Abstract
The immune response against mycobacterial infections is dependant upon a complex interaction between T lymphocytes and macrophages in the context of the granuloma. For this study, we performed the analysis of 18 stage I or II, and 13 stage III or IV granulomas found in lymph nodes from 8 experimentally and 2 naturally infected cattle. T-cell subpopulations (CD3+, CD4+, CD8+, WC1+, CD25+) were investigated by immunohistochemistry. In the majority of stage I/II lesions, CD8+ and CD25+ cells were predominantly found in the lymphocytic outer region of the granuloma, suggesting a possible role for activated CD8+ cells in the initial attempt to restrain the granuloma growth. CD4+ T cells appeared equally distributed in the lymphocytic mantle and in the internal areas of the granulomas. WC1+ cells appeared interspersed among the macrophages. We speculated that this could indicate a role for these 2 subsets in the maintenance and the maturation of the granuloma. In stage III/IV lesions, all of the T-cell subsets investigated appeared interspersed among the mononuclear component of the granulomas. In general terms, there was a higher density of CD8+ cells compared with CD4+ cells. However, there was no sense of rimming effect for any of the investigated cell populations.
Tuberculosis caused by Mycobacterium bovis is an important problem in cattle. This zoonotic disease presents a major barrier to animal-related trade and production. The incidence of this disease is on the increase in bovine herds in Great Britain (http://defraweb/animalh/tb/stats/county.htm), and it is considered one of the priorities in animal health. Our institute runs an active program of research aimed at the development of improved diagnostic tests and vaccines for this infection. To achieve these goals, a greater understanding of the pathogenesis of the disease is required, in particular, of the nature of the specific immune responses. Immunity against mycobacterial infections is essentially cell mediated and is dependant upon a complex interaction between T lymphocytes and macrophages. Most information about the relative contribution of the different T-cell subsets was generated from peripheral blood.10,13,17 In contrast, few studies investigated the local cellular immune response in infected lymph nodes.4,19
A dynamic progression in the T-cell subpopulations was demonstrated in experimental models of bovine tuberculosis and provided evidence that all the major T-cell subsets (αβ-TCR and γδ-TCR cells) play a role in immune responses to mycobacteria.17
Two major subsets of γδ-T cells exist in cattle: the WC1–CD3+CD8+/– and the WC1+CD3+CD8–.14 Anatomical studies showed that WC1+ cells represent less than 10% of cells in healthy lymph nodes (LN) but constitute between 10 and 50% of peripheral blood lymphocytes.5 The WC1–CD8+/– is predominantly found in the red pulp of the spleen, with fewer cells in the secondary lymphoid organs.14 Studies in the bovine model infected with M. bovis show that WC1+ migrate from the peripheral circulation to a primary site of infection4 and that bovine WC1+ γδ-T cells respond in vitro to mycobacterial antigens.21 Also, studies on in vivo models in cattle show that WC1+ depletion does not impact on disease pathology but that this population has a role in modulating the immune response, contributing toward the Th1 bias.10
The release of live M. bovis from infected bovine macrophages by antigen- stimulated CD8+ T cells was demonstrated by using in vitro models.12 In addition, it was shown that bovine CD8+ T cells are able to release γ interferon (IFN) in response to live and soluble mycobacterial antigens.13 Those studies demonstrated that CD8+ T cells may contribute to antimycobacterial immunity by a combination of cytolytic (CTL) activity and immunoregulation of the granuloma environment.
Rhodes et al.19 investigated, in vitro, the proliferative responses of T lymphocytes obtained from peripheral blood and from LN from infected cattle, and concluded that both types of cells had little difference in their responses to dominant mycobacterial antigens. They found a predominant αβ-TCR CD4+ response, also with the contribution of the γδ-T cells, but failed to demonstrate αβ-TCR CD8+ responses. Although the former study showed the potential for memory responses of T cells from LN, it did not identify the spatial distribution of T-cell subsets in the context of the granuloma. Another study focused on the immune reaction at the tissue level, showed that γδ-T cells, neutrophils, CD4+, CD8+, and macrophages sequentially traffic to purified protein derivative (PPD) injection sites and that infiltrating lymphocytes had increased CD25 expression.7
CD25 is the high affinity interleukin (IL)-2 receptor and is a molecule that is involved in the activation of lymphocytes and associated with a regulatory function.3 Memory T cells divide and proliferate by the mediation of CD25.1 In cattle, it was demonstrated that distinct T-cell subsets consistently upregulate CD25 in response to mycobacterial antigen stimulation.21
The aim of our study was to examine T-cell infiltration in LN tuberculous granulomas at various stages of development by using immunohistochemistry (IHC) techniques, with the hope to increase our current understanding of the immunopathogenesis of granuloma formation in bovine tuberculosis.
