Abstract
We have developed a model to explore the early immune response against Mycobacterium avium subspecies paratuberculosis (Map) infection in the bovine calf using subcutaneously placed liquid gel matrix biopolymer (matrigel) containing live Map. Matrigel rapidly polymerizes in vivo, retains recruited cellular infiltrates and soluble immune mediators, and can be rapidly removed 48 hours later and depolymerized for analysis. In this study, we examined early host immune events at matrigel/Map sites; recruited cells were evaluated by histopathology and flow cytometry, and cytokines were measured by flow cytometry, enzyme-linked immunosorbent assay, and Luminex bead immunoassay. Our results demonstrate earlier recruitment of gamma-delta (γδ) T cells to matrigel/Map challenge sites compared to CD4+ T cells. We also show that significantly more γδ T cells were recruited to matrigel/Map sites postinfection day 7 compared to postinfection day 30 and that these cells produced significant amounts of the cytokine interferon gamma. We also provide evidence that peripheral blood-derived γδ T-cell subsets in cattle differentially generate interferon gamma, suggesting distinct roles for these cells. These data provide unique insight into initial antimycobacterial host cellular immune responses following Map infection in calves.
The host immunologic response during Mycobacterium avium subspecies paratuberculosis (Map) infection in cattle is not completely understood. It is thought that perinatal calves are primarily infected via the fecal–oral route, and an initial T-helper type 1 immune response is mediated by interferon gamma (IFNγ) secreting CD4+ T cells. A lengthy subclinical period ranging from months to years follows, during which time there is generally absence of clinical signs, histologic or gross lesions, bacterial shedding, and definitive cellular or humoral evidence of an individual’s infection status. 20,21 Clinical Johne's disease is characterized by wasting, diarrhea, and eventual death of the host, and this is associated with a loss of CD4+ T-cell-mediated immunity, development of Map-specific serum antibodies, and progressive granulomatous enteritis. 30
A significant challenge to studying this disease in cattle is modeling host immunologic responses across multiple disease stages. Immune responses in lymph nodes and intestine 16,32 or peripheral blood 4 during subclinical or clinical Map infection of cattle have been described, yet studying immune function at infection sites remains a difficult research challenge. This knowledge gap has driven recent development of novel modeling systems in cattle with the goal of clarifying factors that may shape development and progression of disease. 1,12,22 The importance of initial host–pathogen interactions are well recognized, 28 and the goal of this study was to evaluate early gamma-delta (γδ) T-cell responses at subcutaneous Map challenge sites in calves. Natural Map infection is an intestinal disease, and clearly, a subcutaneous model is not without limitations; however, we believe that the model is useful to study antimycobacterial host responses at early infection sites.
We are interested in understanding how the initial cellular immune response to Map infection influences development of subsequent adaptive immunity and disease outcome. Specifically, we are interested in the role of γδ T cells during this process. Young calves have large numbers of γδ T cells in peripheral blood; however, their specific functions are poorly defined. These cells have been shown to be important effectors or regulators of immune responses during mucosal infections in mice, 3,26 human, 5,18 and cattle. 13
Subsets of bovine γδ T cells have been described and are largely defined by their expression of the unique workshop cluster 1 molecule (WC1) 17,27 as well as CD2 and CD8 expression. 8 Unique roles for WC1+ and WC1– subsets have been reported, 10,25 but their function has not been completely characterized. Using a subcutaneous infection model in young calves to investigate this, we have shown differential recruitment patterns of WC1+ and WC1– γδ T-cell subsets into experimental Map infection sites leading us to hypothesize that these subsets have distinct functional roles at initial infection sites. 23
The goal of the current study was to collect and analyze cells recruited to Map challenge sites, adding to our image-based studies within developing granulomas over time. We adapted a technique developed by Russell 28 by using a matrigel biopolymer vehicle in a subcutaneous challenge model. An advantage of the matrigel system is that recruited leukocytes can be recovered for ex vivo analysis following matrigel depolymerization. Our data support the hypothesis that bovine γδ T cells are recruited early into Map infection sites where they may regulate local and systemic immunologic responses.
Materials and Methods
Animals and Experimental Design
A total of twelve 5- to 6-week old castrated male Holstein calves were acquired from the Iowa State University dairy research farm (Ames, Iowa), a herd certified free of Map infection. Calves were individually housed in a biosafety level 2 animal care facility during the study and were maintained there at a time for logistical purposes. The Committee on Animal Care and Use at Iowa State University approved all animal protocols prior to the study. Animals were divided randomly into the following groups: uninfected/naive (n = 4), subcutaneous Map infected (n = 4), and Map vaccinated (n = 4). For assays of systemic immunity not dependent on ex vivo cell cultures, samples from all 12 animals were utilized. Unfortunately, during the first group of 3 animals, we experienced unexpected death of cultured cells harvested from matrigel sites due to technical challenges; therefore, for individual cell culture assays, samples from 9 calves yielded useful data and are shown. Animals were administered sham saline (1 ml of sterile 0.9% isotonic saline), live Map (1 × 109 cfu in 1 ml of sterile 0.9% isotonic saline), or Map vaccine (0.5 ml of Mycopar, Fort Dodge Animal Health Company, Fort Dodge, Iowa) in the subcutaneous space of the left cervical region on postinfection day (PID) 0. Calves were subsequently challenged at PID 7, PID 15, and PID 30 with 500 μL of biopolymer vehicle containing 5 × 105 cfu live Map (matrigel/Map) or equal volume of sterile saline injected into the subcutaneous space of the right neck or paralumbar fossa. Forty-eight hours after the final matrigel injection (PID 30), all calves were euthanized with intravascular pentobarbital sodium (Beuthanasia-D, Intervet/Schering Plough Animal Health Company).
Vaccine
M. avium subspecies paratuberculosis bacterin (Mycopar, Fort Dodge Animal Health Company) was used in this experiment. Mycopar is a whole-cell bacterin containing inactivated Mycobacterium paratuberculosis in oil.
