Abstract
In early 2006, the highly pathogenic avian influenza virus (HPAIV) H5N1 of the Asian lineage caused the death of wild aquatic birds in Northern Germany. In the mainly affected areas, a trans-species transmission of HPAIV H5N1 to mammals occurred between birds and domestic cats and 1 Stone Marten (Martes foina), respectively. Here, we report lesions and distribution of influenza virus antigen in 3 cats infected naturally with HPAIV H5N1 A/swan/Germany/R65/06. The hemagglutinin partial nucleotide sequences of the viruses were genetically closely related to a H5N1 HPAIV obtained from a dead Whooper Swan (Cygnus cygnus) of the same area. At necropsy, within the patchy dark-red and consolidated lungs, there was granulomatous pneumonia caused by Aelurostrongylus sp. Histologically, the main findings associated with influenza in all cats were bronchointerstitial pneumonia and marked random hepatic necrosis. In addition, all animals displayed lymphoid necrosis in the spleen and Peyer's patches and necrosis of the adrenal cortex. Immunohistochemically, nucleoprotein of HPAIV was present intralesionally in the lungs, liver, adrenal glands, and lymphoid tissues. Oropharyngeal swabs were shown to be suited to detect HPAIV by quantitative real-time polymerase chain reaction (RT-PCR) in these cats, despite the paucity of influenza virus antigen in the upper respiratory tract by means of immunohistochemistry. The results show that outdoor cats in areas affected by HPAIV in wild birds are at risk for lethal infection. In conclusion, hepatic necrosis was, besides bronchointerstitial pneumonia, the primary lesion, suggesting that in naturally infected cats, damage to the liver plays an important role in the pathogenesis of H5N1 influenza.
Since its first emergence in 1997 in Hong Kong, highly pathogenic avian influenza virus (HPAIV) of the H5N1 subtype has spread in Southeast Asia. In 2005, outbreaks in the Russian Federation and adjacent parts of Kazakhstan and Mongolia have occurred. In the fall of 2005, the virus had reached several countries lining the Black Sea (Turkey, Romania, and Ukraine). These outbreaks occurred not only in domestic poultry but also affected also several wild bird species. In February 2006, outbreaks of influenza due to infection with HPAIV H5N1 occurred simultaneously in 17 countries for the first time. This also included the German coastline of the Baltic Sea, with the first confirmation of HPAIV H5N1 in a Whooper Swan (Cygnus cygnus) and a Mute Swan (Cygnus olor) on February 14. Since then, a total of 158 of more than 1,000 tested wild birds from the Isle of Rügen were diagnosed to harbor HPAIV H5N1 (current situation on September, 25, 2006).3,15,16 Mostly affected were Mute and Whooper Swans as well as several goose species. Furthermore, 1 Stone Marten (Martes foina) was confirmed to be infected with HPAIV H5N1 in Germany. In addition, an isolated outbreak was diagnosed in a commercial medium-sized mixed poultry holding in Saxony.17 Indirect contact with infected wild birds through contaminated fomites has been considered to be the most likely source of infection for this case.3
In addition to a broad number of avian species, HPAIV H5N1 has also been shown to have the ability to infect mammals naturally, especially felids and humans.25 Previously, felids were considered relatively resistant to infection with influenza viruses.6,13,14 In contrast, a few recently published reports describe the transmission of highly pathogenic H5N1 to wild and domestic felids. At first, the infection of 2 leopards and 2 white tigers with HPAIV H5N1 was reported in a zoo near Bangkok. These animals had been infected by ingestion of HPAIV H5N1-contaminated chicken meat and died with signs of a systemic infection, especially involving the respiratory tract.18 Furthermore, horizontal transmission of HPAIV has been confirmed during an outbreak in the largest tiger zoo in Thailand, where 147 tigers died or had to be euthanized.23 Reports of natural H5N1 influenza infections of domestic cats have been confined to 1 confirmed case, although there are further anecdotal reports of 3 possibly infected cats in Thailand.3,20
Susceptibility of domestic cats to HPAIV H5N1 and its horizontal transmission have been studied by experimental intratracheal inoculation of H5N1 virus (A/Vietnam/1194/04) and the feeding of cats with infected chicks.10 While mortality in these experiments was low or not investigated, the main lesions in these cats consisted of diffuse alveolar damage as well as necrosis and inflammation in a variety of extrarespiratory tissues.18 This is unprecedented since earlier infection studies of cats with human and avian Influenza A viruses of the subtypes H3N2, H2N2, H7N3, and H7N7 resulted in infection and viral shedding but did not provoke any clinical signs.6,13,14 These observations were supported by serologic studies in Japan, where 5 of 52 healthy cats were shown to carry antibodies against Influenza A virus subtype H3, demonstrating natural infection.7,12 This is further supported by an anecdotal report from Thailand, where 8 of 111 cats were positive for antibodies against H5N1.2
Here we describe gross lesions and histopathology in 3 domestic cats naturally infected with HPAIV H5N1 of Asian lineage during the recent outbreak among wild birds in Germany. By immunohistochemistry, the distribution of influenza virus nucleoprotein in the infected animals was investigated.
