Abstract
Ten dogs with neuroendocrine carcinoma of the liver were selected for inclusion in the study. Clinical signs were anorexia (7), vomiting (5), polydipsia/polyuria (3), icterus (2), lethargy (2), weight loss (2), paresis (1), ataxia (1), weakness (1), collapse (1), and urinary tract infection (1). Hematologic and biochemical abnormalities included anemia (2/8), leukocytosis (4/8), high liver enzyme activity (serum alkaline phosphatase, 7/9; alanine transaminase, 7/9; aspartate transaminase, 8/9), and high total bilirubin (6/9). Grossly, the tumors were diffuse, involving all liver lobes in six dogs, and two dogs had various-sized nodules in addition to diffuse involvement. Histologically, there were eight tumors with solid or trabecular pattern (group A), one tumor with cords or rows of neoplastic cells (group B), and one tumor with multiple rosette-like structures (group C). immunohistochemical studies revealed that all 10 neoplasms were positive for at least one of the endocrine markers used: neuron-specific enolase (NSE; 8/10), synaptophysin (5/10), and chromogranin-A (3/10). A panel of NSE, chromagranin-A, and synaptophysin detected 100% of the tumors in our series. Electron microscopy confirmed the diagnosis by the presence of intracytoplasmic neurosecretory granules in the two examined cases. our results show that neuroendocrine markers commonly used in humans can be used for the diagnosis of hepatic neuroendocrine carcinoma in dogs, preferably a panel of synaptophysin, chromagranin-A, and NSE because chromogranin-A alone is not as useful in dogs as in humans.
Neuroendocrine cells, which produce peptides and amines, are distributed in many organs and tissues. 3, 9, 22, 28 The most common sites are the respiratory and digestive tracts. In the past, these cells were called “APUD” (amine precursor uptake and decarboxylation) cells. They were thought to be neuroectodermal in origin, arising from the neural crest. 18, 19 At present, these neuroendocrine cells, especially the epithelial-associated cells, are considered to be endodermal in origin and are designated as diffuse endocrine system. 9, 22
The term “carcinoid” was coined by Oberndorfer in 1897 to distinguish a group of mostly benign, small intestinal tumors from aggressive adenocarcinomas. Although the terms carcinoid and “neuroendocrine carcinoma” are interchangeably used, the latter term is considered more appropriate. 10, 25
The clinicopathologic spectrum of neuroendocrine tumors ranges from the benign carcinoid to the aggressive neuroendocrine carcinoma. These tumors are seen in a wide range of organs in humans but have rarely been described in domestic animals. 3, 9, 13, 16, 22, 25 Neuroendocrine tumors arise from the diffuse neuroendocrine system. 9 In humans, the gastropancreatic and bronchopulmonary organs are the most common sites. 3, 9, 22, 25, 26 Primary neuroendocrine carcinoma of the liver is rarely reported, but single cases in middle-aged females predominate. 2, 6–8, 21 Histologically, neuroendocrine carcinomas of the liver in humans are described as having cords, trabeculae, or glandular (rosette-like) patterns. 15 Most of them stain with Grimelius silver stain. Immunohistochemically, they consistently express chromogranin-A, NSE, and synaptophysin, and occasionally cytokeratin, somatostatin, serotonin, and other peptides. 2, 6, 8 Ultrastructurally, these tumors have 40–100 μm intracytoplasmic neurosecretory granules. 2, 6, 8
Neuroendocrine carcinomas have rarely been described in dogs and cats. The sites include the intestines, 17 esophagus, 13 nasal cavity, 11 skin, 17 bile duct, 12 gallbladder, 29 liver, 15 and, more recently, the nasopharynx. 16
The first detailed reports of primary hepatic neuroendocrine carcinoma in dogs were published in 1980 and 1981. 14, 15 A summary of findings from these previous studies showed that 14% (15/110) of primary hepatic neoplasms, exclusive of tumors of nonepithelial origin, were considered to be of neuroendocrine origin because of their clinical, morphologic, and biologic distinctions from the hepatocellular and cholangiocarcinomas in the same series. All the tumors in that series stained positively with Grimelius silver stain. Most of the dogs (71%) were under 10 years of age, and there was no sex or breed predilection. High white blood cell count, low hemoglobin, and higher than normal values for liver function tests were common. Grossly, all liver lobes were involved in 10 dogs with diffuse disease; in the other dogs, the livers were not enlarged, and the lesions were nodular.
The morphologic patterns were similar to those reported in humans with neuroendocrine carcinoma, 2, 7, 9, 15 and three distinct patterns were observed: 1) solid groups of cells with fibrovascular stroma, 2) groups or cords of cells with fibrovascular stroma, and 3) alveolar pattern with multiple rosette-like structures and fibrovascular stroma. Peritoneal carcinomatosis and metastasis to regional lymph nodes were the most common extrahepatic manifestations in most of the dogs. 15 Immunohistochemistry and electron microscopy were not done in this series.
A literature survey done from 1980 shows only one case of hepatic carcinoid in a dog, which had hyper-corticism and hypokalemia. 4 Another case report describes a neuroendocrine carcinoma in the gallbladder of a dog. 29 Single cases of neuroendocrine carcinoma in the liver and extrahepatic bile duct have been described in a study of 47 nonhematopoietic neoplasms of the hepatic and biliary systems of cats. 12
In this study, we describe the clinical, histologic, and immunohistochemical findings in 10 cases and electron microscopic findings in two cases of hepatic neuroendocrine carcinoma in dogs.
Materials and Methods
Ten cases of neuroendocrine carcinoma of the liver in dogs were selected from the pathology department files of The Animal Medical Center. Signalment, results of hematologic and biochemical testing, gross pathologic findings, and follow-up information were collected from the medical and pathology records. All the available slides were examined to confirm the diagnosis and primary location. Only cases in which adequate slides were available to confirm neuroendocrine carcinoma of the liver were included in the study. All tissues had been fixed in 10% buffered formalin, routinely processed, and stained with hematoxylin and eosin.
Morphologically, the neoplasms were divided into three histologic groups on the basis of previously described patterns of neuroendocrine carcinoma in humans and dogs. 15, 16, 25 In cases in which more than one pattern was observed, the predominant component determined the tumor group. In brief, the morphologic groups as described are as follows. Group A tumors consist of solid groups or nests of neoplastic cells separated by fibrovascular stroma, often with a peritheliomatous arrangement of peripheral cells. The cells are round to polygonal, with granular eosinophilic cytoplasm and vesiculated nuclei with prominent nucleoli (Fig. 1). Group B tumors consist of cords of neoplastic cells separated by fibrovascular stroma (Fig. 2). Group C tumors consist of groups of acinar or rosette-like structures, enclosed in similar stroma, and lined by cuboidal or columnar cells (Fig. 3).
