Abstract
Intracellular crystalline deposits of immunoglobulin are occasionally seen in human B-cell lymphoproliferative disorders such as multiple myeloma, chronic lymphocytic leukemia, and various forms of lymphoma. Even more uncommon is the occurrence of immunoglobulin crystals in benign plasma cells or reactive lymphocytes. Here we describe the histologic, immunohistochemical, and ultrastructural features of intracellular immunoglobulin crystals in nonneoplastic plasma cells in a chronic inflammatory lesion in a dog. Microscopically, the intracellular, nonbirefringent eosinophilic crystals were square to rectangular, 2–20 μgm long, and caused nuclear displacement to the periphery. The crystal-containing cells, as well as some of the crystals themselves, were positive for lambda light chain. Ultrastructural findings were consistent with a lattice network of protein-molecule alignment. The cause and significance of the crystals is unknown.
Keywords
Intracellular crystalline deposits of immunoglobulin are seen occasionally in human B-cell lymphoproliferative disorders such as multiple myeloma, chronic lymphocytic leukemia, and various forms of lymphoma.1,7,8 An even more rare occurrence is that of immunoglobulin crystals in benign plasma cells or reactive lymphocytes.3,6 In this report we describe the histologic, immunohistochemical, and ultrastructural features of intracellular immunoglobulin crystals in nonneoplastic plasma cells in a chronic inflammatory lesion in a dog.
A 9-year-old female spayed Beagle presented to the Ohio State University Veterinary Teaching Hospital. The dog had been treated for chronic bilateral otitis externa with Malassezia infection since the age of 1 year. At the time of presentation, a mass in the right ear was noted. The mass was excised, and a total ear canal ablation with a bulla osteotomy was performed. Histologic examination of the mass revealed an inflammatory polyp. The dog did well postoperatively until 7 months later, when a 1-cm painful mass formed at the prior surgical site. At this time the dog had a mild to moderate neutrophilia with a left shift. The total white blood cell count was 23.0 × 103/μl (normal: 4.1 to 15.2 × 103/μl), with 19.1 × 103/μl neutrophils (normal: 3.0 to 10.4 × 103/μl) and 2.3 × 103/μl bands (normal: 0 to 0.1 × 103/μl). Liver enzymes were slightly elevated, which was attributed to oral administration of prednisolone.
The preclinical diagnosis was a neoplasm or abscess. The mass was removed and submitted for histopathologic examination. Tissue was fixed overnight in 10% neutral buffered formalin prior to paraffin embedding. Microscopically there was marked chronic pyogranulomatous cellulitis with widespread infiltrates of degenerate neutrophils, lymphocytes, plasma cells, histiocytes, and epithelioid macrophages (Fig. 1). Reactive fibrosis was present throughout the section, along with formation of granulation tissue at the periphery. Light growth of Staphylococcus aureus and Staphylococcus intermedius was obtained from bacterial culture at the time of surgery. Gram, Gomori methenamine silver, and acid fast stains were negative for microorganisms, and no foreign bodies were seen.
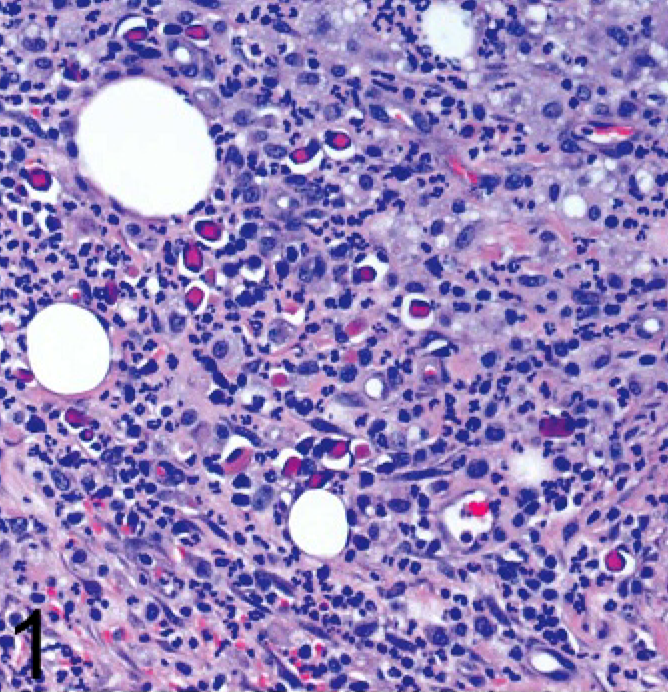
Ear mass; dog. Widespread, chronic, pyogranulomatous inflammation. HE.