Materials and Methods
Case histories
Three domestic Shorthair cats were found dead on the Isle of Rügen, approximately 1 mile from places were HPAIV H5N1-infected wild birds, mainly Mute and Whooper Swans and several species of geese and ducks, were detected continuously during the preceding 3 weeks. Cat No. 1 was an adult male and—according to the owners' report—an outdoor cat, who appeared clinically inconspicuous up to 24 hours prior to death. Five other felines in the same household as cat No. 1 did not develop any clinical signs over the following weeks. No clinical histories of the female, juvenile cat No. 2, and the male, adult cat No. 3, were available. The second HPAIV H5N1-positive cat was found dead about 4 miles distant from cat No. 1, while the third cat was found in 0.5 miles from the site where cat No. 1 had been detected.
Necropsy, histopathology, and immunohistochemistry
Pathologic examinations were performed under biosafety level 3, and tissue samples of the minimally autolyzed carcasses, including brain, soft palate, tongue, retropharyngeal lymph nodes, trachea, lung, intestine, liver, pancreas, adrenal gland, kidney, bladder, spleen, and bone marrow, were taken. Tissues were fixed in 4% neutral-buffered formaldehyde and embedded in paraffin wax. Paraffin wax sections (3 μm) were dewaxed and stained with HE for light microscopy. To detect influenza virus, we mounted nucleoprotein (NP) sections on SuperFrost microscope slides (Menzel, Braunschweig, Germany), dewaxed and incubated with a rabbit anti-NP serum (1:500 in Tris-buffered-saline [TBS], 0.1 M Tris-base, 0.9% sodium chloride, pH 7.6 [kindly provided by E. Mundt, FLI, Insel Riems]).9 To exclude concurrent infection with feline Coronaviruses, we used the monoclonal antibody FIPV3-70 (immunoglobulin G2A [IgG2A] kappa, Custom Monoclonals Int., W. Sacramento, CA, USA), diluted 1:100 in TBS. Rabies and pseudorabies were excluded immunohistochemically using the monoclonal Swiss rabies antibody, diluted 1:80 (kindly provided by Swiss Rabies Center, Bern, Switzerland) and anti-PrV UL19 polyclonal rabbit serum, diluted 1:500 (kindly provided by B. Klupp, FLI). As secondary antibodies, biotinylated goat anti-rabbit IgG1 and goat anti-mouse IgG1 (Vector, Burlingame, CA, USA; diluted 1:200 in TBS) were applied. Rabbit preimmunization serum was used as a negative control. By means of the ABC method and an immunoperoxidase kit (Vectastain Elite ABC Kit, Vector), a bright-red signal was produced from the substrate, 3-amino-9-ethylcarbazole (DAKO AEC substrate-chromogen system, Dako, Carpinteria, CA, USA). Furthermore, sections were counterstained with Mayer's hematoxylin and sealed with aqueous medium (Aquatex, Merck, Darmstadt, Germany).
