Abstract
In September 2002, chronic wasting disease (CWD), a prion disorder of captive and wild cervids, was diagnosed in a white-tailed deer (Odocoileus virginianus) from a captive farm in Wisconsin. The facility was subsequently quarantined, and in January 2006 the remaining 76 deer were depopulated. Sixty animals (79%) were found to be positive by immunohistochemical staining for the abnormal prion protein (PrPCWD)in at least one tissue; the prevalence of positive staining was high even in young deer. Although none of the deer displayed clinical signs suggestive of CWD at depopulation, 49 deer had considerable accumulation of the abnormal prion in the medulla at the level of the obex. Extraneural accumulation of the abnormal protein was observed in 59 deer, with accumulation in the retropharyngeal lymph node in 58of 59 (98%), in the tonsil in 56 of 59 (95%), and in the rectal mucosal lymphoid tissue in 48 of 58 (83%). The retina was positive in 4 deer, all with marked accumulation of prion in the obex. One deer was considered positive for PrPCWD in the brain but not in the extraneural tissue, a novel observation in white-tailed deer. The infection rate in captive deer was 20-fold higher than in wild deer. Although weakly related to infection rates in extraneural tissues, prion genotype was strongly linked to progression of prion accumulation in the obex. Antemortem testing by biopsy of rectoanal mucosal-associated lymphoid tissue (or other peripheral lymphoid tissue) may be a useful adjunct to tonsil biopsy for surveillance in captive herds at risk for CWD infection.
Chronic wasting disease (CWD) is a transmissible spongiform encephalopathy or prion disorder of mule deer (Odocoileus hemionus), 25 white-tailed deer (Odocoileus virginianus), 20 Rocky Mountain elk (Cervus elaphus nelsoni), 26 and moose (Alces alces shirasi). 1 Chronic wasting disease was first recognized in captive cervids in the late 1960s and in free-ranging cervids in the early 1980s in north-central Colorado. 25,26 However, CWD was not found east of the Mississippi River until February of 2002, when 3 free-ranging white-tailed deer bucks harvested in southern Wisconsin were diagnosed with CWD-associated prion protein (PrPCWD) in the retropharyngeal lymphoid tissues and brain. Subsequent surveillance has shown that CWD prevalence in free-ranging white-tailed deer from the core affected area of south-central Wisconsin is approximately 5% to 6%. 10 In contrast to wild populations, CWD has reached remarkably high prevalence in captive cervid populations: 79% (53/67) in mule deer 25 and 52% (88/169) in white-tailed deer at an infected elk farm. 15 In addition, CWD was the primary cause of adult elk mortality in 2 research herds, 13 and 59% prevalence has been reported in a group of 17 elk slaughtered from an infected farm. 16
In September 2002, a 3.5-year-old, male white-tailed deer from a deer farm in Portage County, Wisconsin, was diagnosed with CWD. The farm was quarantined, but depopulation was delayed until January 2006, when the 76 remaining animals were removed in a cooperative effort by the U.S. Department of Agriculture; the Wisconsin Department of Agriculture, Trade, and Consumer Protection; the Wisconsin Department of Natural Resources (WDNR); and the Wisconsin Veterinary Diagnostic Laboratory. After the animals were euthanized by gunshot, the original ear tags from each animal were confirmed, a second unique identification tag was applied, and the carcasses were taken to a WDNR CWD sampling facility. The following tissues were dissected and preserved in 10% neutral buffered formalin: medulla at the level of the obex at the convergence of the dorsal motor nucleus of the vagus nerve (DMNV), where the spinal canal begins, palatine tonsil, and the medial retropharyngeal (RLN), parotid, submandibular, superficial cervical, axillary, prefemoral, popliteal, and inguinal lymph nodes, as well as a portion of recto-anal mucosal-associated lymphoid tissue (RAMALT), and an eye. Brain was frozen for genotyping and an incisor removed for age determination by cementum annuli analysis. a Brain and lymphoid tissues were examined by immunohistochemistry (IHC) using monoclonal antibody F99.97.6.1, following a previous staining technique, 21 except for the initial formic acid treatment. The prion protein (PRNP) genotype was determined by polymerase chain reaction (PCR) and DNA sequence analysis. 15 Genotypes with the sequence encoding glutamine (Q) at codon 95, glycine (G) at codon 96, alanine (A) at codon 116, and Q at codon 226 were considered to be homozygous (wt/wt) for the wild-type allele. Alleles with polymorphisms at codons 96, G to serine S (G96S) and 226, Q to lysine K (Q226K), were observed in the herd; no animals with polymorphisms at codons 95 or 116 were found. This is in contrast to studies conducted on white-tailed deer in western Nebraska, in which approximately 25% of animals had polymorphisms at codon 116 in which G was substituted for A.
