Abstract
The intra-abdominal tumor developing in the uterus and lung of a domestic Shorthair cat was examined histopathologically and immunohistochemically. The tumor showed a proliferation of both endometrial stromal and smooth muscle cells accompanied by prominent vasculature. There were well-differentiated endometrial glands, and tubuli made up a monolayer of eosinophilic cuboidal epithelium. Immunohistochemically, the spindle-shaped cells and half of the stromal-like cells reacted to caldesmon and desmin antibodies. The neoplastic epithelium expressed AE1/AE3 cytokeratin. Feline endometrial stromal tumor has, to the best of our knowledge, not been reported previously and has smooth muscle and glandular components that are a unique variant to the human counterpart.
The majority of tumors developing in the feline uterus are adenocarcinomas. Although leiomyomas and leiomyosarcomas have sometimes been reported, malignant mesenchymal tumors, except lymphomas, occur rarely in the reproductive tract of cats.6 In women, endometrial stromal tumors are known to be rare tumors that have been characterized by the growth of tumor cells resembling nonneoplastic proliferative endometrium.1,4,8,12 Endometrial stromal sarcomas may show a variety of morphologic appearances, including epithelial differentiation, a sex cord-like pattern, smooth muscle differentiation, and fibrous/myxoid appearances. These variants often lead to the difficulties of a differential diagnosis among uterine mesenchymal tumors.10,11 This report describes a novel uterine neoplasm with components of endometrial stroma, smooth muscle, and endometrial glands that developed in the abdominal cavity of a queen and metastasized to the lungs.
A 12-year-old domestic Shorthair queen, weighing about 4.2 kg, was referred a local veterinarian with complaints of depression, anorexia, panting, vomiting, and a purulent vaginal discharge. The abdominal palpation revealed a 3 × 10 cm solid mass in the abdominal cavity. Thoracic radiographs showed that the different-sized masses measured less than 1.5 cm in diameter in the lungs. The mass in hilum pulmonis depressed the heart. The cat died 9 days after the first medical examination.
The resected intra-abdominal and lung masses were fixed in 10% neutral-buffered formalin and embedded in paraffin. Sections were stained with HE and Masson trichrome. Using the streptavidin-biotin method, we performed immunohistochemical staining with the following primary antibodies: AE1/AE3 cytokeratin monoclonal antibody (DakoCytomation N1590, Dako, Kyoto, Japan); vimentin monoclonal antibody (DakoCytomation M0725, Dako); desmin polyclonal antibody (DakoCytomation A0611, Dako); alpha-smooth muscle actin (SMA) monoclonal antibody (DakoCytomation M0851, Dako); estrogen receptor (ER) monoclonal antibody (Novocastra NCL-ER-6F11, Vision Systems Inc., Norwell, MA) and caldesmon monoclonal antibody (Novocastra NCL-CALD, Vision Systems Inc.). A normal feline uterus was used as a positive control of these immunostains.
Grossly, the intra-abdominal mass measured 5.5 × 4.5 × 3.0 cm, was grayish-white on the cut surface, and was contiguous to a portion of the uterus (Fig. 1). Many grayish-white nodules 0.1–1.2 cm in diameter were observed in the lungs. There were no gross changes in the ovaries and mammary glands.