Clinical pathology
Aqueous humor samples were collected at necropsy from all 3 cats and evaluated biochemically according to standard procedures for aspartate aminotransferase (AST), alkaline phosphatase (ALP), alanine aminotransferase (ALT), lactate dehydrogenase (LDH), and gamma glutamyl transferase (GGT). The obtained values were compared with reference data from the literature.1,4
Virological examinations
Nasal and oropharyngeal swabs of the cats were tested for influenza virus RNA by real-time polymerase chain reaction (RRT-PCR). Detection of an Influenza virus A matrix gene fragment by PCR (M-PCR) was followed by further subtype differentiation using RRT-PCRs targeting fragments of the H5 and N1 genes.21 Sequencing of an HA gene fragment spanning the stretch that encodes the endoproteolytic HA cleavage site confirmed presence of HPAIV. Infections with feline immunodeficiency virus (FIV) and feline leukemia virus (FeLV) were examined by the analysis of proviral DNA using PCR. DNA was isolated from peripheral lymph nodes and the spleen of all 3 cats. The DNAs were investigated for the presence of FIV gag and FeLV U3 LTR-specific sequences, using primers and amplification conditions previously published.11,22,24 In addition, lung exudate and cardiac blood derived from cat No. 1 were investigated using a commercial FeLV antigen and FIV antibody test (FeLV/FIV Snap, IDDEX, Westbrook, ME, USA).
Bacteriologic examinations
For bacteriologic examination, probes of lung, liver, and spleen were incubated at 37°C on dextrose blood agar plates and Gassner plates for 24 and 48 hours. For specific detection of Salmonella spp., Kauffmann medium was used.
Results
Gross pathology
At necropsy, cat No. 1 showed hyperemia of the lungs with multiple, mainly peribronchiolar, dark-red, irregularly shaped, consolidated areas. The spleen was markedly enlarged and congested. The liver was dark tan and firm and appeared normal. Within the small intestine, numerous adult ascarid nematodes and a few cestodes were found in the otherwise empty gastrointestinal tract.
The lungs of cat No. 2 were diffusely dark red, with multiple-to-coalescent yellow consolidated nodules. On section, there were mainly peribronchiolar dark-red, irregularly shaped, consolidated areas. Bronchi and bronchioli contained abundant yellow and tenacious mucus. Pulmonary lymph nodes were moderately enlarged and wet on section. Multifocally within the liver, there were a few randomly arranged, sharply demarcated gray-to-white areas of pinpoint size or up to 3 mm in diameter (necrosis). Within the small intestine, numerous adult ascarid nematodes and a few cestodes were found. The heart showed moderate dilation of the right ventricle.
Cat No. 3 showed extensive postmortal loss of the skin, subcutaneous tissue, and parts of the skeletal muscles in the cervical and shoulder region due to scavenging. The mucosa of the nasal cavity, pharynx, and trachea was diffusely hyperemic. There was a hemohydrothorax of about 20 ml. The lungs were severely edematous and showed numerous, sharply demarcated, yellow nodules. Multifocally in the liver, there were few, up to 2-mm-in-diameter large, sharply demarcated light-brown-to-yellow areas (necrosis). Retropharyngeal and pulmonary lymph nodes were moderately swollen with a few ecchymoses. Petechiation or diffuse hemorrhages were found retroperitoneally and intramuscularly in the diaphragm, within the perirenal tissue and the pancreas.
Histopathology
The main histologic findings in all 3 cats were confined to marked, multifocal hepatic necrosis and severe multifocal bronchointerstitial pneumonia and moderate focally extensive necrosuppurative bronchopneumonia. Within the lungs of all animals, alveolar lumina were either diffusely filled with abundant homogeneous pale eosinophilic fluid (alveolar edema), macrophages, erythrocytes, and cellular necrotic debris, or alveoli were collapsed (atelectasis). The bronchiolar epithelium was multifocally degenerated and partially lost, and lumina were filled with sloughed, necrotic epithelial cells as well as karyorrhectic and cellular debris admixed with a few neutrophils and macrophages (Fig. 1A). Peribronchiolar alveolar septa were slightly expanded by beaded fibrillar eosinophilic material (fibrin) and infiltrated by small numbers of neutrophils and lymphocytes. Furthermore, in all cats, alveoli were obliterated multifocally by numerous clustered morulated nematode eggs, nematode larvae, and adult nematodes, characterized by smooth cuticles, polymyarian-coelomyarian musculature, lateral cords, pseudocoelom, and digestive tracts with uninucleate epithelial cells most consistent with Aelurostrongylus sp. Sections of nematodes were surrounded by neutrophils, eosinophilic granulocytes, and rare multinucleated giant cells. Alveolar epithelium in these areas was lost, attenuated, or replaced by large polygonal cells (type II pneumocyte hyperplasia). Alveolar septa were expanded by lymphocytes, eosinophils, and edema. Additionally, cats Nos. 2 and 3 displayed up to 7-μm-thick, eosinophilic hyaline membranes lining the alveolar walls (Fig. 1C). The lung of cat No. 2 showed a mild diffuse suppurative pneumonia, with infiltration of alveolar lumina by numerous neutrophils, degeneration of lining pneumocytes, and mild hyperplasia of pneumocytes type II.
