Abstract
Cardiomyopathies have been rarely described in rabbits. Here we report myocardial necrosis of the ventricular wall in rabbits with experimentally induced rabies. Myocardial lesions were found only in rabbits with brain lesions, and the severity of the cardiac lesions was proportional to that of the brain lesions. Neither the frequency nor the cumulative dose of anesthesia was related to the incidence or the severity of the myocardial lesions. The myocardial lesions were characterized by degeneration and/or necrosis of myocardial cells and were accompanied by contraction band necrosis, interstitial fibrosis, and infiltration of inflammatory cells. The brain lesions due to rabies virus infection were most prominent in the cerebral cortex, thalamus, hypothalamus, brainstem, and medulla. Rabies virus antigen was not found in the hearts of any rabbits. Based on these findings, the myocardial lesions were classified as neurogenic cardiomyopathy.
Domestic rabbits fed a high-fat diet develop hypercholesterolemia and atherosclerosis, which makes them a suitable model for cardiovascular research. 8 Natural and experimentally induced cardiomyopathies have been described in rabbits, 5 and the causes of cardiomyopathy included hypovitaminosis E, infectious agents, drugs, and physical trauma. 5 Anesthesia with a combination of xylazine and ketamine has also been reported as a cause of cardiovascular lesions in Dutch belted rabbits. 5 In humans, the brain-heart interaction has been a focus of much research, particularly subarachnoid hemorrhages, which have been shown to play a crucial role in myocardial damage. 3,6,9 However, the brain-heart interaction has not been reported in rabbits. In this study, we describe neurogenic cardiomyopathy in rabbits affected with experimentally induced rabies.
A total of 20 conventionally housed, Encephalitozoon cuniculi–free New Zealand white rabbits (16 weeks old; Japan SLC, Inc., Japan), weighing 2.92 ± 0.18 kg (mean ± SD), were used in this study. Fourteen rabbits (Nos. 1–14) were intramuscularly inoculated in the hindlimb with 2 ml of the challenge virus standard (CVS) strain of rabies virus (RABV) at a dose of 4 × 10 7 focus-forming units (FFU)/ml. The remaining 6 rabbits (Nos. 15–20) were inoculated in the masseter muscle with 0.2 ml of street RABV (strain 1088) at 5 × 10 6 FFU/ml. The rabbits were kept within biosafety level 2 and 3 facilities and fed commercial rabbit food (CLEA rabbit diet CR-3).
The rabbits were observed daily for clinical signs of rabies after virus challenge. They were anesthetized 4 to 6 times using xylazine hydrochloride (2 mg/kg Selactar; Bayer Health Care, Germany) and ketamine hydrochloride (35 mg/kg Ketalar; Daiichi Sankyo Co., Tokyo, Japan) to collect peripheral blood and cerebrospinal fluid (Suppl. Table S1).
Postmortem examinations were done immediately after death or euthanasia. The vital organs, including brain, heart, and lung samples, were systematically collected, fixed in 20% buffered formalin, embedded in paraffin, and cut into 4-μm sections for routine hematoxylin and eosin (HE) staining. The heart sections were also stained with Masson’s trichrome. Immunohistochemistry (IHC) was performed for both brain and heart sections using a streptavidin-biotin-peroxidase system (SAB-PO kit; Nichirei Bioscience, Tokyo, Japan). Monoclonal mouse antibody against rabies virus nucleoprotein (clone N13-27; kindly provided by Dr. Naoto Ito, Gifu University) was used as the primary antibody. These animal experiments were carried out in accordance with the Animal Care and Use Committee of Hokkaido University (approval number 09-0028) and Oita University (approval number M010002).
Thirteen of the 14 rabbits inoculated with the CVS strain of RABV showed nervous signs of rabies, respiratory distress, and decreased food and water consumption within 8 days postinoculation (dpi). Clinical signs progressed and the rabbits exhibited systemic convulsions by 10 dpi. The remaining rabbit (No. 14) showed no clinical signs. Eight of the 13 rabbits died within 3 to 6 days after showing rabies signs. The other 5 rabbits (Nos. 9–13) recovered and survived until the end of the experimental period (25–34 dpi). All rabbits inoculated with street RABV showed transient clinical sign of rabies such as decreased food and water consumption within 9 to 12 dpi and returned to normal within 4 to 5 days thereafter.
