Abstract
Hermaphroditism was diagnosed in three, 6-month-old, male, chimeric mice generated by microinjection of 129/Ola XY recombinant embryonic stem cells into unsexed C57BL/6 blastocysts. Grossly, mice Nos. 1 and 2 had perigenital masses and hydrometra. All mice had unilateral ovaries and cystic endometrial hyperplasia. Mice Nos. 1 and 3 also had contralateral testes and epididymides. Histologically, mice Nos. 1 and 3 were true hermaphrodites with unilateral ovotestes, while mouse No. 2 was a pseudohermaphrodite with ovarian tissue only. The presence of a uterus with cystic endometrial hyperplasia in these mice resembles XY pseudohermaphroditism in miniature schnauzers. The mice were determined to be 95 to 100% chimeric via haircoat color; however, the presence of both male and female sex organs in these phenotypically male mice suggests otherwise. Published reports note incidences for sex chimeras and hermaphroditism in genetically engineered mice of 50% and 20%, respectively. Hermaphroditism is expected to increase as the numbers of chimeric mice rise with technical advances in genetic engineering.
The population of genetically engineered mice has rapidly increased in research facilities because of decoding of the human and mouse genomes as well as advances in gene insertion and gene “knock-out” technologies. In spite of such progress, the production of transgenic and knock-out or knock-in mice has inherent difficulties, including gene copy number, insertion site, silencing, and germline transmission. The use of embryonic stem (ES) cells to generate knock-out or knock-in mice is efficient because many cells can be altered in a single electroporation event, and recombinant cells can be selected in culture. Chimeric mice are the result of ES cell and blastocyst amalgams. Hermaphroditism can be found in chimeric mice, leading to difficulties in germline transmission of the modified gene(s) of interest.8,11,12 Although a widely accepted phenomena, this finding is uncommonly reported in the current literature: hermaphroditism has been previously reported in up to 20% of chimeric mice, and sex chimerism in up to 50% of mice.12 We present a series of 3 hermaphroditic mice from our gene-targeting facility, and short synopses of chimeric mouse creation and hermaphroditism in the veterinary literature.
Three, 6-month-old, male mice, which were conditional knock-outs for a 3 amino acid loop extension class homeodomain protein, Pbx1, were presented for necropsy owing to the presence of perigenital masses that increased in size over a 2-week period. No treatments had been administered, and differential diagnoses included teratoma, inguinal hernia, and orchitis. The mice were generated by microinjection of 129/Ola XY homologous recombinant embryonic stem cells into unsexed C57BL/6 blastocysts. The animals were determined to be 95 to 100% chimeric via tail DNA analysis and visual assessment of a mixed coat color. At necropsy, large fluid-filled masses extending from the caudal aspect of the right kidney were noted in mice Nos. 1 and 2. These masses, which measured 4.5 × 1.7 × 1.8 cm and 4.0 × 1.9 × 1.7 cm, respectively, were thin-walled and contained abundant pale-yellow, slightly opaque fluid. Attached along the medial aspect of the surface of the mass in both mice was a small amount of adipose tissue with a 2-mm, dark-yellow, firm nodule interpreted to be an ovary. The penis with prepuce, hypoplastic seminal vesicles, and urinary bladder were identified on gross examination of both mice. In addition, a presumptive left testis and epididymis were noted in mouse No. 1 (Fig. 1). Upon gross evaluation of mouse No. 3, no perigenital mass was evident. Urogenital structures noted grossly in mouse No. 3 included the penis with prepuce, right uterine horn and ovary, left testis with epididymis, seminal vesicles of normal size, and a urinary bladder. Representative tissues from all mice were fixed in 10% neutral-buffered formalin, routinely processed, and embedded in paraffin. Sections (5 μm) were stained with hematoxylin and eosin (HE) and examined by light microscopy.