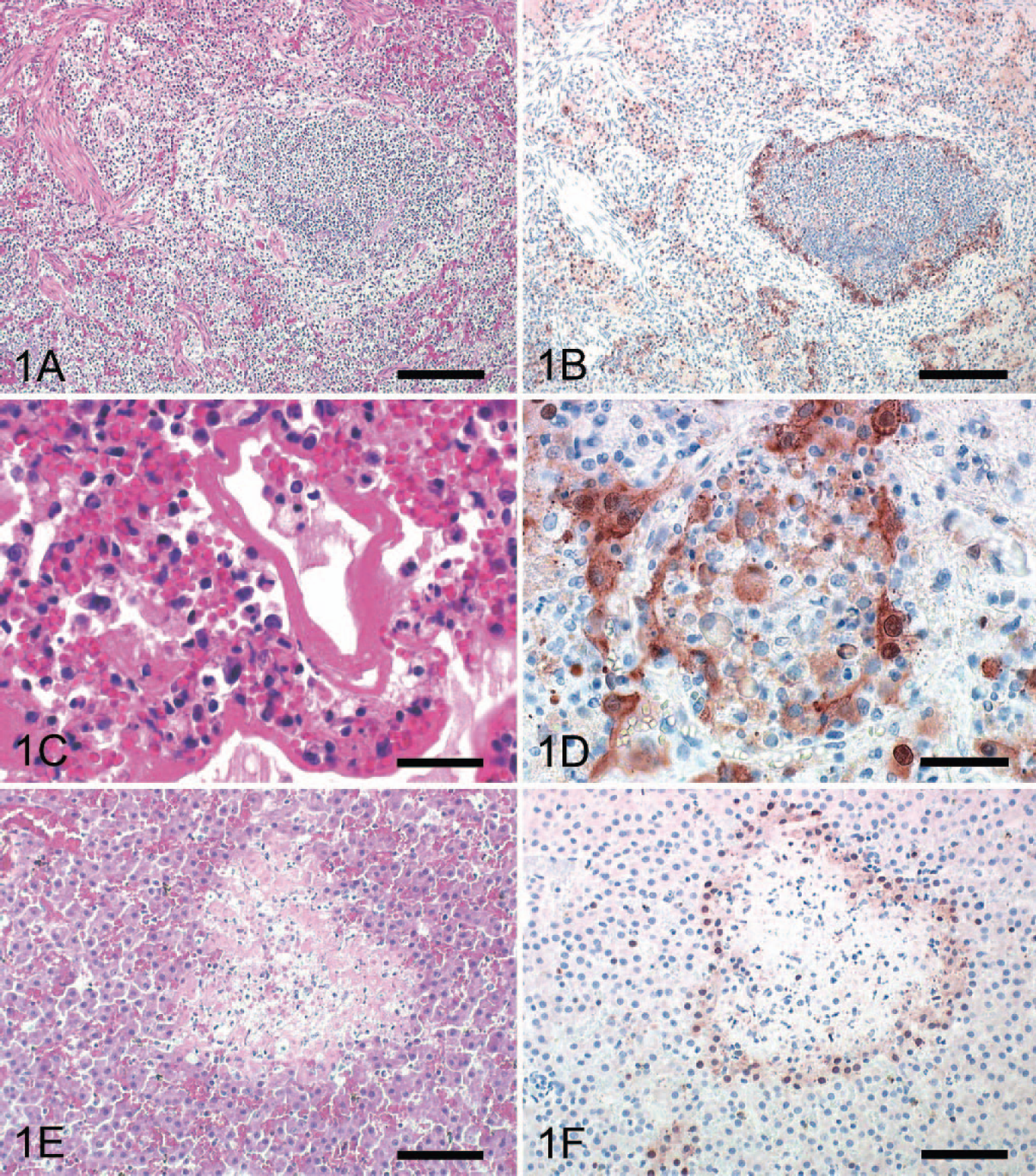
Lung: cat No. 3. Bronchiolar epithelium is partly lost and necrotic, and lumen
is completely obliterated by cellular debris. Peribronchiolar alveoli are
obscured by numerous intra-alveolar macrophages, debris, and thickening of
alveolar septa. HE. Bar = 500 μm.
