Abstract
Incisional iris biopsy was performed for diagnosis of an unusual opaque white mass protruding from the right ventrolateral iris of a 10-year-old neutered male Great Dane dog. Histopathology revealed a diagnosis of bone formation within otherwise normal iris tissue. No underlying etiology was identified. Osseous metaplasia or heterotopic bone formation may be an additional differential diagnosis for a nonneoplastic mass in the eye of a dog.
A 10-year-old, 55-kg, neutered male Great Dane was presented for evaluation of the right eye because of discomfort and color change. The dog was under treatment with oral antibiotics for resolving pneumonia and had a well documented 3-year history of a right iridal mass. Examination of the right eye revealed mild blepharospasm, bulbar conjunctival hyperemia, serous discharge, and third eyelid elevation. The right eye was clinically blind, as it lacked a menace response, exhibited only an equivocal dazzle reflex, and had minimal direct and indirect pupillary light responses. Slit-lamp biomicroscopy of the right eye revealed multiple focal dense accumulations of pigment on the ventrolateral corneal endothelium, mild ventrolateral corneal edema, and moderate aqueous flare with red blood cells and a small fibrin clot. Two well demarcated, raised, grey-white, 3-mm-wide masses were identified extending in arcuate fashion 4 mm and 10 mm, respectively, over the regions of the lateral and ventrolateral right iris collarette (Fig. 1). A focal dorsolateral left chorioretinal scar was identified by binocular indirect ophthalmoscopy.
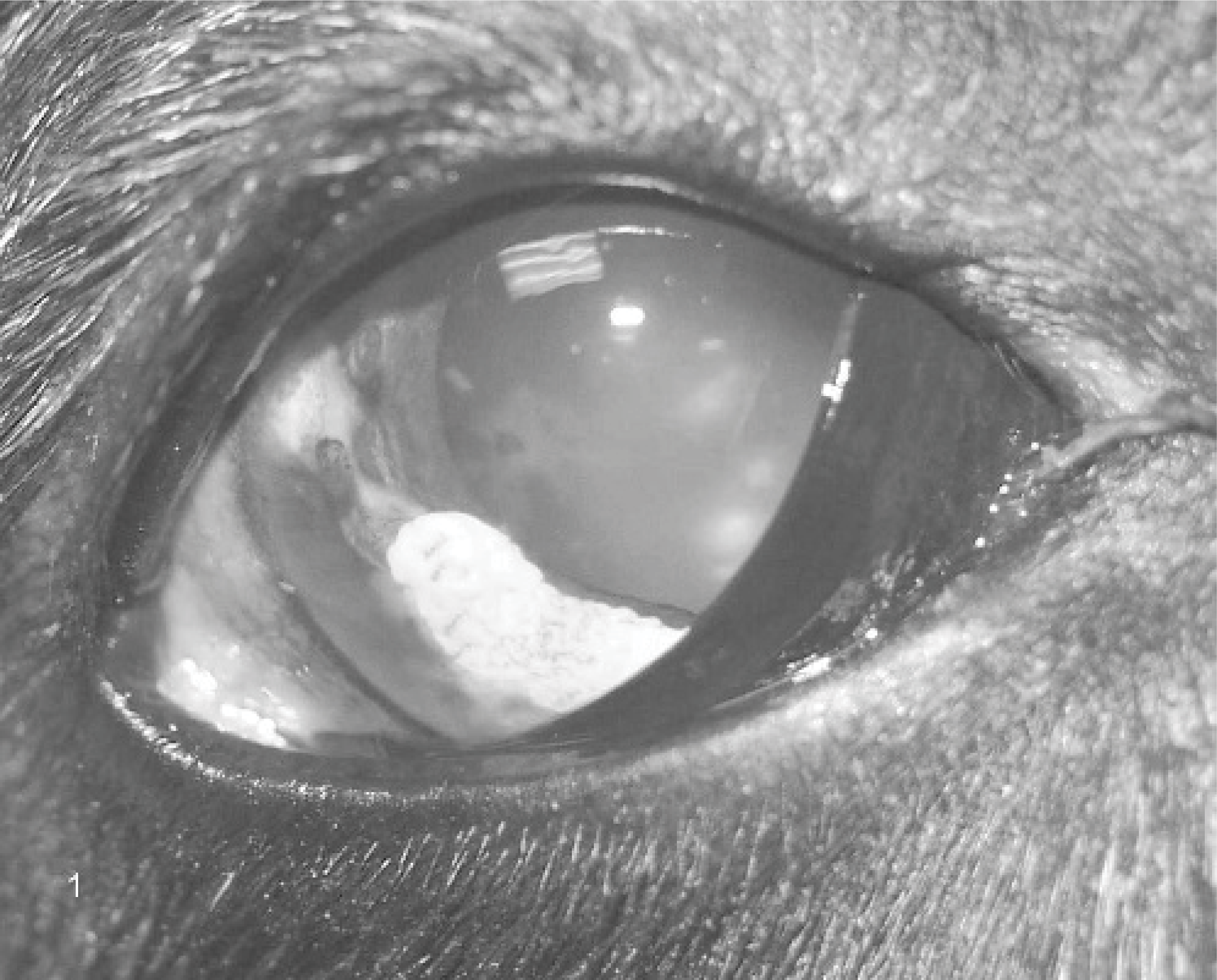
Clinical external photograph; 10-year-old Great Dane. A well demarcated dense white mass is demonstrated overlying the right ventrolateral iris. There are prominent multifocal endothelial pigment accumulations, moderate bulbar conjunctival hyperemia, mild segmental corneal edema, and ventrally located fibrin and blood clots. The third eyelid is partially elevated. Extension of the opacity laterally and dorsally to a second focus is present, but not easily appreciated in this photograph.