Generation of Map Antigen (Whole Cell Sonicate)
Late-growth-phase live Map strain K10 bacteria were collected from culture stock in our laboratory. Bacteria were sonicated briefly with 3 cycles of 10-minute bursts (18 watts) separated by 10-minute chilling periods. Protein concentration was determined using the Pierce BCA protein assay (Pierce Biotechnology, Rockford, Illinois). Whole cell sonicate (WCS) was streaked onto blood agar to check for contamination prior to use.
Bacterial Inoculum Preparation
The Map strain K10 was obtained from the National Animal Disease Center (Ames, Iowa) and is maintained in Middlebrook 7H9 broth supplemented with mycobactin J in our laboratory. Bacteria were prepared for inoculation as previously described. 22
Matrigel
BD Matrigel Basement Membrane Matrix (Matrigel, BD Biosciences, Bedford, Massachusetts) is a commercially available purified solubilized basement membrane preparation extracted from the Engelbreth-Holm-Swarm mouse sarcoma. Matrigel was stored at –20°C and thawed at 4°C by placing on ice 24 hours prior to use. Immediately prior to challenge, live Map (5 × 105 cfu) or an equal volume of sterile saline was added to 500-μL aliquots of thawed matrigel.
Matrigel Cell Collection and Processing for Flow Cytometry, Culture, or Histology
Forty-eight hours following each matrigel/Map challenge, calves were heavily sedated (0.1 mg/kg of xylazine intramuscularly, followed by 2.0 mg/kg of ketamine intravenously) and placed in lateral recumbency for removal of subcutaneous matrigel plugs. A cutaneous incision over each challenge site exposed matrigel material, which was collected and placed into 5-ml sterile isotonic saline on ice for depolymerization. Incisions were sutured and the calves recovered uneventfully. Cell pellets were collected by centrifugation, cell number was determined using a Beckman Coulter counter (Beckman Coulter, Inc., Miami, Florida) and cells were resuspended in sterile PBS (flow cytometry) or complete RPMI (cell culture) at a final concentration of 1 × 106 cells per 100 μL of media. For histopathology, matrigel was placed into 10% neutral-buffered formalin and stored at room temperature prior to paraffin-embedding, sectioning (4 μm) and staining with hematoxylin and eosin for evaluation.
Peripheral Blood Mononuclear Cell Collection for Flow Cytometry or Culture
Peripheral blood mononuclear cells (PBMCs) were collected by jugular venipuncture at PID 0 and PID 30, and the buffy coat was separated from the whole blood. Individual γδ T-cell subsets were sorted from PBMCs in 2 consecutive steps using an autoMACS cell separation column per the manufacturer’s instructions (MiltenyiBiotec Inc., Auburn, California). Sorting events were based on expression of the surface molecules workshop cluster 1 (WC1) and the γδ T-cell receptor (TCR) to achieve 2 γδ T-cell populations with purity > 85% (verified by flow cytometry, data not shown): γδTCR+/WC1– and γδTCR+/WC1+.
Monoclonal Antibodies and Antigens
Mouse anti-bovine monoclonal primary antibodies and fluorescence-tagged secondary antibodies were used to label cell surface markers and characterize T-cell phenotype by multicolor flow cytometry (Table 1). For intracellular cytokine staining, cells were fixed in 2% paraformaldehyde for 20 minutes after surface marker staining. Fixed cells were then permeabilized with permawash buffer (0.1% saponin, 0.1% sodium azide in 1× phosphate buffered saline) prior to antibody staining. Stained cells were stored in 2% paraformaldehyde until analysis by flow cytometry.
Antibodies for Flow Cytometry.

aVMRD, Inc., Pullman, Washington.
bSerotec, Inc., Raleigh, North Carolina.
cInvitrogen Molecular Probes, Inc., Carlsbad, California.
Flow Cytometric Data Collection and Analysis
Flow cytometric data were collected using a FACSCanto flow cytometer (Becton Dickinson Biosciences, San Jose, California), and FlowJo cell analysis software (Tree Star Inc, San Carlos, California) was used to analyze data.
Cell Culture
After harvest from matrigel sites, total cells were suspended in RPMI 1640 complete medium (1 × 106 cells per well) in 96-well plates. Map antigen (WCS, 5 μg) was added to appropriate wells prior to culture for 6 days at 37°C. In addition for peripheral blood-derived γδ T-cell subset cultures, recombinant human interleukin-2 (rhIL2, Peprotech Inc., Rocky Hill, New Jersey) was added at a concentration of 1 ng/ml to appropriate wells prior to culture for 6 days at 37°C.
Luminex Immunoassay
Gel or culture supernatants were incubated with agitation for 2 hours at room temperature, then overnight at 2°C to 8°C with biotinylated mouse anti-bovine IFNγ-coupled Luminex beads. After addition of detection antibodies (streptavidin-PE) and incubation at room temperature for 30 minutes, mean fluorescence intensities of individual samples (in duplicate) were compared to a concurrently generated standard curve to determine cytokine concentrations.
Whole Blood IFNγ Assay
IFNγ enzyme-linked immunosorbent assay (ELISA) was performed according to the manufacturer’s instruction (Mycobacterium bovis gamma-interferon test kit for cattle, Bovigam, Prionics USA, Inc., LaVista, Nebraska) with one modification. In place of the kit-supplied M. bovis antigen, we used Map K10 strain WCS antigen (10 μg/ml), which was generated in our laboratory.
Delayed-Type Hypersensitivity Reaction
Delayed-type hypersensitivity was assessed by measuring skin pinch thickness 72 ± 6 hours following intradermal injection (in the cervical region) of 0.1 ml of purified protein derivative (Johnin PPD = 4.6 mg/ml, National Veterinary Services Laboratories, Ames, Iowa).