Within the liver of all cats, there were numerous, randomly arranged, sharply demarcated areas of coagulative necrosis with loss of hepatic architecture and cellular detail and replacement by karyorrhectic and cellular debris admixed with abundant fibrin and numerous erythrocytes (Fig. 1E). In cats Nos. 1 and 3, these foci were surrounded by a small number of lymphocytes. Additionally, all cats displayed multifocal areas of necrosis of adrenocortical cells and, to a lesser extent, areas of lymphoid necrosis with replacement by karyorrhectic debris and fibrin in the spleen and the Peyer's patches of the small intestine (Fig. 2A, C). The hearts of all animals showed mild myocardial hemorrhage and in cat No. 3, a mild fibrinous epicarditis.
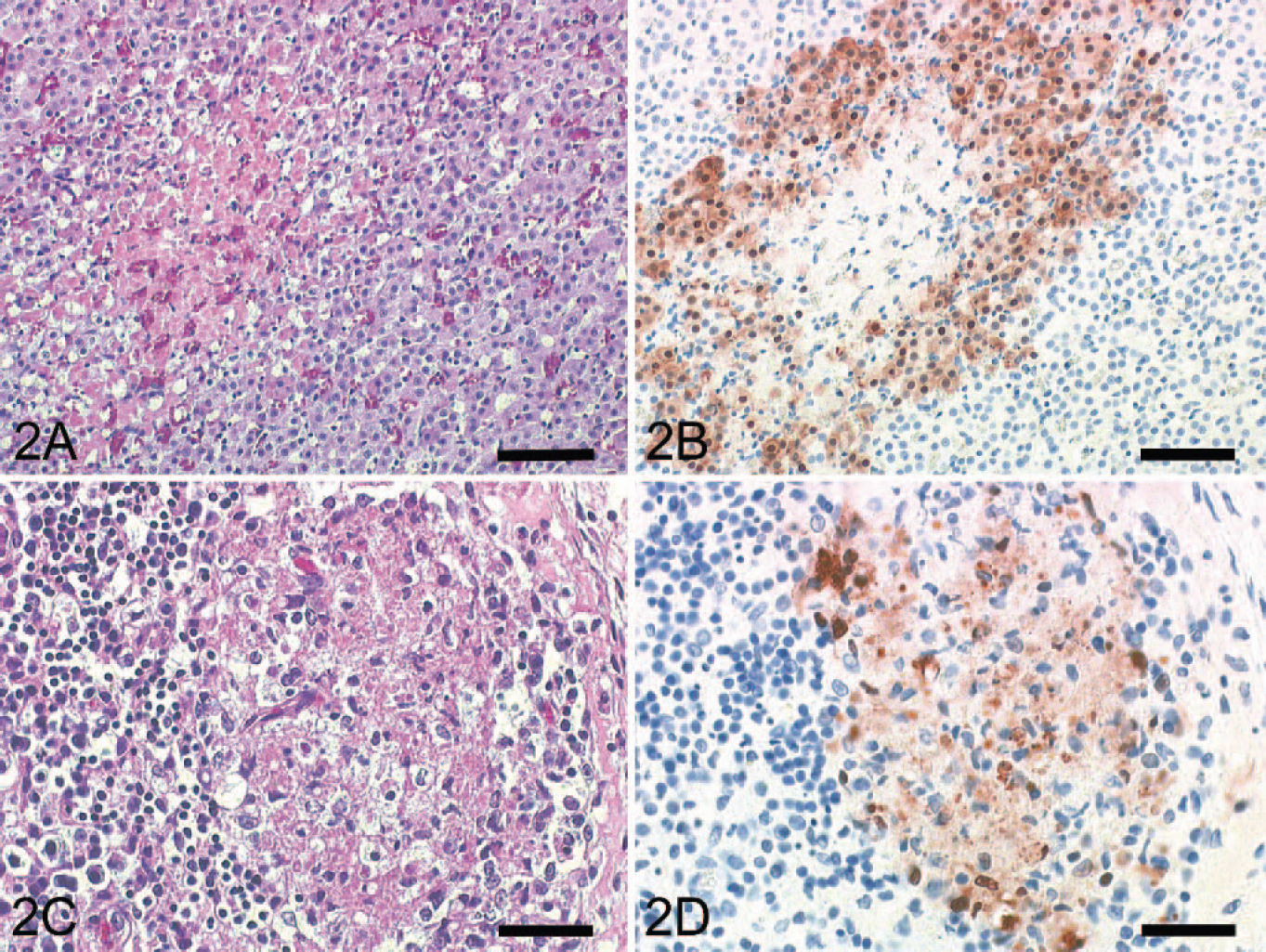
Fig. 2A. Adrenal gland: cat No. 1. Zona fasciculata with sharply demarcated
areas of necrosis is seen. HE. Bar = 200 μm.
Immunohistochemistry
In all 3 cats, AIV antigen was present in the cytoplasm and nucleus of intact, degenerating, and necrotic or sloughed ciliated and nonciliated bronchiolar epithelial cells (Fig. 1B, D). Furthermore, moderate numbers of type I and II pneumocytes, large round cells with abundant cytoplasm, presumed to be intra-alveolar macrophages and interstitial mononuclear cells, were strongly positive. Corresponding to the histologic findings, strong intracytoplasmic and intranuclear immunohistochemical signals were found in degenerating hepatocytes adjacent to hepatocellular necrosis in all cats (Fig. 1F). Furthermore, adrenocortical cells surrounding areas of necrosis were immunohistochemically