Abstract
Nongestational ovarian choriocarcinoma (NGCO) is a tumor of germ cell origin seldom described in nonhuman species. Few spontaneous cases are reported in macaques and mice, with the B6C3F1 strain overrepresented. This report describes 2 cases of ovarian choriocarcinoma in nulliparous female mice with conditional loss of Trp53 under the Tie2 promoter. The mouse line was maintained on a mixed genetic background including Crl: CD1(ICR) and 129X1/SvJ strains. In both cases, affected ovary was partially replaced by blood-filled lacunae lined by neoplastic trophoblast-like giant cells. Immunohistochemically, neoplastic cells expressed folate-binding protein and prolactin and were invariably negative for p53. To the authors’ knowledge, this is the first report characterizing this entity in a genetically engineered mouse (GEM) line. Considering that germ cells (the cell population from which NGCO originates) constitutively express Tie2 receptor, it can be speculated that Tie2-driven deletion of Trp53 may have played a role in the development of these tumors.
Choriocarcinoma is a rare highly malignant hormonally functional tumor. This entity currently encompasses 3 distinct neoplastic conditions: (1) gestational choriocarcinoma of trophoblastic origin primarily located in the uterus and arising after a gestational event, (2) gonadal or extragonadal (usually midline locations) nongestational choriocarcinoma of germ cell origin, and (3) nongestational choriocarcinoma arising in the context of a poorly differentiated somatic carcinoma. 4
Nongestational choriocarcinoma of the ovary (NGCO) is an extremely rare tumor that accounts for less than 1% of all human ovarian malignancies. 4 NGCO most commonly affects children and women younger than 20 years of age. 4 NGCO frequently coexists with other neoplasms of germ cell origin such as seminoma, embryonal carcinoma, yolk sac tumor, and teratoma in the same or contralateral ovary, with the pure form being exceedingly rare. 4 As seen also for the other gestational and nongestational variants of choriocarcinoma, the development of NGCO is typically accompanied by high levels of human chorionic gonadotropin (hCG) in serum and urine. The distinction between NGCO and the gestational counterpart is warranted because of the worse prognosis of NGCO, mainly due to its metastatic potential to brain and lungs. 4
Primary ovarian choriocarcinomas are also extremely rare in laboratory animals. Surveys of ovarian tumors in rats have failed to reveal any cases. 2 Primary unilateral ovarian choriocarcinomas arising spontaneously have been reported only twice as incidental findings in nonhuman primates. 7 Reports of ovarian choriocarcinomas in mice are exceedingly rare, with B6C3F1 strain overrepresented and just 1 study mentioning (but not further characterizing) a single case in genetically engineered mice (GEM) carrying a targeted null mutation of both Hic1 and Trp53 on a 129S4/SvJae and C57BL/6 mixed genetic background. 1,3,8,12,14
The objective of this study is to characterize the distinctive pathological and immunohistological features of this rare neoplasm in 2 mice not related to the B6C3F1 strain and genetically engineered to carry both Trp53 alleles conditionally deleted under the control of Tie2 promoter.
Both mice included in this study were 6-month-old nulliparous females genetically engineered to have both Trp53 (p53) alleles deleted in Tie2 expressing cells. This specific mouse line is maintained on mixed genetic background primarily including Crl: CD1(ICR) and 129X1/SvJ strains and it has been recently described as valuable model for studying spontaneous hemangiosarcoma development. 10 A complete necropsy was performed on each mouse and representative tissues/organs were obtained for histopathological examination. Samples were fixed in 10% buffered formalin, paraffin-embedded, sectioned at 4 µm and stained with hematoxylin and eosin. Immunohistochemical analyses and periodic acid Schiff (PAS) stain were also performed to better characterize origin and pathogenesis of the ovarian lesions. Details concerning the panel of immunohistochemical stains applied are reported in Table 1. Sections from normal E14.5 murine placenta were included in the immunohistochemical study as comparison.
