstitial cellularity (Figs. 2, 3). The latter was mainly due to the presence of mononuclear cells with macrophage morphology, that is, round- to spindle-shaped cells with a moderate to high amount of cytoplasm and an oval to round nucleus (Fig. 2). The majority of these cells exhibited strong Iba-1 (Fig. 4a) and MHC II expression (Fig. 4b) but were calprotectin-negative (Fig. 4c), suggesting that they were resident or formerly blood-derived macrophages that had proliferated locally, rather than recently blood-derived macrophages. 34 A few individual cells in the interstitium were identified as T cells (CD3+) and B cells (CD20+). Occasional small- to medium-sized intramural vessels showed mild medial hypertrophy, without significant narrowing of the lumen.
The quantitative assessment (Tables 2, 3) confirmed a significant increase in interstitial Iba-1-positive cells in HCM cats across all 3 assessed regions (LV, IVS, RV) and an increase in interstitial small- to medium-sized vessels, which was significant in IVS and LV but not RV. As well, the amount of interstitial collagen was significantly higher in the IVS of HCM cats compared to controls.
In HCM, the Myocardium Frequently Exhibits Focal Areas of Cardiomyocyte Replacement by Cell-Rich Fibrous Tissue
In 8 of the 18 HCM hearts (44%), the myocardium exhibited one or more poorly delineated areas where cardiomyocytes were replaced by a cell-rich infiltrate (Fig. 5). The periphery of these areas contained degenerate cardiomyocytes (ie, with shrinkage, loss of striation, hypereosinophilia, intracytoplasmic vacuolization) and rare necrotic cardiomyocytes (Fig. 6). The core of these lesions contained densely packed collagen bundles with spindle-shaped cells (fibroblasts) and mononuclear cells (Fig. 7). The mononuclear cells had a morphology similar to those found in increased numbers in the interstitium, and they were Iba-1-positive (Fig. 8a) and MHC II-positive but calprotectin-negative. Scattered T cells (CD3+) and B cells (CD20+) were also present. Between the Iba-1-positive cells