positive for AIV antigen in all cats (Fig. 2B). Within the congested spleen of all cats, a few large polygonal mononuclear cells and reticular cells of the periarteriolar sheaths and the red pulp displayed an intracytoplasmic and intranuclear staining. Similar positive cells were found in pulmonary and mesenteric lymph nodes and Peyer's patches of all cats (Fig. 2D). While neurons and glial cells of the cerebellum and the cerebrum were consistently negative in all cats, few intravascular mononuclear cells showed an immunoreaction for influenza virus antigen. Immunohistochemistry of the soft palate, tongue, trachea, intestine, pancreas, kidney, and bladder revealed no AIV antigen. Immunohistochemistry for feline Coronavirus (brain, liver, intestinal tract, lymph nodes), pseudorabies virus (brain), and rabies virus antigen (brain) was negative in all cats.
Clinical pathology
The results of the clinical chemistry in the aqueous humor samples are shown in the Table. Marked increase of enzyme activities of AST, ALT, and LDH indicated hepatocellular leakage and necrosis in all cats. Furthermore, enzymatic activity of ALP (not specified as liver isoenzyme) was increased in cat No. 2, indicating hepatic lipidosis or extrahepatic disease, since cholestasis could be excluded by GGT values, which were within the reference intervals. Furthermore, the increased ALP value of cat No. 2 could be at least partly attributed to the age of this juvenile cat.
Postmortem analysis of aqueous humor(anterior chamber, eye) of 3 cats infected naturally with highly pathogenic avian influenza virus H5N1.