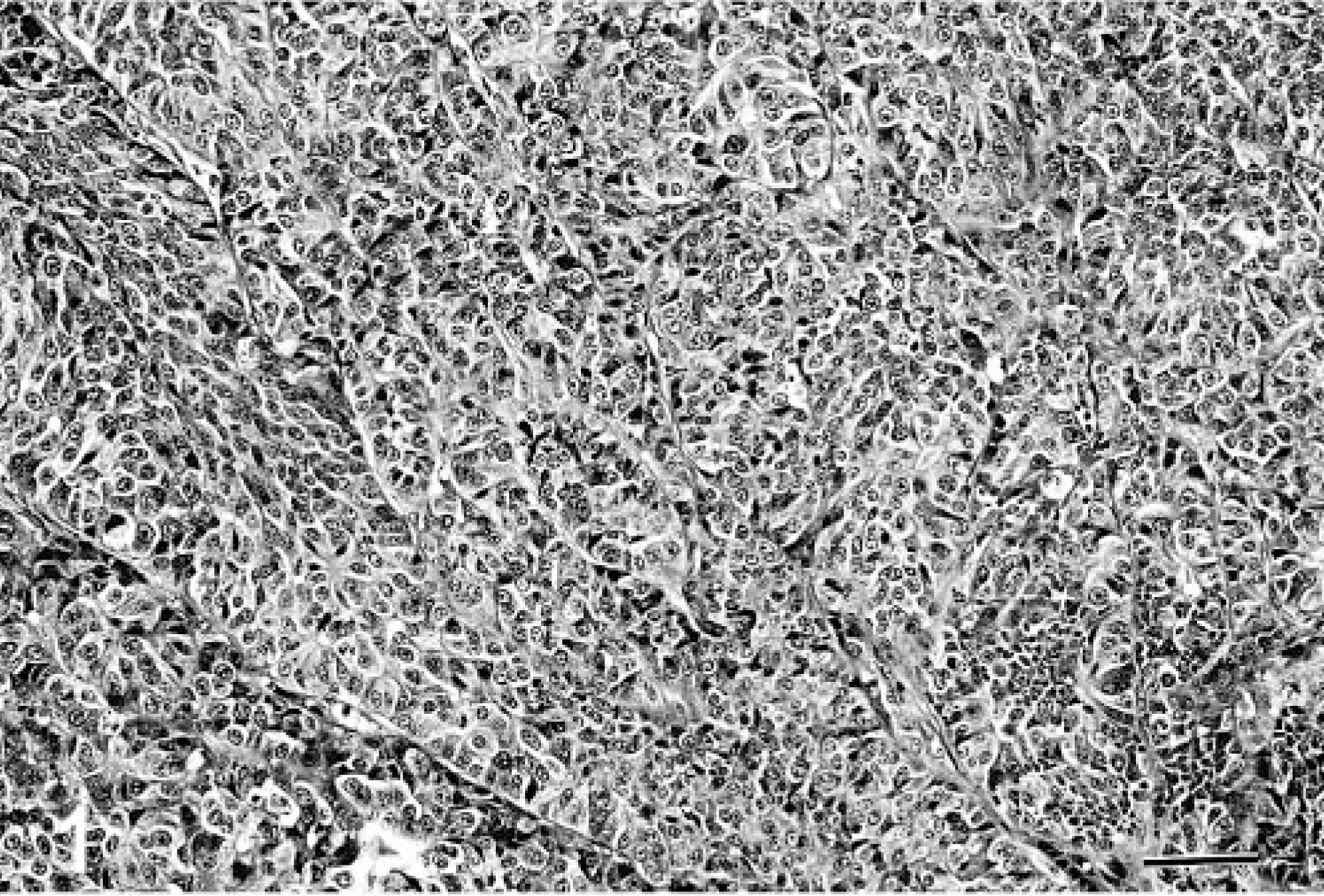
Liver; canine. Neuroendocrine carcinoma with trabecular cell arrangement. HE. Bar = 217 μm.
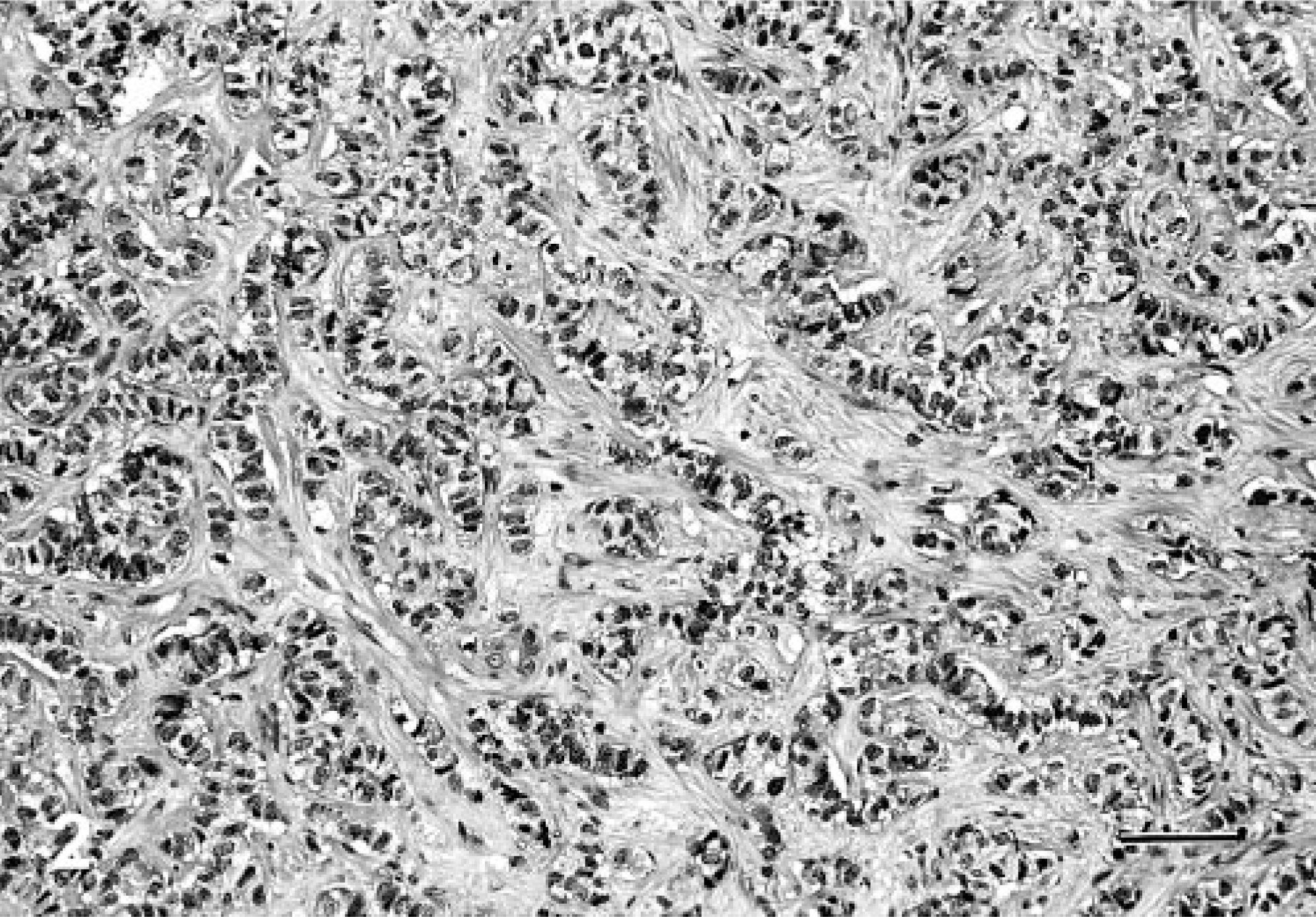
Liver; canine. Neuroendocrine carcinoma with cells arranged in rows. HE. Bar = 217 μm.