were abundant small- to medium-sized vessels, confirmed by the ring-like structures containing Factor VIII–positive endothelial cells, as well as α-SMA-positive pericytes and smooth muscle cells of the vascular wall (Fig. 8b). A few individual spindle-shaped cells expressed α-SMA (myofibroblasts) (Fig. 8b). Degeneration of individual cardiomyocytes in the periphery of the focal lesions was confirmed by TEM (Figs. 9–12). Individual or multiple cardiomyocytes were embedded and walled off by dense and partially irregularly arranged collagen fiber bundles (Figs. 10, 11) and showed myofiber disarray and disruption of intercalated discs with marked disorganization and interdigitation of Z-lines (Fig. 12).
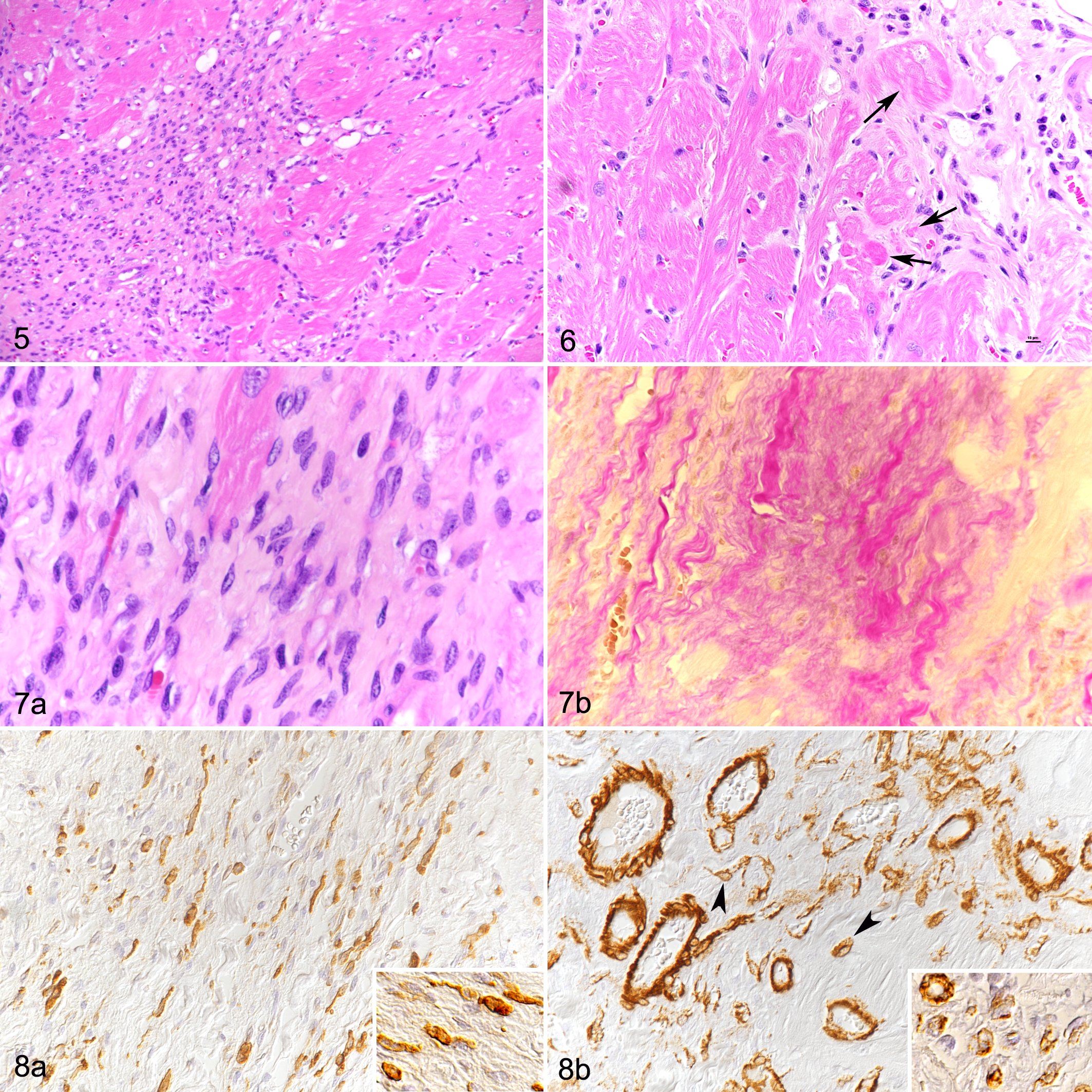
Hypertrophic cardiomyopathy (HCM), left ventricular myocardium, cat.
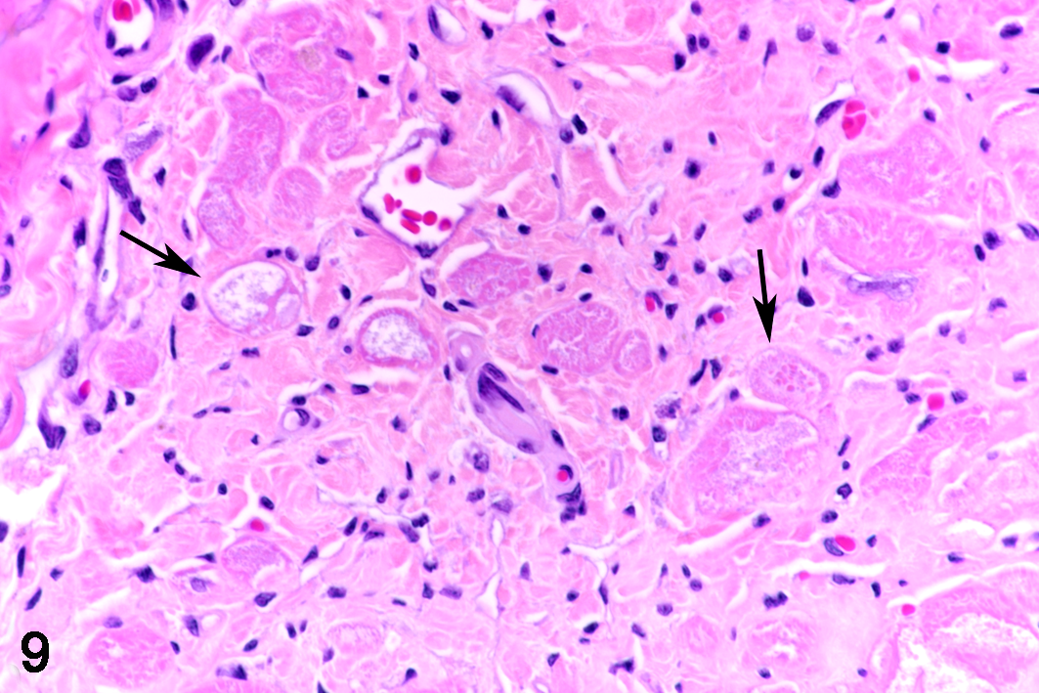
Hypertrophic cardiomyopathy (HCM), left ventricular myocardium, cat, case 1.9. Focal lesion. At the periphery, individual cardiomyocytes embedded among collagen fibers display degenerative changes, that is, shrinkage, loss of striation, hypereosinophilia, vacuolization, and fragmentation of the cytoplasm (arrows). HE.