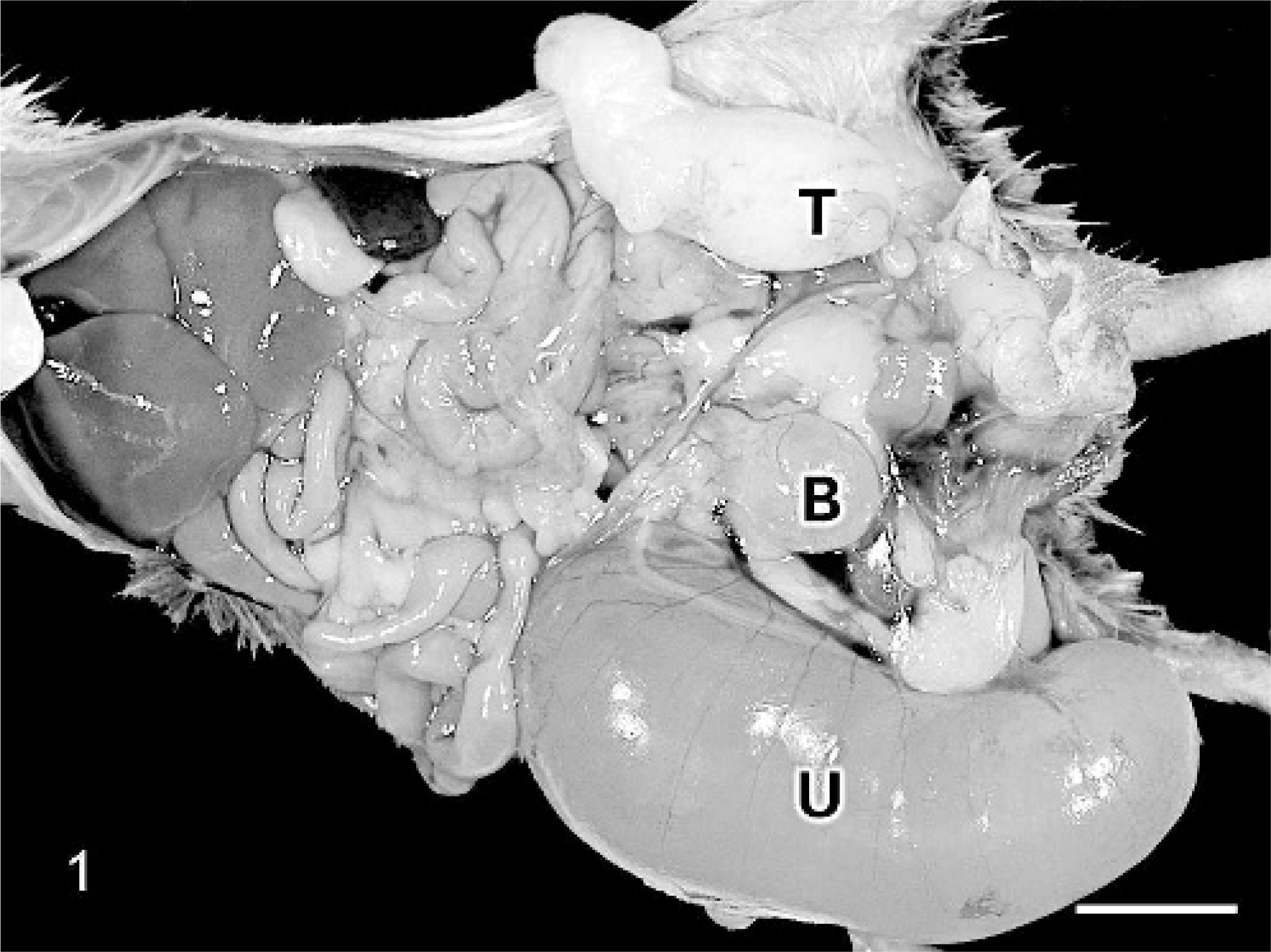
Caudal abdomen; C57BL/6 chimeric mouse No. 1. A large, fluid-filled, cystic uterine horn (U) caudal to the right kidney and contralateral testis (T) with epididymis are present. Urinary bladder (B). Bar = 1 cm.