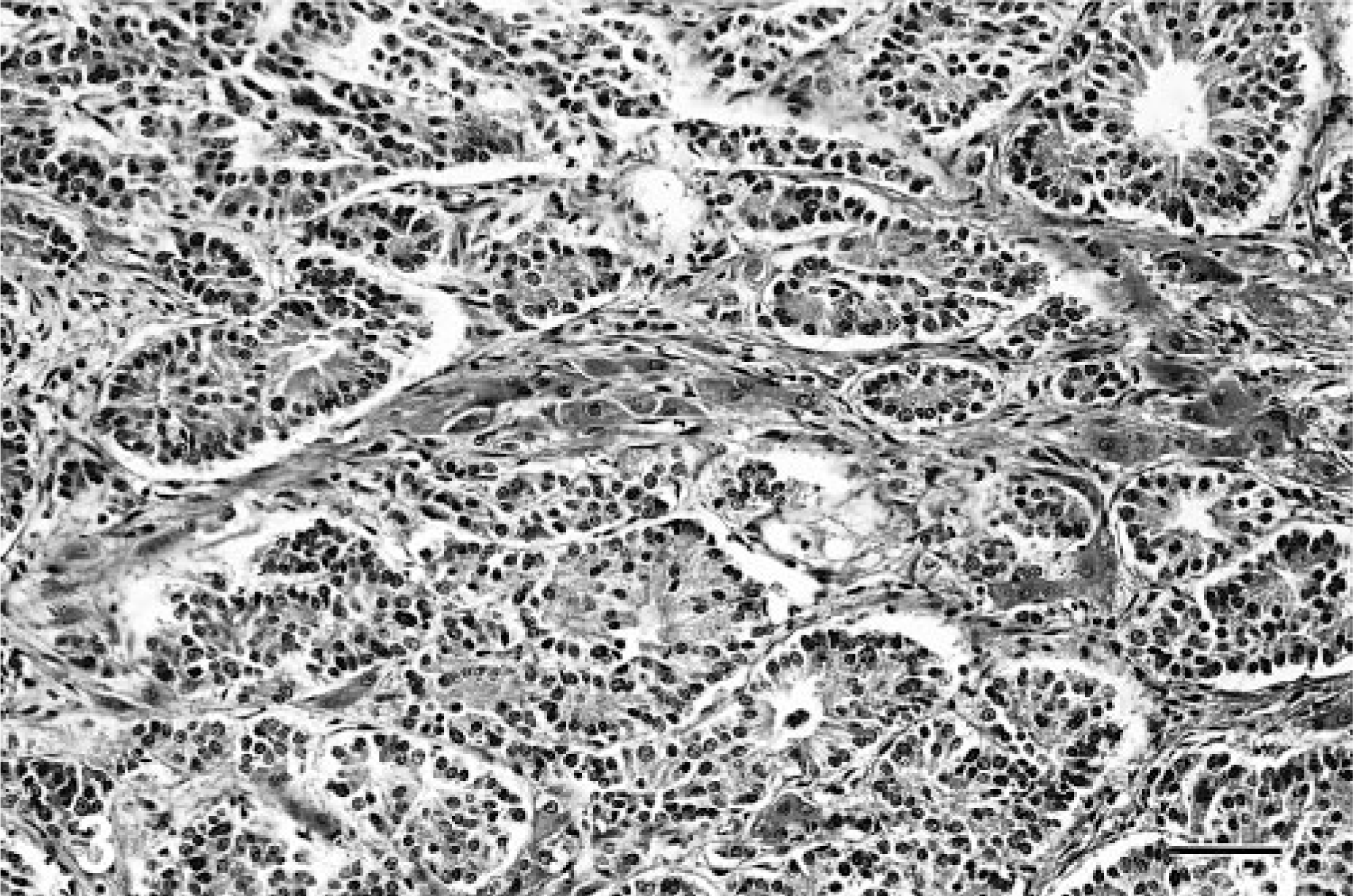
Liver; canine. Neuroendocrine carcinoma with cells arranged in rosettes. HE. Bar = 217 μm.