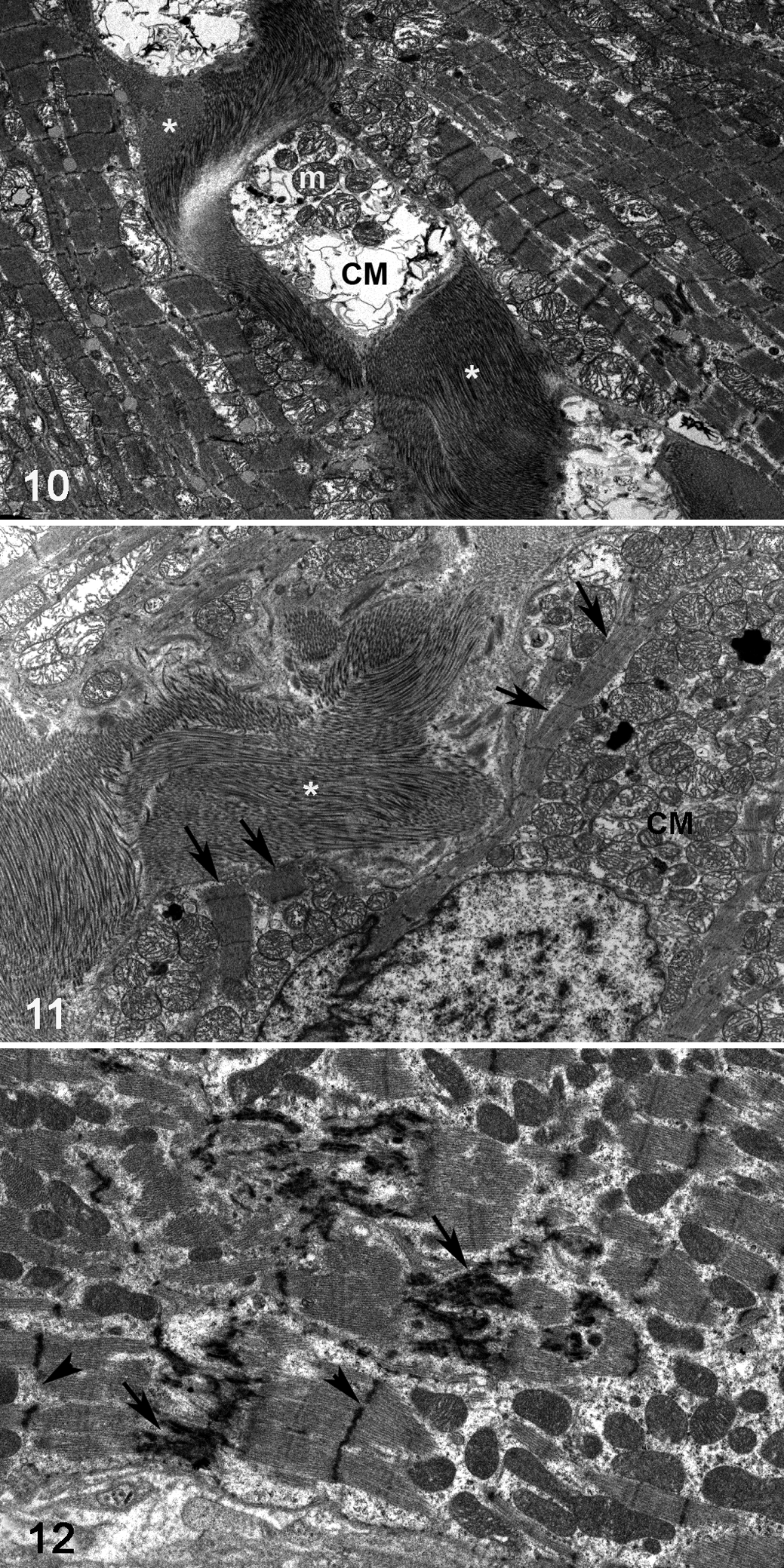
Hypertrophic cardiomyopathy (HCM), left ventricular myocardium, cat, case 1.9. Focal lesion. Transmission electron microscopy.
Pro-inflammatory and Pro-fibrotic Mediators Are Up-Regulated in the Myocardium of HCM Cats
RT-qPCR results confirmed the constitutive expression of cytokines, MMPs, and TIMPs in the myocardium of all cats. However, compared to the controls, cats with HCM showed significantly higher transcription levels for IL-1, IL-6, IL-18, TNF-α, TGF-β, MMP-13, and TIMP-1 (Table 5). As previously reported, control cats exhibited a moderate positive correlation of age and myocardial TGF-β transcription and a moderate negative correlation of IL-4 mRNA levels and age. 10 For HCM cats, a strong negative correlation with age was observed for IL-8, MMP-2, and MMP-13; a moderate negative correlation for IFN-γ, TGF-β, MMP-3, and TIMP-2; and a weak negative correlation for IL-2 and IL-4 (Table 6).
