Abstract
A 10-year-old, female, pregnant alpaca (Vicugna pacos) presented with a discolored, swollen right eye. The eye did not respond to treatment and was enucleated. Sectioning of the fixed eye revealed an ocular mass replacing the aqueous and vitreous humors, with multiple regions of bone formation. Histopathological, immunohistochemical, and transmission electron microscopy confirmed the tumor was an intraocular melanoma with bone formation. Osteogenic melanoma is a rare variant of melanoma not reported in the eye before.
A 10-year-old, female, pregnant alpaca (Vicugna pacos) was examined on-farm for the development of a discolored, enlarged right eye. Reexamination of the eye 7 days later revealed no clinical improvement, so the eye was enucleated (Fig. 1). The globe was fixed in 10% neutral buffered formalin solution and submitted to Gribbles Veterinary (Palmerston North, New Zealand) for examination.
The enucleated globe was turgid, firm, and swollen. On palpation, a hard mass could be felt through the sclera. When the eye was sectioned, a 25 × 6-mm semicircular piece of bone was revealed, surrounded by the mass and extending across the vitreous humor in the region of the iris. The aqueous and vitreous humors were replaced by a firm, dense, grey-to-black mass adherent to the iris, retina, and cornea. The lens could not be identified. Sectioning the mass was difficult because of hard, gritty material within it.
Tissue sections were routinely processed, embedded in paraffin, sectioned at 5 μm, and stained with hematoxylin and eosin and Fontana stains. Later, additional sections were stained with primary mouse monoclonal antibodies to vimentin, S100, melan A, AE1/AE3, and CK1 a using an automated avidin-biotin complex method. b Control tissues were of human origin.
For transmission electron microscopy, selected areas of the formalin-fixed tumor were postfixed in buffered 2% paraformaldehyde and 2% glutaraldehyde at pH 7.2, postfixed in 1 % osmium tetroxide, and processed into epoxy resin. Thick sections were stained with 1 % toluidine blue, and thin sections were stained with uranyl acetate and lead citrate, before examination on an electron microscope. c
Histopathological examination of the eye revealed multinodular proliferations of neoplastic melanocytes replacing the iris and extending through the ciliary body as well as into the vitreous and aqueous humors. Spindle-shaped tumor cells were arranged in sheets on a light fibrovascular stroma. Many of the cells contained thickly clumped, intracytoplasmic, Fontana-positive melanin pigment. Nuclei were moderately pleomorphic, round to oval, with densely clumped chromatin and individual nucleoli. Mitotic figures ranged from 3 to 10 per 400× field, and atypical mitotic figures were frequent (Fig. 2). Neoplastic cells infiltrated the lens remnants and adhered to the corneal endothelial layer. Mineralized trabeculae adjacent to the tumor nodules were closely associated with loose melanin pigment (Fig. 3). Necrotic debris, hemorrhage, degenerate neutrophils, and fibrin were adherent to the tumor nodules. Tumor cells were strongly positive for vimentin, melan A, and AE1/AE3, moderately positive for CK1, and negative for S100. Ultrastructural examination confirmed the presence of pleomorphic, cytoplasmic melanosomes within the tumor cells, characteristic of malignant melanoma cells 3 (Fig. 4).
