Abstract
Cytologic evaluation of aspirate slides from a small, <1-cm, interdigital mass on a 9-y-old, spayed female Yorkshire Terrier revealed a proliferation of discrete, round cells containing few-to-many, variably sized, round, eosinophilic, cytoplasmic inclusions. The top differentials based on the cytologic findings were either a plasma cell tumor or a B-cell lymphoma with Mott cell differentiation. The unencapsulated, well-demarcated, multilobulated round-cell neoplasm was completely excised. Immunohistochemical stains were performed to further characterize the neoplasm, which had immunolabeling for multiple myeloma oncogene 1 and vimentin, but did not react with CD3, CD20, melan A, or ionized calcium–binding adapter molecule 1, nor with a Giemsa special stain. Ultrastructurally, the cytoplasmic granules had Russell body–like morphology. A solitary, cutaneous plasmacytoma with Mott cell differentiation has not been described previously in veterinary medicine, to our knowledge.
Keywords
Cytologic slides from a 9-y-old, spayed female Yorkshire Terrier were submitted to the Iowa State University Clinical Pathology Laboratory (Ames, IA, USA) from aspirates of a small, <1-cm, ulcerated mass on the right front paw between digits 2 and 3. The slides were stained with modified Wright stain and evaluated by an ACVP-certified veterinary clinical pathologist (CL Auch). The smears contained many individualized, rounded cells that had moderate-to-abundant, moderately basophilic cytoplasm and eccentric, round nuclei with finely stippled to stippled chromatin and variably visible to prominent nucleoli. Anisocytosis and anisokaryosis were marked with nuclear size of ~1–3× the size of a neutrophil (~10–35-µm). Many (~75%) of the cells contained few-to-many, variably sized, eosinophilic, round, cytoplasmic inclusions. There were occasional bi- and multinucleate cells. Rare cells had macronucleoli, approximately the same size as a red cell.
The cytologic findings (Fig. 1A) were interpreted as consistent with a round-cell tumor, and the top differentials were either a plasmacytoma or a B-cell lymphoma with Mott cell differentiation. An inflammatory condition, such as plasmacytic pododermatitis, was not considered a strong differential given the species, gross description of the lesion, and the presence of markedly atypical cells without increased numbers of other inflammatory cells, such as macrophages or small lymphocytes; however, that differential could be considered for other cases. The tumor was excised by the veterinarian and submitted to the Iowa State University–Veterinary Diagnostic Laboratory (Ames, IA, USA) for histologic evaluation by an ACVP-certified veterinary anatomic pathologist (A Michael) ~2 wk after cytologic evaluation of the aspirates. On H&E-stained digitally scanned slides, an unencapsulated, well-demarcated, multilobulated neoplasm expanded the dermis and elevated the ulcerated epidermis, and was composed of sheets and cords of cells within native stroma. The neoplastic cells had moderate amounts of cytoplasm containing moderate-to-large numbers of <1- and up to 2-µm eosinophilic inclusions. The nuclei were round with coarsely stippled chromatin and 1–2 large nucleoli. Nuclear pseudoinclusions (Dutcher bodies) were moderately prevalent, and there was moderate anisocytosis and anisokaryosis with nucleolomegaly and occasional karyomegaly. The mitotic count was 4 per 10 hpfs (total area of 2.37 mm2). With the exception of mildly hyperplastic apocrine glands, the mass was devoid of adnexa. The overlying epidermis was ulcerated with dense neutrophilic infiltration and formation of immature granulation tissue. Where intact, the neoplastic cells were separated from the epidermis by a narrow but well-defined Grenz zone.
