Abstract
Mortality in wild aquatic birds due to infection with highly pathogenic avian influenza viruses (HPAIV) is a rare event. During the recent outbreak of highly pathogenic avian influenza in Germany, mortality due to H5N1 HPAIV was observed among mute and whooper swans as part of a rapid spread of this virus. In contrast to earlier reports, swans appeared to be highly susceptible and represented the mainly affected species. We report gross and histopathology and distribution of influenza virus antigen in mute and whooper swans that died after natural infection with H5N1 HPAIV. At necropsy, the most reliable lesions were multifocal hemorrhagic necrosis in the pancreas, pulmonary congestion and edema, and subepicardial hemorrhages. Major histologic lesions were acute pancreatic necrosis, multifocal necrotizing hepatitis, and lymphoplasmacytic encephalitis with neuronal necrosis. Adrenals displayed consistently scattered cortical and medullary necrosis. In spleen and Peyer's patches, mild lymphocyte necrosis was present. Immunohistochemical demonstration of HPAIV nucleoprotein in pancreas, adrenals, liver, and brain was strongly consistent with histologic lesions. In the brain, a large number of neurons and glial cells, especially Purkinje cells, showed immunostaining. Occasionally, ependymal cells of the spinal cord were also positive. In the lungs, influenza virus antigen was identified in a few endothelial cells but not within pneumocytes. The infection of the central nervous system supports the view that the neurotropism of H5N1 HPAIV leads to nervous disturbances with loss of orientation. More investigations are necessary to clarify the mechanisms of the final circulatory failure, lung edema, and rapid death of the swans.
Introduction
Epidemic outbreaks of highly pathogenic avian influenza (HPAI) caused by viruses of the subtype H5N1 of Asian lineage emerged since 2003 in several countries of Southeast Asia. To contain the epidemic in poultry, millions of birds were culled. Moreover, 224 confirmed cases of human H5N1 HPAI virus (HPAIV) infections, with a total of 154 fatalities, were reported to the World Health Organization until 29 November 2006 (http://www.who.int/csr/disease/avian_influenza). The majority of human cases so far were traced to direct intense contact with domestic poultry infected with HPAIV H5N1. However, exposure risks in several recent cases reported from Azerbaijan were associated with contact (defeathering) with infected wild swans.
The occurrence of HPAIV H5N1 of Asian lineage in wild birds in northeastern Germany on 14 February 2006 was part of a rapid transcontinental spread of the virus across Asia in wild and domestic birds that possibly commenced with the mass mortality among bar-headed geese at Lake Qinghai, China, in spring 2005. During late winter and early spring 2006, HPAI was officially reported to the OIE from numerous European countries: Bulgaria, Greece, Italy, Slovenia, Austria, Germany, Bosnia-Herzegovina, France, Hungary, Slovakia, Switzerland, Serbia-Montenegro, Poland, Denmark, and Sweden (http://www.who.int/csr/disease/avian_influenza/timeline.pdf).
In Germany, increased mortality among mute and whooper swans, as well as other wild aquatic birds, has been observed since the beginning of February 2006 along the northwestern shores of the Island of Ruegen, which is part of a national park. This area is characterized by open coastal landscape, shallows, and inlets of the Baltic Sea, which are a temporary home for thousands of migratory and resident aquatic birds. Since its first detection in February on the Island of Ruegen, HPAIV H5N1 has been found by means of a H5N1-specific real-time reverse transcriptase polymerase chain reaction (RRT-PCR) in aquatic wild birds, such as whooper (Cygnus cygnus) and mute swans (Cygnus olor), Canada geese (Branta canadensis), and tufted ducks (Aythya fuligula), and scavenging species, such as seagulls (Larus marinus, L argentatus), hawks (Accipiter gentilis), and common buzzards (Buteo buteo). A total of 344 cases have been identified throughout Germany, not only from the Island of Ruegen but also scattered focally to geographically widely separated regions (covering 8 of 16 federal states).