Eight castrated 8-month-old male calves (Friesian cross) were infected by using an intratracheal route,24 with 104 colony forming units of a UK field isolate of M. bovis. In addition, 2 tuberculin field-reactor cattle were also included in the study. The experimentally infected animals were euthanatized 5–12 weeks after infection. All the animals in the study were subjected to detailed postmortem examination.6 A sample of all LN that showed tuberculosis-like lesions, was collected and individually homogenized by using a rotating-blade macerator system, decontaminated with (N-acetyl-L-cysteine)-NaOH, inoculated onto Lowenstein-Jensen slopes with pyruvate and/or glycerol and incubated at 37°C between 8 and 12 weeks. Identification of isolates was done by standard biochemical and molecular methods.
LN tissues were fixed in zinc salts fixative (ZSF), as described previously,8 and transferred to 70% alcohol for 3 hours 15 minutes for decontamination before tissue processing to paraffin wax. Six serial sections (4 μm) were cut, 1 section was stained with hematoxylin and eosin (HE) for histopathologic examination, and the rest were used for the IHC runs.
For IHC, tissue sections were processed as described before,25 with the only modification being that antigen retrieval was not necessary. Details for the primary antibodies are given in Table 1. An analysis of the sections was performed by using a semiquantitative scoring system based on examination of at least 3 400× fields of 0.14 mm2 (–, no immunostaining; +, 1–10 positive cells/field; ++, 10–20 positive cells/field; +++, 20–50 positive cells/field; ++++, >50 positive cells/field). Because of the specific interest in T cells, the analysis focused in the lymphocyte-rich areas of the granulomas. In the stage I/II lesions, the focus was in the lymphocyte rim population immediately adjacent to the epithelioid macrophages; for the stage III/IV lesions, the focus was on the lymphocytic infiltration situated internally to the fibrotic capsule but externally from the central necrosis/calcification area. Three separate randomly selected microscopic fields, representative of the stage of granuloma, were used for the analysis. When a clearly stronger signal was perceived in the mantle region compared with the epithelioid macrophage-rich internal area of the granuloma, the result was recorded as rim signaling (Table 2).
Antisera used for immunohistochemistry.
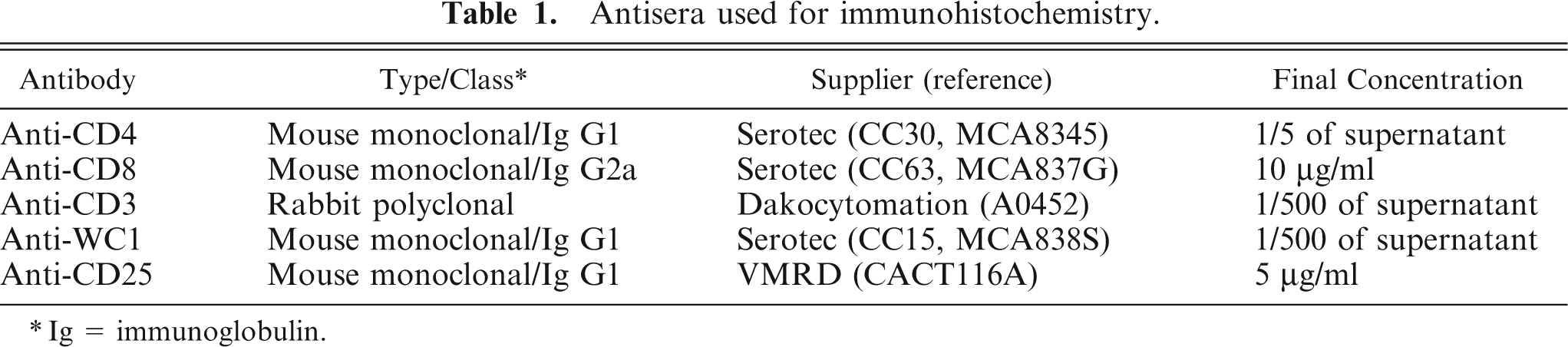

Ig = immunoglobulin.