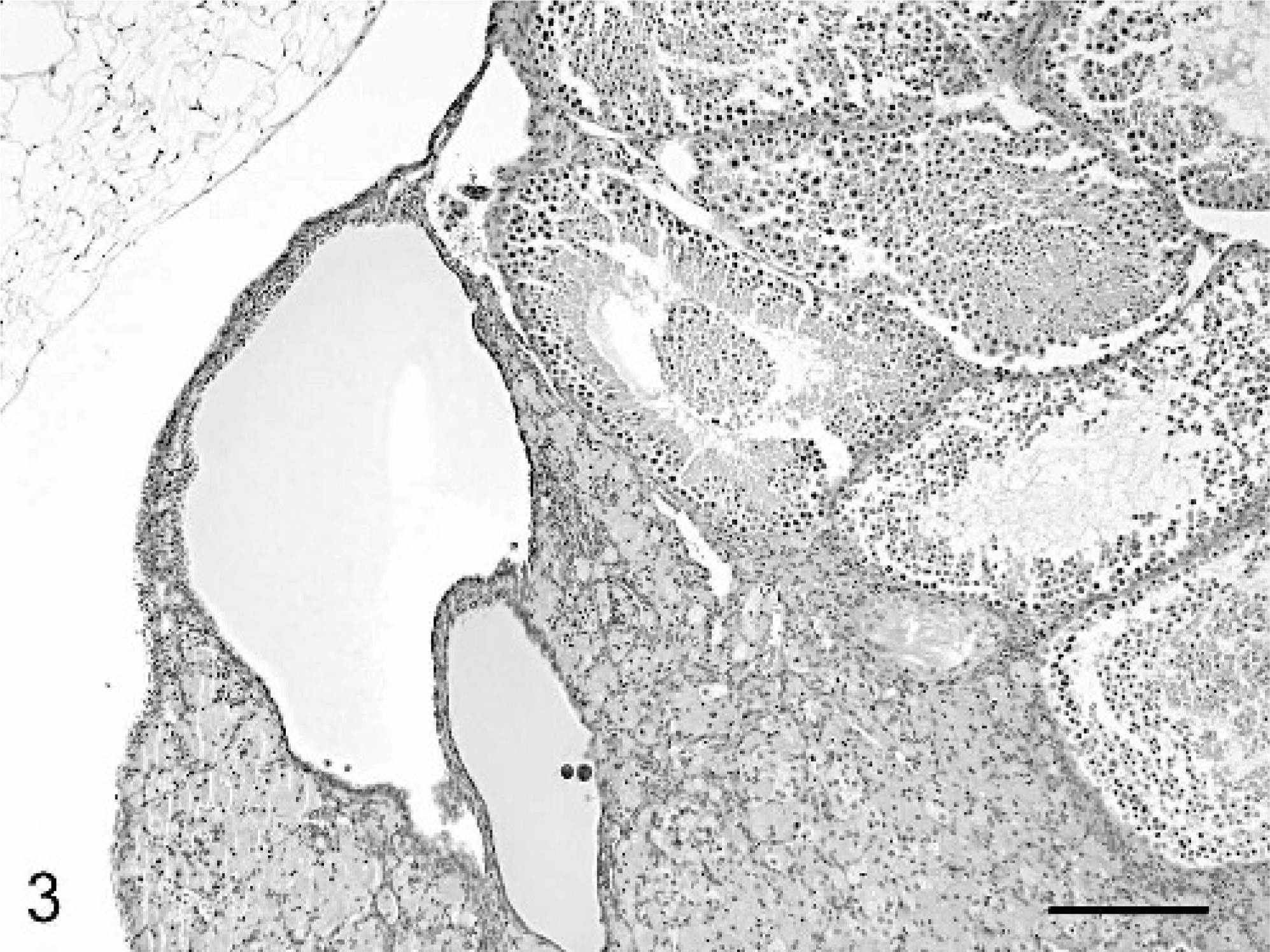
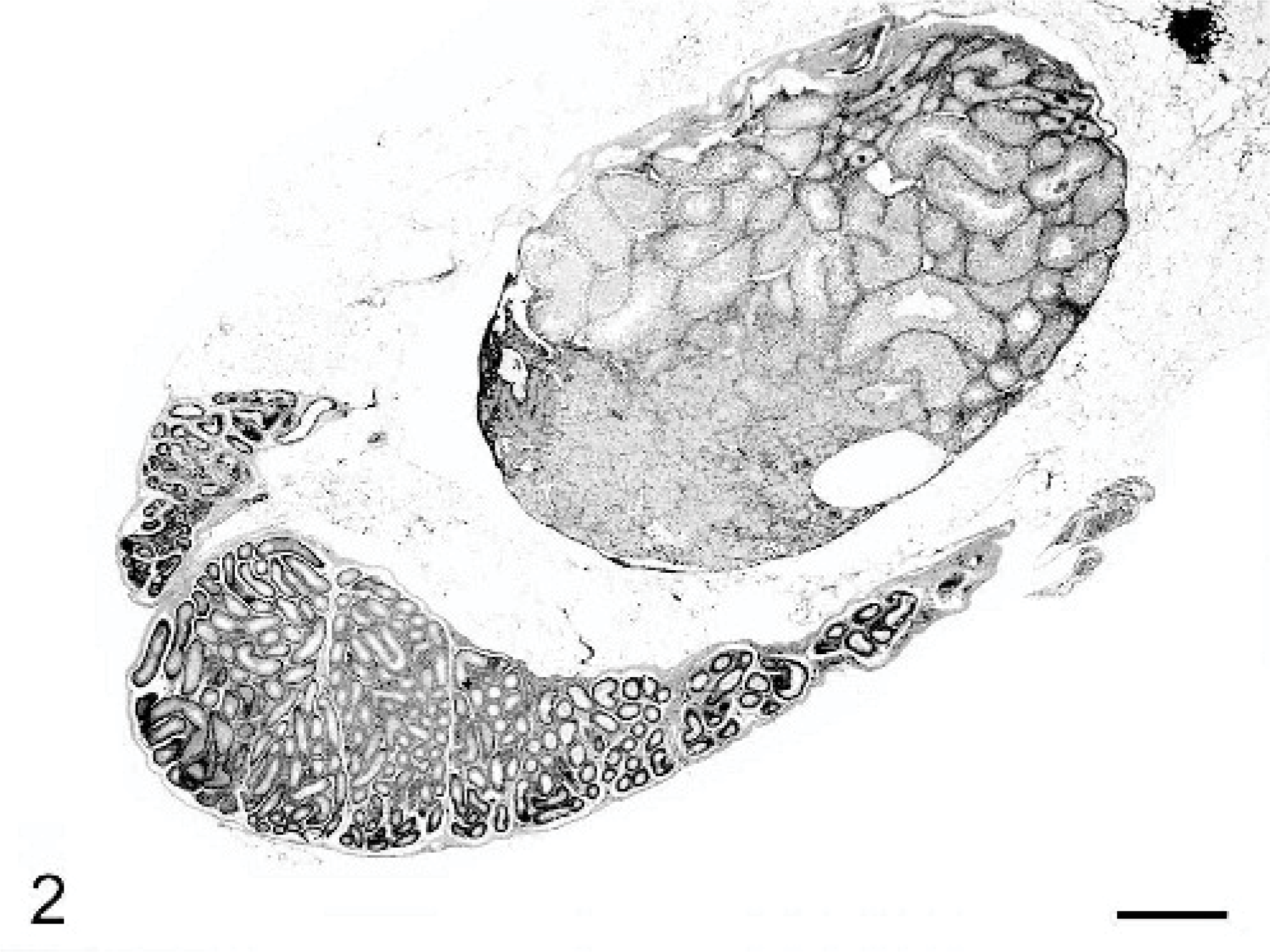
Histologically, an ovary with numerous follicles in various stages, as well as corpora lutea and an attached epididymis were present in mice Nos. 1 and 2. Mouse No. 3 had an atrophic ovary with attached epididymis. Unilateral ovotestes with attached epididymides were noted in mice Nos. 1 and 3 (Figs. 2, 3). The fluid-filled masses in mice Nos. 1 and 2 corresponded histologically to cystic uterine horns with hydrometra. There was cystic endometrial hyperplasia in mouse No. 3, which had not yet progressed to macroscopically evident cystic dilation. Male accessory sex glands including seminal vesicles, coagulating glands, prostate glands, and bulbourethral glands were identified histologically in all 3 mice. Mice Nos. 1 and 3 were diagnosed as true hermaphrodites due to the concurrent presence of ovarian and testicular tissue. Although male external genitalia and internal accessory sex glands were found in mouse No. 2, only ovarian gonadal tissue was identified, consistent with pseudohermaphroditism.
Ovotestis; C57BL/6 chimeric mouse No. 1. Histologically, the ovotestis is partially lined by simple low cuboidal epithelial cells and contains seminiferous tubules as well as large numbers of interstitial cells with golden brown to yellow pigment and few degenerative follicles as in an atrophic ovary. HE stain. Bar = 200 μm.
Ovotestis and epididymis; C57BL/6 chimeric mouse No. 1. Ovarian tissue is evident at low magnification within the distal third of the testis. HE stain. Bar = 600 μm.