Animals were considered positive for CWD if any one of the tissues examined contained detectable PrPCWD. On the basis of the positive PrPCWD immunostaining in the medulla at the level of the obex where the fourth ventricle enters the spinal canal, the positive animals were assigned an obex score: 0 = no PrPPCWD staining present, 1 = scant PrPPCWD staining involving <50% of the DMNV, 2 = PrPPCWD staining present in >50% of the DMNV with no spillover into surrounding tissue, 3 = DMNV totally filled with PrPPCWD stain with some spillover into surrounding tissue, and 4 = heavy PrPPCWD staining within the DMNV and surrounding tissue, as described in elk 19 (Fig. 1). Lymphoid tissue was considered positive if PrPPCWD deposits were detected in lymphoid follicles (Fig. 2). The eye was considered positive if PrPPCWD deposits were detected in the retina (Fig. 2).
Sixty animals (79%) were positive by IHC staining of at least one tissue. Of the PrPPCWD-positive animals, 58 of 60 (96.7%) had PrPPCWD in RLN, 49 of 56 (87.5%) had detectable PrPPCWD staining in the obex, and 48 of 58 (82.7%) had detectable PrPPCWD in RAMALT (Table 1); 48 (85.7%) had deposits of PrPPCWD in the obex and lymphoid tissues, 7 (12.5%) had deposits only in lymphoid tissue, and 1 (1.8%) had deposits only in brainstem. To the authors' knowledge, this is the first report of a white-tailed deer with PrPPCWD deposits limited to the brainstem, although the pattern is relatively high (14%) in Rocky Mountain elk. 19 Interestingly, this animal was a fawn of wt/wt genotype, and the PrPPCWD staining was minimal, with very small amounts of stain around 2 neurons. Thirty-two of the 37 adult animals (86.5%), 12 of 16 yearlings (75.0%), and 15 of 22 fawns aged 6 to 9 months (68.2%) were positive. There did not appear to be a difference in the distribution of CWD antigen in tissues among the various age groups. Infection rates between males (23/29, 79.3%) and females (37/47, 78.7%) were not significantly different. Highest rates of PrPPCWD were found in the RLN and tonsil tissues (Table 1), a finding that is consistent with earlier reports in mule deer, 5,22–24 white-tailed deer, 11 and Rocky Mountain elk. 19 Prion protein was detected in the retina in only 4 animals, all with marked prion accumulation in the obex, as indicated by an obex score of 4. Prion protein was primarily located in the inner and outer plexiform layers of the retina; all other regions within the eye were free of prion.
Of the 51 deer with wild-type prion genotype 15 (wt/wt), 44 (88.0%) had positive immunostaining in at least one tissue (Table 2). Twenty-one deer had the prion genotype wt/G96S, and 14 of these (66.7%) were positive in at least one tissue. Two adult deer had the prion genotype G96S/G96S. One of these was negative in all tissues examined, and the other had positive PrPPCWD staining in the RLN only. To the authors' knowledge, this is the first report of a CWD-positive white-tailed deer with this prion genotype in Wisconsin, indicating that there are no truly resistant genotypes to this disease. One animal with the PRNP genotype wt/Q226K was positive in lymphoid tissue, but this animal did not have an obex sample suitable for scoring. A second deer with genotype G96S/Q226K was negative in all tissues. Prion genotype (wt/wt vs. wt/G96S) was a weak predictor of CWD RLN infection (X 2 = 3.45, P = 0.07). The odds of CWD RLN infection were 3.1-fold higher (95% confidence interval [CI]: 0.984–10.5) in deer with the wild-type prion genotype (wt/wt) compared to deer with the wt/G96S prion genotype. However, prion genotype was a strong predictor of CWD obex infection (X 2 = 8.54, P = 0.004). The odds of CWD obex infection were 12.3-fold higher (95% CI: 2.28–65.7) in deer with the wt/wt prion genotype than in deer with the wt/G96S prion genotype. Prion genotype was also an important predictor of the extent of CWD staining in the brain (F 1,50 = 9.3, P < 0.01). Forty-two wt/wt deer with suitable obex had a mean obex score of 2.5 (95% CI: 2.16–2.84) and 7 wt/G96S deer had a mean obex score of 1.3 (95% CI: 0.49–2.13), indicating more advanced stages of CWD infection in wild-type deer. These findings are consistent with those reported in captive 15 and free-ranging 9 white-tailed deer, in which animals with the wt/wt genotype were overrepre-sented in the CWD-positive population compared to those with the wt/G96S genotype.