Details Concerning Primary Antibodies and Procedures Used for the Immunohistochemical Examination of NGCOs Reported in This Study.
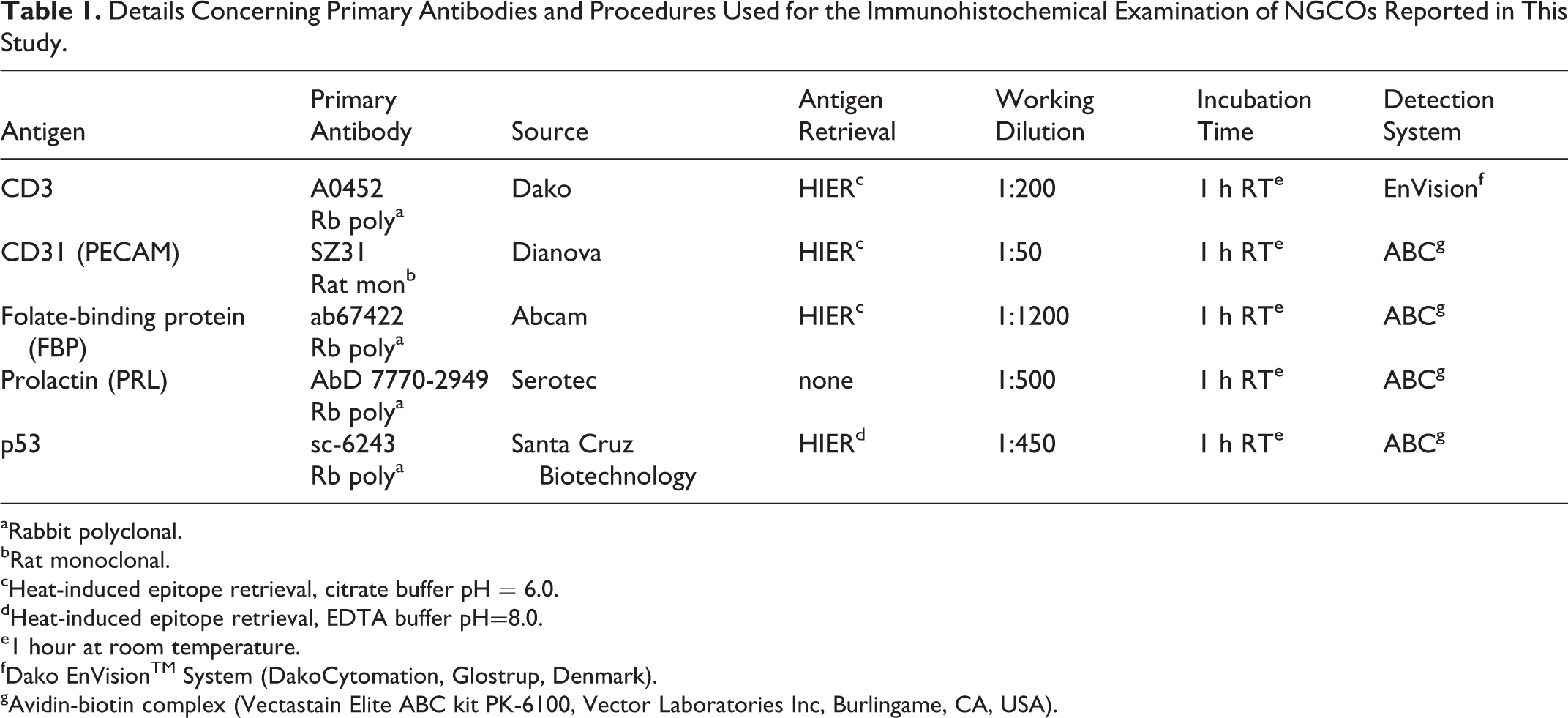
aRabbit polyclonal. bRat monoclonal. cHeat-induced epitope retrieval, citrate buffer pH = 6.0. dHeat-induced epitope retrieval, EDTA buffer pH=8.0. e1 hour at room temperature. fDako EnVisionTM System (DakoCytomation, Glostrup, Denmark). gAvidin-biotin complex (Vectastain Elite ABC kit PK-6100, Vector Laboratories Inc, Burlingame, CA, USA).