Statistical Analysis
Statistical analysis was performed using JMP 8.0.2 (SAS Institute, Cary, North Carolina). Data are presented as the mean values ± standard error of the mean. Student t test and one-way analysis of variance were used for the statistical analysis, unless otherwise specifically stated. Group mean differences were considered significant if the P value was < .05 and highly significant if the P value was < .01.
Results
Systemic Immune Response
To measure systemic immune response, we measured Map-specific IFNγ production from PBMCs in all calves at PIDs 0 and 30 by ELISA. Minimal IFNγ was generated at PID 0 in all groups of calves (Fig. 1a, clear bars). Compared to all other calves, PBMCs from Map-vaccinated calves generated significant IFNγ only at PID 30 (Fig. 1a, P = .0103). We also measured systemic immune memory at PID 30 by delayed-type hypersensitivity reaction, a test commonly utilized for evaluating cell-mediated responses to mycobacteria in humans and animals. Skin thickness at the site of intradermal Map PPD injection at PID 30 was statistically different between each treatment group (Fig. 1b, P < .01 for each group).
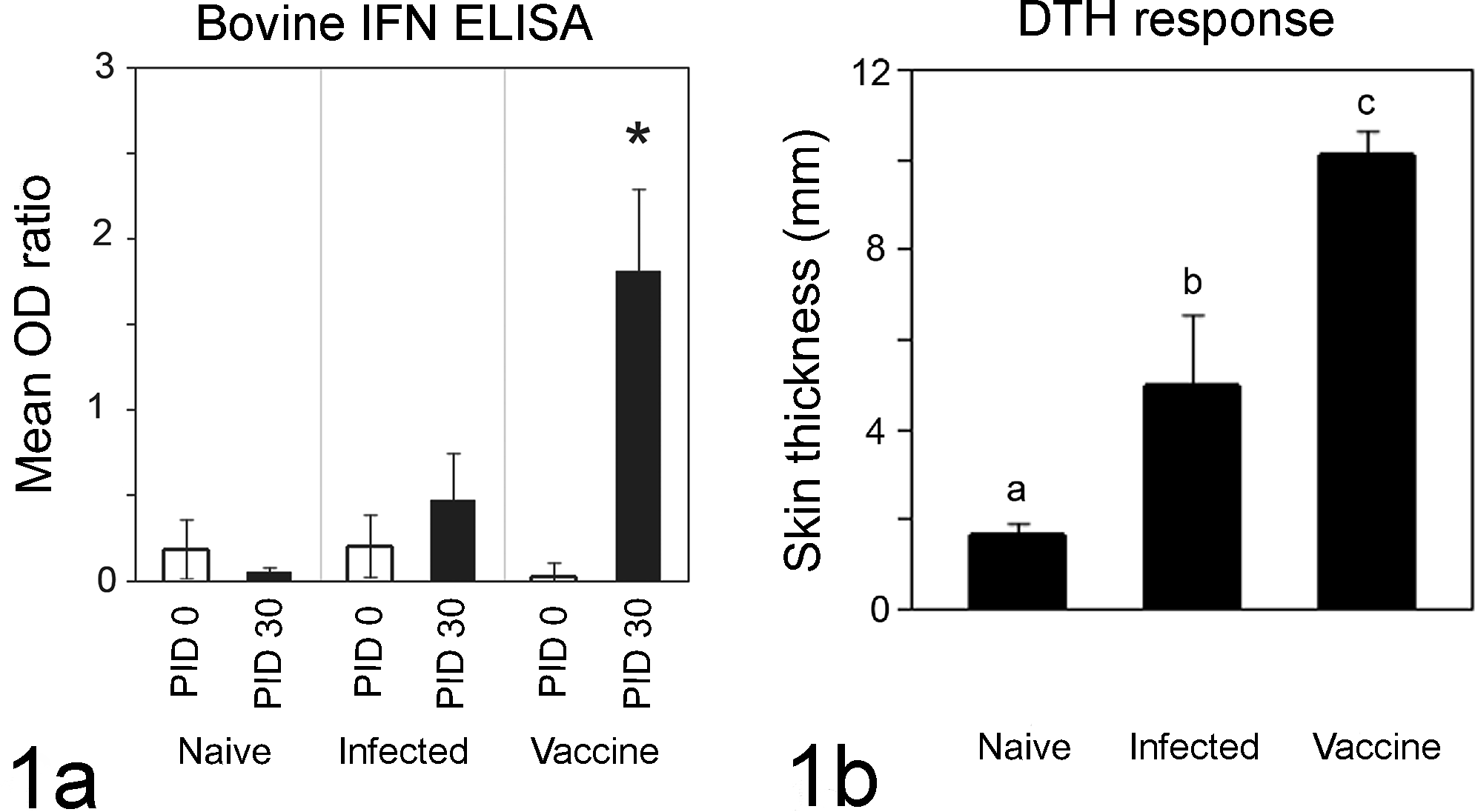
Systemic immune responses. Bovine interferon gamma (IFN)γ ELISA (Bovigam) data show that significantly more Map-specific IFNγ was generated by peripheral blood mononuclear cells from Map-vaccinated calves at PID 30 compared to PID 0 and to all other calves at PID 30 (a, P = .0103). Mean optical density ratio (OD ratio is calculated as sample OD/kit-supplied positive control OD) from 4 calves per group at each time point (n = 12 total) is shown. Delayed type hypersensitivity responses are shown in panel b, and skin thickness at purified protein derivative (PPD) injection sites was significantly different between groups (naïve vs Map infected vs Map vaccinated) at PID 30 (b, P < .01 between all groups). Mean skin thickness from 4 calves per group at PID 30 (n = 12 total) is shown.