Routine ophthalmic diagnostic tests, including corneal fluorescein staining and intraocular pressure (IOP) measurements by applanation tonometry, were within normal limits, as were physical and neurologic examinations. A complete blood count, serum chemistry panel, endemic infectious disease titers, and urinalysis were submitted to rule out systemic diseases that may have caused secondary uveitis. All tests, including serologies for Histoplasma capsulatum, Aspergillus spp., Coccidioides immitis, Blastomyces dermatitidis, Ehrlichia canis, Borrelia burgdorferi, and Rickettsia rickettsii, were unremarkable. Thoracic radiographs revealed no evidence of current pneumonia or neoplasia.
Vision loss was suspected to be caused by the consequences of uveitis, including aqueous flare, anterior chamber fibrin and cells, corneal endothelial opacities, and corneal edema. The clients declined enucleation or evisceration, but accepted iris mass biopsy as an alternative means of obtaining a diagnosis. Because a dazzle reflex was present and IOP was not currently elevated, this was considered a reasonable alternative. However, it was considered less likely to result in a definitive diagnosis and more likely to result in complications than enucleation with submission of the globe en bloc for histopathology. Iris biopsy was performed through a dorsolateral perilimbal clear corneal incision with the aid of an operating microscope, with the patient placed in dorsal recumbency under general anesthesia. The mass was exceptionally hard. Excisional biopsy was deemed infeasible, but an incisional one was obtained and submitted in 10% buffered formalin to a veterinary pathologist. The formalin-fixed tissue was decalcified, embedded in paraffin, routinely sectioned, and stained with hematoxylin and eosin. Histopathology revealed well differentiated lamellar osteoid emanating from segments of otherwise unremarkable iris (Fig. 2). No evidence of neoplasia and minimal inflammation were identified within the sections. These findings were consistent with osseous metaplasia or heterotopic bone formation. Unfortunately, this patient developed postoperative glaucoma and gastric dilatation with volvulus. Despite aggressive medical and surgical treatment, the dog was euthanized because of declining health 6 weeks later, and necropsy was not performed.