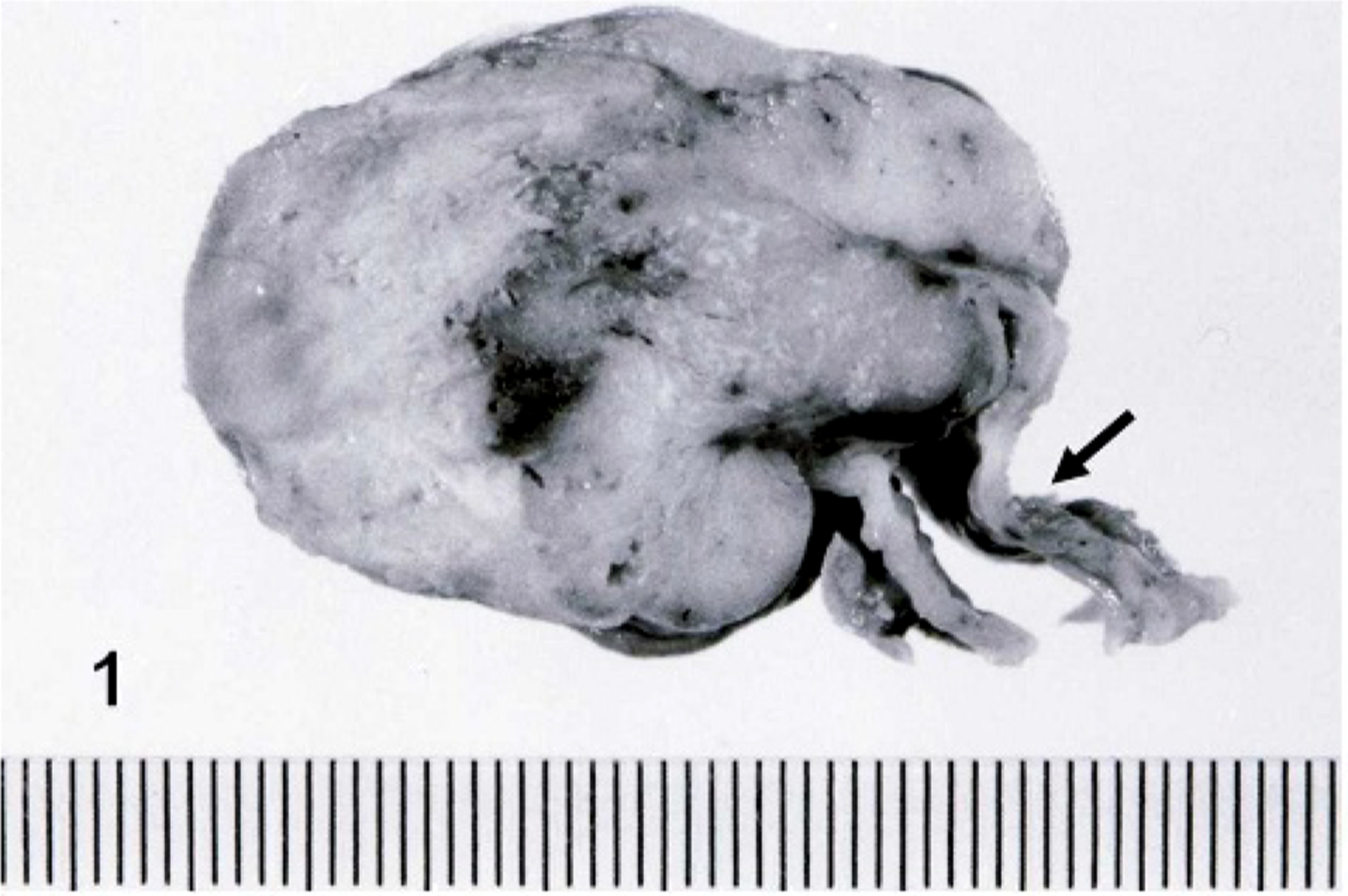
Uterus; cat. The cut surface of intra-abdominal tumor shows a solid and grayish-white mass derived from the uterine wall (arrow).
Histologic low-power evaluation showed that the intra-abdominal mass developed from the endometrium and invaded the myometrium (Fig. 2). The tumor showed a proliferation of both endometrial stromal and smooth muscle cells with focal tumor cell necrosis. The endometrial stromal component had small, ovoid, and spindle-shaped cells with scanty cytoplasm and was arranged in a diffuse pattern accompanied by prominent vasculature, mostly arterioles (Fig. 3a). There were edema and hyaline deposition around blood vessels. The endometrial stromal cells showed moderate nuclear pleomorphism and no mitotic figures. In these areas, well-differentiated endometrial glands and tubuli were present and contained neoplastic epithelial cells and cell debris within the lumen. The endometrial glands were made up a monolayer of eosinophilic cuboidal epithelium (Fig. 3b). Small groups of neoplastic cells were observed within blood and lymphatic vessels. Along with the epithelial components, long spindle cells were present and arranged in interwoven fascicles with abundant fibers stained by Masson trichrome (Fig. 3c). As well as the intra-abdominal mass, the lung tumors were composed of uniform cells with round-to-elongated nuclei, growing densely in a nodular pattern. The focal necrosis was often present in the center of the nodular mass. Additional features included myxoid stroma present with epithelial cells in a glandular or ductal differentiation (Fig. 3d).