Transcription of Cytokine Genes and Genes Involved in Remodeling in the Myocardium of Cats With and Without Hypertrophic Cardiomyopathy.a
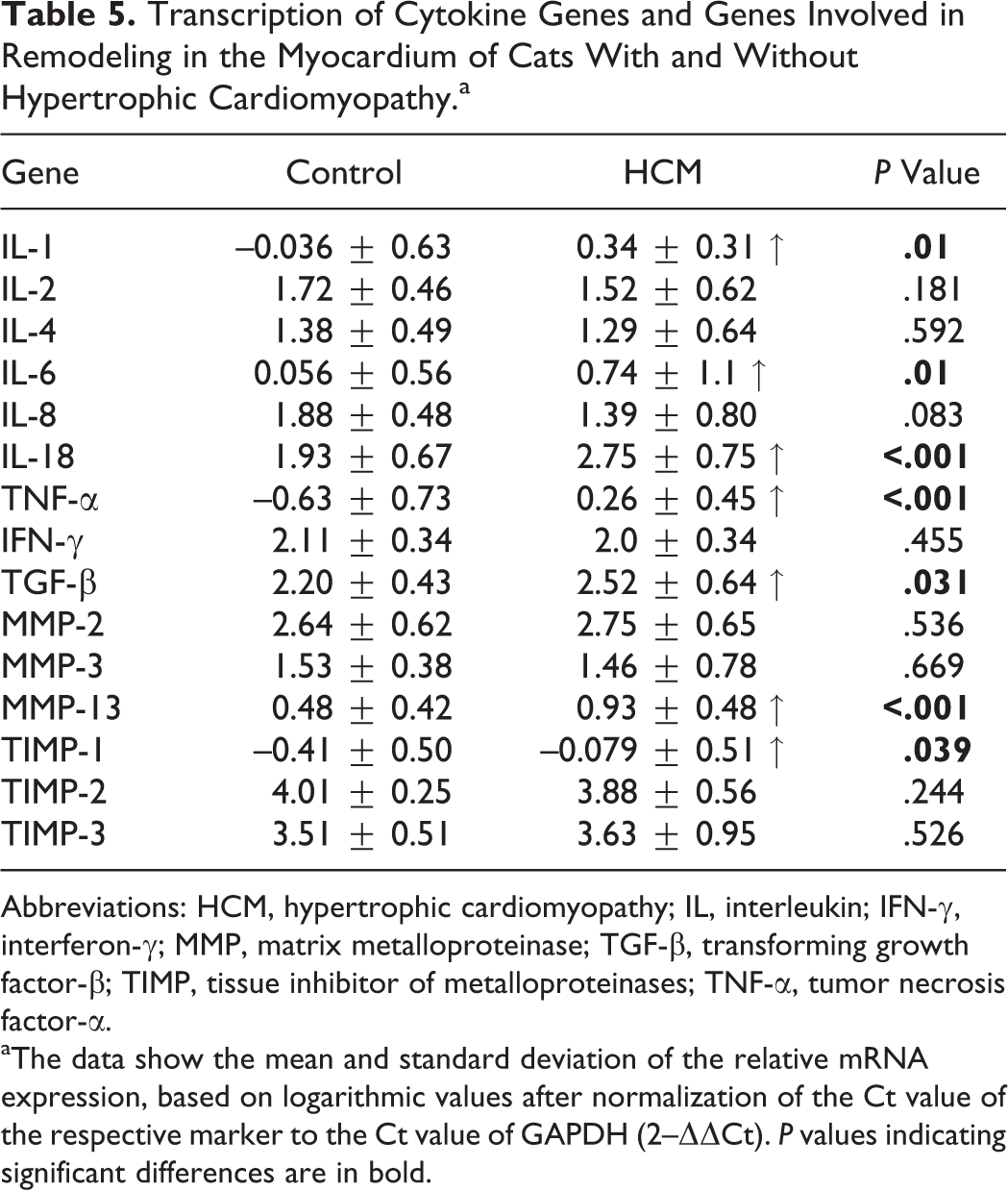
Abbreviations: HCM, hypertrophic cardiomyopathy; IL, interleukin; IFN-γ, interferon-γ; MMP, matrix metalloproteinase; TGF-β, transforming growth factor-β; TIMP, tissue inhibitor of metalloproteinases; TNF-α, tumor necrosis factor-α.
aThe data show the mean and standard deviation of the relative mRNA expression, based on logarithmic values after normalization of the Ct value of the respective marker to the Ct value of GAPDH (2–ΔΔCt). P values indicating significant differences are in bold.
Correlation of Relative mRNA Expression of Cytokine Genes and Genes Involved in Remodeling With Age in Control Cats and Cats With HCM, Based on the Available Data for All 3 Cardiac Regions.a
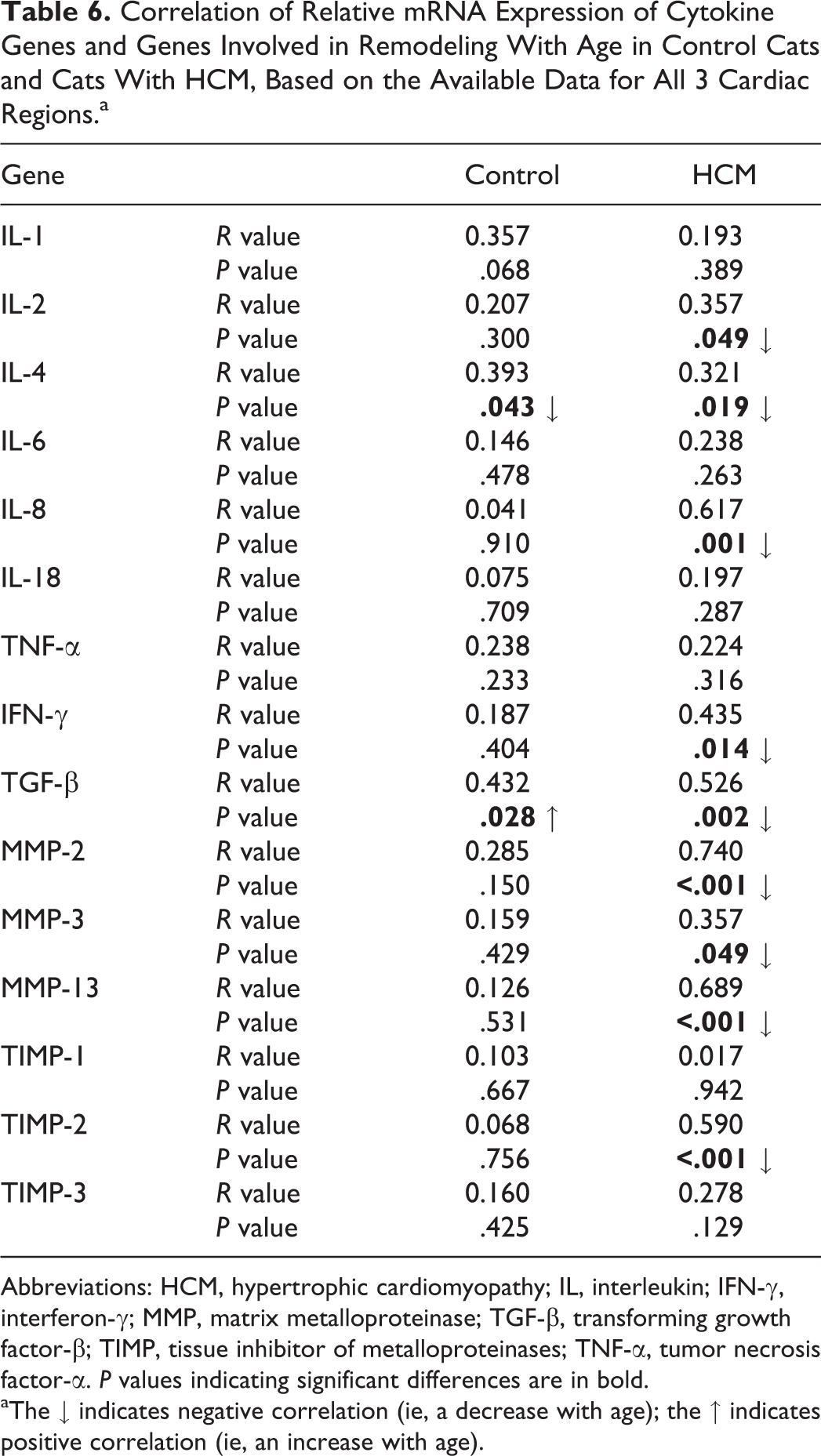
Abbreviations: HCM, hypertrophic cardiomyopathy; IL, interleukin; IFN-γ, interferon-γ; MMP, matrix metalloproteinase; TGF-β, transforming growth factor-β; TIMP, tissue inhibitor of metalloproteinases; TNF-α, tumor necrosis factor-α. P values indicating significant differences are in bold.
aThe ↓ indicates negative correlation (ie, a decrease with age); the ↑ indicates positive correlation (ie, an increase with age).
Discussion
Feline HCM is a disease with a complex pathophysiology that is yet far from clear.
We have recently shown that the normal myocardium of cats actively transcribes a range of inflammatory and remodeling mediators, with a clear age- and sex-related variation and a shift from a pro-inflammatory state in young age to a pro-fibrotic state with increasing age; the former being more pronounced in male cats. 10 Since HCM is a disease particularly of older, intact or neutered male cats, 1,25,29 we aimed to further investigate the pathological features of the myocardium in HCM with particular emphasis on the presence and extent of remodeling processes.