Antisera used for immunohistochemistry.
All the experimentally infected animals examined showed microscopic lesions typical of tuberculosis in at least one of the LNs of the thorax (mediastinal and/or tracheobronchial). For the field reactor cases, the lesions were found in mesenteric and hepatic LNs of 1 case and in retropharyngeal LNs of the second case. A detailed examination was carried out to characterize the level of lesion advancement into 4 stages, I, II, III, or IV, according to the scheme developed in our laboratory.25 The results of the histopathologic examination of 23 tissue sections are shown in Table 3. On several occasions, LN sections contained granulomas of different stages. For the purpose of the IHC in this study, we identified and studied, in detail, 18 stage I/II and 13 stage III/IV granulomas from the examined tissue sections. All samples examined in this study were bacteriologically confirmed as M. bovis positive.
Detailed histopathologic analysis of lymph-node sections.
Cd Med = caudal mediastinal, Cr Med = cranial mediastinal, Cr TB = cranial tracheobronchial, L B = left bronchial, Mesen = mesenteric, RPh = retropharyngeal, Hep = hepatic.
The results of the microscopic analysis of the immunostained LN sections are summarized in Table 3. The pan–T-cell marker anti-CD3 detected a strong presence of T-lymphocytes in all lesions investigated. In addition, all T-cell subsets (CD4+, CD8+, and CD25+) were found in stage I/II and stage III/IV lesions, with the exception of 3 stage III/IV lesions from a single animal, where CD4+ cells could not be identified. It is theoretically possible that some of the CD8+ cells in our study may be γδ-T cells, however, previous studies showed that this subset only represents a minority population in secondary lymphoid organs.5,14 ZSF enables improved antigen survival in comparison with buffered formalin-fixed tissues.8 In our study, anti-CD4, anti-CD8, and anti-CD25 antibodies did not work on formalin-fixed tissue, however, they provided strong specific staining in the ZSF-fixed tissues.
Stage I/II granulomas are characterized by a lack of fibrotic capsule and no, or minimal, necrosis. As presented in Fig. 1, IHC showed the diffuse distribution of CD4+ and WC1+ T cells in both internal and external areas of the granuloma. In contrast, we found abundant CD8+ and CD25+ T cells prominent in the outer boundary zone, with a smaller number of these cells in the macrophage-rich internal area. In 18 of 18 and in 10 of 18 stage I/II lesions, a clear strong rim of CD8+ and CD25+ cells, respectively, was observed around the macrophage areas.