To compare infection rates of wild versus captive deer and for prion genotypes, the annual probability of CWD infection (incidence rate) in deer <2 years of age was determined by estimating the instantaneous force of infection 2–4,7,8 based on deer age. Data from the fawn and yearling deer were used to ensure that all animals had been exposed to CWD infection since birth and to avoid complications in estimating force of infection with potential disease-related mortality in older infected animals. Prion genotype (wt/wt vs. wt/G96S) did not significantly affect the annual rate of CWD RLN infection in deer <2 years of age (t =-1.15, P = 0.25). Wild-type deer (wt/wt) had an annual probability of RLN infection (0.75, 95% CI: 0.14–0.85), which was similar to the annual probability for deer with the wt/G96S prion genotype (0.56, 95% CI: 0.0–0.72), although confidence intervals on these incidence rates were large. Overall these young deer had a 0.68 (95% CI: 0.43–0.79) annual probability of RLN infection, substantially higher than infection rates for wild fawn (<0.005) and yearling (0.03–0.04) deer in Wisconsin. 6 Prion genotype had a significant effect (t = −2.67, P = 0.01) on annual probability of obex infection. Wild-type deer (wt/wt) had a higher annual probability of obex infection (0.69, 95% CI: 0.22–0.80) than deer with wt/G96S prion genotype (0.19, 95% CI: 0.0–0.35).
The present results indicate that infection rates in these captive deer were at least 20-fold higher than rates observed in wild deer. In this captive herd, the infection rate in young deer was almost as high as in adults. This extremely high infection rate is a striking feature of CWD in captive white-tailed deer herds and is likely caused by rapid disease transmission and/or high exposure of this captive herd to CWD prions. However, the absence of clinical disease on this farm, despite widespread distribution of abnormal prion in obex tissues, was surprising. Captive studies on mule deer indicate that environmental prion reservoirs are likely an important source of infection in high-density captive situations. 12 The high infection rate for captive fawns compared with wild fawns and the rapid rate of RLN infection in challenged fawns 18 indicate that infection rate in wild fawns is likely limited by exposure to CWD prions in wild populations rather than disease incubation period. Postmortem surveillance of deer of any age by examination of the RLN and brain may be useful in identifying infected captive herds. In contrast, surveillance in wild white-tailed deer populations has focused on testing lymph nodes in adult deer 17 because of low rates of CWD infection in wild deer fawns 6 and the lack of identification of an obex-only-positive wild white-tailed deer. 11 Although there are apparent differences in CWD infection between deer with wt/wt and wt/G96S prion genotypes, the primary difference appears to be in the rate of abnormal prion accumulation and spread after extraneural infection. Both genotypes have similar rates of early infection in RLN tissues, but incubation rates and PrPCWD progression to the obex appear much slower in wt/G96S genotype deer. Interestingly, the proportion of infected captive deer with positive obex tests (87.5%) is not substantially higher than that found in wild deer in Wisconsin (82%), indicating that the rate of abnormal prion progression to the brain following extraneural infection may not be strongly related to the level of CWD exposure. Further studies to clarify how prion genotype affects the rate of CWD infection and PrPCWD progression are recommended. The high rates of CWD infection in captive deer herds may provide opportunities for epidemiological research to understand CWD incubation processes, prion shedding, and transmission. Although the sensitivity of IHC for CWD from RAMALT biopsy (and other peripheral lymphoid tissues) is lower than that of RLN or tonsil, the relative ease of collecting RAMALT from live deer using disposable instrumentation indicates that whole-herd testing may be a suitable adjunct to tonsil biopsy and necropsy surveillance, particularly for farmed deer with suspected risk of environmental exposure 14 or exposure to infected captive stock.