Grossly, all HCM hearts exhibited the typical diffuse thickening of the LV and/or the IVS and dilation of the left atrium. 11 Histopathology confirmed the presence of several previously described features, that is, cardiomyocyte disarray and degeneration and interstitial fibrosis, as well as leukocyte infiltration, in most cases. 2 –4,17,20 Other studies described marked medial hypertrophy, fibrosis, and elastosis of intramural vessels in feline HCM. 12,20 These features were not prominent in our HCM cohort where the vascular changes were restricted to mild medial hypertrophy in small- to medium-sized intramural vessels. However, the present study identified several additional quantitative changes affecting the interstitium that were rather subtle but consistent and statistically significant: an overall increase in the numbers of Iba-1-positive mononuclear cells, the numbers of small- and medium-sized vessels, and the amount of collagen. The differences were most reliably demonstrated in the IVS, followed by the LV, whereas in the RV the quantitative changes were restricted to the increased amount of interstitial Iba-1-positive cells. Interestingly, the increase in interstitial cells was not associated with an increase in overall cell numbers, suggesting a reduction of cardiomyocytes, which is indirect evidence of some degree of cardiomyocyte injury and loss.
Immunohistochemistry confirmed the mononuclear interstitial cells as macrophages, as these were in the vast majority not only Iba-1- but also MHC II–positive. The lack of calprotectin expression suggests that they were resident or locally proliferated macrophages, rather than recently recruited from the blood into the myocardium. 34 T cells and B cells were not more numerous in HCM than in the control myocardium.
The focal lesions, which were found in addition to the diffuse changes in a large proportion of HCM hearts, were most prominent in IVS and LV, that is, the regions that show the characteristic thickening and were found to exhibit the most severe diffuse changes. These focal infiltrates, with their core of fibrous tissue and abundant embedded macrophages and new vessels, were reminiscent of infarct-like lesions and disorderly arranged granulation tissue. 4,7,13 The presence of dying cardiomyocytes in the periphery, where they seemed to be walled off by collagen fibers, suggests that these infiltrates were actively expanding. Their random distribution indicates that they are initiated by focal tissue damage, that is, cardiomyocyte death potentially caused by ischemia.
The findings suggest that HCM is not a primary inflammatory condition that occurs as a consequence of persistent recruitment and interaction of inflammatory cells in the myocardium. Instead, also in light of the significant increase in interstitial small vessels and the diffuse interstitial collagen deposition, a diffuse macrophage-driven remodeling process is suspected. The myocardium itself could initiate the recruitment and proliferation of interstitial macrophages, representing a secondary inflammatory component. Cardiomyocytes are a known source of cytokines, constitutively transcribing inflammatory and profibrotic mediators (eg, IL-1, Il-6, TNF-α, INF-γ, and TGF-β). 10 Expression of these genes can be enhanced by cardiomyocyte injury. 27 Activated macrophages could then release angiogenic and fibrogenic mediators that induce the formation of new interstitial vessels, fibroblast proliferation and activation, development of myofibroblasts, and deposition of interstitial collagen. Indeed, we found a significant increase in the transcription of inflammatory (IL-1, IL-6, IL-18, TNF-α) and profibrotic (TGF-β, MMP-13, and TIMP-1) mediators in HCM. Involvement of inflammatory cytokines in myocardial remodeling in feline HCM is therefore likely, and both the cardiomyocytes and abundant macrophages might be the sources. 10,14,33
New vessel formation, fibroblast proliferation, and collagen deposition in the focal lesions in HCM are all key events of granulation tissue formation. 19 Angiogenesis is fundamental for any repair processes and critical for the maintenance of the required blood flow at sites of injury or ischemia. 36 Fibroblasts that migrate into the damaged tissue produce the collagen-rich connective tissue to maintain the structural integrity. 19,36 The observed diffuse increase in interstitial vessels could be due to new vessel formation, which can be triggered by hypoxia. 18 Low oxygen levels lead to up-regulation of various pro-angiogenic pathways promoting increased vessel sprouting. 18 In humans, it has been shown that compared to their normoxic counterparts, cardiomyocytes exposed to constant hypoxia have elevated secretion of inflammatory cytokines, including IL-1, IL-6, and IL-8, as well as certain growth factors and chemokines. 15