Macroscopically, the 2 rabbits that survived the CVS challenge showed multifocal white streaks in the myocardium of the left ventricle and interventricular septum (Fig. 1). The lungs revealed multifocal dark red discoloration with foamy exudate in the trachea of rabbits that died (Nos. 1–8). The other organs were grossly normal.
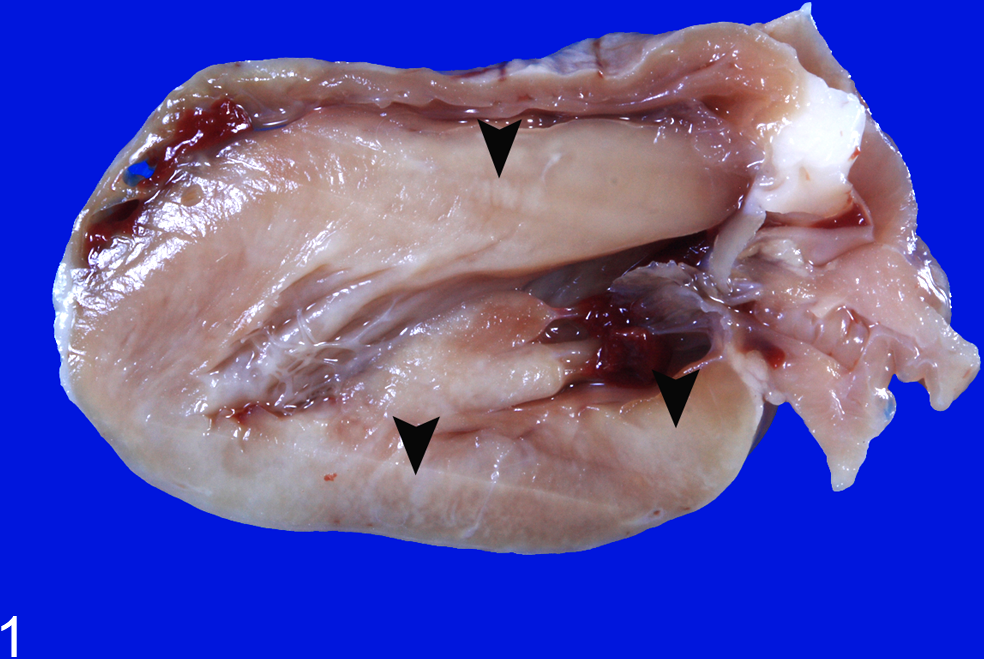
Heart; rabbit No. 12. Formalin-fixed specimen showing multiple white streaks (arrowheads) in the myocardium of the left ventricle and interventricular septum.
Microscopically, the 8 rabbits (Nos. 1–8) that died of the CVS challenge showed neuronal necrosis with occasional neuronophagia (Fig. 2) and a large amount of RABV antigen in the nerve cell bodies/projections (Suppl. Fig. S1) throughout the central nervous system (CNS). These lesions were most severe in the cerebral cortex, thalamus, hypothalamus, ascending nuclei, and reticular formation of the brainstem, cerebellar vermis, dorsal horn and intermediate substance of the gray matter of the spinal cord, and lumbar and sacral dorsal ganglia. Malacic foci were occasionally found in the medulla. In addition, perivascular infiltration by lymphocytes and proliferation/hypertrophy of microglia were observed in these lesions. In the 5 surviving rabbits (Nos. 9–13), neuronal necrosis was most obvious in the Purkinje cells and granular cells of the cerebellar vermis, the pontine reticular nuclei, and the tegmental areas of the brainstem. Small malacic foci, mild astrogliosis, lymphoplasmacytic meningitis, and perivascular cuffing were also found in these areas. RABV antigen was rarely detected in the brains of these rabbits. On the other hand, no brain lesion or RABV antigen was observed in 1 CVS-inoculated rabbit (No. 14) that survived without clinical signs.