Procedures involving animals and their care were conformed to institutional guidelines in compliance with national (D.L.vo 116/92 and following additions) and international (EEC Council Directive 86/609, OJ L 358, 1, 12-12-1987; NIH Guide for the Care and Use of Laboratory Animals, U.S. National Research Council 1996) laws and policies.
At necropsy, a unilateral ovarian enlargement with red discoloration was observed in both mice. Microscopically, in both cases up to 40% of the ovarian parenchyma was effaced by a well-demarcated, partially encapsulated, loosely cellular, hemorrhagic, pseudocystic mass, bulging from the organ’s surface, compressing adjacent parenchyma, and focally invading ovarian bursa (Fig. 1). The neoplasm was primarily composed of large polygonal cells up to 200 µm in diameter with variably distinct cell borders, low nuclear to cytoplasmic ratio and abundant bright eosinophilic cytoplasm (Fig. 2), often containing PAS-positive droplets (Fig. 3). These cells had round to multilobed paracentral nuclei up to 100 µm in diameter with vesicular to coarsely granular chromatin and 1-2 evident magenta nucleoli (trophoblast-like giant cells). Evident findings of cytologic atypia were observed, including marked anisocytosis and anisokaryosis, multiple irregular nucleoli, multinucleation, bizarre mitotic figures, and erythrophagocytosis. In the peripheral portion of the tumor, trophoblast-like giant cells were interspersed with occasional round to polygonal cells measuring 60 to 80 µm in diameter and characterized by distinct cell borders, high nuclear to cytoplasmic ratio, scant amount of pale eosinophilic homogeneous cytoplasm, and single round central nucleus with finely stippled chromatin and no evident nucleoli (cytotrophoblast-like cells) (Fig. 2). Cytotrophoblast-like cells exhibited less prominent findings of atypia compared to trophoblastic-like giant cells. Intralesional areas of hemorrhages and necrosis, scattered apoptotic cells and multifocally large