Melanoma is used to describe all malignant melanocytic tumors, whereas melanocytoma refers to the benign form. 9 Intraocular melanoma has only been described once before in an alpaca. 4 In the current case, a melanoma with a high mitotic index and bone formation was diagnosed. Bone formation within a melanoma is a very rare occurrence, 8 although cases have been reported in humans 5 and in dermal 6 and oral 2 melanomas in dogs, but has not been described in an ocular melanoma in any species. In all previous reports, the melanomas were characterized as malignant. In the current case, the high mitotic rate is consistent with a malignant melanoma. A mitotic rate of >3 mitoses per high-powered field has been used to predict early recurrence and possible metastasis of oral melanomas. 8
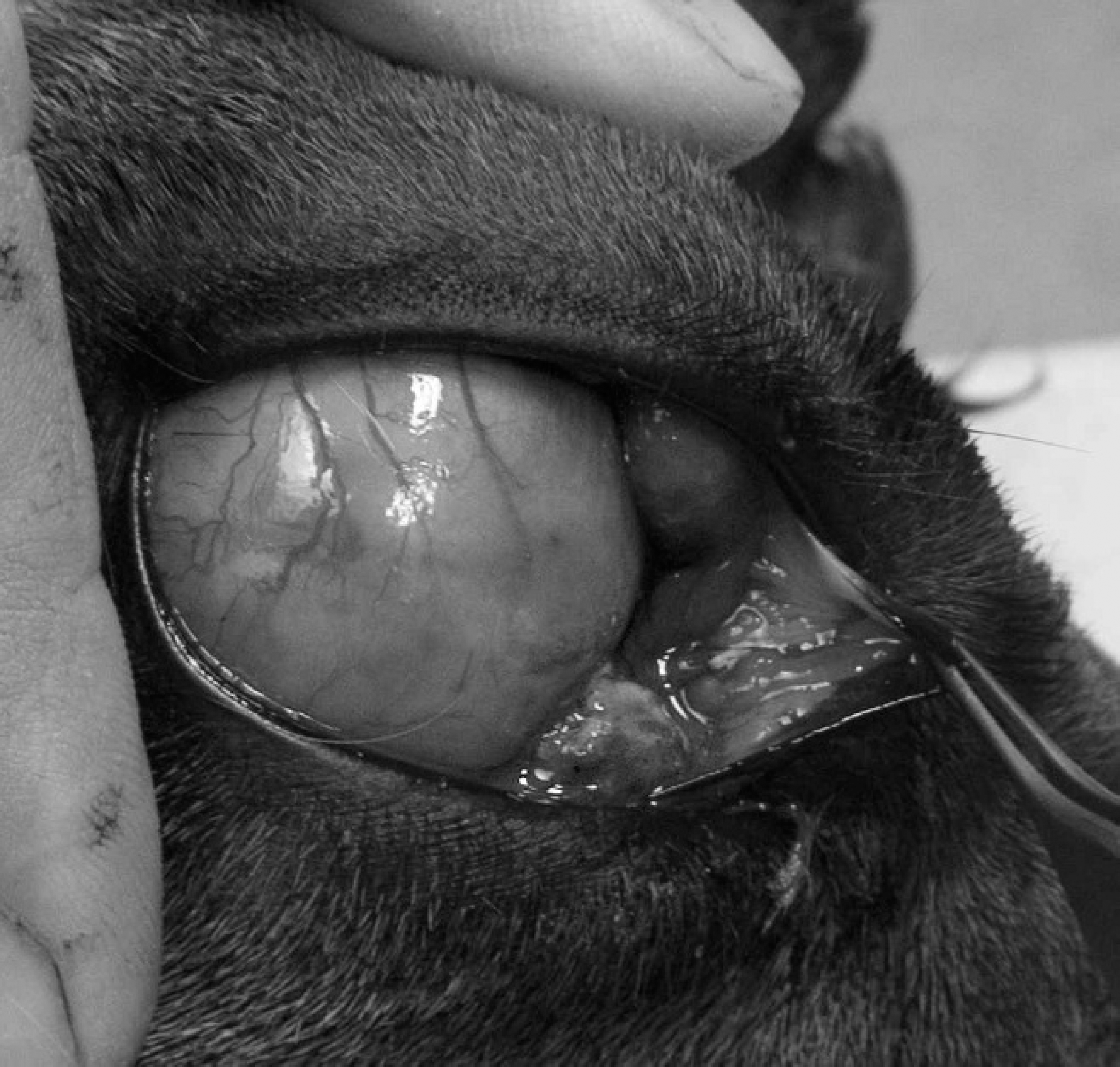
Eye, alpaca. The swollen, opaque eye just prior to surgical enucleation.