In several areas there were clusters of cells with one to several brightly eosinophilic, hyaline, square to rectangular, nonbirefringent, intracytoplasmic inclusions (Fig. 2). These inclusions ranged from 2–20 μm on the long axis, and the larger ones expanded the cytoplasm and displaced the nucleus to the periphery. The cell type could not be distinguished with certainty on the hematoxylin and eosin–stained slide. The tinctorial properties of the inclusions resembled Russell bodies found in Mott cells, but the morphologic appearance was quite different.
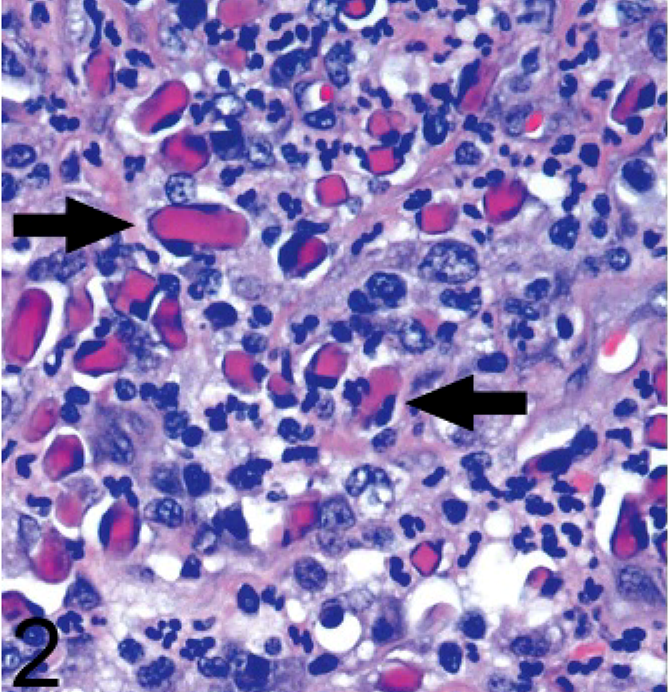
Ear mass; dog. Numerous cells contain one to several brightly eosinophilic crystals (arrows) that expand the cytoplasm and cause nuclear displacement. HE.
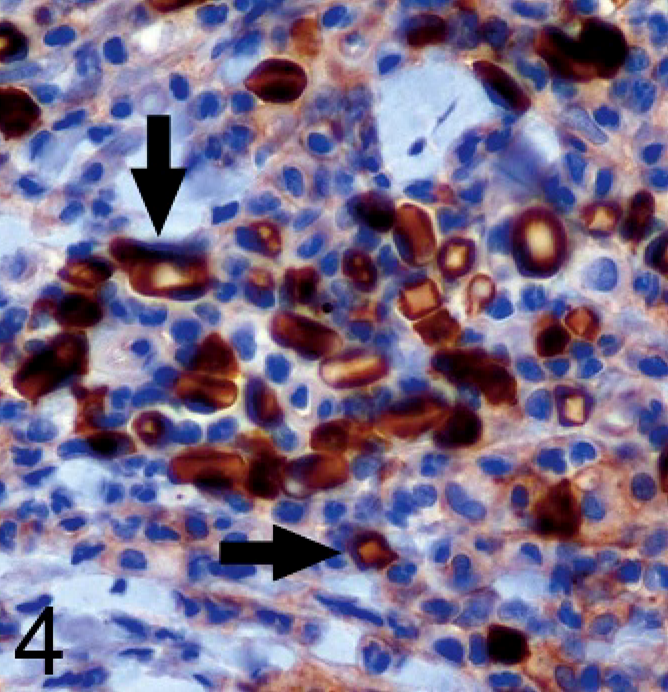
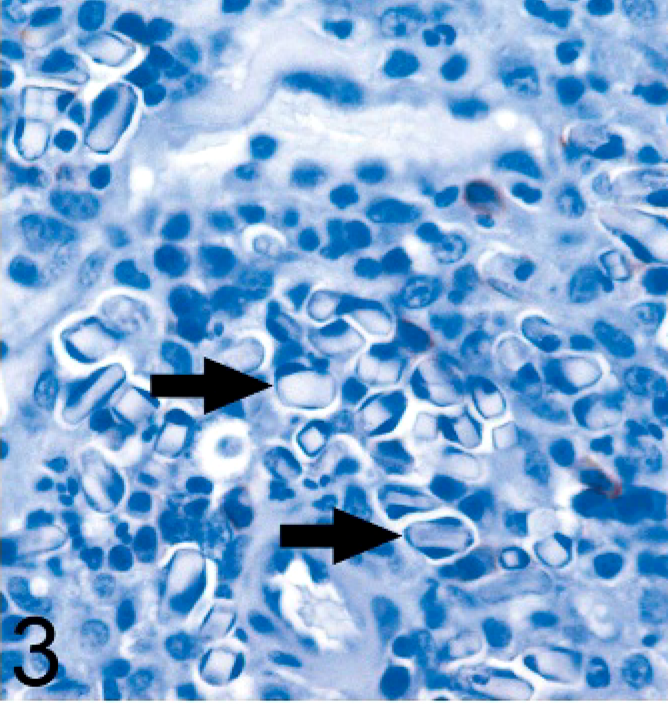
Immunohistochemical staining was performed using a 1 : 100 dilution of anti-CD3 antibody (Dako, Carpinteria, CA), a 1 : 20 dilution of anti-CD18 antibody (clone CA16.3C10, provided by Peter Moore at the University of California, Davis, CA), 1 : 200 dilution of anti-CD20 antibody (NeoMarkers, Fremont, CA), and a 1 : 3000 dilution of anti–lambda light chain antibody (Dako). The cells containing the inclusions were negative for CD3, CD18, and CD20 (Fig. 3). The cells had positive cytoplasmic staining for lambda light chain, and there was variable positive staining of the inclusions themselves (Fig. 4).
Ear mass; dog. The cells containing intracytoplasmic crystals (arrows) are intensely positive for lambda light chain, as are portions of some of the crystals. Avidin–biotin peroxidase method, hematoxylin counterstain.