laminated fibrinocellular thrombi were also present. The mass was infiltrated at the periphery by moderate numbers of both viable and necrotic (karyorrhectic) neutrophils. Morphologically the tumor was consistent with a diagnosis of primary ovarian choriocarcinoma with a prevalent trophoblastic giant cell component. Based on the fact that the affected females were nulliparous, both neoplasms were classified as NGCO.
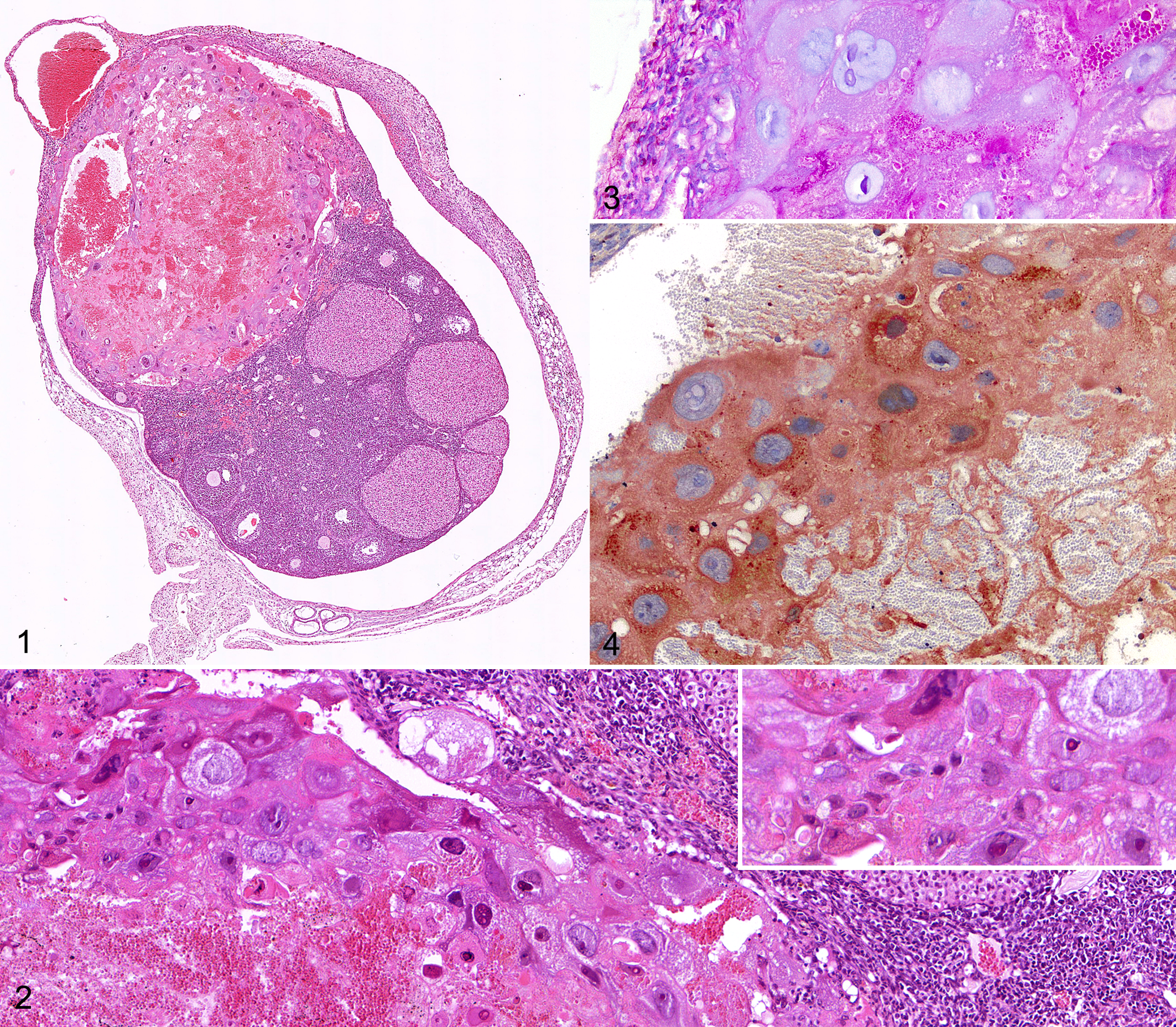
Nongestational ovarian choriocarcinoma, ovary, female mouse with conditional loss of Trp53 driven by the Tie2 promoter.
Immunohistochemically, atypical trophoblastic-like giant cells from both the ovarian neoplasms were diffusely positive for folate-binding protein (FBP) (Fig. 4). Tumor cells also displayed variable degree of prolactin (PRL) immunoreactivity, but they were invariably negative for p53. CD31 immunohistochemistry failed to detect endothelial cells lining intratumoral blood-filled lacunae. As NGCO typically displays a complex trophoblastic differentiation, normal E14.5 murine placenta was included as reference (control) tissue for immunohistochemistry. Trophoblastic giant cells in the normal placental tissue were immunohistochemically positive for FBP and PRL and had scattered nuclei positive for p53. The immunohistochemical profile of NGCOs and normal E14.5 murine placenta are summarized in Table 2.
Summary of the Immunohistochemical Phenotype Displayed by the Nongestational Ovarian Choriocarcinomas (NGCOs) Reported in This Study and Normal E14.5 Murine Placenta Included as Comparison.

In addition, both mice affected by NGCO had concomitant occurrence of other tumor phenotypes. One mouse had a multicentric T cell lymphoma/leukemia with infiltrates of CD3-positive neoplastic lymphoid cells invading the ovary concomitantly developing the NGCO, whereas the other mouse had a solitary hemangiosarcoma originating from the soft tissues of the thoracic wall and a mediastinal lymphoma, likely of thymic origin.