Cells Recruited to Matrigel Map Challenge Sites
During surgical removal of the matrigel sites, we observed differences in gross appearance of matrigel sites (pale pink and clear) versus matrigel/Map sites (opaque, white, and firm). Histopathologic examination of these sites revealed that matrigel sites contained homogeneous eosinophilic matrigel material with rare inflammatory cells (Fig. 2a), while matrigel/Map sites were characterized by marked infiltration of inflammatory cells and fibrin (Fig. 2b).
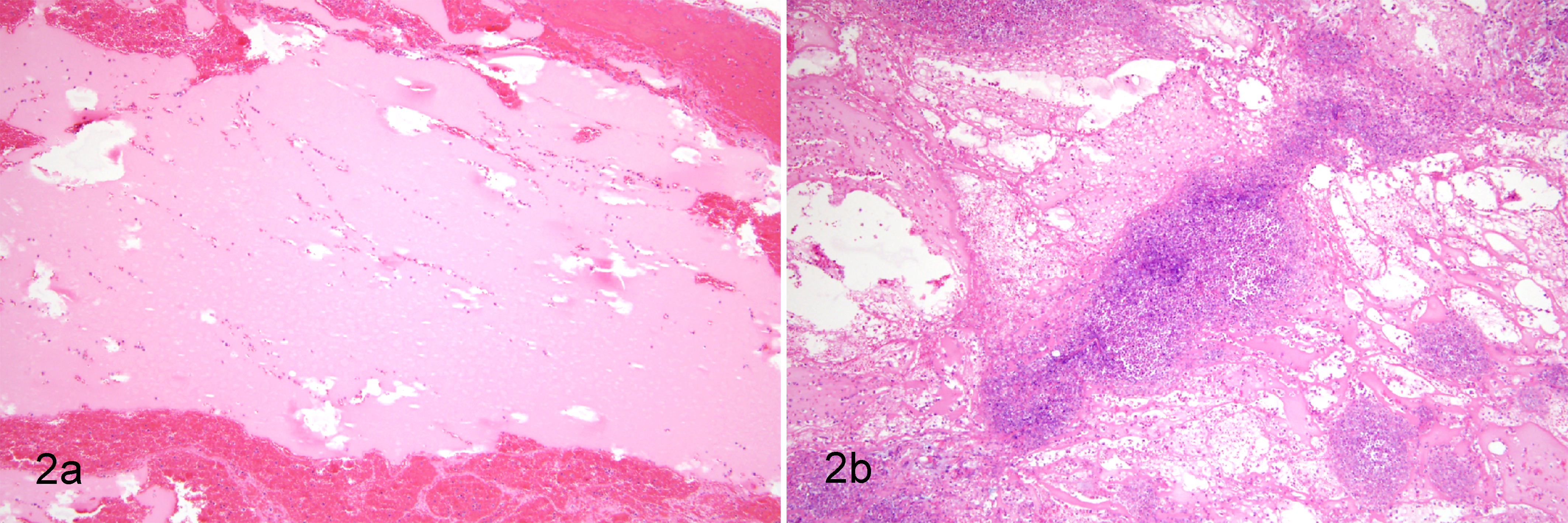
Histology of matrigel sites. Histologic appearance of matrigel alone (a, HE stain) and matrigel with live Map (b, HE stain) are shown. Matrigel appears pale eosinophilic and homogenous. Few leukocytes were recruited to matrigel only sites (a), while many leukocytes were recruited to matrigel with Map sites (b).
Phenotype of Lymphocytes Recruited to Matrigel/Map Sites
To understand patterns of T-lymphocyte recruitment into early Map challenge sites in this model, we used flow cytometry to examine phenotype of T cells collected from matrigel/Map sites. In all calves, significantly more γδ T cells were recovered from matrigel/Map sites at PID 7 compared to PID 15 or PID 30 (Fig. 3, black bars, P < .0001). In contrast significantly more CD4+ T cells were recovered from matrigel/Map sites at PID 30 compared to PID 15 (Fig. 3, grey bars, P = .0073). Matrigel/Map sites at PID 30 contained significantly more CD4 T cells than γδ T cells (Fig. 3, P < .05).
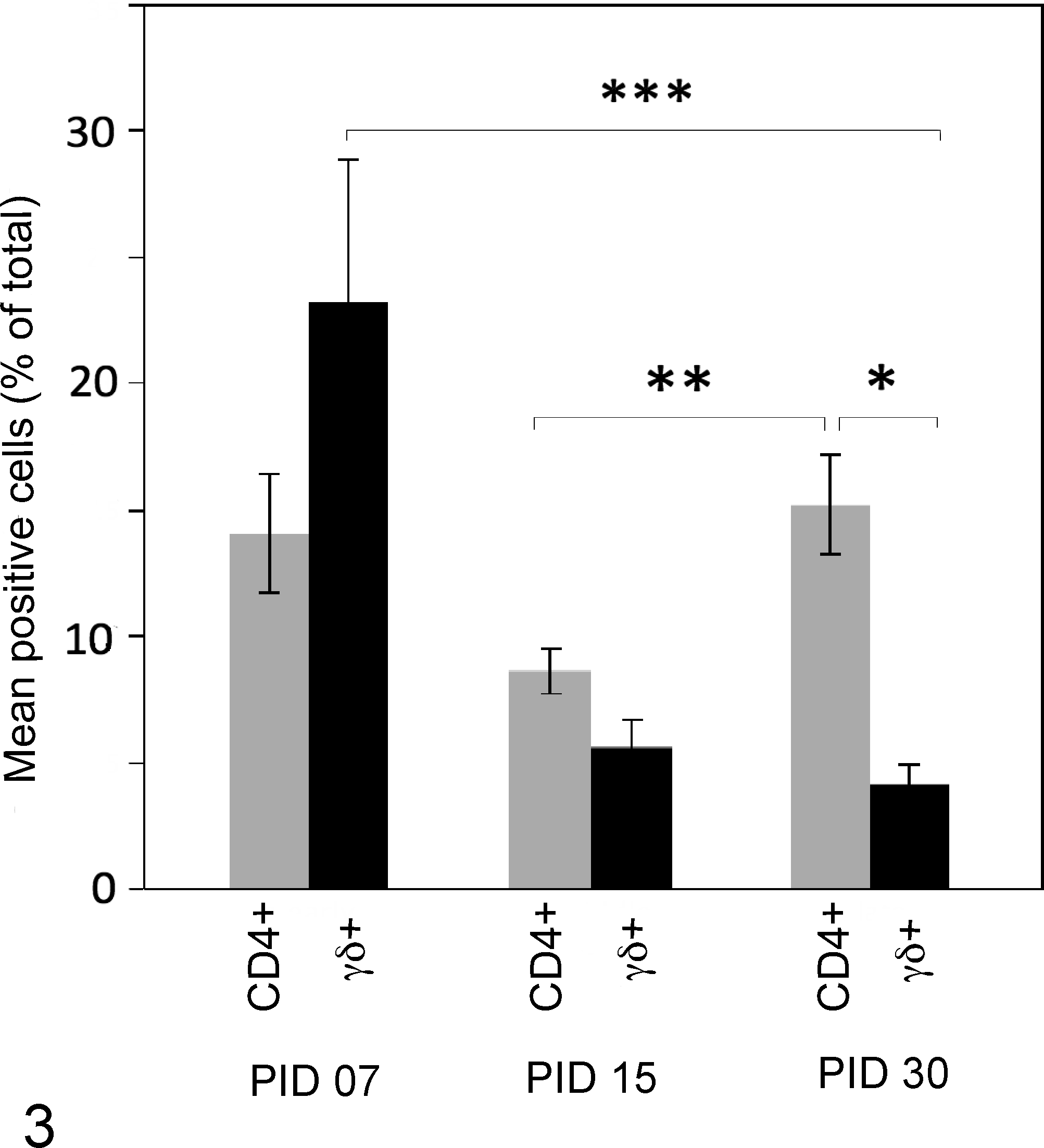