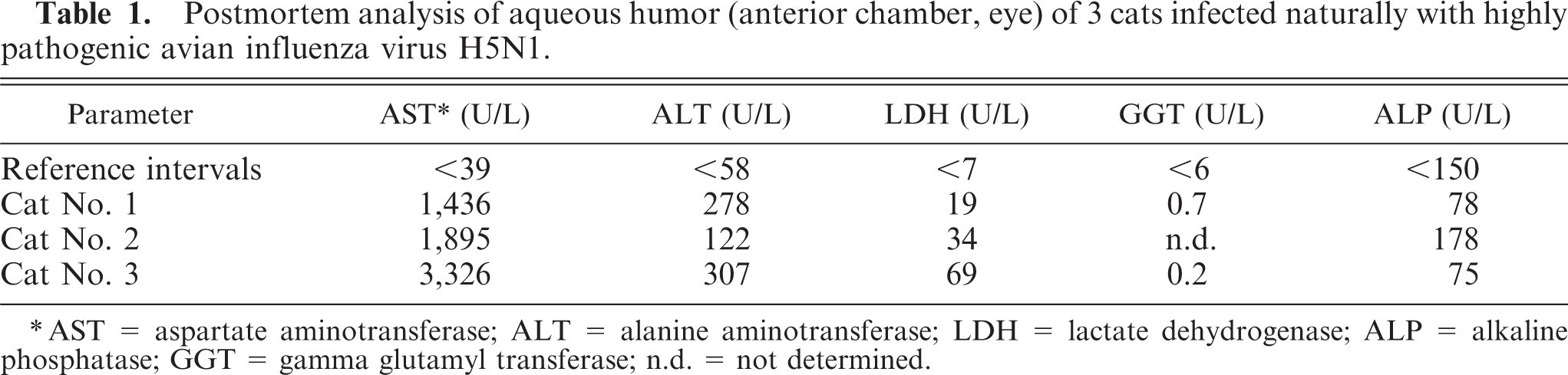

AST = aspartate aminotransferase; ALT = alanine aminotransferase; LDH = lactate dehydrogenase; ALP = alkaline phosphatase; GGT = gamma glutamyl transferase; n.d. = not determined.
Virologic examinations
M-PCR and H5N1-specific RRT-PCRs with samples from all 3 cats were positive. RRT-PCRs showed a viral load of 109.0 embryo (egg) infective dose 50% (EID50) in the lung and 105.5 EID50 in the brain per 0.2 ml of a 10% tissue suspension. Molecular pathotyping confirmed the presence of HPAIV H5N1 of the Asian lineage in all 3 cats and revealed the closest relationship to a H5N1 HA partial sequence obtained from a dead Whooper Swan from the Isle of Rügen (not shown). All 3 cats were negative by PCR for the detection of FIV gag-specific and FeLV U3 LTR-specific proviral DNA. Using lung exudate and cardiac blood derived from cat No. 1, we did not detect FeLV-specific antigen or FIV-specific antibodies.
Bacteriological examinations
Bacteriology revealed a moderate growth of Pasteurella multocida in the lungs of cats Nos. 1 and 2, whereas cat No. 3 showed minimal growth of Streptococcus spp. in the lungs. No relevant bacteria were detected in the livers of the 3 cats. Spleen, kidney, heart, and brain were bacteriologically negative or contained only small-to-moderate numbers of Escherichia coli.
Discussion
Epidemiologic data, results of virologic investigations, and, most strikingly, the intralesional detection of influenza virus antigen strongly indicate the lethal course of natural HPAIV H5N1 infection in all 3 cats. We show that HPAIV H5N1 replicated in hepatocytes, pulmonary epithelium, and adrenocortical and mononuclear cells. According to the hepatocellular necrosis that was co-localized with influenza virus antigen, acute hepatic disease may play a major role in the pathogenesis of the disease in these cats. The bronchointerstitial pneumonia as a result of infection with HPAIV H5N1 in association with pulmonary aelurostrongylosis has certainly contributed to the severity of the clinical course. The evidence of P multocida in the lungs is assumed to be secondary to HPAIV infection.