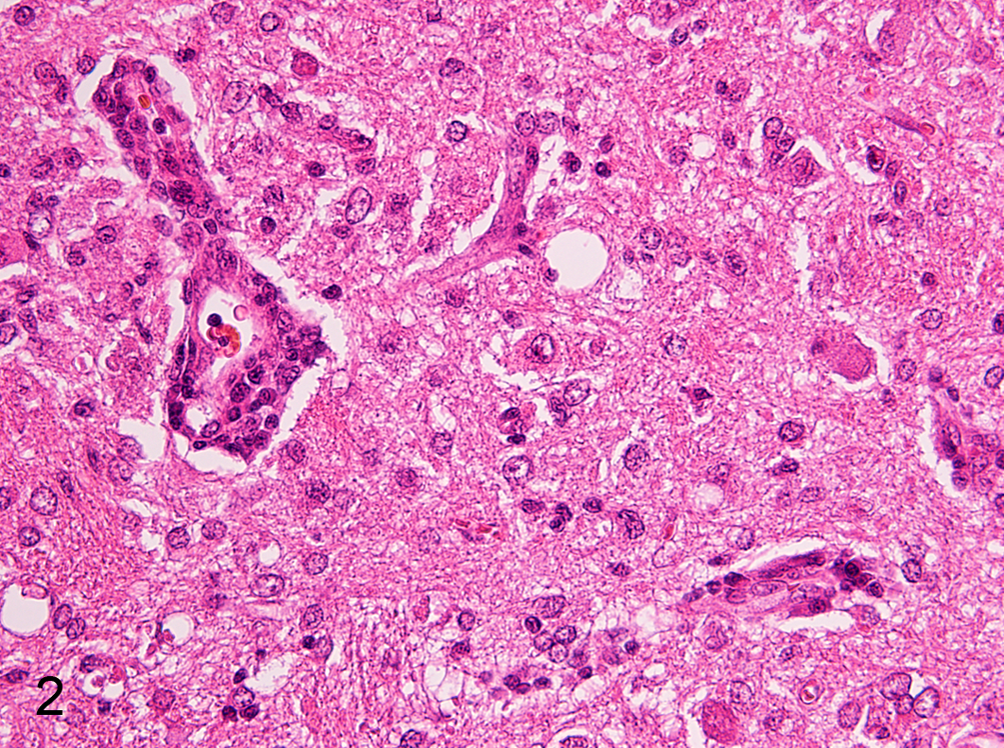
Brainstem; rabbit No. 2. Nonsuppurative encephalitis with encephalomalacia. Hematoxylin and eosin.
In street RABV-inoculated rabbits (Nos. 15–20), mild perivascular infiltration by lymphocytes and plasma cells was seen in the meninges of the brain, but RABV antigen was not detected immunohistochemically in the brains of these rabbits (Suppl. Fig. S1).
Mild to severe, multifocal myocardial necrosis was found in all 8 rabbits (Nos. 1–8) that died of CVS RABV inoculation and in 5 (Nos. 9–13) of the 6 rabbits that survived the challenge. The necrotic foci were mainly distributed in the myocardium of the left ventricle and interventricular septum. The necrotic myocardial cells were characterized by hyalinized and/or vacuolated cytoplasm, loss of cross-striations, and occasional cytoplasmic contraction bands (Fig. 3). The arrangement of myocardial cell was distorted in these areas (Fig. 4). The severity of the myocardial lesions corresponded to the severity of the brain lesions in each rabbit (Suppl. Table S1, Suppl. Fig. S3). Myocardial fibrosis was found only in the surviving rabbits and was not found in rabbits that died. The surviving rabbits persisted for 21 to 28 days after showing neurological signs of rabies, and the magnitude of the fibrosis was consistent with that of myocardial necrosis. The fibrotic tissue of the surviving rabbits was well organized with remarkable production of Masson’s trichrome–positive collagen fibers (Fig. 4). However, the fibrotic tissue invariably contained necrotic myocardial fibers and inflammatory cells, suggesting that the myocardial damage was ongoing. RABV antigen was not detected in the hearts of any of the rabbits. Mild to moderate congestion and edema accompanied with a small number of intra-alveolar hemosiderin-laden macrophages (heart failure cells) were found in rabbits that died (Suppl. Fig. S2), and it appeared also in 5 surviving rabbits (Nos. 9–13) that had myocardial necrosis. Other organs were unremarkable, and none of the rabbits showed either pathological changes or clinical signs suggestive of E. cuniculi infection or vitamin E deficiency.