Since its first appearance in Hong Kong in 1997, H5N1 HPAIV infection of swans has been described only rarely.2 Nevertheless, during the recent outbreaks in Europe, swans appeared to be primarily affected and were designated as an indicator species of the presence of HPAIV H5N1 in wild birds.3,11
Here we report the results of postmortem examinations and describe the distribution of histologic lesions and viral antigen of HPAIV in mute and whooper swans that were naturally infected and succumbed to fowl plague during the recent outbreak along the German Baltic Sea coast.
Materials and Methods
Case material and sampling
Fourteen juvenile and adult mute and 6 adult whooper swans that were in good postmortem condition analyzed in this study were found dead at the Baltic Sea coastal area in late February 2006. Tracheal swabs were taken and analyzed for HPAIV H5N1 RNA by RRT-PCR and nucleotide sequencing. Eighteen animals were found positive and necropsied. The 2 HPAIV-negative juvenile mute swans served as controls. A panel of tissue samples (Table 1) was fixed for 48 hours in 4% phosphate-buffered neutral formaldehyde and processed for paraffin embedding.
Distribution of histological lesions and influenza virus antigen in mute and whooper swans after natural infection with HPAIV A/swan/Germany/R65/06 (H5N1).

RRT-PCR and DNA sequencing
RNA was isolated from tracheal swabs of all 20 swans of this investigation using the QIAamp Viral RNA Mini Kit (Qiagen). Diagnosis and characterization of H5N1 HPAIV by RRT-PCR followed a cascade style recommended by the Diagnostic Manual issued by the European Commission: 1) Generic influenza PCR: Detection of an influenza A virus matrix (M) gene fragment by duplex RRT-PCR, including an externally added extraction and inhibition control10; 2) Specific detection of subtypes H5 and/or H7: RRT duplex PCR for the detection of subtypes H5 and N1 and exclusion of subtype H7 by uniplex RRT-PCR6,10; 3) Molecular pathotyping of H5/H7 subtypes: Confirmation of the highly pathogenic type by nucleotide sequence analysis of an HA gene fragment encoding the endoproteolytical HA cleavage site. In addition, aiming at further characterization, the full-length HA and matrix genes were sequenced.
Histopathology and immunohistochemistry
Paraffin-wax sections (3 μm) were dewaxed and stained with HE. For immunohistochemistry, sections were mounted on charged SuperFrost® Plus microscope slides (Menzel, Braunschweig, Germany), dewaxed, and rehydrated. To detect influenza virus antigen, sections were incubated with a rabbit antinucleoprotein serum in a dilution of 1:500 in Tris-buffered saline (TBS, 0.1 M Tris-base, 0.9% NaCl, pH 7.6).5 A biotinylated goat anti-rabbit IgG1 (Vector, Burlingame, CA; diluted 1:200 in TBS) was used as linker-antibody for the avidin-biotin-complex (ABC) method. As negative controls, the preimmunization serum of the same rabbit was applied. A bright red signal was produced with an immunoperoxidase kit (Vectastain Elite ABC Kit, Vector) and the substrate 3-amino-9-ethylcarbazole (DAKO AEC substrate-chromogen system; Dako, Carpinteria, CA, USA). The sections were counterstained with Mayer's hematoxylin and sealed with aqueous medium (Aquatex; Merck, Darmstadt, Germany). Positive and negative control tissues of chicken, infected experimentally with H5N1 HPAIV, were included for each immunohistochemistry procedure.
Results
RRT-PCR and DNA sequencing
M-PCR and H5N1-specific RRT-PCRs with tracheal swabs and brain and lung samples from 12 mute and 6 whooper swans were positive. Partial nucleotide sequencing of the HA gene confirmed the presence of HPAIV in all animals by a typical deduced amino acid sequence (PQGERRRKKR∗GLF).