The term intersex, in which there is a discordance between genetic, gonadal, and/or phenotypic sex, has been reported in goats, swine, horses, cattle, mice, rats, and dogs, and includes both true and pseudohermaphroditism.3 True hermaphrodites are defined as individuals with both ovarian and testicular tissue in the form of separate organs or a combined ovotestis.1 Pseudohermaphrodites have gonads of one sex and external genitalia of the opposite sex and are classified as male or female depending on the type of gonad present. Sex chimerism indicates the presence of both XX and XY cells in the same individual. Sex chimeras frequently lack detectable anatomic abnormalities and may be fertile.9 The 3 mice reported herein are true and pseudohermaphrodites derived from blastocysts injected with genetically modified ES cells.
Although spontaneous occurrences of hermaphroditism within a mouse breeding colony have previously been identified, current reports associate chimeras and hermaphroditism.8,11,14 Initial reports of hermaphroditism in chimeric mice were published in the 1960s as researchers fused pre-implantation embryos from mothers of different strains and colors. The resulting mice had a variety of coat colors, ranging from solid to multicolored. The multicolored subpopulations had increased male ° female ratios, with occasional intersex characteristics (hermaphrodites). It was expected that half of the fused embryos would yield sex chimeras due to the inability to identify blastocysts as XX or XY.6
A common method of generating chimeric mice includes either aggregation of, or microinjection of genetically modified ES cells into a blastocyst. ES cells maintained in culture are typically genotyped as XY. These cells are propagated under stringent conditions and electroporated with DNA constructs designed for the gene(s) of interest. The cells are briefly cultured with various antibiotics to positively