CD4+ and γδ T-cell recruitment patterns to matrigel/Map sites. CD4+ and γδ T cells display inverse recruitment patterns into matrigel/Map sites over time. More γδ T cells were recruited to matrigel/Map sites at PID 7 compared to PID 30 (black bars, P < .0001). More CD4+ T cells were recruited to matrigel/Map sites at PID 30 compared to PID 15 (Fig. 3 gray bars, P = .0073). At PID 30, extracted matrigel/Map contained more CD4+ T cells than γδ T cells (P < .05). Graphed data at each point represent mean percentage marker-positive cells (CD4+ or γδ T+ of total live cells) harvested from matrigel/Map sites of 9 calves.
IFNγ Production at Matrigel Sites
As an additional measure of the local immune response, we measured secreted IFNγ within matrigel and matrigel/Map sites from calves. Compared to PID 7 (Fig. 4a), more IFNγ was detected from matrigel/Map sites at PID 30 (Fig. 4b, note y-axis scale difference between panels 4a and 4b). More IFNγ was detected at matrigel/Map sites compared to matrigel sites in all calves throughout the study (Fig. 4, black bars); however, statistical significance was observed in Map-infected calves at PID 7 (Fig. 4a, P = .0339, Wilcoxon/Kruskal-Wallis test) and Map-vaccinated calves at PID 30 (Fig. 4b, P < .035).
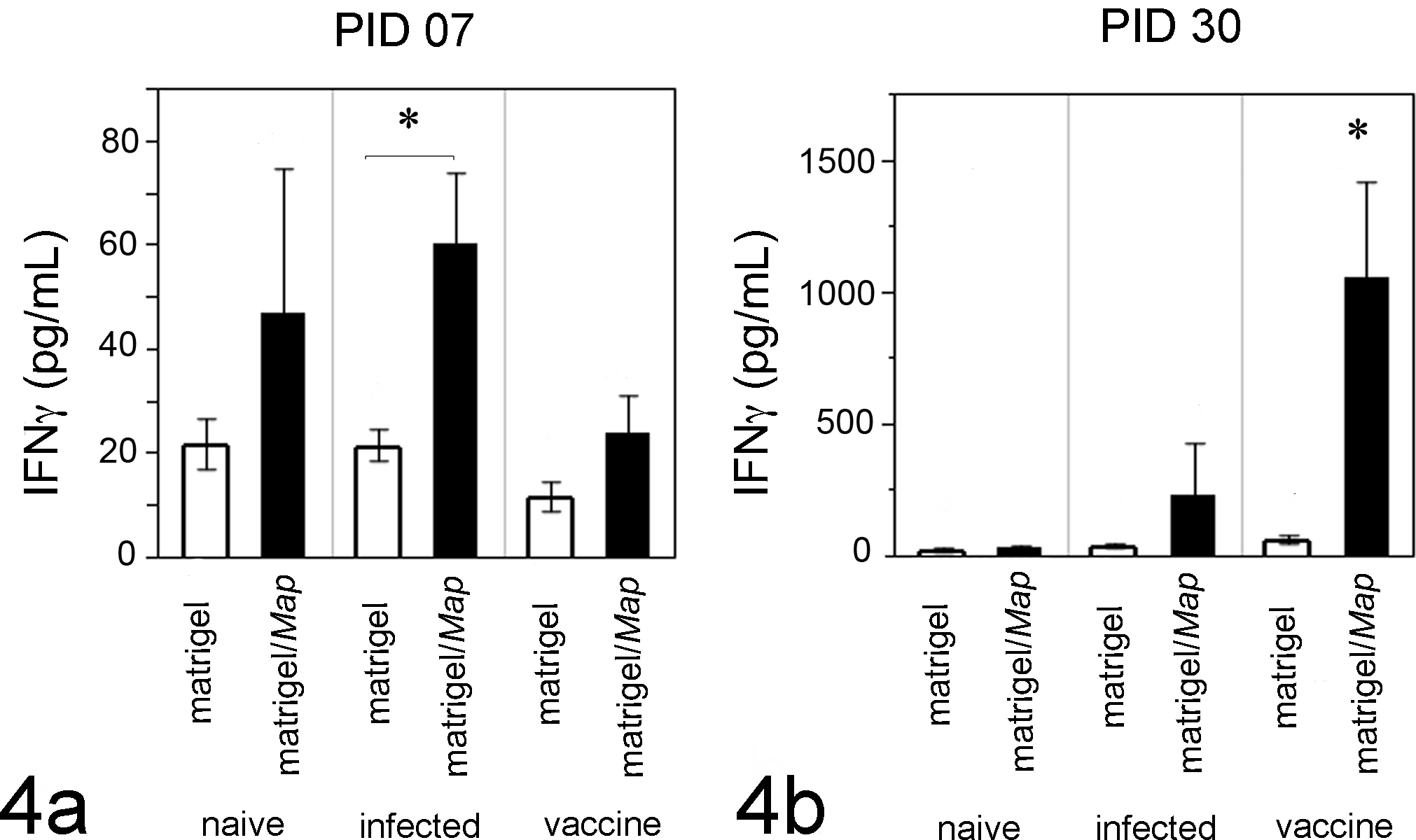
Secreted IFNγ at matrigel and matrigel/Map sites. At PID 7 (a), more IFNγ was detected within extracted matrigel/Map sites (black bars) compared to matrigel sites (clear bars); however, statistical significance was achieved only in Map-infected calves (a, P = .0339, Wilcoxon/Kruskal-Wallis test). At PID 30 (b), significantly