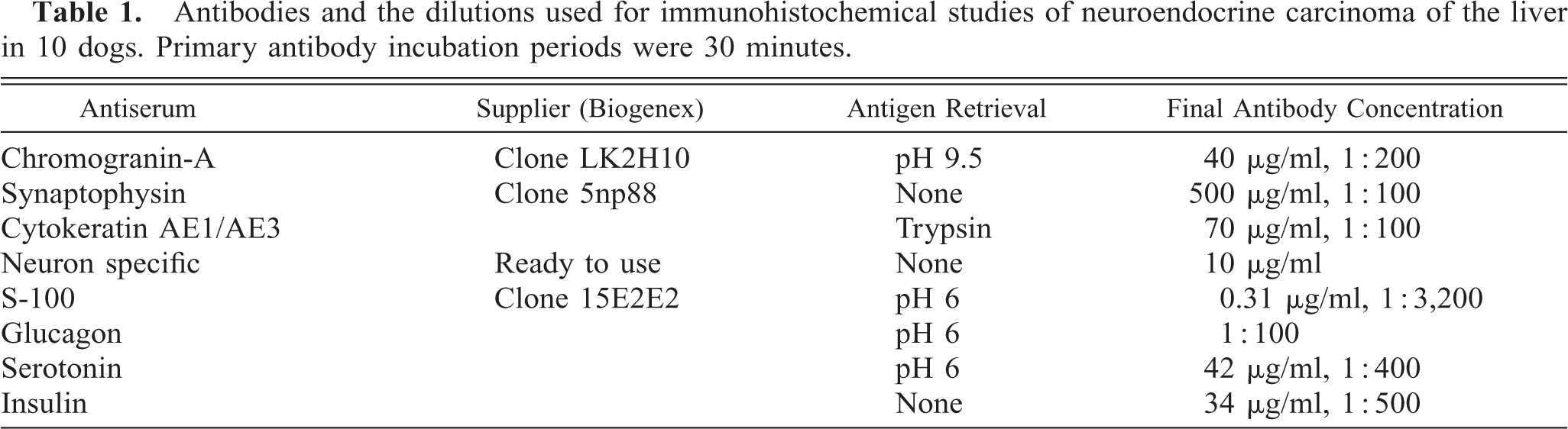
Selected slides from each case were immunohistochemically stained using a computer-controlled automated system (Optimax Plus, Biogenex Laboratories, San Ramon, CA), within the Flaherty Comparative Oncology Laboratory at the Animal Medical Center. Antigen-retrieval pretreatment was carried out either by heating the tissue sections in buffered solutions (pH 6.0 or 9.5) or by enzymatic proteolytic digestion with trypsin (Table 1). Tissue sections were treated with 3% hydrogen peroxide, washed in buffer, and avidin and biotin block applied (Biocare Medical, Walnut Creek, CA). Sections were incubated with the primary antibody for 30 minutes at room temperature. Chromagen staining was achieved by use of a modified, biotin-streptavidin amplified detection system (Biogenex Laboratories), and color was developed by 3,3′-diaminobenzidine. Sections were then counterstained with hematoxylin. Negative controls were produced by replacing the primary antibody with polyclonal rabbit antisera raised against an irrelevant antigen (negative-control sera, Biogenex Laboratories). Paraffin-embedded arrays containing different canine tissues (specifically pancreatic islets, stomach, adrenal medulla, and skin) were used as controls to ensure correct positive and negative expression. A list of the antibodies used and their dilutions is given in Table 1.
Antibodies and the dilutions used for immunohistochemical studies of neuroendocrine carcinoma of the liver in 10 dogs. Primary antibody incubation periods were 30 minutes.
Immunohistochemically processed slides (for which at least one of the endocrine markers was positive) were evaluated by two of the authors (AP and SJN) both individually and jointly. The degree of expression was graded as 0, 1+, 2+, or 3+ on the basis of the approximate number of cells that were expressing the antigen of interest—grade 3+, more than 50% of the cells positive; grade 2+, 25–50% of the cells positive; grade 1+, less than 25% or rare cells positive; and zero, when there was no observed staining.
Tissues for electron microscopy were fixed in a buffered (pH 6) formaldehyde and gluteraldehyde mixture and processed according to standard procedures.
Results
Ten dogs were included in this study. The number of males and females was equal. Breeds represented included mixed breed (3), Boxer (2), Dalmatian (1), Doberman (1), Golden Retriever (1), Shetland Sheep-dog (1), and Poodle (1). The age range was 3–16 years (median age, 10 years and mean age, 9.8 years). Clinical signs included: anorexia (7), vomiting (5), polydipsia/polyuria (3), lethargy (2), weight loss (2), paresis (1), ataxia (1), weakness (1), and collapse (1). Associated conditions included: icterus (2) and urinary tract infection (1).
Results of hematologic and biochemical testing are given in Table 2. Abnormalities included anemia (2/8), leukocytosis (4/8), high liver enzyme activity (serum alkaline phosphatase, 7/9; alanine transaminase, 7/9; aspartate transaminase, 8/9), and high total bilirubin (6/9). High lactate dehydrogenase and low plasma proteins were each seen in one dog (1/9).
Results of hematologic and biochemical testing in 10 dogs with hepatic neuroendocrine carcinoma.
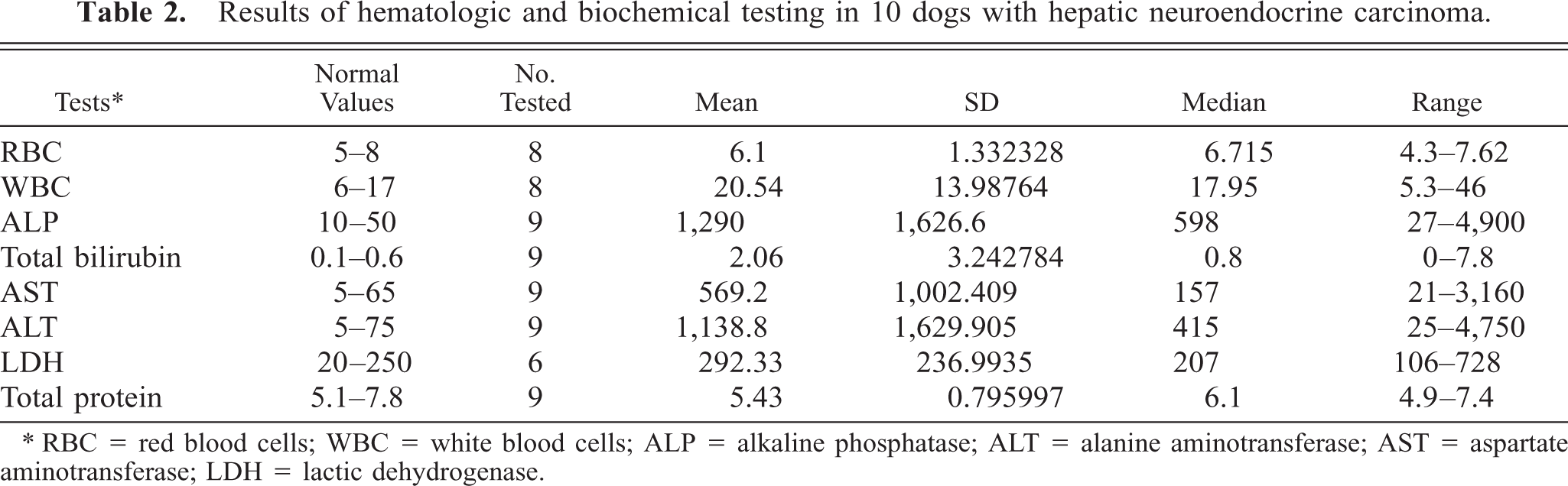

∗ RBC = red blood cells; WBC = white blood cells; ALP = alkaline phosphatase; ALT = alanine aminotransferase; AST = aspartate aminotransferase; LDH = lactic dehydrogenase.