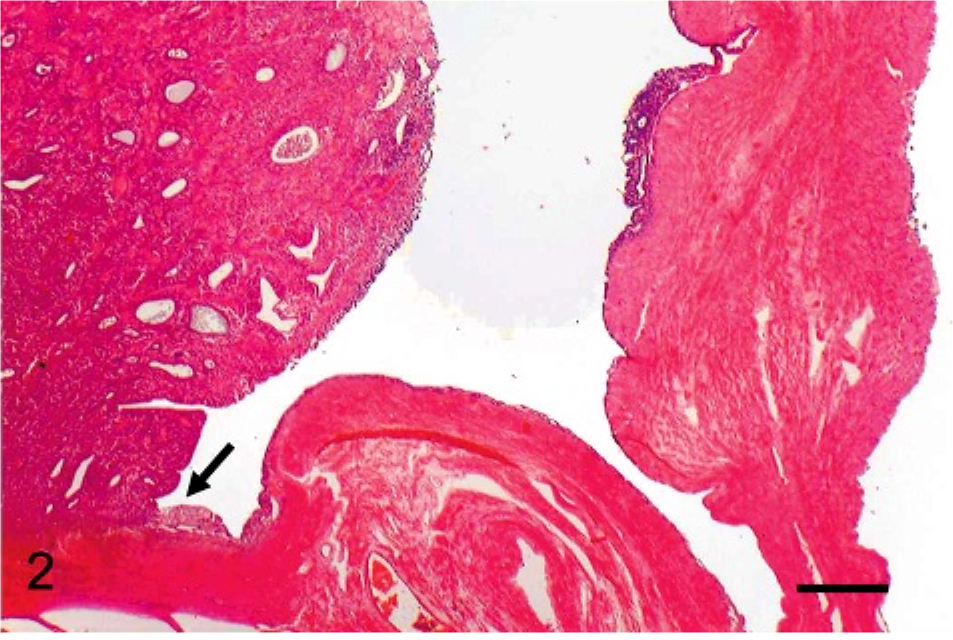
Endometrial stromal sarcoma, uterus; cat. The intra-abdominal mass developed from the endometrium (arrow) and invaded the myometrium. HE. Bar = 500 μm.
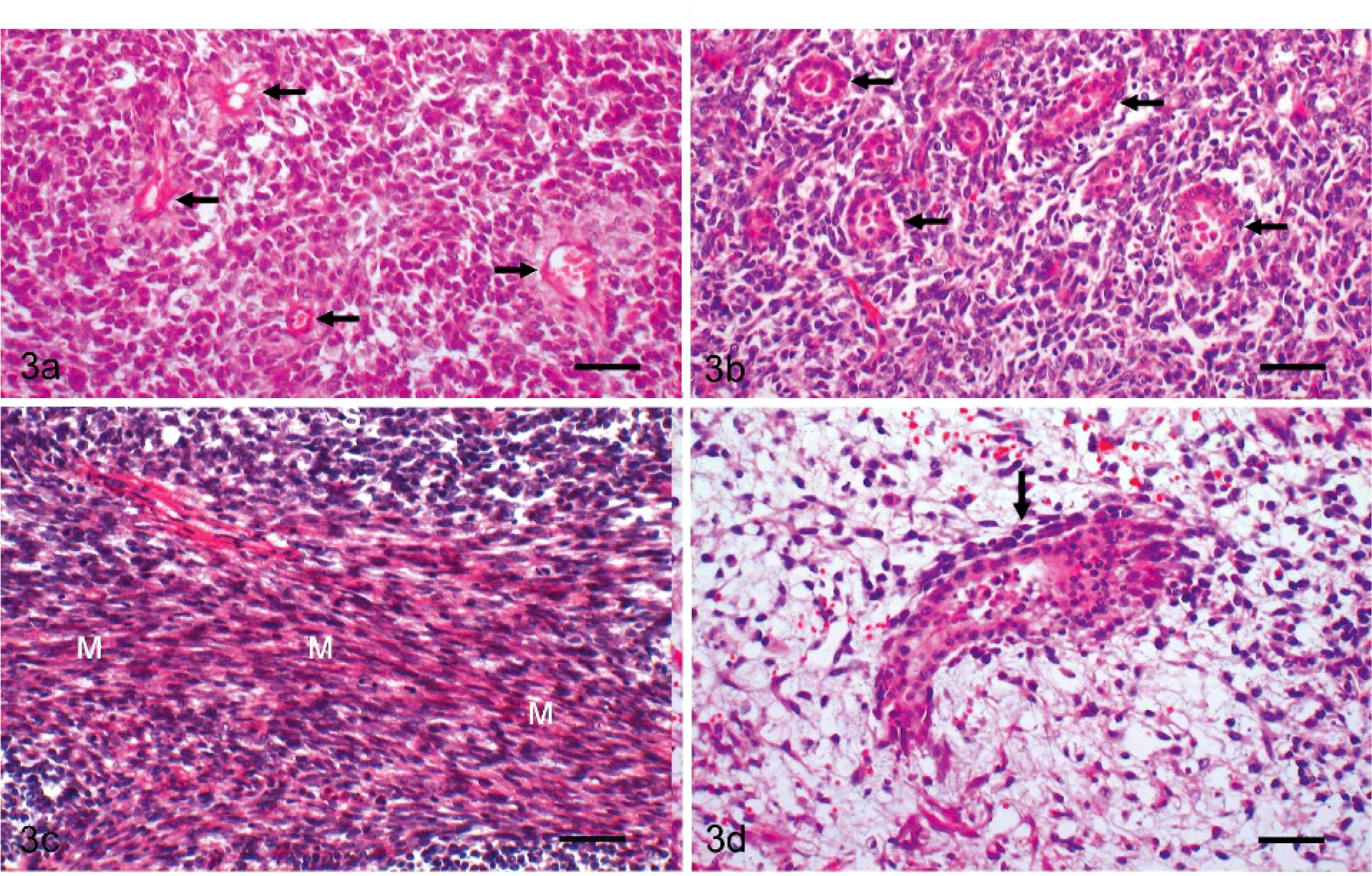
Endometrial stromal sarcoma, uterus; cat. The endometrial stromal cells were
uniform, small, ovoid, and spindle in appearance with scanty cytoplasm,
accompanied by arterioles (arrows). HE. Bar = 100 μm.
Immunohistochemical staining with vimentin antibody was strongly positive on the spindle-shaped cells and stromal cells, whereas the tumor cells of the endometrial glands did not react to the antibody. The spindle-shaped cells and half of the stromal-like cells reacted to caldesmon and desmin antibodies, but glandular cells were negative (Fig. 4). Morphologic differentiation between smooth muscle cells and endometrial stromal cells was ambiguous in some areas. Therefore, the cells positive to desmin and Caldesmon antibodies were regarded as the neoplastic cells differentiated from smooth muscle cells. The neoplastic epithelium showing glandular formation expressed AE1/AE3 cytokeratin (Fig. 5). All types of tumor cells did not react to the SMA and ER antibodies. The same immunostaining profiles were obtained from both the intra-abdominal and lung masses (Table).
Results obtained from immunochemical staining of cell types in feline endometrial stromal sarcoma.

AE1/AE3 = AE1/AE3 cytokeratin; – = negative; + = positive; SMA = alpha-smooth muscle actin; ER = estrogen receptor.