The initial cause of myocardial damage in feline HCM that would trigger the observed remodeling processes is still unknown, but hypoxia is a possibility considering the present findings. 4,11 Latent myocardial hypoxia due to so-far unknown structural or functional myocardial alterations could lead to individual cardiomyocyte degeneration and death. With focally pronounced or chronic hypoxia, a larger area of cardiomyocyte necrosis could develop and eventually lead to the observed focal lesions. Here, the release of chemokines and cytokines and the presence of dead cardiomyocytes could induce recruitment and proliferation of macrophages, which subsequently clear the cellular debris and secrete those chemokines and cytokines that are responsible for the other observed processes. 5,30 This hypothesis is supported not only by the observed increase in vessels in our case cohort but also by the fact that a hypoxic environment can induce a macrophage-driven inflammatory response, where recruited, blood-derived monocytes differentiate into macrophages that reside in areas of hypoxic damage. 26
In human HCM patients, myocardial ischemia is the result of changes in the coronary arteriolar microvasculature. 21,22,35 In our cohort, however, there was no evidence of a similar scenario in cats. 12
We have previously shown that the myocardium of young, male cats is in a pronounced pro-inflammatory and potentially primed state. 10 This might provide a favorable environment for cardiomyocyte damage, if appropriately triggered. Alternatively, susceptible cats might exhibit persistent endogenous up-regulation of cytokines, which would render cardiomyocytes more prone to injury even after mild hypoxic events.
We have also shown that the pattern of myocardial transcription shifts from the pro-inflammatory state in young animals to a pro-fibrotic state in age. 10 The morphometric analysis of the present study now adds data on age-related changes in the composition of the myocardium: with age, the cellularity, the amount of interstitial blood vessels and macrophages, and the proportion of contractile tissue decrease, suggesting a decreased reactivity and with it, possibly, decreased repair capacity of the myocardium in older cats. Such impaired cardiac repair, alongside continuous, slow disease progression, could lead to the observed and more pronounced damage with reduced tissue and immune reaction in older animals. This might also promote atrial thrombus formation, as the cats with an atrial thrombus in our HCM cohort were older than those without.
A limitation of the present study is the sex imbalance between the overall case group and the animals in which gene expression was evaluated. Some of the observed differences might reflect sex differences. However, as we compared a primary cardiac disease with macroscopic myocardial changes with cats without cardiac disease that do not show structural cardiac changes, we believe that the observed differences in myocardial gene expression are primarily influenced by HCM and that potential sex differences play a minor role.
In conclusion, the results of our study provide further evidence that the increase in wall thickness and relative cardiac weight in feline HCM is not the result of true myocardial hypertrophy but due to a diffuse expansion of the interstitium by vessels, macrophages, and collagen as a consequence of degeneration and repair processes triggered by a still-unknown cause. In HCM, the hearts seem to exhibit a long-term, progressive myocardial remodeling process. On a cellular level, this process is likely driven by cardiomyocytes and local macrophages. Further studies are required to identify the type and source of these macrophages and further specify their role in the pathogenetic process of feline HCM.
Supplemental Material
Supplemental Material, DS1_VET_10.1177_0300985819837717 - Feline Hypertrophic Cardiomyopathy: The Consequence of Cardiomyocyte-Initiated and Macrophage-Driven Remodeling Processes?
Supplemental Material, DS1_VET_10.1177_0300985819837717 for Feline Hypertrophic Cardiomyopathy: The Consequence of Cardiomyocyte-Initiated and Macrophage-Driven Remodeling Processes? by Sarah Kitz, Sonja Fonfara, Shelley Hahn, Udo Hetzel and Anja Kipar in Veterinary Pathology
Footnotes
Acknowledgements
We thank the technical staff of the Histology Laboratory and the TEM Unit, Institute of Veterinary Pathology, Vetsuisse Faculty, University of Zurich, as well as the staff of the Histology Laboratory, Faculty of Veterinary Medicine, University of Helsinki, for excellent technical assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