more IFNγ was detected in extracted matrigel/Map sites from Map-vaccinated calves compared to all other calves (b, P < .035). Significantly less IFNγ was detected in extracted matrigels at PID 7 compared to PID 30; note the scale difference between panels a and b. Graphed data at each point represent mean IFNγ detected in matrigel or matrigel/Map sites of all calves (n=12).
IFNγ Production by γδ T-cell Subsets
We investigated IFNγ generation by γδ T cells recruited to matrigel sites, which we hypothesize will provide insight into their role during early host immunity of mycobacterial infection. Total recruited cells from matrigel sites were cultured and stimulated with Map antigen (5μg WCS); γδTCR and IFNγ were measured by flow cytometry. Cells collected from matrigel sites showed limited ability to generate IFNγ in our system (Fig. 5). After culture, significantly more cells recovered from matrigel/Map sites at PID 7 expressed γδTCR and IFNγ compared to PID 30 (Fig. 5a, P = .005; Fig. 5b, P = .0087).
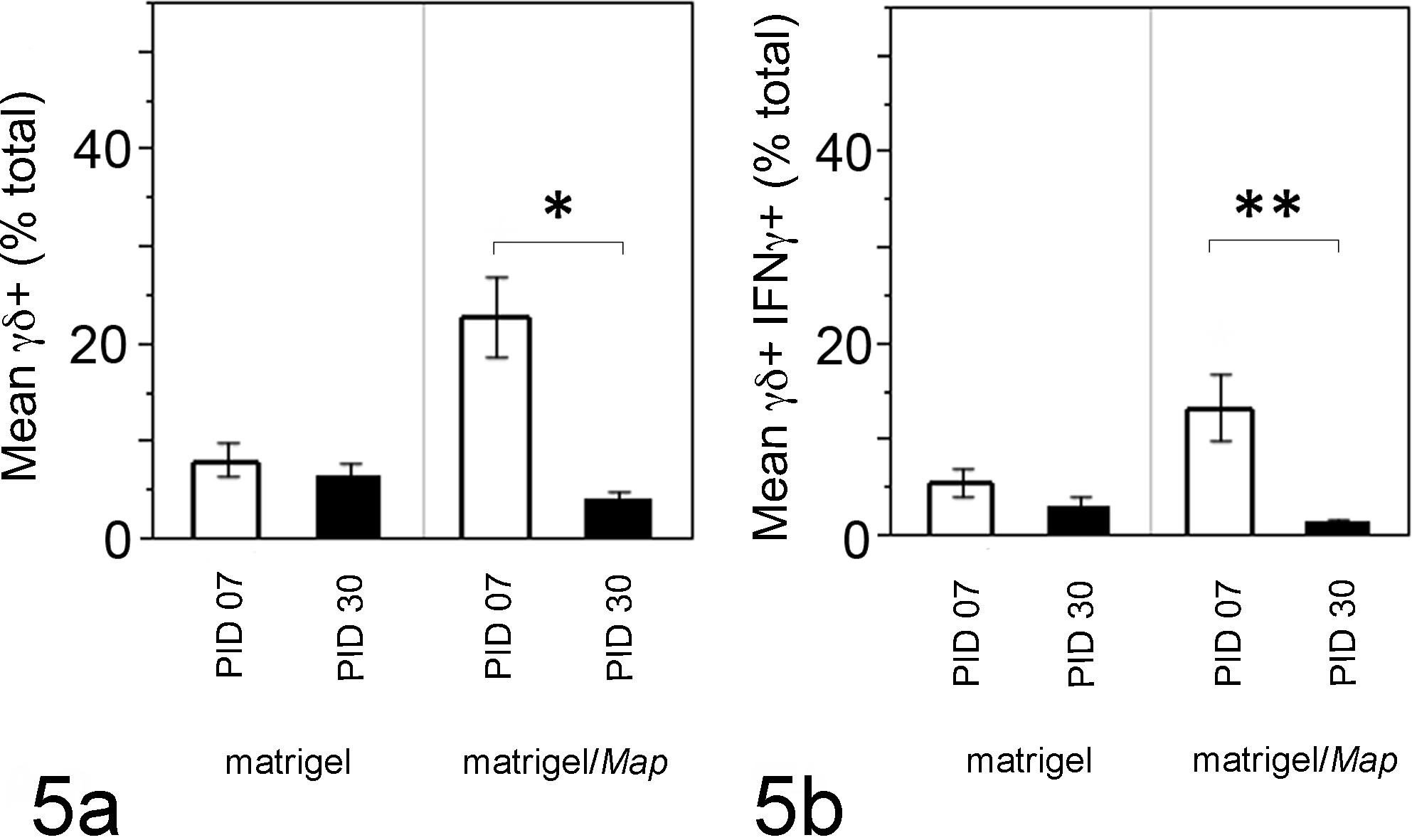
Cultured γδ T cells recruited to matrigel/Map sites generate IFNγ. After culture and Map antigen stimulation, significantly more cells recovered from matrigel/Map sites at PID 7 expressed γδTCR and IFNγ compared to PID 30 (a, P = .005; b, P = .0087). Few cultured cells from matrigel sites at PID 7 or PID 30 expressed γδTCR and/or IFNγ. Graphed data at each point represent mean positive cells (percent γδ TCR+ or IFNγ+ of total live cells by dual color flow cytometry) collected from matrigel and matrigel/Map sites of 9 calves.