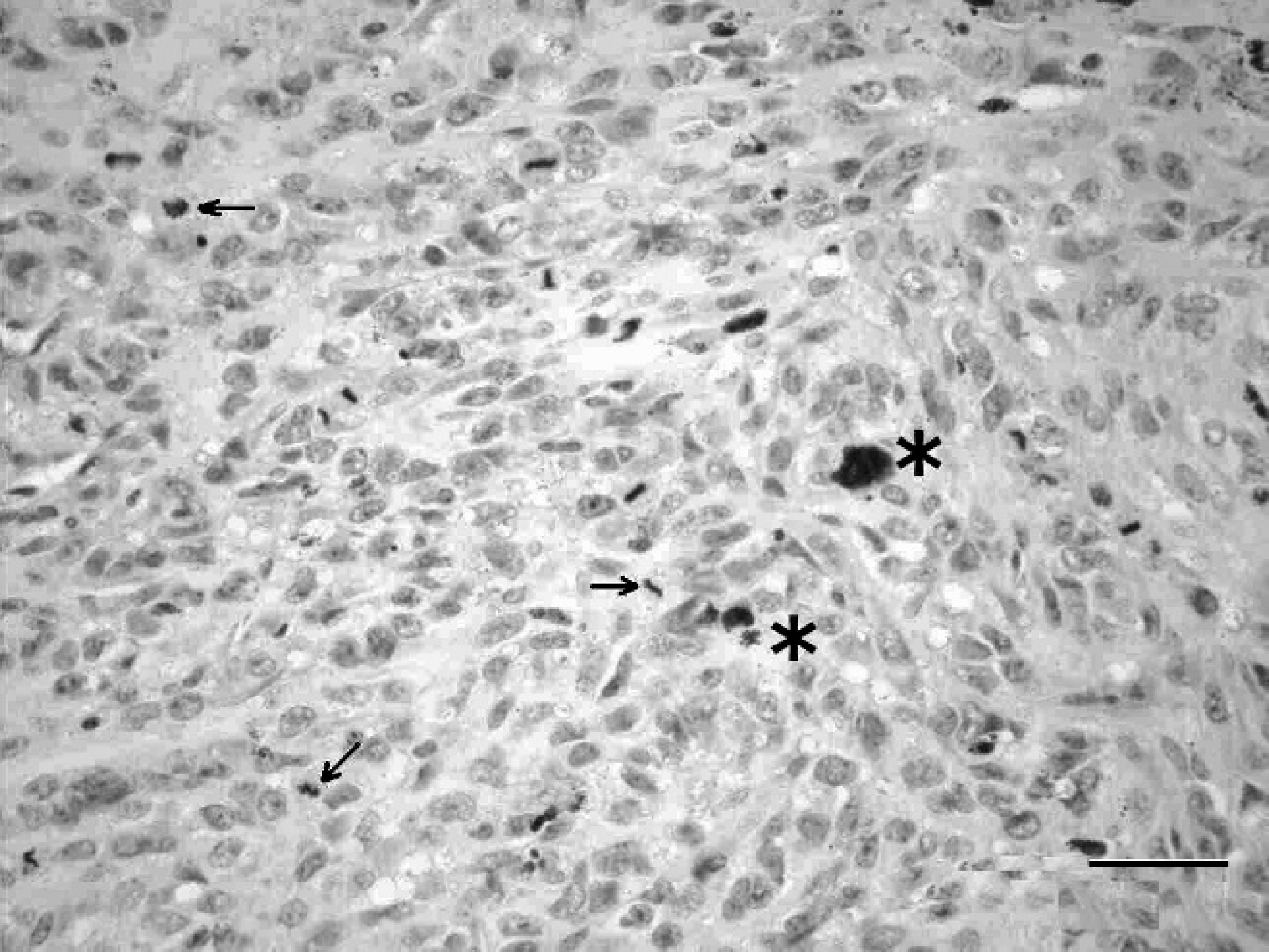
Eye, alpaca. Tumor cells within the eye of an alpaca containing intracytoplasmic melanin pigment (*) and atypical mitotic figures (arrows). Hematoxylin and eosin. Bar = 50 μm.