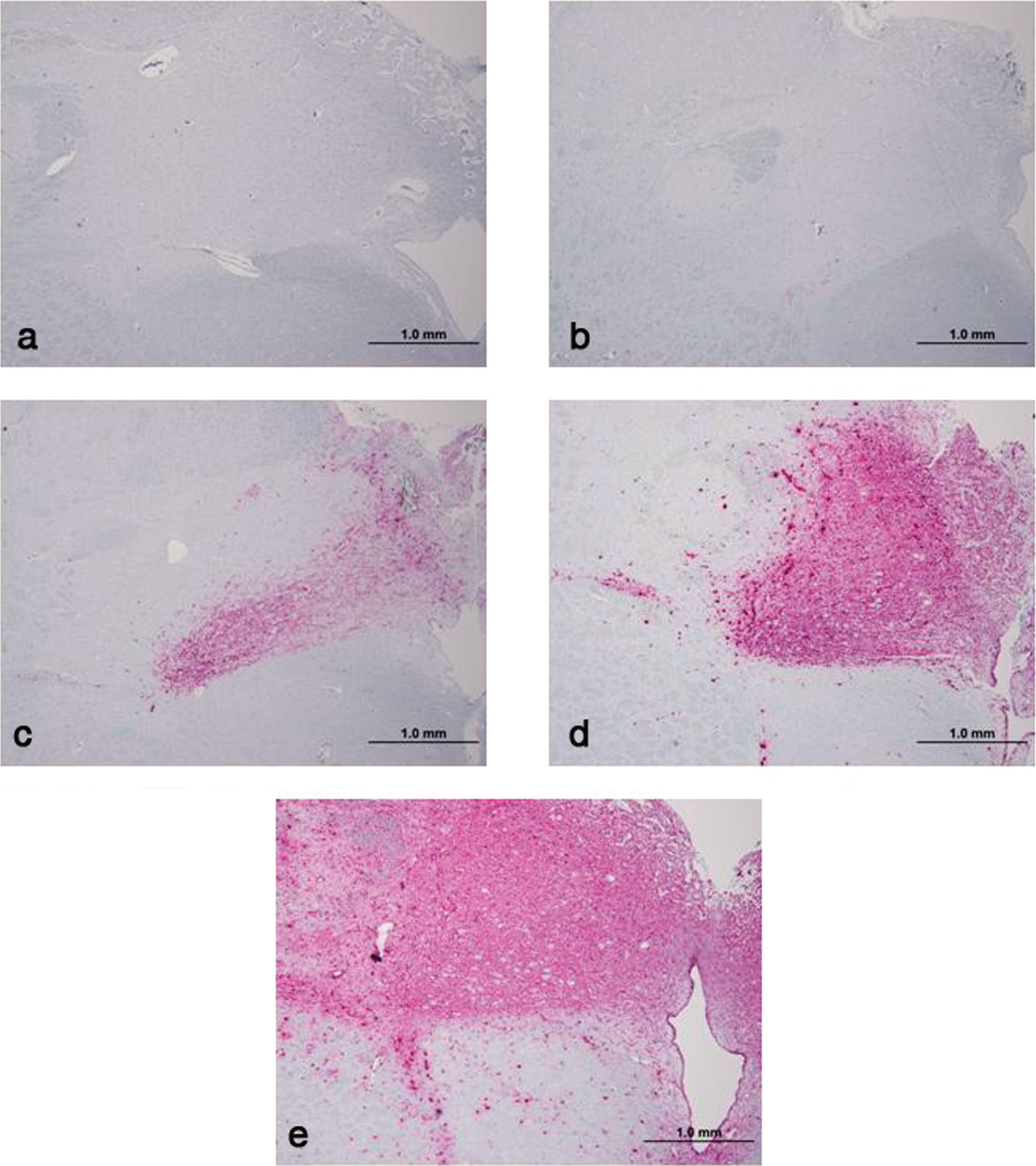
Photomicrographs of immunohistochemistry of obex tissue at the convergence of the dorsal motor nuclei of the vagus (DMNV) using monoclonal antibody F99/97.6.1.