We also examined the ability of peripheral blood derived γδ T cell subsets (WC1+γδTCR+ and WC1–γδTCR+) from these calves to generate IFNγ in vitro over time. γδ T-cell culture supernatants from resting (media) or stimulated (media + rhIL2) cell subsets were analyzed for secreted IFNγ, and data are shown in Figure 6. Supplementation with rhIL2 was required, 31 as minimal IFNγ was detected in the absence of rhIL2 (Fig. 6, media, IFNγ range = 3.27–9.24 pg/ml). Addition of Map WCS did not further enhance IFNγ production in our system (data not shown). Distinct responses to rhIL2 stimulation were observed between WC1+ and WC1– γδ T cells at PID 0: naïve WC1– cells generated significantly more IFNγ than WC1+ cells (Fig. 6a, P < .02). At PID 30 we observed distinct responses in γδ T cells from Map-vaccinated but not unvaccinated calves. Specifically WC1– cells from vaccinated calves generated more IFNγ compared to WC1– cells from unvaccinated calves (Fig. 6b, P < .02). We detected IFNγ secretion from WC1+ cells of Map-vaccinated calves at PID 30, but no significant difference was detected between WC1+ and WC1– γδ T cells’ ability to generate IFNγ at PID 30 (Fig. 6c).
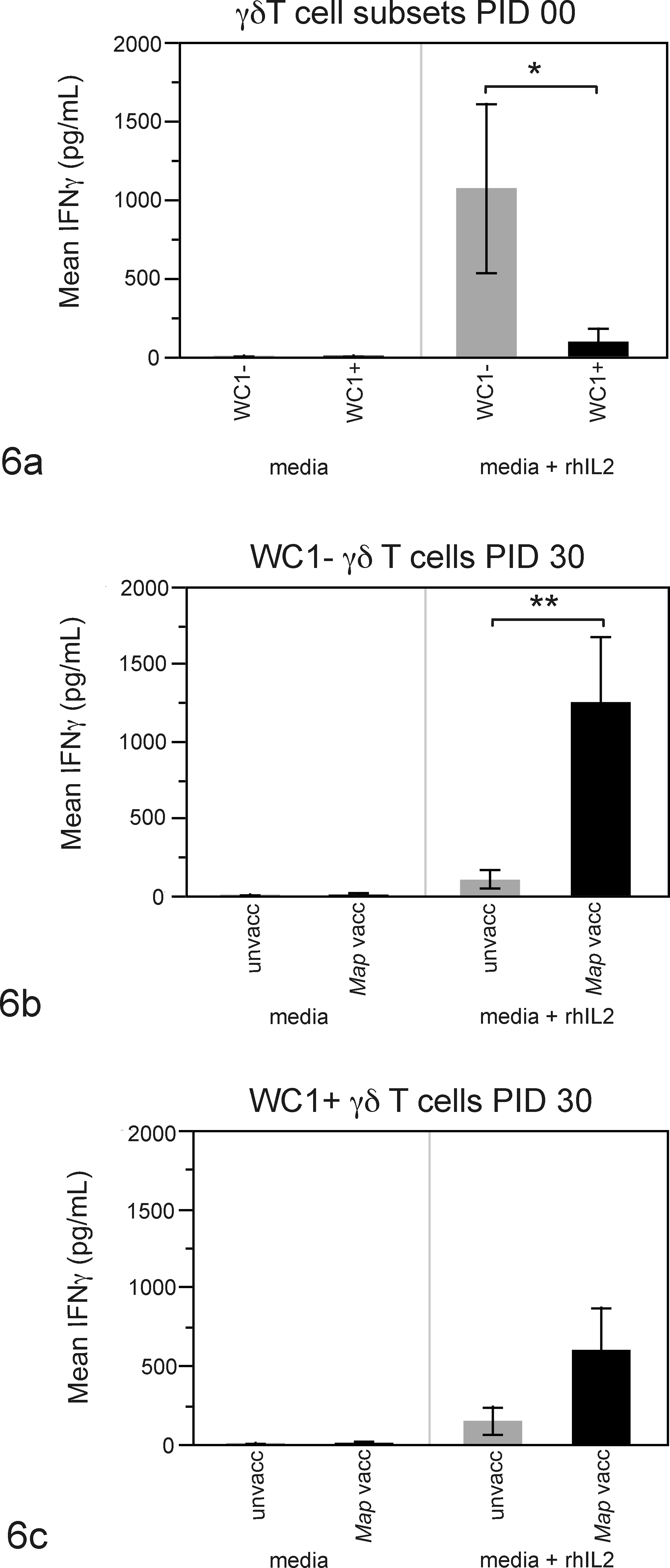
Differential IFNγ production by cultured peripheral blood γδ T-cell subsets. Cultured peripheral blood derived WC1– cells at PID 0 generated significantly more IFNγ than WC1+ cells (a, P < .02). At PID 30, cultured WC1– cells from Map-vaccinated calves secreted significantly more IFNγ compared to cultured WC1– cells from unvaccinated calves (b, P < .02). We detected IFNγ secretion from cultured WC1+ cells of vaccinated calves only at PID 30; however, this was not statistically significant (c). Data in a represent mean IFNγ detected in supernatants from cultured peripheral blood-derived γδ T-cell subsets from 9 calves at PID 0. In b and c, each point represents mean of 6 unvaccinated or 3 Map-vaccinated calves (n = 9 total) at PID 30.