To date, avian influenza in felids following ingestion of contaminated poultry has been confirmed in a small number of cases of natural and experimental infections.8,18,20 Furthermore, horizontal transmission of HPAIV H5N1 was demonstrated for domestic cats and hypothesized for tigers.10,23
The cats in this report were found in close spatiotemporal correlation with an outbreak of HPAI in wild birds. In these areas, carcasses of wild swans, ducks, and geese had been accessible to both avian and mammalian scavengers. Therefore, infected wild birds were assumed to be most likely the sources of infection. From this, it follows, that the respiratory or intestinal tract might be the possible portal of entry in these cats, followed by a viremia. That corresponds to recent reports of ingestion of virus-infected chicks or a naturally infected pigeon resulting in infection of felids.18,20 Similar exposure and infection of scavenging mammalian species is also most likely in a confirmed HPAIV H5N1 infection of a Stone Marten found in the same area. Following the exhaustive removal of bird carcasses from the shores, coinciding with the order to keep cats indoors and to take dogs on the leash, no further cases in mammals have been noticed. In addition, evidence for horizontal transmission among felids has not been obtained so far. Although other species like red foxes (Vulpes vulpes) and raccoon dogs (Nyctereutes procyonoides) have been included in the ongoing monitoring programs, so far, neither virologic nor serologic evidence for the involvement of these mammalian species has been obtained.
With regard to feline retrovirus infections, all 3 animals belong to the high-risk group of stray outdoor cats. Because FIV and FeLV infections are associated with severe immunodeficiencies that might have favored infection with HPAIV H5N1 in these cats, we investigated different lymphoid tissues for provirus. None of the cats was positive in this assay, which was supported in cat No. 1 by negative FeLV antigen and FIV antibody test results.
Under experimental conditions, cats have been shown to excrete HPAIV H5N1 influenza viruses oronasally and by the fecal route.18 In our cats, immunohistochemistry revealed no influenza virus antigen in laryngeal, tonsillar, and tracheal tissues or the mucosa of the gastrointestinal tract. Nevertheless, all 3 cats were diagnosed as HPAIV- positive by nasopharyngeal and tracheal swabs and revealed a substantial viral load in these samples. Expectorated bronchial mucus with cellular debris or infection of the nasal respiratory mucosa seems to be responsible for the oronasal excretion. Samples of nasal mucosa were not available for immunohistochemistry. However, massive virus replication was detected in the bronchial epithelium but not in the trachea and the oral mucosa. These observations agree with reports describing the pattern of HPAIV attachment to pneumocytes type II, nonciliated bronchiolar epithelium, and alveolar epithelium lining the lower respiratory mucosa of cats and their absence in the mucosa of the trachea.26 Oropharyngeal swabbing may be the most promising sample for monitoring felids alive for HPAIV H5N1.
Acute hepatic damage appears to be a hallmark in the course of the infection of cats. Marked elevation of ALT and AST activity in the aqueous humor supports the clinical relevance of hepatocellular necrosis in cats.4 While rectal swabs were not available for testing, the excretion of virus through feces even in the absence of active viral replication in the gastrointestinal mucosa might be possible through bile excreted from the heavily infected and damaged liver. Biliary excretion of infectious agents has also been shown for rabbit hemorrhagic disease virus or Listeria monocytogenes.5,19 So far, HPAIV has been detected in fecal swabs of experimentally infected cats and in the submucosal and myenteric plexus in the ileum and duodenum, while intestinal epithelium, as in our cats, was not infected.18 Further experimental studies are needed to finally clarify the source and relevance of fecal shedding of HPAIV by infected cats. Although it was described in the literature, we were not able to demonstrate infection of the cerebrum and cerebellum, myocardium, kidney, and enteric nervous plexus in any of the 3 cats.18,20 Predisposing factors, like the severe coinciding verminous bronchopneumonia or differences in the amount of ingested virus between field infections and experimental settings, might be responsible for the interference with virus spread, a different virus-host-interaction, and the temporal course of disease.18
In summary, we show that outdoor cats in areas with HPAIV infection in wild birds are at risk for fatal infection. Hepatocellular damage obviously plays a major role in the pathogenesis of the disease in felids. Further investigations are necessary to explain if this hepatotropism is a unique characteristic of this particular virus strain or more related to a specific susceptibility of the host. Because of its consistency in all 3 cats, we recommend including HPAI as a differential diagnosis for cases of random and widespread hepatocellular necrosis with necrotizing bronchointerstitial pneumonia in felids.
Footnotes
Acknowledgements
We thank G. Czerwinski, K. Steffen, and K. Wink for the excellent technical assistance and T. Kuiken for providing control feline tissues for immunohistochemistry.