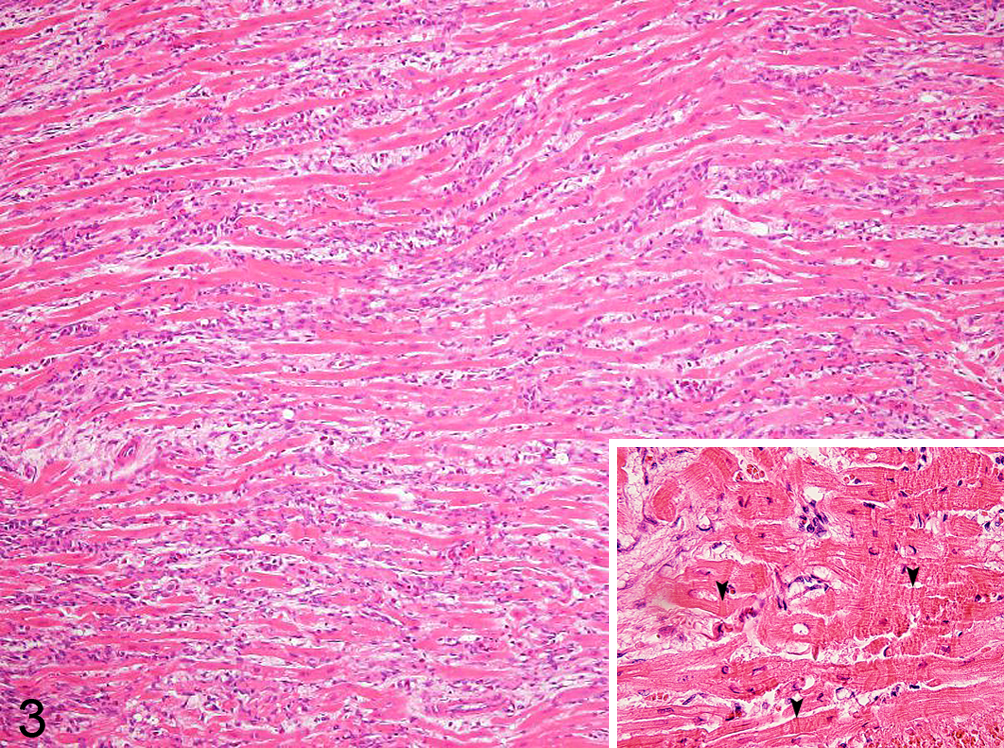
Left ventricle; rabbit No. 11. Myocardial degeneration is characterized by hypereosinophilic and vacuolated cytoplasm and contraction band necrosis. The arrangement of the myocardial cells is distorted. Infiltration of mononuclear cells and heterophils and fibrosis are seen in the interstitial area. Inset: Higher magnification; myocardial cells with contraction band necrosis (arrowheads). Hematoxylin and eosin.