All 10 dogs were euthanatized soon after diagnosis (1–8 days; mean, 3 days). In six dogs, the tumors were described grossly as diffuse, involving all liver lobes, with no nodular lesions. In two dogs, the liver had various-sized nodules in addition to diffuse involvement. In the other two dogs, gross descriptions were not available. Histologically, there were eight group A tumors (solid), one group B tumor (cords or rows of neoplastic cells), and one group C tumor (multiple rosette-like structures).
The results of immunohistochemical studies are given in Table 3. None of the neoplasms expressed AE1/AE3, S-100 protein, glucagon, or gastrin. All 10 neoplasms were positive for at least one of the endocrine markers used: NSE (8/10, Fig. 4), synaptophysin (5/10, Fig. 5), and chromagranin-A (3/10, Fig. 6). One neoplasm expressed all the three markers. Three neoplasms stained only for NSE and one only for chromagranin-A. In addition, three neoplasms stained for serotonin (3/10) and one for insulin (1/10).
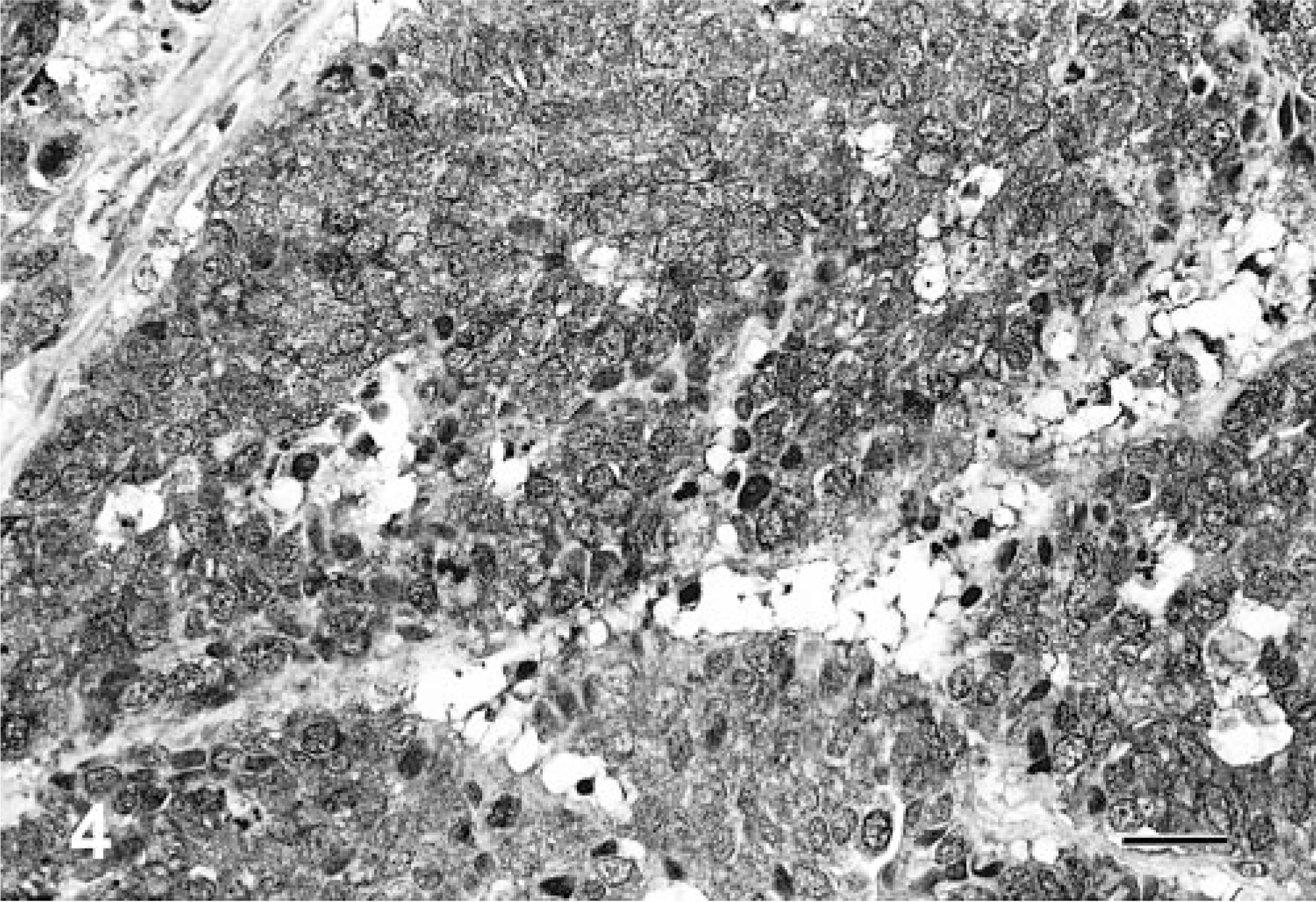
Liver; canine. Neuroendocrine carcinoma cells demonstrating positive expression for neuron-specific enolase (NSE). Avidin–biotin–peroxidase complex method, hematoxylin counterstain. Bar = 108 μm.