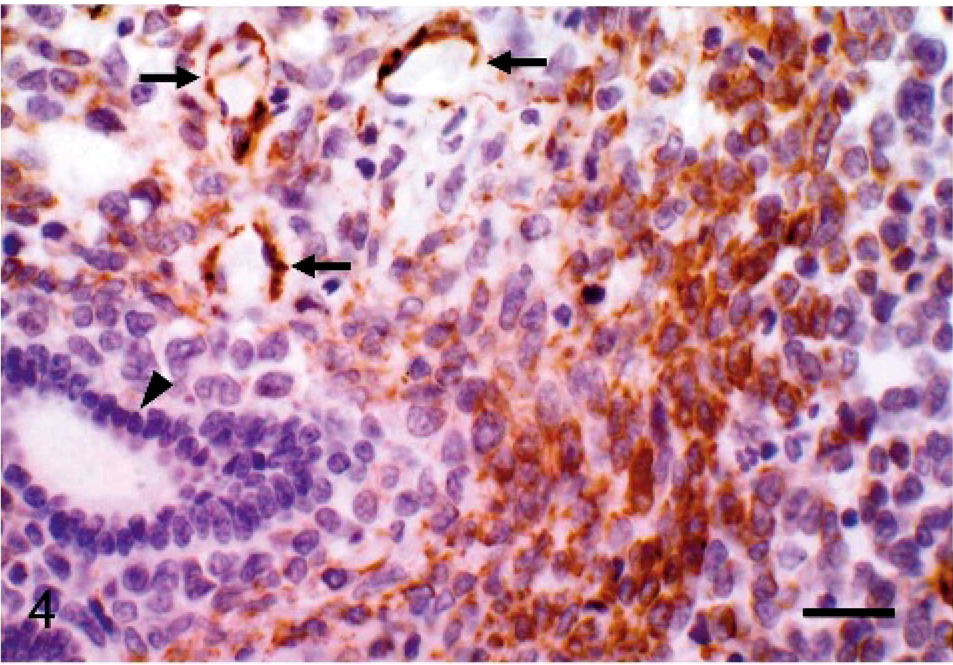
Endometrial stromal sarcoma, uterus; cat. Immunohistochemical staining with caldesmon antibody is strongly positive on the spindle-shaped cells and arteriolar smooth muscle (arrows), whereas the tumor cells of endometrial glands have not reacted (arrowhead). Avidin-biotin peroxidase method. Mayer's hematoxylin counterstain. Bar = 50 μm.
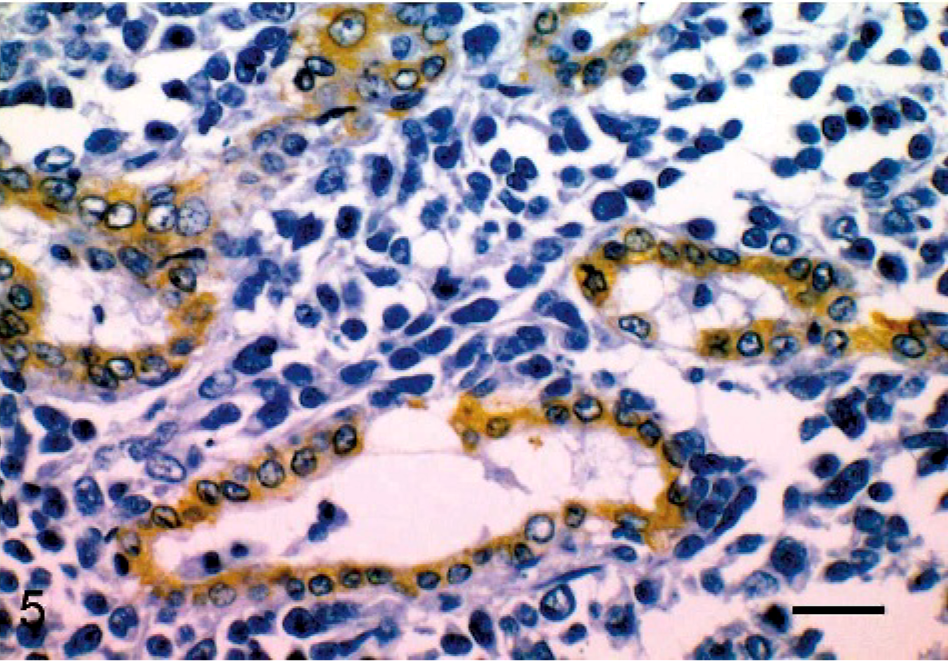
Endometrial stromal sarcoma, lung; cat. The neoplastic cuboidal epithelium shows that glandular formation expressed AE1/AE3 cytokeratin. Avidin-biotin peroxidase method. Mayer's hematoxylin counterstain. Bar = 50 μm.