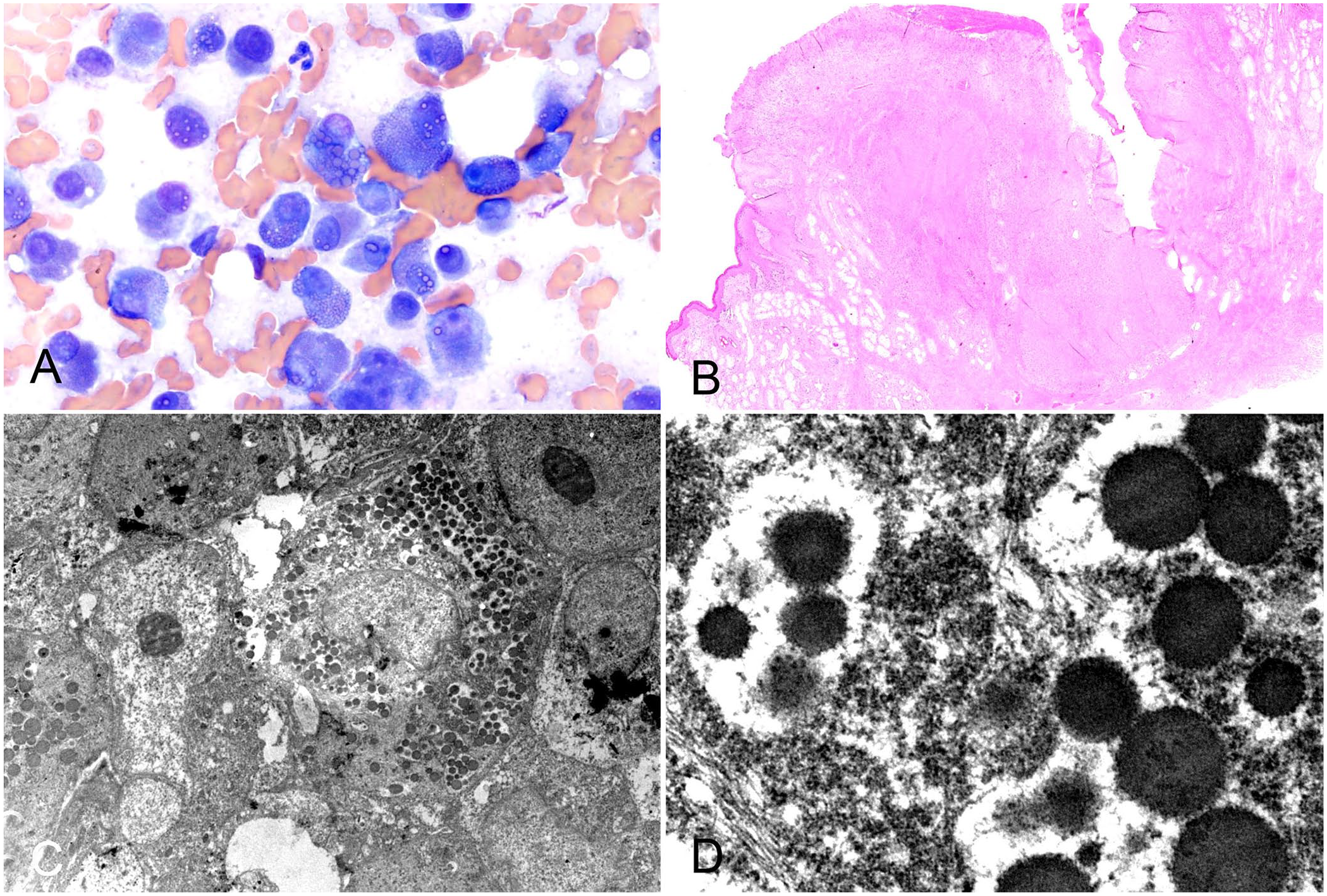
Cytologic, histologic, and ultrastructural morphology of an interdigital plasmacytoma.
Our histologic findings (Fig. 1B) were consistent with a completely excised round-cell tumor, and the top differentials were plasmacytoma or lymphoma with Mott cell differentiation. Electron microscopy was performed, and ultrastructural examination of cytoplasmic granules revealed uniformly electron-dense intracytoplasmic bodies with a relatively smooth surface (Fig. 1C, 1D). 20 Russell bodies are typically contained within ectatic cisternae of the endoplasmic reticulum. 14 Despite the appearance of electrolucent clearance around larger aggregates, higher magnification failed to reveal clear evidence of membranous or cisternal containment of either clustered or individual inclusions. This may be a function of suboptimal formalinized tissue preservation.
Round-cell tumor panels to aid with further classification typically include markers for B- and T-lymphocytes, melanin, plasma cells, and cells of monocyte-macrophage origin. 17 In our case, the panel included multiple myeloma oncogene I/interferon regulatory factor 4 (MUM1; Fig. 2A), vimentin (Fig. 2B), CD20 (Fig. 2C), ionized calcium–binding adapter molecule 1 (Iba1; Fig. 2D), CD3 (Fig. 2E), and melan A (Fig. 2F).