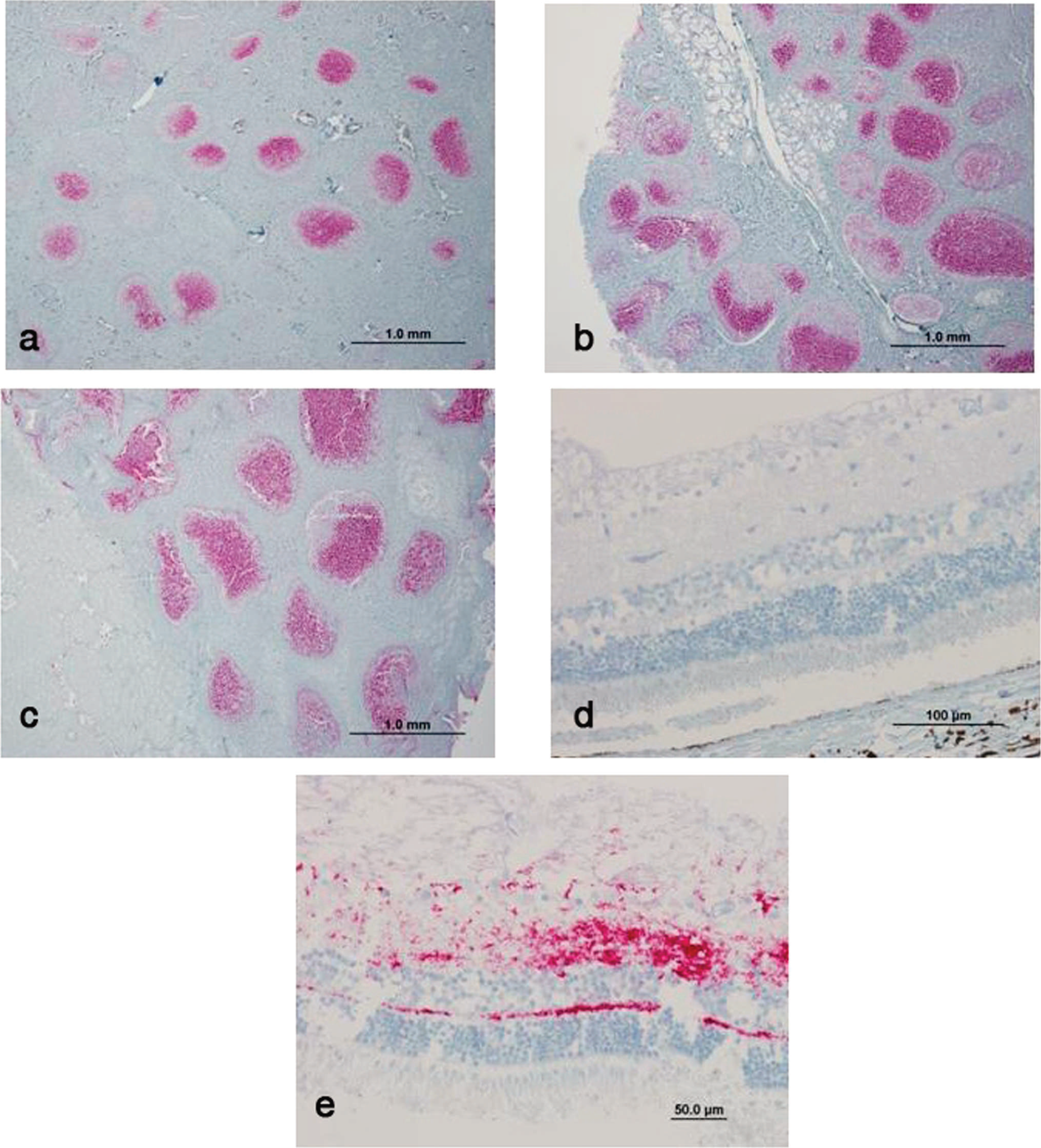
Photomicrographs of immunohistochemistry using monoclonal antibody F99/97.6.1 to detect chronic wasting disease (CWD)-associated prion protein (PrPCWD) in the
Number and percent of chronic wasting disease (CWD)-positive deer with positive immunohistochemical staining for CWD-associated prion protein (PrPCWD) in lymphoid tissues and obex.