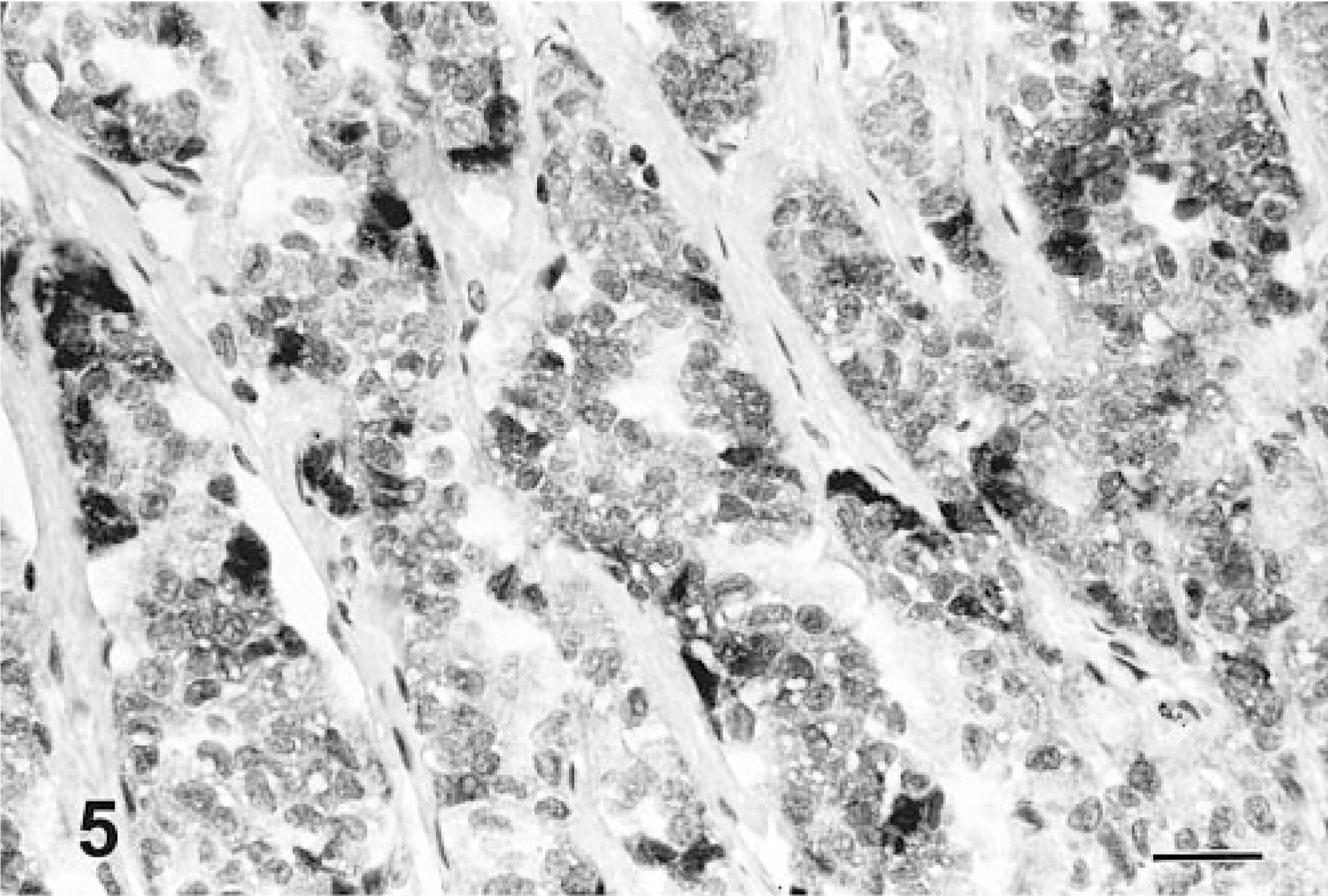
Liver; canine. Neuroendocrine carcinoma cells demonstrating positive immunohistochemical expression for synaptophysin. Avidin–biotin–peroxidase complex method, hematoxylin counterstain. Bar = 108 μm.
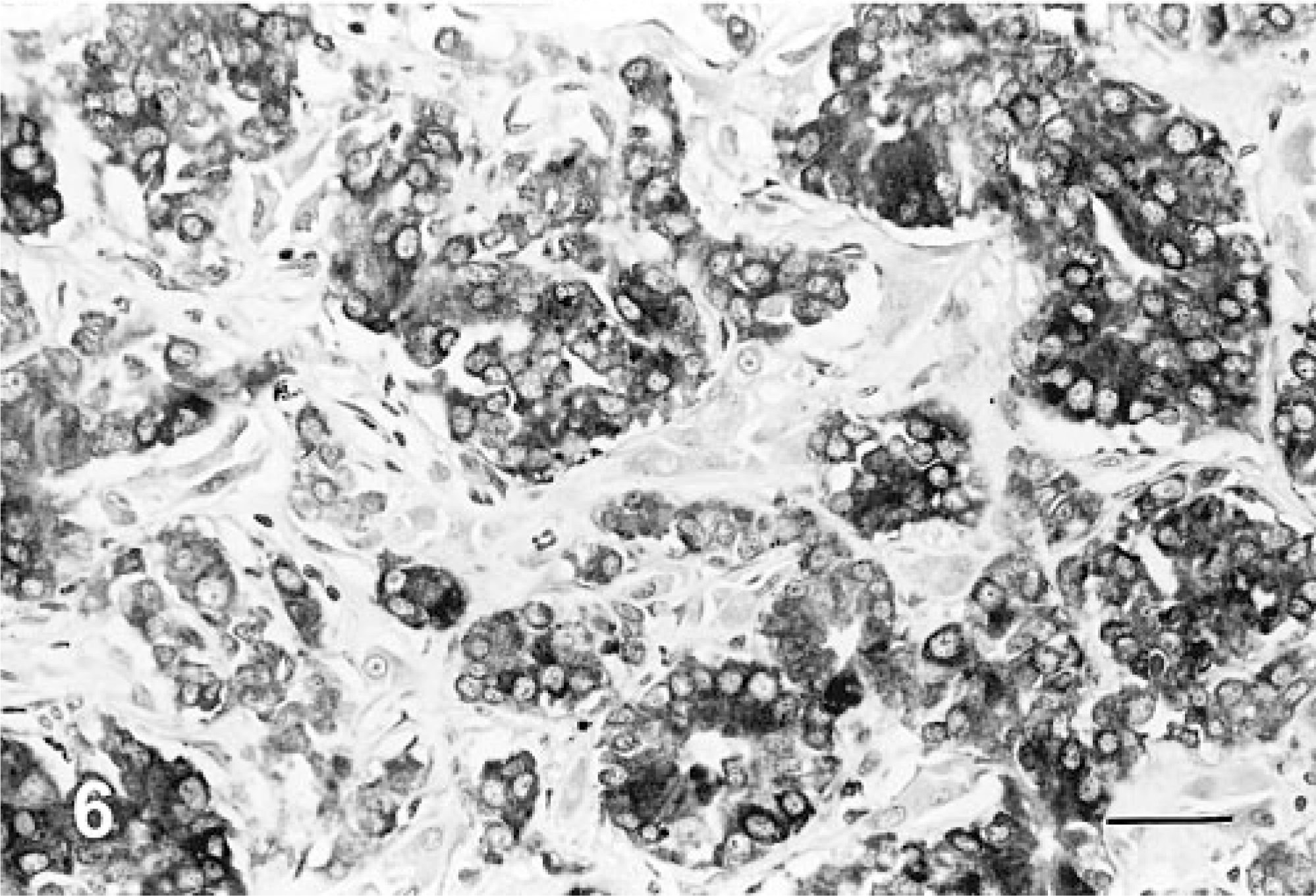
Liver; canine. Neuroendocrine carcinoma cells demonstrating positive immunohistochemical expression for chromagranin-A. Avidin–biotin–peroxidase complex method, hematoxylin counterstain. Bar = 108 μm.
Results of immunohistochemical studies in 10 dogs with neuroendocrine carcinoma of the liver.∗.