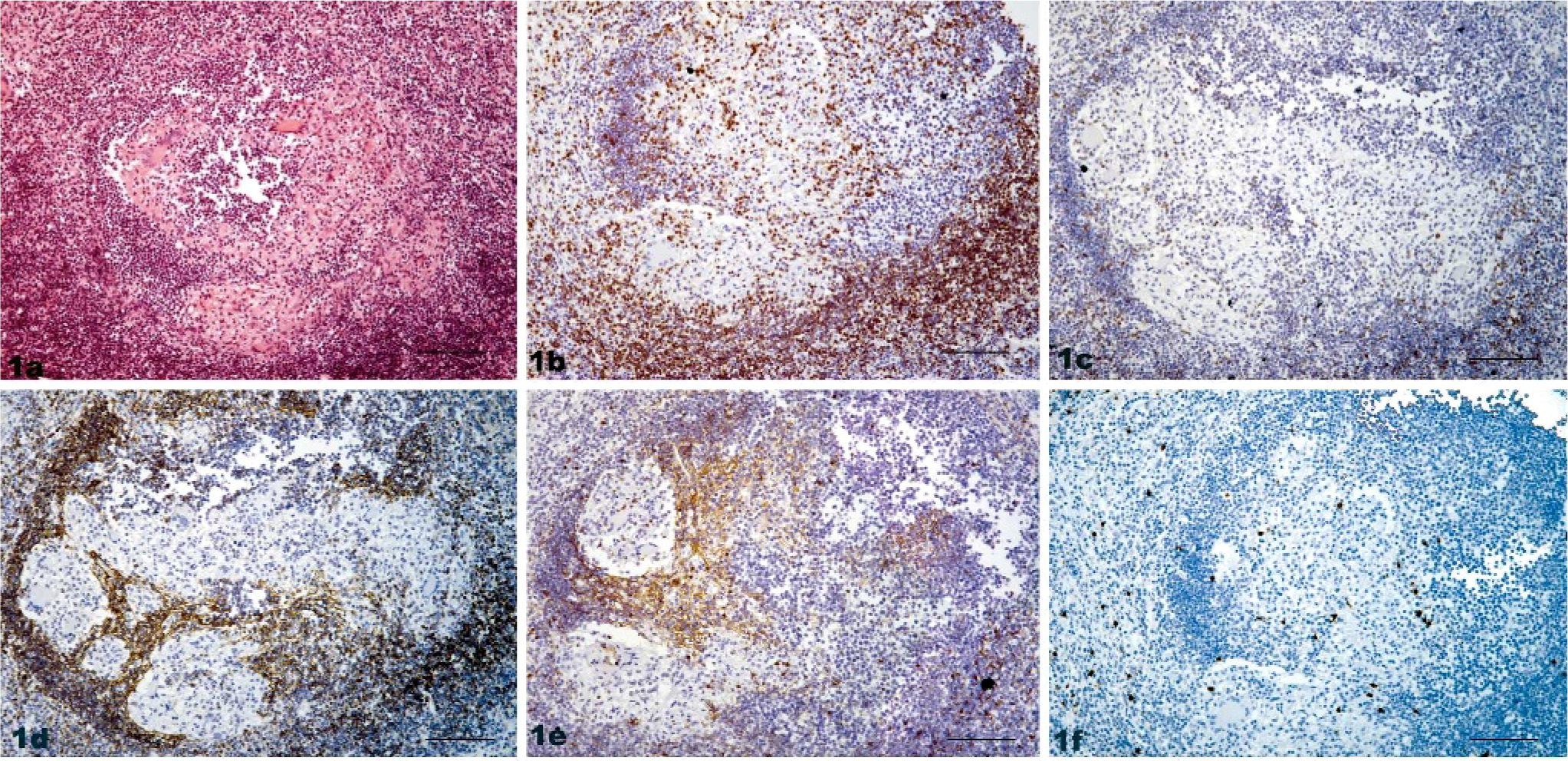
Lymph node, bovine tuberculosis; animal No. 8199. Stage I granuloma.
Stage III/IV granulomas are characterized by the presence of a well-formed fibrotic capsule, and they present a central core of caseous necrosis, often with mineralization. As presented in Fig. 2, all T-cell subsets investigated appeared interspersed among the mononuclear component of the granuloma. In general terms, there was a higher density of CD8+ cells compared with CD4+ cells. However, there was no sense of rimming effect for any of the investigated cell populations, as noted in stage I/II granulomas. The level of CD25+ staining was lower in comparison with the stage I/II lesions.