Endometrial stromal sarcomas may arise not only from endometrial stromal cells but also from endometriotic stromal cells and peritoneal coelomic epithelium and may develop in the ovary, fallopian tube, vagina, rectovaginal septum, and greater omentum.5 In veterinary references, to our knowledge, a tumor has not been reported that originated from endometrial stromal cells. A variety of mesenchymal tumors occur in the uterus of domestic animals, but only the tumors originating from smooth muscle cell and fibroblasts.6 Human endometrial stromal sarcomas have been classified grossly, microscopically, and prognostically. Norris and Taylor8 originally classified endometrial stromal tumors into stromal nodules, endolymphatic stromal myosis (low-grade endometrial stromal sarcomas), and stromal sarcomas (high-grade endometrial stromal sarcomas). Both low-grade and high-grade endometrial stromal tumors show an invasion of the myometrium and hematogenous and lymphogenous metastasis. The differentiation among these tumors is based on the degree of invasion, atypicality of the cells, and the number of mitotic figures.4
Because the single abdominal mass arose from the endometrium, it was definitely from the uterus. A continuity between the mass and uterine wall grossly and microscopically was confirmed. The typical morphology of tumor cells resembling stromal cells and the presence of many arterioles were in agreement with characteristic features in human endometrial stromal sarcoma.1,2,4,8,9,12 In addition, a well-differentiated glandular formation is apparent evidence of the endometrial stromal origin. It has been known that the formation of glands or tubules may be present in low-grade endometrial stromal sarcoma in human patients.3,8 The hematogenous metastasis sometimes occurs in human endometrial stromal sarcomas, developing in 12 of 182 patients.5 Like high-grade endometrial stromal sarcoma, low-grade sarcoma can metastasize in human patients.4 The tumor definitely had malignant features such as local invasion and pulmonary metastases but did not apparently have malignant morphology because it had no mitotic index, as well as moderate nuclear pleomorphism and well-differentiated glands. Norris and Taylor8 have proposed that tubular formations and uniformed cells indicate low-grade endometrial stromal sarcoma.8 Our case had malignant biologic behavior but may be classified as a low-grade tumor.
Immunohistochemical examination successfully differentiated among the components of the normal feline uterus. The glandular epithelium reacted to AE1/AE3 cytokeratin, and the other tissues reacted to the vimentin. The smooth muscle cells in the myometrium reacted to the desmin, SMA, and caldesmon antibodies, as well as the vascular smooth muscle cells. The nuclei of the glandular epithelium, smooth muscle cells, and endometrial stromal cells reacted to ER antibody. A distinction between endometrial stromal tumor and smooth muscle tumor is always challenging in human patients because the immunohistochemical profiles of these tumors can overlap.2,10 Morphologically, typical smooth muscle cells always reacted to vimentin, desmin, and caldesmon antibodies; thus, they do not pose a diagnostic problem. However, with spindle-shaped cells in some areas, it was difficult to differentiate stromal cells from smooth muscle cells on HE stain.
A lack of desmin expression is sufficient to support endometrial stromal cells, but desmin expression is insufficient to exclude endometrial stromal tumors.11 In fact, half of the tumor cells showing desmin and Caldesmon expression in our patients apparently indicated the smooth muscle differentiation, although it is difficult to explain the lack of SMA expression. Interestingly, the neoplastic epithelium showing glandular formation expressed only AE1/AE3 cytokeratin, but not vimentin, desmin and Caldesmon. The CD10 antibody is known to be a positive marker for endometrial stromal tumor in humans.10 We used the CD10 monoclonal antibody (Novocastra NCL-CD10, Vision Systems Inc.) to differentiate from smooth muscle cells. However, the staining was nonspecific on feline tissue as well as progesterone receptors (Novocastra NCL-PGR-AB, Vision Systems Inc.).
McCluggage et al.7 reported human endometrial stromal sarcoma with smooth muscle and glandular differentiation as a unique variant that was similar to our case. It is still unknown whether our case has typical features of feline endometrial stromal tumor histologically and immunohistochemically. It is necessary to investigate more cases to design a classification outline of endometrial stromal tumor, but this report may serve as a first case of feline endometrial stromal sarcoma.