∗ Syn = Synaptophysin; NSE = neuron-specific enolase; Chr-A = Chromagranin-A.
Electron microscopy showed that both neoplasms had conspicuous, intracytoplasmic, dense-core neuro-secretory granules, which ranged from round (Fig. 7) to pleomorphic (Fig. 8). Both showed epithelioid morphology, closely opposed cell membranes, and continuous basement membrane. The nuclei were round or slightly irregular, with small nucleoli.
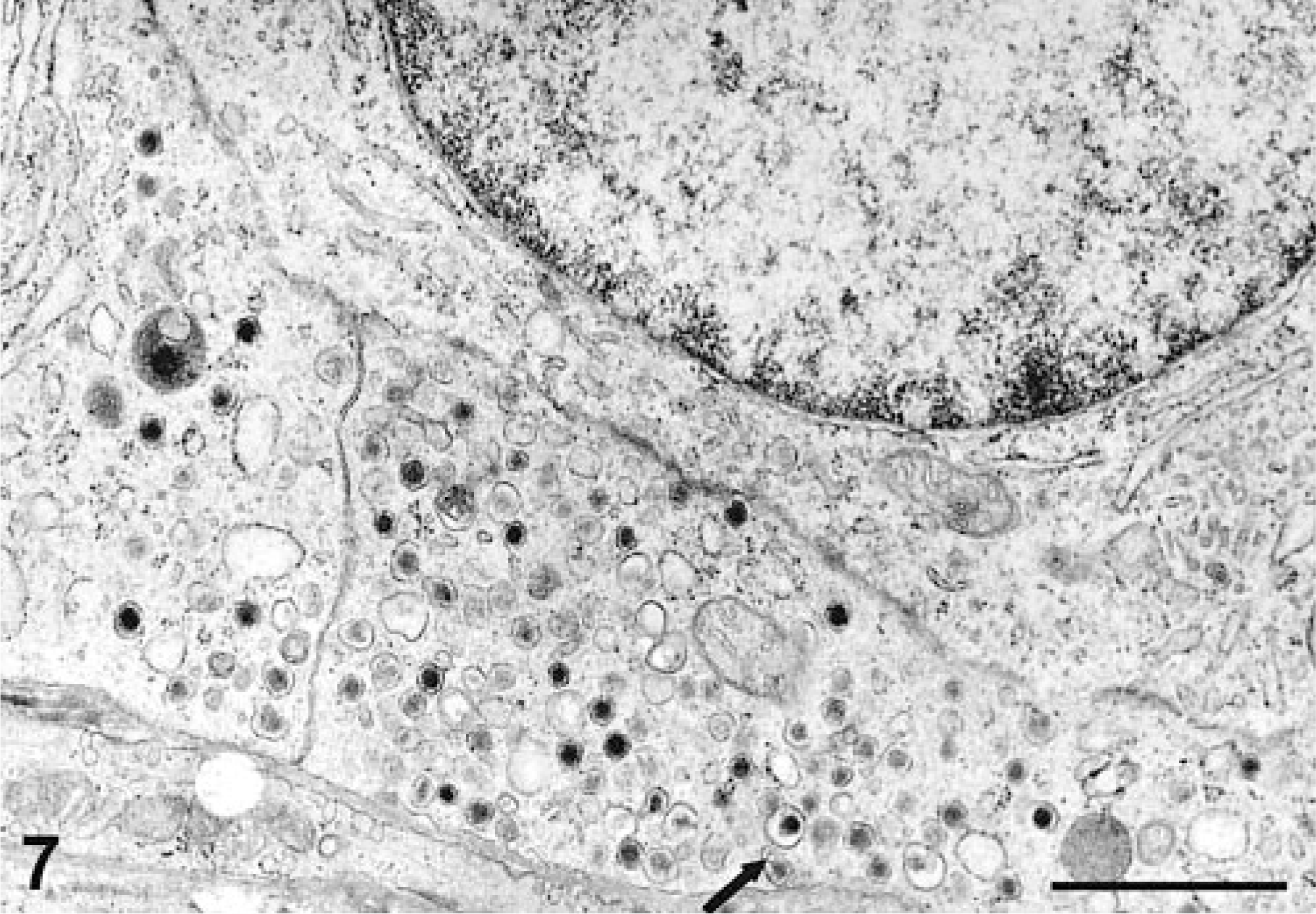
Liver, canine. High-power electron micrograph showing abundant, round, membrane-bound neurosecretory-type granules with prominent halos concentrated in the elongated cell processes (arrow). Uranyl acetate and lead citrate counterstain. Bar = 1 μm.
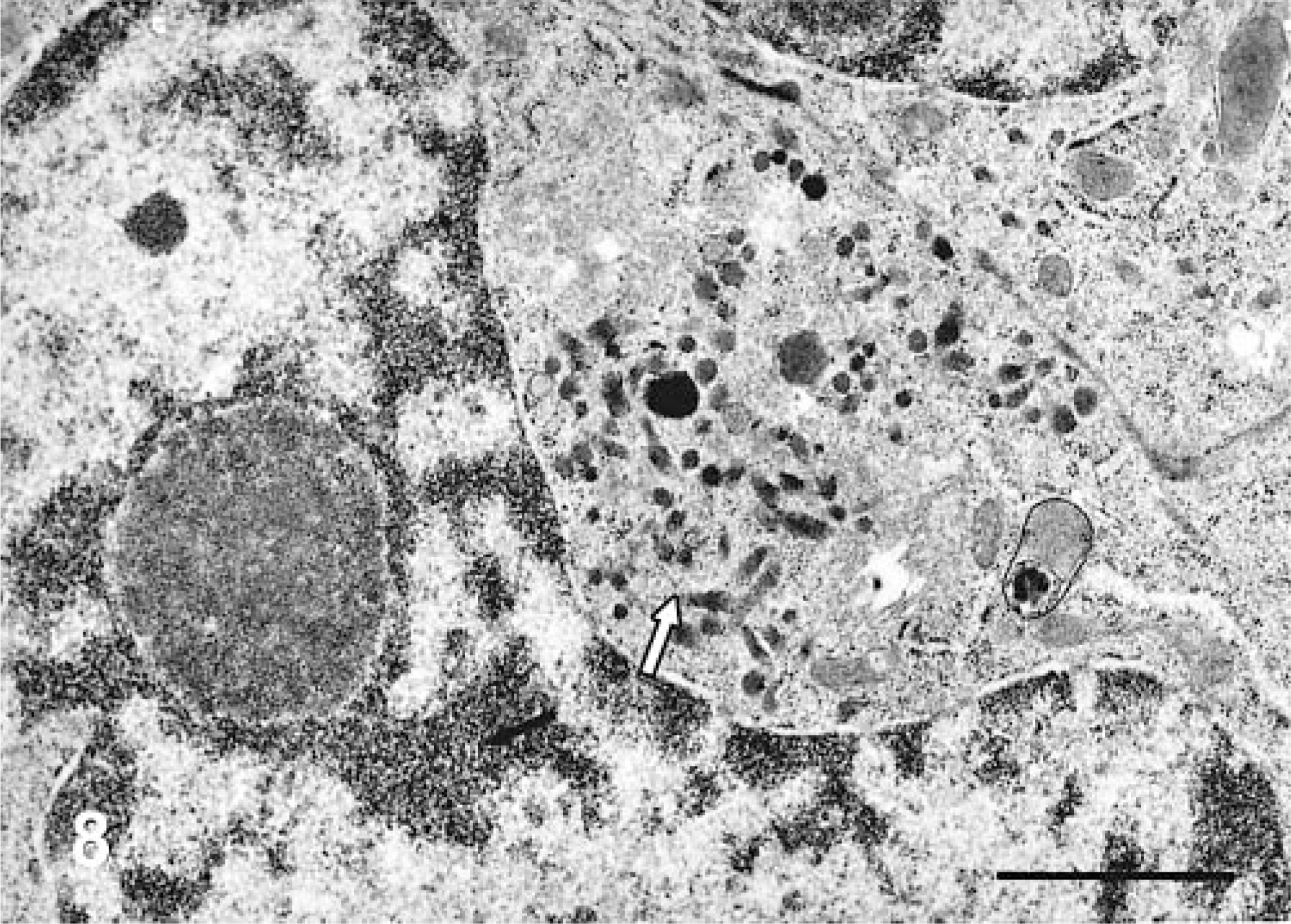
Liver; canine. Electron micrograph of a portion of an epithelial cell showing a cluster of oval and pleomorphically shaped, dense-core granules (arrow). Uranyl acetate and lead citrate counterstain. Bar = 1 μm.
Discussion
One additional case of hepatic neuroendocrine carcinoma in the dog 4 has been described since 1980, when the first studies of this tumor were published. 14, 15 This group of hepatic tumors, about 14% of the population of hepatic neoplasms in the 1980 study, may be underreported or less well recognized; and possibly these neoplasms are being diagnosed as cholangiocarcinoma or hepatocellular carcinoma. However, the distinct morphologic features of neuroendocrine carcinoma distinguish it from other epithelial neoplasms of the liver. The signalment of the 10 dogs of this study was similar to that of the 1980 group, but because we had no reference population for the dogs of this study, this comparison cannot be validated. 14, 15 Immunohistochemical studies were not reported in the 1980 study, but in all cases, the presence of neurosecretory granules was confirmed by use of modified Grimelius silver stain.
Immunohistochemical studies of hepatic neuroendocrine carcinoma of dogs have not been done, except for a single case report, in which none of the antibodies used were positive. 4 Results of this study reveal that the commonly used neuroendocrine markers for neuroendocrine carcinoma in humans, synaptophysin, NSE, and chromagranin-A, can also be used to distinguish these tumors in dogs. All 10 neoplasms were positive for at least one of three markers (Table 3). Eight of 10 and five of 10 were positive for NSE and synaptophysin, respectively. Only three of 10 were positive for chromogranin-A, and only three of 10 were positive for serotonin; but when the results for serotonin and insulin (1/10) were included, most of the tumors in this series (6/10) expressed more than two peptides or amines. The possibility that insulin and serotonin expression in a small percentage of tumors might represent metastatic tumor from primary pancreatic neoplasms was excluded by examination of surgical records and histopathologic samples. Chromogranin-A, which is one of the most common antibodies used as an endocrine marker in humans, 3, 9, 21 is less useful than synaptophysin and NSE in dogs and cats. 13, 16, 17 A combination of neuroendocrine markers would be a better choice than chromagranin-A alone, in these species. A panel of NSE, chromagranin-A, and synaptophysin detected 100% of the tumors in this series, whereas combinations of NSE and synaptophysin or NSE and chromagranin-A detected 90% of cases, respectively.