Immunostains used to confirm melanomas in cats, dogs, and humans include vimentin, S100, and melan A. 9 In the present alpaca, vimentin and melan A were positive, but S100 was negative. In addition, the keratin immunostains AE1/AE3 and CK 1 were positive. Pancytokeratin reactivity has been seen once before in a horse 7 and in humans with melanoma, 1 as is the case in this alpaca. Explanations for the reactivity suggest intermediate filaments in the melanoma cells are staining positively for epithelial cell markers. 7
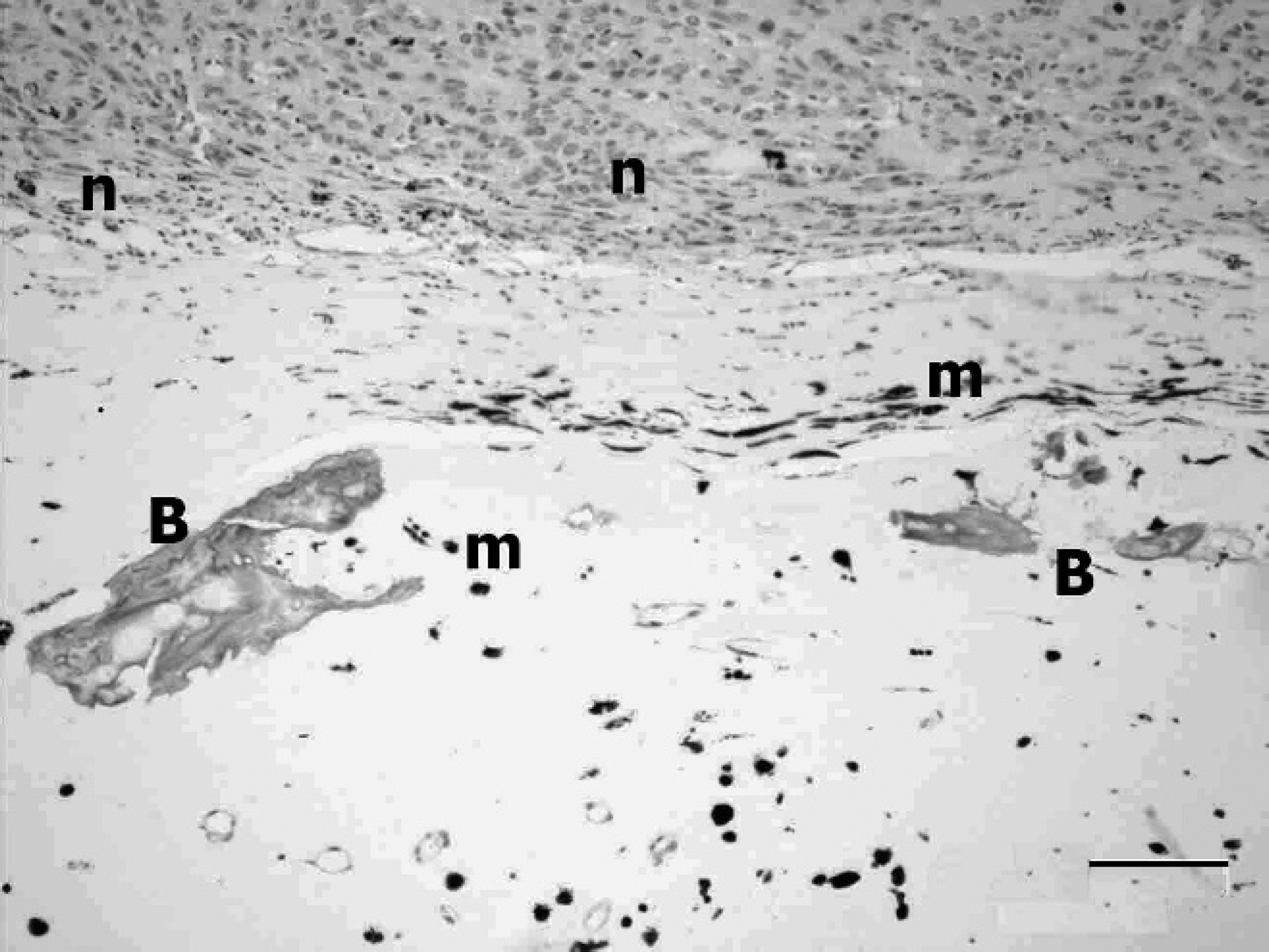
Eye, alpaca. Bone formation (B) associated with neoplastic cells (n) in an intraocular melanoma. Melanin pigment (m) is scattered throughout the tumor and surrounds the bone. Hematoxylin and eosin. Bar = 100 μm.