Gross pathology
In general, juvenile mute swans were in bad constitution, emaciated with severe muscle atrophy, and often dehydrated, whereas adult mute and whooper swans presented as well-nourished with sufficient body fat reserves. Conspicuously, most adult swans showed no external gross lesions. A small number of juvenile mute swans had subcutaneous edema of the head with swelling of the periorbital region. Few animals had diarrhea and showed green discolored feathers around the cloaca. At necropsy, the most consistent and predominant lesions in both the mute and the whooper swans were multifocal, sharply demarcated, and partly coalescent hemorrhage with necrosis in the pancreas (Fig. 1a). Characteristic but not present in all infected animals was a severe congestion of the lungs with pronounced alveolar and bronchiolar edema (Fig. 1b). Subepicardial and scattered intramyocardial hemorrhages or petechiation in adipose tissue were additional but rather inconsistent findings (Fig. 1b). In many cases, liver and spleen were moderately enlarged and congested.
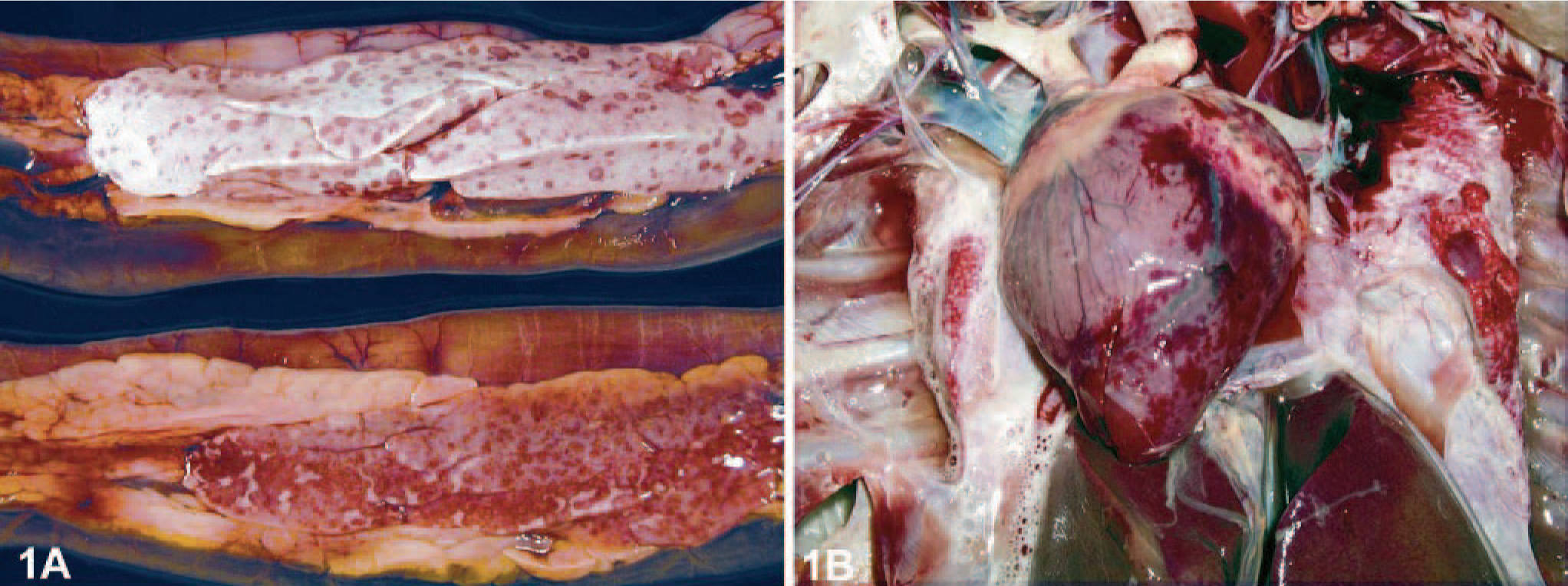
Pancreas; whooper swans. Multifocal to coalescent necrosis and hemorrhage.