Discussion
To address knowledge gaps regarding early host immunity during intestinal Map infection, we have developed a subcutaneous model of Map infection in young calves with a specific goal to understand the role of bovine γδ T-cell subsets during initial host–mycobacterial interactions. Though γδ T cells are generally considered to function during early stages of the host immune response during infections in cattle and humans, 6,19 in vivo studies evaluating their recruitment into infection sites are limited. 18,23,33 In cattle, WC1+ γδ T cells are numerous in peripheral blood and are the most widely studied, yet most γδ T cells lining the gastrointestinal (and other mucosal sites) are thought to lack WC1 expression. 17,34,35 It seems logical that γδ T cells preferentially localized along mucosal surfaces are involved with innate immunity during infections of these body systems; however, recruitment and function of γδ T cell subsets at mycobacteria-induced lesions remain poorly understood.
Using the matrigel vehicle in a model of bovine Map infection in this study, we identified patterns of T-cell recruitment into early artificial subcutaneous Map infection sites of cattle. We found that γδ T cells were recruited to matrigel/Map sites earlier than CD4 T cells and that significantly more IFNγ was generated at matrigel/Map sites compared to matrigel sites in the model. We therefore hypothesized that IFNγ generation by recruited γδ T cells could account for this difference. To test this hypothesis, we collected cells in matrigel and matrigel/Map sites and stimulated them with crude Map protein preparation (WCS) to show that γδ T cells recruited to matrigel sites with Map generated significant amounts of IFNγ. This is an important finding, because of implications for innate immune cells recruited initially to mycobacterial infection sites of cattle with ability to generate significant amount of the cytokine IFNγ. Early production of IFNγ for activation of other T cells, dendritic cells, and macrophages is known to be a critical step during early host defense against pathogens and in particular against invading intracellular agents. 14,29 More work is needed to clarify the mechanisms of γδ T-cell recruitment to and activation at infection sites and what potential roles they may play during development of adaptive immunity. This research represents an initial step toward potential identification of key factors mediating either success or failure of host immunity during intracellular infections and ultimately leading to pathogen clearance/healing or establishment of persistent infection, respectively.
Much of the current understanding of γδ T-cell immunobiology is based on in vitro data from peripheral blood-derived WC1+ γδ T cells, and to our knowledge, attempts have not been made to correlate peripheral blood cell data with in vivo findings. We therefore examined peripheral blood-derived γδ T-cell subsets from these calves over the course of the study and found that they displayed distinct in vitro response patterns. Our data show that in naïve calves, WC1– cells generated significant IFNγ, suggesting that at least during initial Map infection, WC1– cells are capable of IFNγ production, though this appears to be IL2-dependent and Map antigen-independent. This finding is particularly intriguing as it relates to host defenses against Map, because of the population of WC1– γδ T cells found along gastrointestinal mucosal surfaces. 35 In contrast to WC1– cells, we detected IFNγ in WC1+ cell cultures in this study only from Map-vaccinated calves (Fig. 6c). Though in our study this difference was not statistically significant, the data are similar to a study by Blumerman et al suggesting that memory WC1+ cells can be induced by killed bacterin. 2
A lack of antigenic specificity in naive γδ T cells of cattle is consistent with previous reports. γδ T cells are nonmajor histocompatibility restricted and thus directly respond to pathogen-associated molecular patterns. This is termed antigenic-independent priming, and primed cells have been defined by their upregulation of interleukin 2-receptor α (IL2-Rα). 11 Primed γδ T cells become fully activated and are strong producers of proinflammatory cytokines in response to secondary signals such as IL2 with or without specific antigens. 9,11 Early priming of naive γδ lymphocytes during enteric salmonellosis in calves was recently demonstrated by IL2-Rα gene and protein upregulation; however, γδ T-cell proliferation or proinflammatory cytokine secretion was not described. 7 Distinct priming and activation stages were not clearly defined in γδ T cells of our study, yet the data suggest that γδ T cells are recruited to Map challenge sites where they influence host immunity via their generation of cytokines, such as IFNγ. Jutila et al have hypothesized that a localized response by primed γδ T cells may be sufficient to control the initial infection, at which point the primed γδ T cells would then return to a resting state. 11
It is currently unknown how γδ T cells recruited to and activated at early mycobacterial infection sites in calves influence the outcome of Map infections; however, potential downstream benefits include (1) increasing bacterial killing by macrophages at the infection site 15 or (2) promoting protective immunity via modulating important adaptive immune cells such as αβ T cells or dendritic cells. 24 These pathways need further investigation in cattle and other species, because they could have profound implications for understanding the immunopathology of early mycobacterial infections of cattle. Subsets of γδ T cells in humans and animals continue to be characterized and their functions clarified, and we recognize that a definitive link between γδ T cells of peripheral blood and those of various mucosal sites has not been demonstrated. However, we believe that the subcutaneous model is useful for defining initial features of the host antimycobacterial response, and this should prove beneficial for improvement of current diagnostic tests or development of novel diagnostic strategies during early Map infection in cattle.
Footnotes
Acknowledgments
We thank Dr Shawn Rigby and Christine Deal of the Iowa State University Cell Facility for wise counsel, technical assistance, and help with flow cytometry data processing during this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