Acknowledgements. The authors are grateful to all those from the U.S. Department of Agriculture; the Wisconsin Department of Agriculture, Trade, and Consumer Protection; the Wisconsin Department of Natural Resources (WDNR); and the Wisconsin Veterinary Diagnostic Laboratory who were involved in the organization and implementation of this depopulation. Special thanks to J. Langenberg and C. Johnson for their critical review of this paper.
Numbers of white-tailed deer positive for chronic wasting disease-associated prion protein (PrPCWD) in each genotype and age class.
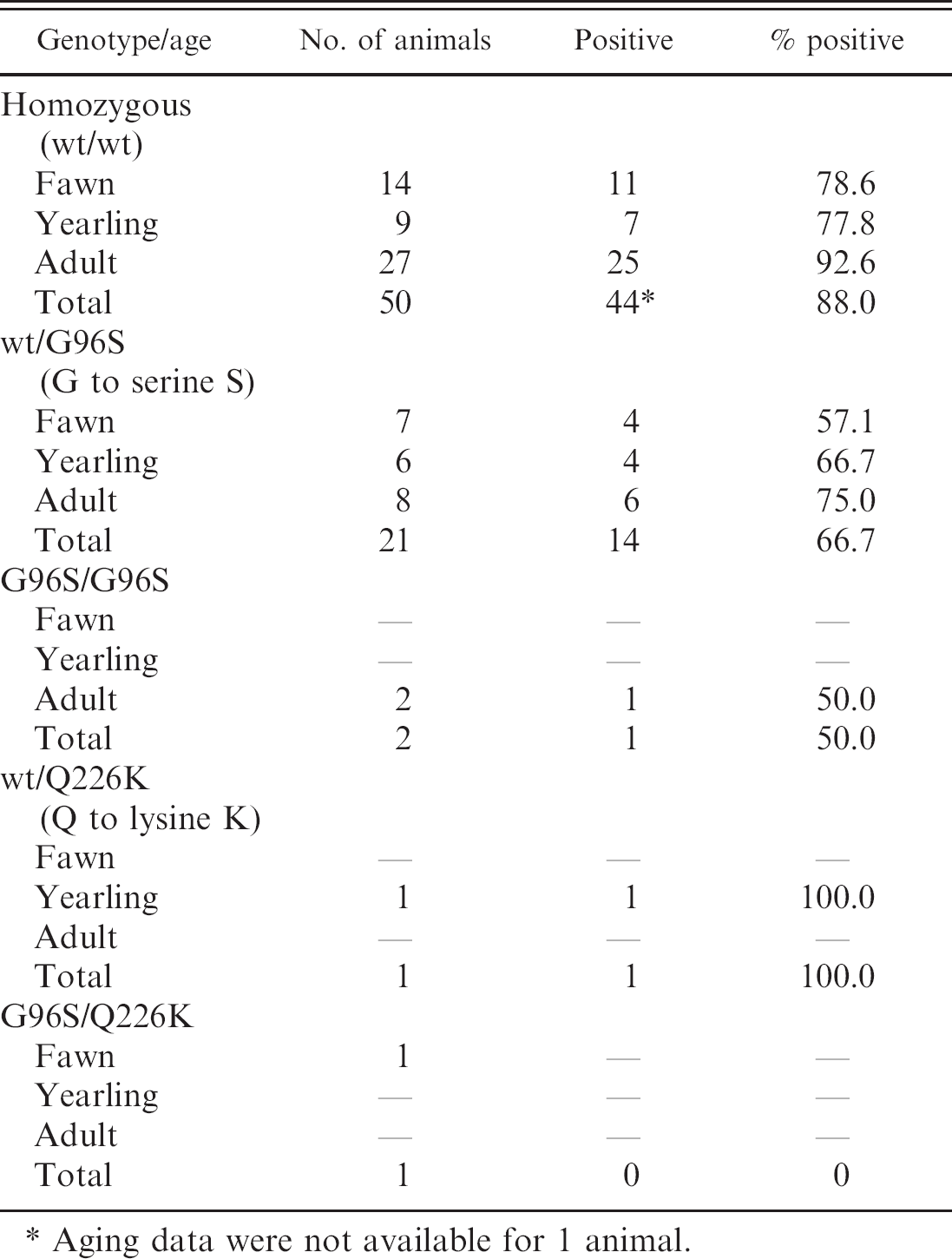
Aging data were not available for 1 animal.
Footnotes
a.
Matson's Lab, Milltown, MT.