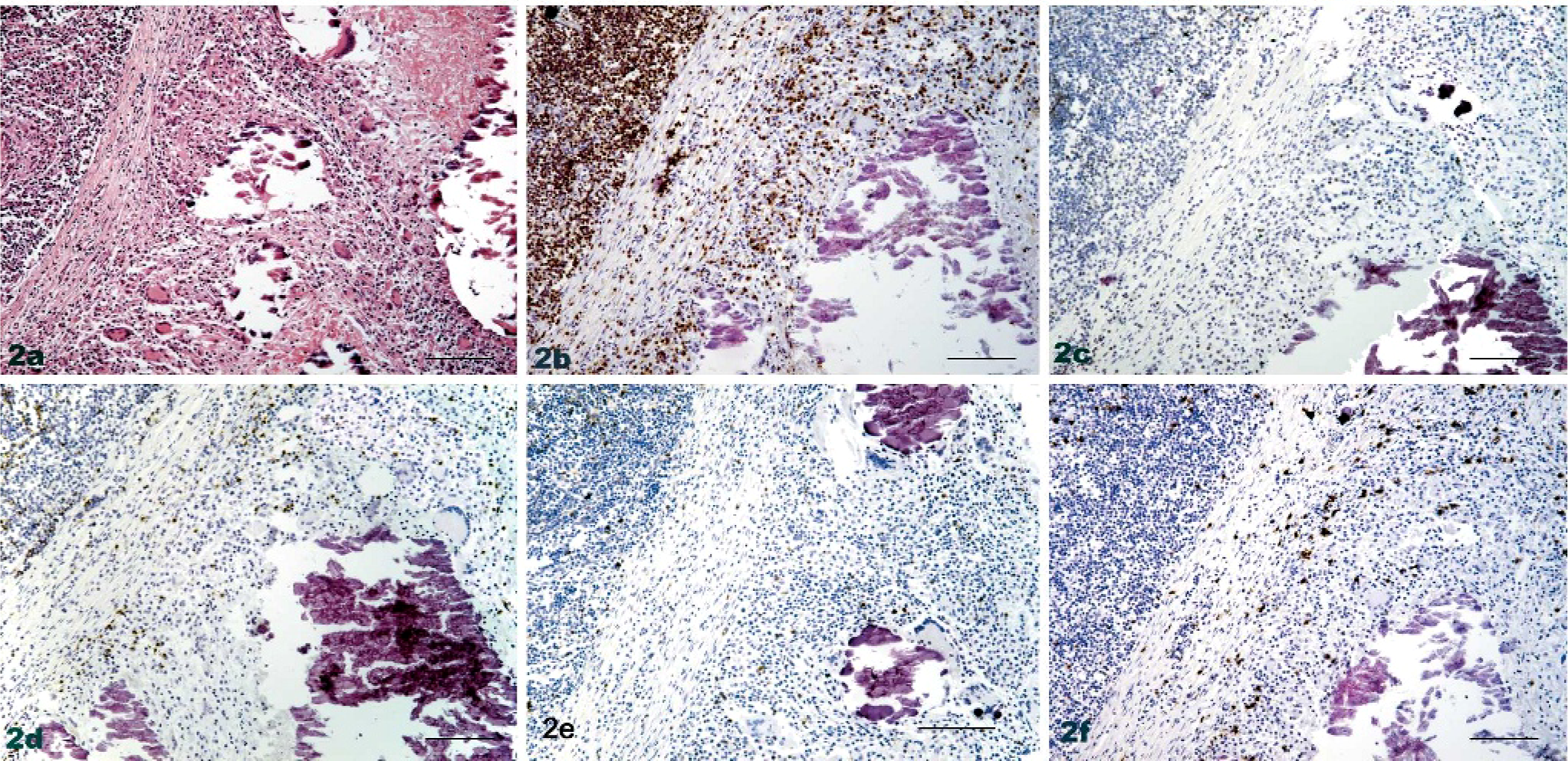
Lymph node, bovine tuberculosis; animal No. 8193. Stage IV granuloma.
Immunohistochemical labeling identified a greater density of CD8+ cells compared
to CD4+ cells generally, and less distinct peripheral margin distribution, as
noted in stage I/II granulomas; fewer CD25+ cells were identified in comparison
with stage I/II lesions.
In total, for 22 of 31 lesions (70%) investigated, the intensity of CD8 staining was higher than that of CD4 staining (ratio CD4:CD8, <1). These represented 15 of 18 stage I/II lesions (83%) and 8 of 13 stage III/IV lesions (61%).