Histopathology
Major histologic lesions and the incidence of viral antigen detection in the analyzed tissue samples are indicated in Table 1. The predominant lesions were found in the brain, pancreas, and liver. Almost all animals displayed multifocal and severe lymphoplasmacytic encephalitis in the cerebrum and severe necrosis and loss of Purkinje cells in the cerebellum (Fig. 2a). Within the neuropil there was vacuolation of the white and gray matter (edema), especially around Virchow-Robins spaces, which were expanded by marked cuffs composed of lymphocytes, histiocytes, and plasma cells (Fig. 2c). Randomly distributed foci of neuronal necrosis and neuronophagia with increased numbers of partly swollen glial cells were scattered throughout the neuropil. Focal hyperemia and hemorrhage were present inconsistently in the gray matter. Rarely, spinal cord ependymal cells were hypereosinophilic, necrotic, and sloughed. The liver showed randomly distributed and partly coalescent coagulative necrosis surrounded by various numbers of heterophils and lymphocytes (Fig. 3a). In the pancreas multifocal necrosis of the acini was present constantly (Fig. 3c). The inflammatory reaction was only mild and preferentially confined to the necrotic exocrine pancreatic acini but not affecting the islets of Langerhans. Adrenals displayed multifocal, well-circumscribed areas of cortical and medullary cell necrosis that were infiltrated by few heterophils (Fig. 3e). Sections of the spleen and Peyer's patches showed multifocal, mild lymphocyte necrosis with replacement by cellular debris and fibrin (Fig. 4a, b). In the lungs