select for cells containing the appropriate DNA introduced via homologous recombination and eliminate unmodified or incorrectly modified cells. The recombinant cells are then introduced into unsexed blastocysts, obtained from superovulated wild-type female mice, and implanted into pseudopregnant recipient mice. Chimeric mice are typically identified through the use of coat color. Stem cells derived from a mouse with a contrasting coat color than the blastocysts will yield chimeric mice with a mixture of the 2 coat colors. It is presumed that chimeras with the greatest proportion of coat color (95–100% preferred) derived from the ES cells are more likely to carry the gene mutation in the germline.6 Chimeric mice are raised to sexual maturity and mated to propagate the line of genetically modified mice. The pups (F1) of the chimeric mouse are genotyped to verify that the modified gene was passed via germline transmission and are, in turn, bred to maintain the line of genetically engineered mice. Hermaphrodite or pseudohermaphrodite chimeric mice may develop when XY ES cells are introduced into an XX blastocyst, and both cell types have significant contributions to the gonads of the resulting embryo.8 In the cases presented, it appears that XX blastocysts were recipients of the 129/Ola XY ES cells. The presence of ovotestes and uteri indicates that XX blastocyst cells were able to differentiate in the developing gonad adjacent to introduced XY ES cells. When ES cells have a low contribution to the gonad, either a female, hermaphrodite, or pseudohermaphrodite chimeric mouse may develop. If the ES cells have a high contribution to the developing gonad, the result will be fertile males that carry the modified gene in their germline.