Our observation that, for the majority of stage I/II lesions, CD8+ and CD25+ T cells were predominantly located in the lymphocytic peripheral margin of the granuloma could suggest a role for activated CD8+ T cells in the initial attempt to restrain the growth of the granuloma. This speculation is based on the fact that CTL was traditionally considered the main function for this subset. It was suggested that specific lysis of chronically infected macrophages would enable uptake of extracellular bacilli by infiltrating activated monocytes, with better bactericidal potential helping to resolve the inflammation.9
In contrast, CD4+ T cells appeared equally distributed in the lymphocytic mantle and in the internal areas of the stage I/II granulomas, and WC1+ cells appeared interspersed among the macrophages. We speculate that this could indicate a role for these 2 subsets in the maintenance and the maturation of the granuloma. This is based on previous reports that showed that the main function for these subsets is to secrete cytokines, such as IL-2 and γ-IFN that contribute to stimulation of Th1 and inhibition of Th2 responses, and activation of natural killer cells and macrophages, all of which contribute to the development of the granuloma.16,21 Our findings are in agreement with previous studies of human lymphadenitis that show that CD4+ T cells are distributed diffusely throughout the granulomatous lesions, whereas CD8+ T cells are found mainly in the outer part of the granulomas.23 The role of the CD4+ subset in formation, maturation, and maintenance of the granulomatous inflammation was previously proposed, based on the fact that most T cells in the internal areas of granulomas are CD4+ memory T cells.20 This is regarded as an indication of the existence of some persistent etiologic agent.
Previous studies of human tuberculosis and sarcoidosis 11,22 show that, despite an increase of γδ-T cells in peripheral blood in human patients with granulomatous disease, practically all the T lymphocytes that promote granuloma formation had an αβ type T-cell receptor (TCR) and not a γδ-type TCR. Our results are in contrast with the former studies, as we saw in the bovine, a substantial presence of WC1+ T cells in both stage I/II and III/IV tuberculous granulomas. In agreement with our findings, Cassidy et al.4 reported that there is an association of WC1+ T cells with the development of early stage lesions in bovines. This discrepancy could be explained by the different role of this population of T cells in the bovine immune response. Although WC1+ depletion experiments in cattle show no effect on severity of pathology, it is believed that this subset plays a role in modulating the immune response.2,10 Further studies with dual labeling will be necessary to clarify what cytokines are released by these cells in situ at the lesion level.
In contrast with results reported by Kita et al.11 in human sarcoids, we saw that lymphocytes in the peripheral area of the granuloma often present a high degree of CD25 expression that suggested an aggregation of highly activated cells most probably of the CD8+ lineage. A lower level of CD25 expression was found in stage III/IV lesions compared with stage I/II lesions. This could potentially indicate that the stage I/II lesion is a more changing and active environment, whereas, the stage III/IV lesion may represent a more static environment.
Rhodes et al. 19 showed a predominant CD4+ T-cell proliferation in vitro to mycobacterial antigens, with cells originating from LN tissue and suggested that CD4+ cells are the major T-cell subset involved in the acquired immune response. This contrasts with the elevated ratio CD8:CD4 found in our in situ studies, indicating that CD8+ cells may have a more important role than previously thought. Randhawa18 reported, by IHC, on tuberculous granulomas in human cervical LN, that the proportion of CD4+ cells is much lower than that seen in the normal circulation, with a ratio CD4:CD8 of 1.1 in discrete lesions; this is in agreement with a concentration of CD8+ at the sites of inflammation. Muller and Takeshita15 report that epithelioid macrophage granulomas in human tuberculosis showed higher numbers of CD8+ T cells in their mantle zones and that epithelioid macrophage formation was strongly correlated with CD25+. Although, in our study, we observed CD25+ cells in the interior of the granuloma, in a good proportion of stage I/II lesions this population appeared predominately in the mantle area.
In summary, our results suggest that different T-cell subsets participate in granuloma formation in bovine tuberculosis and display different spatial distribution, depending on the lesion stage. We observed that outer boundary T cells surrounding the internal macrophage-rich area are mainly composed of CD8+ and CD25+ cells, and speculated that these may fulfill the function of restraining granuloma advancement. This study constitutes the first observation of in situ T-cell subset distribution within tuberculous bovine LN granulomas.
Footnotes
Acknowledgement
This work was funded by the United Kingdom Department for Environment, Food and Rural Affairs (Defra) project SE3225.
© British Crown Copyright, 2007