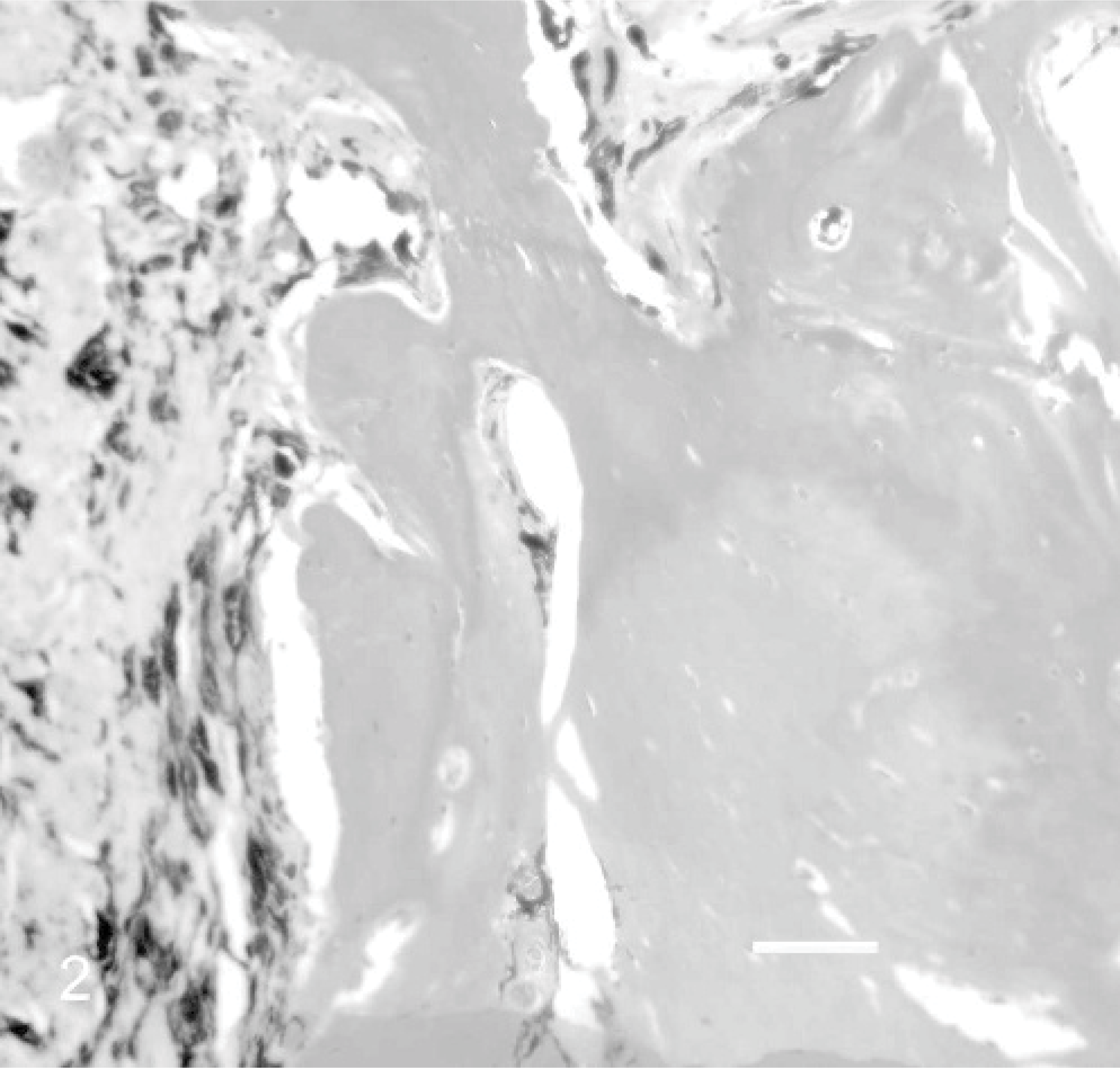
Incisional biopsy of right iridal mass; 10-year-old Great Dane dog. Note the lamellar osteoid formation within a segment of iridal tissue, showing no evidence of neoplasia and minimal inflammation. HE. Bar = 75 μm. Photograph courtesy of Dr. Richard Dubielzig, University of Wisconsin-Madison.
Although bone is used as a supportive structure within the sclera of many bird and reptile species, it is not a normal component of the mammalian eye. Certain primary or metastatic intraocular neoplasms, including osteosarcoma and feline post-traumatic ocular sarcoma, may contain bone.4,8 Ossification of normal ocular structures is rare, but has been reported in both man and guinea pig.1,3,7,9,10,12,13 Macroscopic osseous metaplasia of the iris is a condition that has not previously been reported in the dog to the authors' knowledge.