A specific type of spontaneous pseudohermaphroditism known as XX sex reversal has been described in mice.7,13 The “sex-reversed” (Sxr) gene is located on an abnormal Y chromosome and contains a duplicated Sry gene encoding for testis-determining factor. During male meiosis, 1 copy of the Sry gene is translocated to the male's own X chromosome or pseudoautosomal region of the Y chromosome long arm resulting in sperm that may carry the XSxr or YSxr genotype. Progeny from XSxr sperm will be males with the XXSxr genotype and may have transient ovotestes during embryogenesis. Mice with the genotype T16H/XSxr have been characterized as having inactivated Sxr sequences in some cells of the gonadal primordium, leading to hermaphroditism. These hermaphroditic mice are described as having a predominantly male phenotype, although “female” hermaphrodites with ambiguous genitalia were also described. Masculinization of the external genitalia was explained by the influence of androgens produced by the fetal testis. Phenotypic males were determined to have a left gonad that was significantly more masculine than the right. Although the cases reported here were not of the XXSxr or T16H/XSxr genotype, it is interesting to note that we also report male phenotypes with more masculine characteristics found on the left of midline.
In domestic animal species, true hermaphroditism is most common in swine and goats, while various forms of pseudohermaphroditism are described in mice, pigs, goats, and dogs.5 Male pseudohermaphroditism is common in goats in which it is also associated with polledness.2 Affected XX goats have testes and masculinization of external genitalia including clitoromegaly. Sry negative sex reversal most often occurs in American Cocker Spaniels and has been reported in other purebred dogs.10 XY gonadal dysgenesis, also known as androgen insensitivity or testicular feminization syndrome, is a form of male pseudohermaphroditism described in cats, mice, rats, cattle, and horses. Affected individuals have XY genotypes, normal or elevated testosterone production, and female external genitalia. In contrast, XY Miniature Schnauzers with this syndrome have hypoplastic male external genitalia, cryptorchid testes prone to sertoli cell tumor development with epididymides, and a hypoplastic uterus.4 Endometrial hyperplasia and hydrometra/mucometra often develop in the residual uterus. It is interesting to note that all 3 of the male mice described herein had uteri with cystic endometrial hyperplasia ± hydrometra and normal to hypoplastic male accessory sex glands.
Despite the paucity of published reports in genetically engineered mice, hermaphroditism is a widely accepted phenomena associated with genetic engineering. It is anticipated that documentation of these intersex mice will assist laboratory animal veterinarians and veterinary pathologists in further characterizing this condition, the incidence of which is expected to increase as the numbers of chimeric mice rise with technical advances in genetic engineering.
Footnotes
Acknowledgements
The authors wish to thank Dr. Willie Mark for valuable discussion regarding transgenic technologies and Mr. Sunny Yeung for assistance with photography.