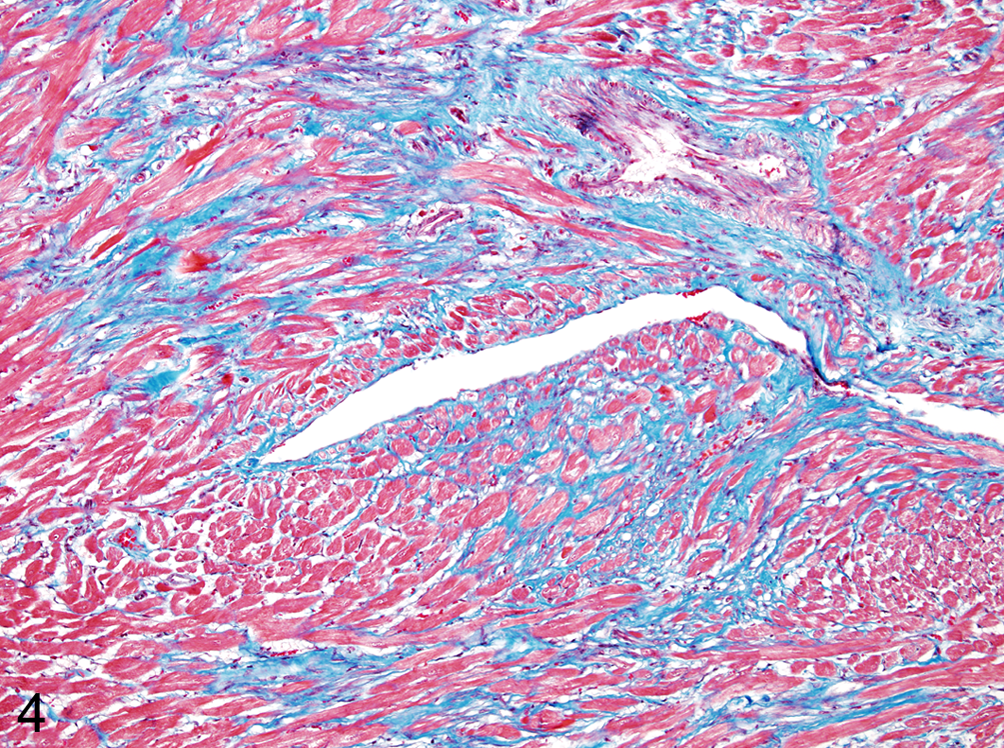
Left ventricle; rabbit No. 12. Diffuse interstitial fibrosis accompanied by distorted arrangement of the myocardial cells. Masson’s trichrome.
After reaching the CNS via the peripheral nerves, street RABV disperses centrifugally to various organs, including the heart, and causes myocarditis. 1,3,7 On the other hand, the CVS strain, a neurovirulent and fixed RABV strain, does not descend from the CNS to the periphery and does not infect the myocardium. It was confirmed in the present study by IHC for the RABV antigen, suggesting that RABV infection was not the cause of the myocardial lesion. Anesthesia using a combination of xylazine (300–470 mg) and ketamine (1300–1800 mg) was reported to be a possible cause of cardiomyopathy in rabbits, 5 and the vasoconstriction caused by xylazine or xylazine-like agents has been proposed as a mechanism. 4 In the present study, we used far lower doses (24–36 mg of xylazine and 420–630 mg of ketamine) for anesthesia of rabbits. Furthermore, the myocardial lesions appeared only in rabbits with brain lesions, irrespective of the frequency or cumulative dose of the anesthetics, and the severity of the myocardial lesions correlated well with that of the neurological lesions in each rabbit. In humans, brain-heart interaction has been well documented, particularly in the cases involving the insular cortex, the hypothalamus, and/or the brainstem. 2,6 Brain damage causes the release of catecholamine, which is directly secreted into the heart via the nerves and induces myocardial degeneration. 9 The myocardial change caused by brain lesions is characterized by hypereosinophilic and/or vacuolated cytoplasm, loss of cross-striation, contraction bands necrosis, fibrosis, and infiltration of inflammatory cells, as seen in this study. 3,6,9 In particular, contraction band necrosis of myocardial cells has been reported as a marker of the catecholamine surge. 9 Hypovitaminosis E and E. cuniculi infection were unlikely as the cause of the myocardial lesions due to the health status of the rabbits and diet used in the present study. In fact, no rabbits showed pathological changes suggestive of E. cuniculi infection, such as interstitial nephritis and hepatitis. 5,8 In conclusion, the myocardial lesion of the rabid rabbits was classified as neurogenic cardiomyopathy, and myocardial necrosis should be recognized as a complication of brain lesions due to RABV infection.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a Grant-in-Aid for Scientific Research from the Ministry of Education, Culture, Sports, Science, and Technology (MEXT) (23380171 to T.U. and 23780306 to Y.S.) and by the Global Center of Excellence Program and the Program of Founding Research Centers for Emerging and Reemerging Infectious Diseases, Japan.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