there was marked congestion, edema, and hemorrhage with slight endothelial cell swelling. Scattered throughout the myocardium was mild hemorrhage. Nasal mucosa, trachea, gastrointestinal tract, cloaca, bone marrow, kidneys, gonads, thyroids, and skin had no lesions.
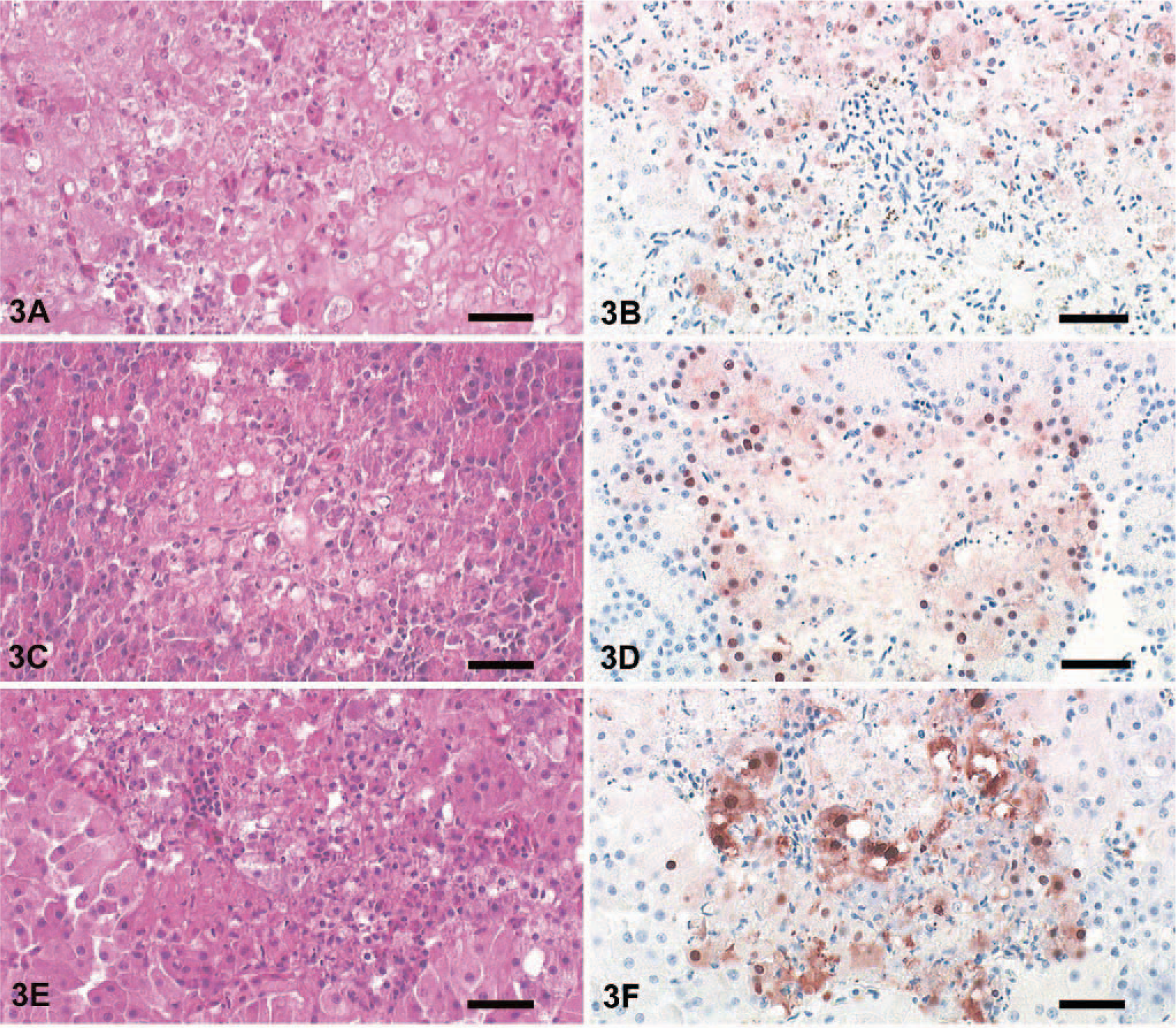
Liver; mute swan. Foci of hepatic necrosis with loss of cellular detail, cytoplasmic hypereosinophilia and replacement of hepatic cords by cellular debris. HE. Bar = 70μm.
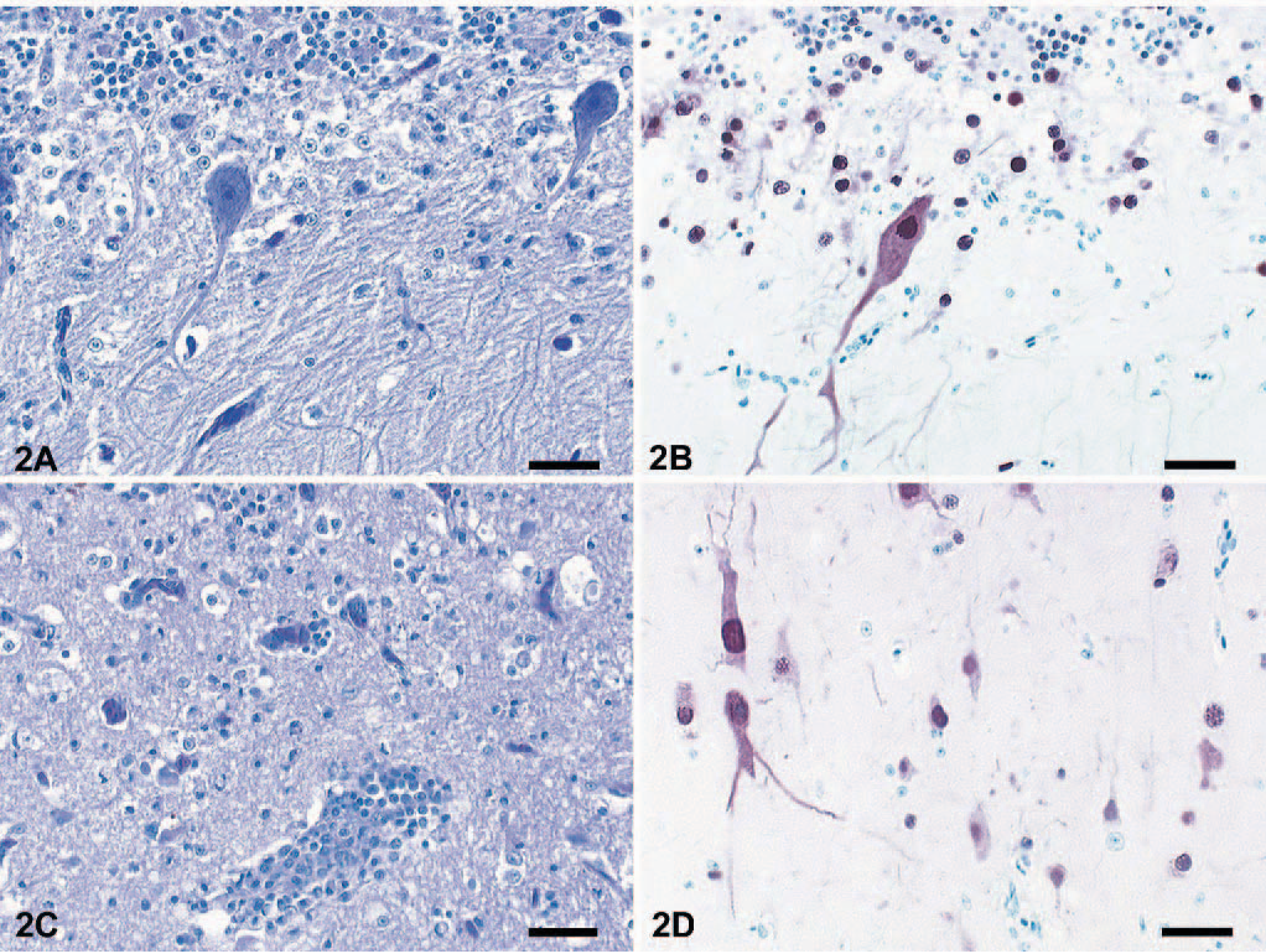
Cerebellum; mute swan. Paucity of Purkinje cells and mild neuropil edema. HE. Bar = 70μm.
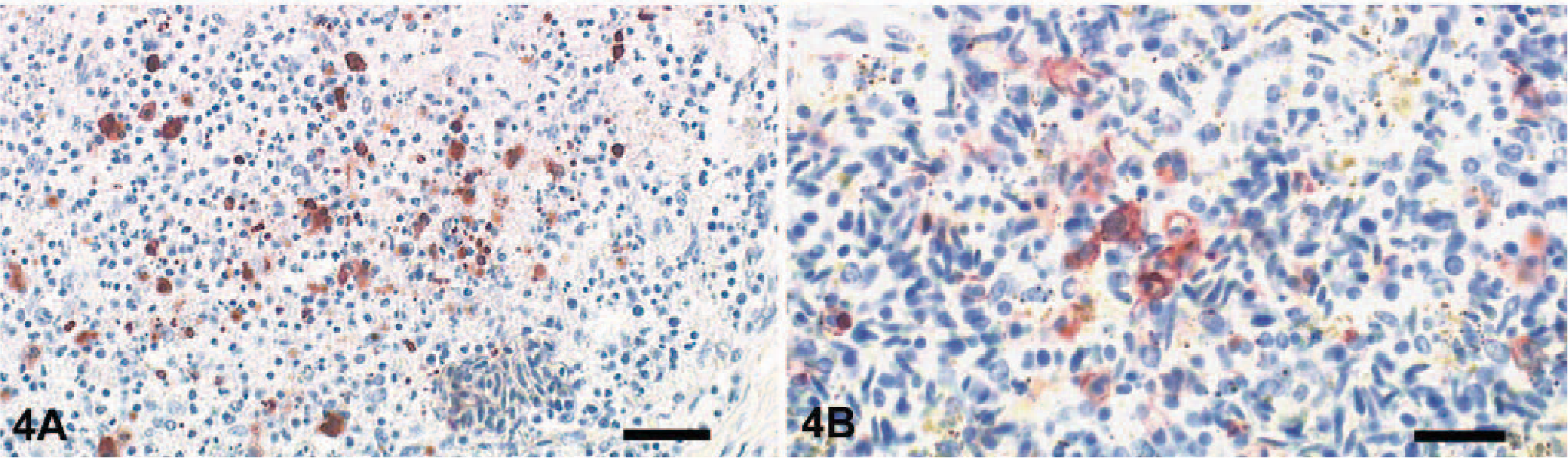
Peyer's patch; mute swan. Focal necrosis of mononuclear cells associated with detection of AIV nucleoprotein. ABC method with hematoxylin counterstain. Bar = 100μm.