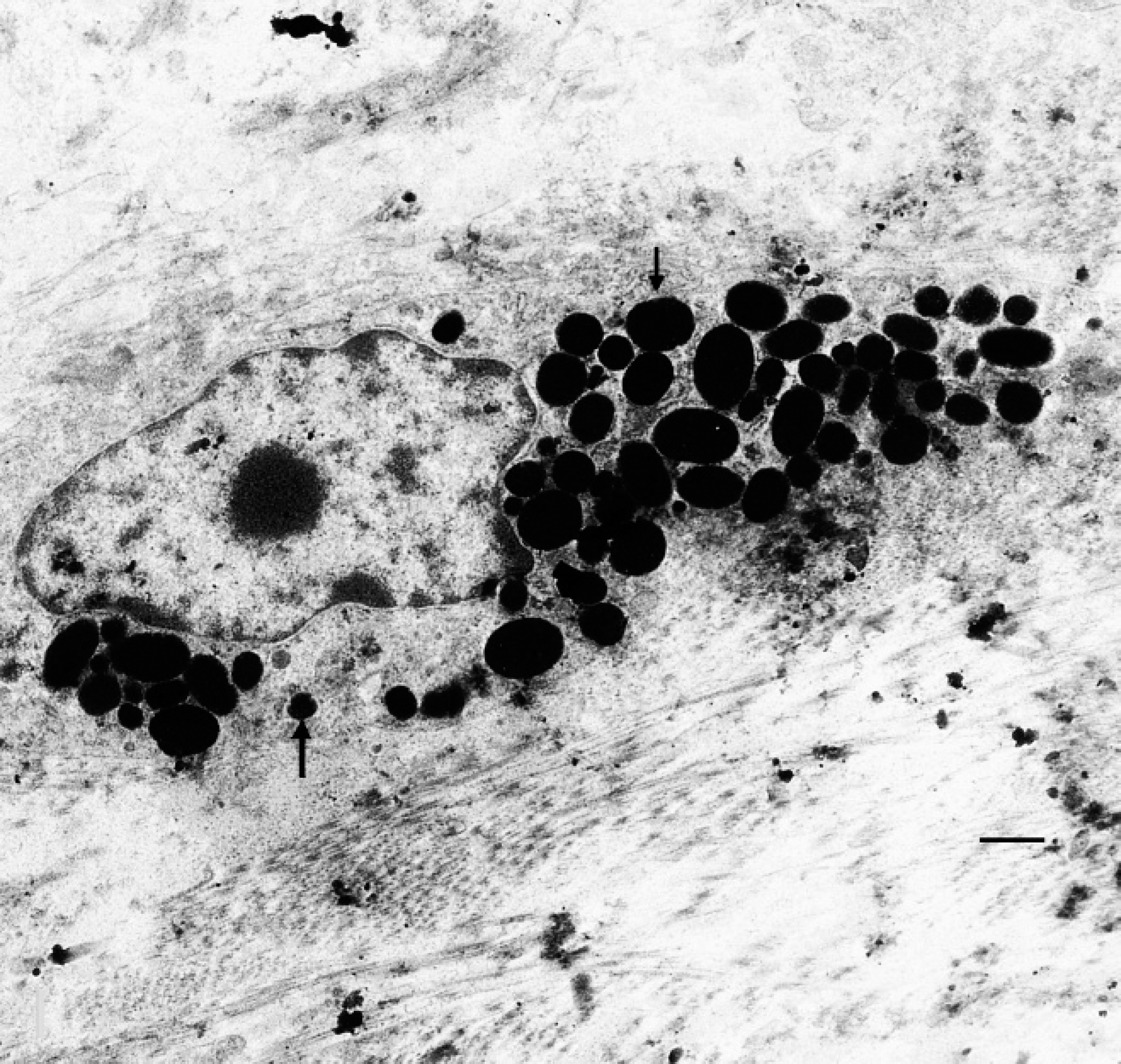
Eye, alpaca. Pleomorphic melanosomes (arrows) within the cytoplasm of a neoplastic melanocyte. Uranyl acetate and lead citrate. Bar = 1 μm.
The origin of the osteogenic stimulus is unknown and could be a result of differentiation of neoplastic melanocytes or metaplasia of the supporting stroma. Heterotopic bone formation can occur in chronically inflamed tissues; intestinal adenocarcinomas of cats, sheep, humans, and horses; and prostate adenocarcinomas in humans. 2 Positive immunolabeling of the osteoid for S100 and melan A in 2 studies 6,8 suggested neoplastic melanocytes were the progenitor cells, whereas negative S100 staining in another study 2 suggested they were not. Neither the melanoma cells nor the bone were S100 positive in the current case, and the bone appeared adjacent to the tumor nodule. This would suggest that local osteo-inductive factors produced by the neoplastic melanoma cells induced the bone formation in the supporting stroma.
Acknowledgements. The authors thank Dr. Craig Hassell for surgical assistance, Dr. Monique Churcher for the image of the eye, and Dr. Alastair Johnstone for the electron micrographs.
Footnotes
a.
Dako Denmark A/S, Glostrup, Denmark.
b.
Bond™ System, Leica Microsystems GmbH, Wetzlar, Germany.
c.
Philips CM10, Philips, Eindhoven, The Netherlands.