In this series, none of the tumors expressed AE1/AE3. A similar observation has been made in previous reports of neuroendocrine carcinoma in dogs and cats. 13, 16, 17 AE1/AE3 can possibly be used to differentiate adenocarcinoma from neuroendocrine carcinoma in dogs and cats. 6
Hepatic neuroendocrine carcinoma in humans expresses more peptides than was demonstrated in this study, although the most commonly reported peptides are chromogranin-A, chromagranin-B, NSE, synaptophysin, serotonin, gastrin, and insulin. 2, 6, 8, 21 In humans, high serum concentrations of NSE, chromogranin, and 5-hydroxyindole acetic acid are being used to diagnose neuroendocrine carcinoma, and this may be contemplated in dogs. 2, 8, 21 A panel of NSE, chromagranin-A, and synaptophysin is recommended to diagnose hepatic carcinoids in dogs because 100% of our cases were detected with this panel. Use of single markers such as NSE, synaptophysin, and chromagranin-A detected 80, 50, and 30%, respectively. Histologic differentiation of hepatic neuroendocrine carcinoma from biliary adenocarcinoma and hepatocellular carcinoma is essential to the pathologist, and the former neoplasm may be associated with shorter survival/euthanasia intervals (mean 3 days in this study).
As to the cell of origin of hepatic neuroendocrine carcinoma, studies of various hepatic diseases in humans and results of animal experiments suggest that the canal of Hering harbors facultative stem cells. 1, 26, 27 The facultative stem cells give rise to hepatocytes and small oval cells, and the oval cells act as bipotential progenitor cells giving rise to hepatocytes and bile ductular cells. 5 The ductular cells stain with cytokeratin 19, a biliary epithelial marker. The proliferating bile ductular cells, which are intermediate cells between hepatocytes and bile ductular cells, immunohistochemically express chromogranin-A, and electron microscopic studies reveal dense-core neuroendocrine granules. 20, 23, 24 These pluripotent cells may be the precursor cells of neuroendocrine carcinoma in the liver. It has been suggested that the oval cells and ductular cells develop into hepatocellular carcinoma and cholangiocarcinoma. 20, 26 There is also growing evidence that bone marrow stem cells may contribute to the regeneration of hepatic cells. 1, 20, 27
Footnotes
Acknowledgements
We thank A. Christine MacMurray for editorial support.
Presented in part as an abstract at 2003 Veterinary Cancer Meeting, Madison, WI.
1Present address: Department of Pathology, College of Veterinary Medicine, University of Tennessee, Knoxville, TN.