Immunohistochemistry
Immunohistochemical demonstration of influenza virus antigen in pancreas, adrenals, liver, and brain was strongly consistent with histologic lesions (Table 1). In the cerebrum a large number of viable and degenerating neurons and glial cells showed strong intranuclear and intracytoplasmic staining for NP antigen. Purkinje cells and cells of the granular layer of the cerebellum also reacted positively for viral antigen (Fig. 2b, d). Furthermore, ependymal cells lining the central canal of the spinal cord were strongly positive but only in a few animals. Areas of coagulative necrosis in the liver, pancreas, and adrenals were surrounded by rims of numerous antigen-positive cells (Fig. 3b, d, f). Within spleen, bone marrow, and Peyer's patches, few mononuclear cells, arteriolar endothelium, and rare lymphocytes were infected (Fig. 4a, b). In the lungs, NP antigen could be identified in a few capillary and arteriolar endothelial cells. Individual animals displayed antigen-positive cells in the heart (cardiomyocytes), kidney (tubular epithelium), proventriculus (mucosa), thyroid (follicular epithelium), and trachea (mucosal epithelium).
Discussion
High mortality in wild aquatic birds due to infection with HPAIV has been a rare event and was restricted to only a few individual birds adjacent to holdings where poultry infected with HPAIV was kept.1,2 Swans, belonging to the order Anseriformes, are assumed to play a role like ducks and geese as a natural, asymptomatically infected reservoir for influenza viruses and had, so far, not been afflicted by HPAIV.4,8 However, during the recent outbreaks in different regions of Europe, swans were the most affected species.11 So far, there are only very limited data on influenza virus pathology in this species in the literature.
During the recent outbreak in Germany, the most reliable gross lesions were multifocal pancreatic necrosis and hemorrhage and lung edema. Severe additional histologic changes were noted consistently in the brain and liver and were in close relation with the results of virologic investigation. These lesions were peracute and, except for the central nervous system, associated with mild inflammatory reactions. Due to the massive infection of cerebrum, cerebellum, and brainstem, a pronounced neurotropism of H5N1 HPAIV may have led to nervous disturbances with loss of orientation and final bulbar paralysis. However, the circulatory failure, lung edema, and rapid death of these animals might also have been caused by an intense cytokine storm as result of systemic infection.
In contrast to chicken and other gallinaceous species, passerine birds, and budgerigars, we were not able to detect influenza virus antigen within pneumocytes.5,7,9 Few endothelial cells of the lung showed immunoreactivity for NP. The fact that virological investigations by means of quantitative RRT-PCR of the lungs revealed a high viral load in this tissue can be explained by the severe pulmonary congestion during the viremic spread of HPAIV.
The distribution and character of lesions in the juvenile and adult mute and whooper swans suggest an acute course of disease. The lethal outcome is attributed to the systemic viral infection. No concurrent diseases have been identified in the animals. Nevertheless, it is clear that not all swans found dead during February on the Island of Ruegen were victims of the virus infections. Many of them, especially emaciated juveniles, simply starved to death, which is not unusual at this time of the year. When the first positive swans were found on the Island of Ruegen, arctic cold spells had led to a situation in which large parts of the shallows and inlets were frozen and the land was covered with snow. In places where strong tidal currents hampered the formation of sea ice, large concentrations of water birds of different species (swans, geese, and diving ducks) were observed. However, significant mortality attributable to HPAIV infection has only been noticed in swans and Canada geese in this area. A bias in this observation can not be excluded because carcasses of swans and larger goose species are more likely to float and be recognized compared with smaller diving ducks. In fact, a substantial number of duck carcasses were found on ice further off the shores. However, these carcasses were impossible to retrieve for analysis. Still, there seems to be a predisposition of swans to a fulminant course of HPAIV H5N1−associated disease. This would render these species an interesting indicator animal suitable in early warning systems signaling the presence of HPAIV H5N1 of Asian lineage in wildlife. Yet many questions concerning the pathogenesis remain and should be addressed in experimental infection studies before recommending mute swans as indicators.
Footnotes
Acknowledgements
We are grateful for the excellent technical assistance provided by G. Czerwinski and C. Illing. This work is dedicated to Dr. Ortrud Werner, an outstanding veterinary virologist and committed diagnostician in the field of aviary diseases.