The terms “osseous metaplasia,” “heterotopic bone formation,” and “osseous choristoma” are used to describe bone formation in abnormal locations. The term osseous choristoma refers to microscopically normal bone in an abnormal location, but usually implies a congenital or developmental tumor. Deposition of bone into nonosseous tissues, or heterotopic bone formation, can occur secondary to metastatic calcification from systemic disease or dystrophic calcification from local tissue disease, but this is uncommon. An example of the former is kidney disease causing systemic electrolyte disturbances. The latter can occur secondary to local trauma or neoplasia. More commonly, ectopic mineralization occurs without lamellar bone formation. Osseous metaplasia implies alternative differentiation of local cells into osteogenic cells as a response to some insult (eg, traumatic, neoplastic, metabolic), and is thought by the authors to be the most appropriate term to describe the ocular changes in this dog.
Osseous metaplasia occurs not uncommonly in the guinea pig. Reports of bone formation in multiple soft tissues of this species exist.1,3,6,7,11 Elderly guinea pigs may be predisposed to bone formation in the ciliary body region.7 Osseous metaplasia of the ciliary body region has been reported in 17 guinea pigs.1–3,7,10 Six had grossly visible lesions, and the remaining 11 had microscopically identifiable osseous metaplasia. One author suggested that occult lesions predispose older guinea pigs to glaucoma.10 At least 3 of the animals with macroscopic ciliary body lesions also had mineralization of other tissues.1,3,7 Two of these 3 had documented renal insufficiency.1,7 These data are consistent with previous reports suggesting heterotopic bone formation in this species occurs at sites of metastatic calcification because of electrolyte disturbances caused by renal disease.11 Dietary factors may also play a role.6 Brooks et al. suggested a unique quality of the guinea pig ciliary body microenvironment may predispose this location.1
In humans, extraskeletal calcification occurs in chronic renal failure patients on dialysis. Multiple tissues may be affected, including the cornea and conjunctiva.12 Additionally, intraocular ossification in humans has been correlated with retinal detachment, hemorrhage, fibrovascular proliferation, gliosis, inflammation, trauma, glaucoma, congenital disease, and neoplasia.9,13 As in the guinea pig, the uveal tract appears predisposed in humans, but the choroid, rather than the ciliary body, is more commonly affected.5
Although neither excisional biopsy nor necropsy was performed on this patient, the 3-year history of the mass and lack of histopathologic evidence made malignant neoplasia unlikely. The lesions were not identified until well into adulthood and so were unlikely to be congenital, although later advancement of a subclinical congenital lesion could not be excluded. The dog's diet was unremarkable. No evidence of metabolic disease was identified on screening tests.
Moderate anterior uveitis was identified clinically and mild iritis identified histopathologically. Because the iridal opacities were present for years asymptomatically, it seems likely that the current anterior uveitis was either caused by progression of the masses or was coincident with it, and it is unlikely that pre-existing uveitis caused a localized change in microenvironment resulting in the osseous metaplasia. Although the submitted titers were negative, all infectious causes of uveitis cannot be definitively excluded. The reason for development of the original mass lesion years earlier is unclear, but could have been related to unobserved trauma. Traumatic ocular injury could have caused anterior uveitis, which is associated with blood aqueous barrier (BAB) breakdown. Breakdown of the BAB can be prolonged and predispose the eye to recurrent uveitis secondary to unrelated systemic disease, such as the pneumonia in this patient. Pre-existing unilateral BAB breakdown could also account for the asymmetry of presentation. Regardless of etiology, osseous metaplasia appears to be a rare differential diagnosis for an iris mass lesion in the dog. Presumably, the ciliary body or choroid could also be affected.
Footnotes
Acknowledgements
We extend sincere thanks to Drs. Jeff Bowersox, Rebecca Burwell, Julius Brinkis, and Douglas Esson, for their clinical involvement in this case over 3 years, as well as Dr. Richard Dubielzig for providing his histopathology expertise and photomicrographs.