Ear mass; dog. The cells containing intracytoplasmic crystals (arrows) are negative for CD20. Avidin–biotin peroxidase method, hematoxylin counterstain.
Transmission electron microscopy was performed to further evaluate the inclusions. Because of the limited distribution of the plasma cells with inclusions, we were unable to locate them in the remaining formalin-fixed tissue. Therefore, sections were taken from the original paraffin-embedded block. Removal of paraffin was carried out by reverse processing using gradual concentrations of xylene, ethyl alcohol, and pure water. The sample was pre-fixed with 3.5% gultaraldehyde and postfixed with 1% OsO4, followed by dehydration and embedding in an epon–resin mixture. Thin sections were counterstained with uranyl acetate and then lead citrate. Samples were observed with a Phillips 300 (Phillips, Eindhoven, The Netherlands) at an accelerating voltage of 60 kV.
Most of the fine structures were partially destroyed, but the crystal-containing cells were not completely damaged. The electron density of the crystalline material was slightly higher than that of the neighboring structures. Abundant rough endoplasmic reticulum was present frequently on the periphery of the crystals, supportive of the plasma cell origin (Fig. 5). The nucleus could sometimes be seen on the periphery of the cell. Fine structure of the crystalline material was difficult to discern due to paraffin embedding, but some sections had periodic parallel lines with a periodicity of 13 nm, consistent with a lattice network of protein–molecule alignment (Fig. 6).

Ear mass; dog. This section demonstrates longitudinally oriented protein–molecule arrangements with a periodicity of approximately 13 nm. Electron micrograph. Bar = 50 nm.