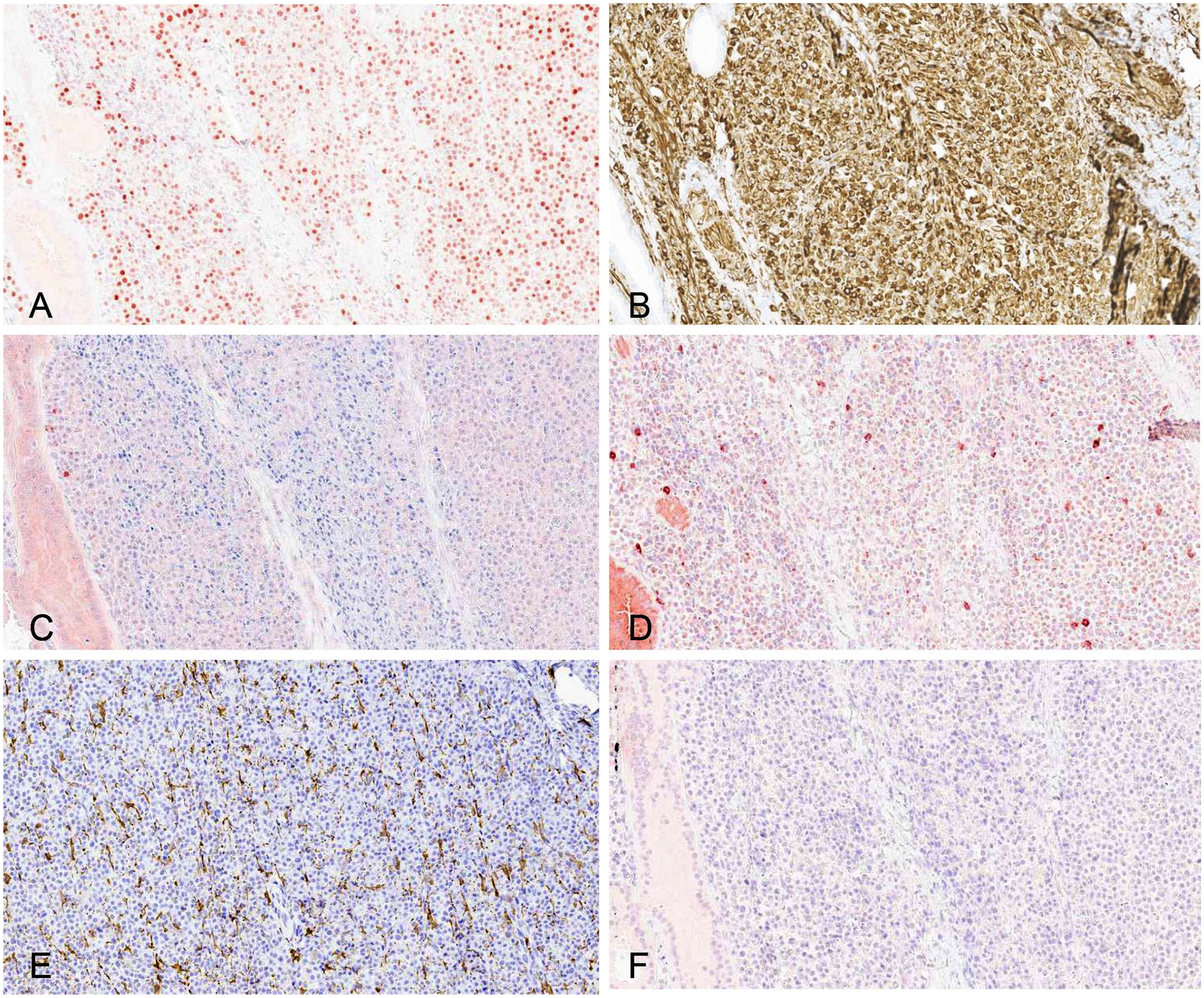
Immunohistochemical stains of an interdigital plasmacytoma from a dog.
The tumor cells were positive for vimentin, confirming non-epithelial lineage. The tumor cells also displayed strongly positive nuclear staining for MUM1, a transcription factor associated with B-cell lymphocyte maturation and, therefore, often used to confirm the diagnosis of plasma cell neoplasms. 16 However, activated T-cells and some subsets of macrophages and dendritic cells can also express MUM1, and labeling has been reported in both canine cutaneous histiocytomas and Langerhans cell histiocytosis. 9 The cells of monocyte-macrophage lineage should demonstrate strong cytoplasmic labeling of Iba1, whereas plasmacytomas do not. 13 The neoplastic cells in our case expressed MUM1, but did not label with Iba1, supporting exclusion of canine cutaneous histiocytoma and Langerhans cell histiocytosis as differentials. 13
In our case, the tumor cells did not express CD20, an immunomarker typically expressed by pre–B- and activated B-cells (both normal and neoplastic), but CD20 is not often expressed by normal or neoplastic plasma cells. 15 CD3 was also negative, which is expected to be expressed by T-cell lymphomas, including epitheliotropic lymphoma. 13 Lastly, the cells did not express melan A.