To the authors’ knowledge, this is the first report describing in detail the immunohistological features of NGCOs in a GEM line.
Both NGCOs here examined were composed of 2 cell populations, with large prevalence of trophoblastic giant cells, admixed with lesser numbers of cytotrophoblast cells. Although NGCOs affecting nonhuman species are usually composed of 3 types of cells (cytotrophoblasts, multinucleated syncytiotrophoblasts, and trophoblastic giant cells), neoplasms with 1 (ie, trophoblastic giant cells) or 2 (ie, cytotrophoblasts and syncytiotrophoblasts) of the 3 aforementioned cell types have already been reported in mice and nonhuman primates, respectively. 1,7 In contrast to that reported for previous descriptions of NGCO in B6C3F1 mice where PAS-positive material was mainly observed in the cytoplasm of syncytiotrophoblasts, 1 in the cases intracytoplasmic PAS-positive droplets were consistently detected within trophoblastic giant cells.
Examined cases did not coexist with other neoplasms of germ cell origin in either ovary, suggesting NGCOs here described are pure forms, known to be exceedingly rare in human beings. 4 The concurrent presence of other tumors of germ cell origin has been reported in nonhuman primates 7 and it has recently been described in the testes of a Swiss albino mouse, 11 but there is no information so far regarding murine ovarian choriocarcinomas. Furthermore, no metastases were observed in any of the examined cases, although metastatic dissemination to multiple organs has been described in NGCOs affecting nonhuman primates 7 and humans. 4
To confirm the diagnosis of ovarian choriocarcinoma, human chorionic gonadotropic (hCG) immunohistochemistry was performed, but the antibody failed to work properly on formalin-fixed paraffin-embedded murine tissues. Thus, as a second option, we applied FBP and PRL, markers known to be constitutively expressed in normal placental tissues and in choriocarcinomas from both humans and rodents. 9,13,16 NGCO immunophenotype was further characterized by lack of CD31 expression, thus confirming that blood filled lacunae within the mass were lined by neoplastic trophoblastic giant cells rather than by endothelial cells. This peculiar feature, known as “vasculogenic mimicry,” has been described in choriocarcinomas. 15
It was unclear whether or not the tumors here described resulted from the specific genetic manipulation applied to the mice or should be rather regarded as incidental lesions. Considering that primordial murine germ cells, which are the specific cellular population from which NGCO originates, constitutively express Tie2, 6 it is possible that Tie2-driven deletion of Trp53 played a role in the development of the NGCOs here reported. Further supporting this hypothesis, deletion of p53 has been already associated with the development of murine choriocarcinomas of putative germ cell origin 3,5 and loss of function mutation of p53 is frequently found in human choriocarcinomas. 4 In this context, complete lack of p53 expression observed in the NGCOs here described may suggest that Tie2-driven deletion of Trp53 has actually occurred in neoplastic cells and may therefore have played a role in tumor development. The demonstration of Tie2 expression in the 2 NGCOs reported in the present work would have further supported the role of conditional Trp53 deletion in tumor development.
In conclusion, (1) there is strong evidence that the uncommon ovarian tumors described in this study are nongestational choriocarcinomas; (2) examined tumors arose in a complex pathological scenario, but neither concurrent nor contralateral gonadal neoplasms or metastatic dissemination were observed; (3) the lack of p53 expression by the tumor cells is highly suggestive that tumor development was related to the genotype of these mice.
Footnotes
Acknowledgements
We gratefully acknowledge Cory Brayton, Phenotyping Core, Molecular and Comparative Pathobiology, Johns Hopkins University, Baltimore, Maryland, USA, for her invaluable advice and supervision; and Robert Cardiff, Neil E. Hubbard, and Pan Soonsawad, Center for Comparative Medicine, University of California, Davis, USA, for their assistance with immunohistological procedures. The manuscript was prepared according to the Uniform Requirements format.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