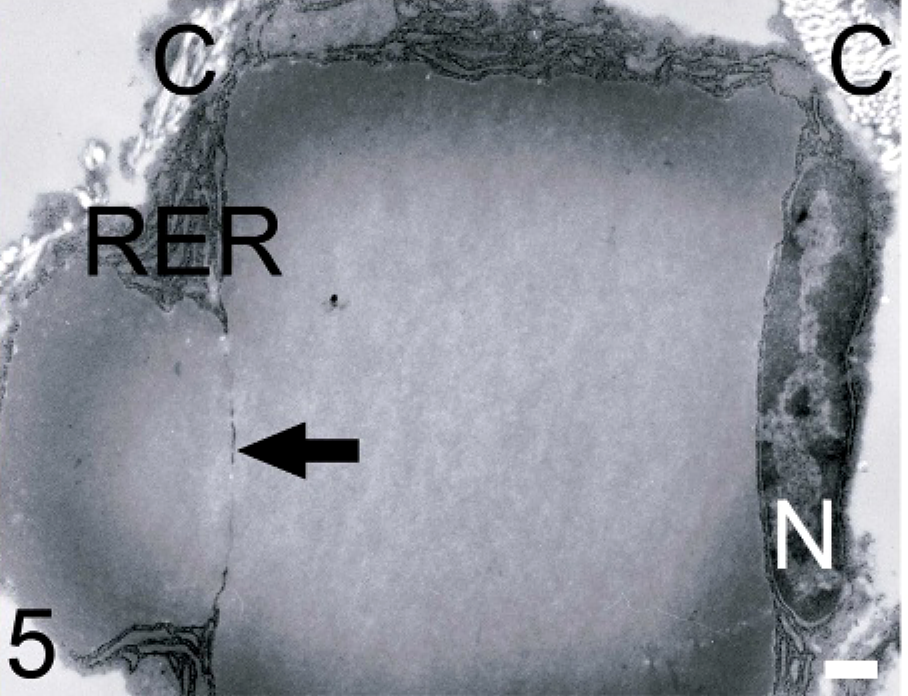
Ear mass; dog. This cross-section of a plasma cell shows 2 tightly grouped crystals. The crystals are surrounded by rough endoplasmic reticulum (RER), and the nucleus (N) has been displaced to the periphery. The arrow shows the continuity of the RER that separates the crystals. Collagen fibers (C) are adjacent to the upper left and right portions of the cell. Transmission electron micrograph. Bar = 0.5 μm.
Together, these results indicate that the cells containing inclusions are plasma cells, and the intracytoplasmic material is consistent with crystalline deposits of immunoglobulin light chain. The cause of the crystal formation in plasma cells and lymphocytes is not known. Possible mechanisms that have been speculated include overproduction of immunoglobulins, defects in immunoglobulin processing and/or secretion, and mutations in the immunoglobulin VH region that lead to an altered protein with a propensity to self-aggregate.3 Immunoglobulin crystals also have been documented within phagocytic histiocytes, leading to a condition called crystal-storing histiocytosis.1
Previous reports of crystals in reactive plasma cells in people indicate that this is a rare occurrence.3,6 One case of swine follicular lymphoma with intracytoplasmic Russell body globules has been described, but we found no reports of intracytoplasmic immunoglobulin crystals in other domestic animals.2 The shape of crystals can vary considerably, ranging from needlelike to square to rhomboid.4,8 Light chain restriction of the crystal-forming cells also has been reported. It has been suggested that lambda light chain is more frequently associated with lymphomas, but others have found a high percentage of kappa light chain–producing lymphoma cells with crystals, so the significance of this is uncertain.1,4 We showed lambda light chain production in the crystal-forming cells of this case but found no evidence of a neoplastic process in the dog. Although ultrastructural analysis was hampered by the use of paraffin-embedded tissue, we were able to demonstrate a crystalline structure similar to what has been described elsewhere.5,8 While the underlying cause of immunoglobulin crystal formation in this dog was not determined, it is possible that it was related to years of local antigenic stimulation and chronic inflammation.