The cytologic, histologic, and ultrastructural findings were considered consistent with a cutaneous plasmacytoma, a type of extramedullary plasma cell tumor, with Mott cell differentiation. Extramedullary plasma cell tumors are uncommon, with ~86% being cutaneous.1,15 Most cutaneous extramedullary plasma cell tumors (also called plasmacytomas) are solitary. 1 Multiple cutaneous plasma cell tumors, termed cutaneous plasmacytosis, are rare in dogs, and have been associated with lymph node and visceral metastasis. 1 Very few (<1%) cutaneous plasmacytomas are associated with multiple myeloma. 1
Grossly, solitary cutaneous plasmacytomas often appear as discrete, raised, alopecic, round 1–2-cm masses. They develop most frequently on the head and limbs, particularly the pinna and ventral paw, but can occur in other locations. 1 Cutaneous plasmacytomas typically exfoliate readily with fine-needle aspiration and can display substantial atypia, such as anisocytosis, anisokaryosis, and multinucleation, while still having a benign clinical behavior. 4 Some tumors are associated with extracellular eosinophilic fibrillar material consistent with amyloid, which is a fibril-forming protein associated with other non-fibrillogenic components. 4 Solitary cutaneous plasmacytomas are not typically associated with a monoclonal gammopathy. 19 Around the time of presentation, a clinical chemistry panel was performed by the referring veterinarian in this patient, and the total protein, albumin, and globulin concentrations were within their respective RIs.
Approximately 8 mo after excision of the tumor, the patient was presented to the primary veterinarian with a 5-d history of diarrhea, labored breathing, and anorexia. On radiographic imaging, the patient was noted to have bicavitary effusion and suspected intestinal wall thickening. Neither thoracocentesis with fluid analysis nor urinalysis were performed. However, a chemistry panel revealed severe hypoproteinemia (26 g/L [2.6 g/dL]) due to severe hypoalbuminemia (<10 g/L [<1.0 g/dL]). Based on the signalment, history of diarrhea, and presence of severe hypoalbuminemia with bicavitary effusion, a presumptive diagnosis of protein-losing enteropathy was made. Progressive plasma cell disease was not suspected by the primary veterinarian. Unfortunately, the patient died shortly after this diagnosis, and an autopsy was not performed.
Mott cells are B-cell lineage lymphocytes or plasma cells that contain cytoplasmic inclusions of accumulated non-secreted immunoglobulins called Russell bodies, which have been found in association with both neoplastic and non-neoplastic conditions, such as inflammatory and autoimmune conditions.3,4 In non-neoplastic conditions, Russell bodies are thought to be formed when immunoglobulins are produced at a rate faster than what can be secreted from differentiated plasma cells. 18 Russell bodies may also be produced by neoplastic plasma cells or lymphocytes that have not differentiated into plasma cells. 18 In veterinary medicine, Mott cell–differentiated B-cell lymphomas have been described in dogs, ferrets, hedgehogs, and cats, and typically include diffuse involvement of the lymphoid tissues or gastrointestinal tract.2,3,5,6,8,10,11,18 Extramedullary plasma cell neoplasms have been described in the canine gastrointestinal tract, including the colon, rectum, 12 and tonsils 7 ; however, Russell bodies are not described as a distinctive feature of the tumors.
The cytologic and histologic findings, special stains, immunohistochemical labeling, and ultrastructural findings support the diagnosis of an uncommon tumor (i.e., cutaneous plasmacytoma) with novel morphology (i.e., Mott cell differentiation). Specifically, the defining features for this tumor include discrete, plasmacytoid, round cells with cytoplasmic inclusions that stained positively with MUM1 and did not immunolabel with markers for lymphocytes, melanocytes, and cells of macrophage and monocyte origin. EM was useful to confirm the cytoplasmic inclusions to be Russell bodies; however, EM is not a routine diagnostic test, and we suspect that the history, cytologic and histologic findings, special stains, and immunohistochemistry are likely sufficient for most similar-appearing tumors. In a routine diagnostic setting, EM would not be necessary for the diagnosis of this tumor. We retrieved no described cases of a cutaneous Mott cell–differentiated plasmacytoma in the literature for any species after searching Google Scholar, PubMed, and Web of Science, which suggests that a cutaneous plasmacytoma with Mott cell differentiation has not been described previously in veterinary medicine.
Footnotes
Acknowledgements
We thank Judith Stasko, the Supervisory Electron Microscopist at the U.S. Department of Agriculture–National Animal Disease Center for providing the electron microscopy images, and Dr. Maggie Wilson for providing clinical details and pathology samples.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
