Abstract
Degenerative changes in the aorta are commonly observed in both dogs and humans, and those changes that occur with age morphologically overlap with those observed in genetic or degenerative diseases. Therefore, recognition of age-related aortic changes is important for diagnosticians, as such histologic findings should be distinguished from lesions of specific diseases. The aortas from 37 dogs without clinical cardiovascular disease ranging in age from 2 months to 15 years were divided into 3 cohorts to assess age-relatedness, and evaluated histologically using standardized nomenclature and diagnostic criteria adapted and modified from the human literature. We found that the histopathologic severity scores for intimal thickening, translamellar medial fibrosis, loss of smooth muscle cell nuclei, and medial microcalcification were higher in older dogs, whereas the scores for both intralamellar and translamellar mucoid extracellular matrix accumulation (“cystic medial necrosis”) were not different among age groups. Dogs with translamellar medial fibrosis and aortic medial microcalcification were significantly older compared with dogs without these findings, while the presence of aortic medial chondro-osseous metaplasia was not related to age. Taken together, we demonstrate a range of age-related aortic histologic changes in dogs without clinical cardiovascular disease and suggest that integration of signalment and clinical data can aid in the differentiation of such findings from non-age-related disease processes.
Keywords
Degenerative changes are a common feature of aortic diseases in dogs and humans. Aortic changes in humans caused by certain genetic syndromes and normal aging processes have overlapping morphologic features. 9 Mutations or defects of certain genes, such as fibrillin-1 in Marfan syndrome in humans and cattle, lead to aortic degenerative changes that can cause aortic aneurysm and dissection. 8,16 Aortic aneurysm, dissection, and dissecting hematomas in young dogs less than 1-year-old have been reported, in which cystic medial degeneration or necrosis, elastic fiber fragmentation and disorganization, and accumulation of mucopolysaccharides were described. 2,5,12 Although specific genetic diseases of the aorta have not been definitively diagnosed in dogs, a hereditary basis was suggested in those cases because the disease onset was early in life.
Aortic medial degeneration is thought to be the basis for clinical manifestations of inherited aortic diseases; however, there is controversy on the contribution of medial degeneration to aortic dissecting aneurysm in aged humans. Medial degeneration is proposed to cause weakening of the aortic wall and ultimately leads to fatal aortic dissection. However, other studies suggest that medial degeneration is an age-related change and might not result in aortic dissecting aneurysm, based on the observation of similar morphologic features in the normal aging human aorta. 3,19 The same uncertainty exists in the veterinary literature as aortic medial degeneration is described in both healthy aged dogs and aged dogs with aortic aneurysm or dissection. 1,13,25
This study aimed to characterize age-related histologic changes in the canine aorta. We hypothesized that degenerative changes such as aortic aneurysm and dissection are present in aged dogs without clinical evidence of cardiovascular disease, and that the severity of degenerative changes is associated with age. Because aortic degenerative changes in dogs are morphologically similar to those in humans, 13 we adapted a standardized nomenclature and grading scheme for reporting human aortic histopathology for qualitative and semiquantitative scoring of the canine aorta. 9 Consistent use of terminology to describe the spectrum of age-related aortic morphologic changes would be useful to more accurately distinguish incidental findings from disease- or treatment-related lesions.
Materials and Methods
Animals
Proximal aortas from client-owned dogs submitted to the autopsy service at The Ohio State University College of Veterinary Medicine were retrospectively searched. Included cases were dogs that were euthanized or had died for various reasons unrelated to the heart or aorta. Selected cases must have had available 1 to 2 sections of aorta perpendicular to the aortic valve annulus, including the proximal ascending aorta (at least 1–2 cm distal to the sinotubular junction), aortic sinus, aortic valve, and valve annulus. Cases were excluded if there was clinical evidence of cardiovascular disease (such as congenital abnormality, murmur, hypertension, heart failure, thromboembolic disease, cardiac enlargement, or cardiac neoplasm) based on the review of clinical records, or evidence of cardiovascular disease on postmortem examination (including atrial or aortic rupture, cardiac neoplasm, thromboembolic disease, or severe endocardiosis). Signalment, clinical, and pathology data including breed, sex, body weight, heart weight, and relevant medical history were obtained (Supplemental Table S1).
Histopathology and Scoring Scheme
Samples of aorta were fixed in 10% neutral-buffered formalin for at least 48 hours, processed, and stained with hematoxylin and eosin (HE). One representative section of the aorta from a young dog and one from an aged dog with degenerative changes were additionally stained with Verhoeff-van Gieson (VVG). Some cases were additionally stained with von Kossa and Alcian blue.
Aortic lesions were scored using a modification of a consensus grading scheme for human aorta. 23 The terminology and scoring scheme are detailed in Supplemental Tables S2 and S3. Representative histologic findings with different severity scores are shown in Supplemental Figures S1 to S18. Scoring of HE-stained sections was performed by a veterinary pathology resident (CY) while blinded to the clinical history and age of the dog. Scoring was subsequently reviewed in a blinded manner by a board-certified veterinary pathologist (RK) and a consensus was determined.
Statistical Analysis
Statistical analysis was performed using GraphPad Prism software. Data were compared using Mann-Whitney test, Student’s t test, and Kruskal-Wallis test with Dunn’s post hoc test, and χ2 test, where appropriate. Differences with P ≤ .05 were considered statistically significant.
Results
The study was based on proximal aortas from 37 dogs aged 2 months to 15 years (mean age 6.9 years). The cases included 22 (60%) male dogs, 13 (59%) of which were castrated, and 15 (40%) female dogs, 9 (60%) of which were spayed. Twenty breeds were represented in addition to mixed-breed dogs. Cases were ≤2 years of age (n = 9, 24%), 2 to 7 years of age (n = 10, 27%), and >7 years of age (n = 18, 49%). The aortas from young dogs less than 2 years old were used as controls for comparison with the aortas from older dogs.
Aortic Architecture
Three layers of the aorta were present: intima, media, and adventitia (Fig. 1a, b). The intima formed a single smooth layer of endothelium, and little connective tissue separated the endothelium from the first lamella of the aortic media (Fig. 2a, b). The media was composed of regularly spaced lamellar units, defined as a single layer of smooth muscle cells and a small amount of collagen and proteoglycans stacked between fenestrated elastic laminae; these features were highlighted by the VVG stain (Fig. 3a, b). The adventitia contained loose connective tissue, vasa vasorum, and vasa nervorum. Thinning of the proximal aortic wall associated the origin of a coronary artery was seen in some sections.
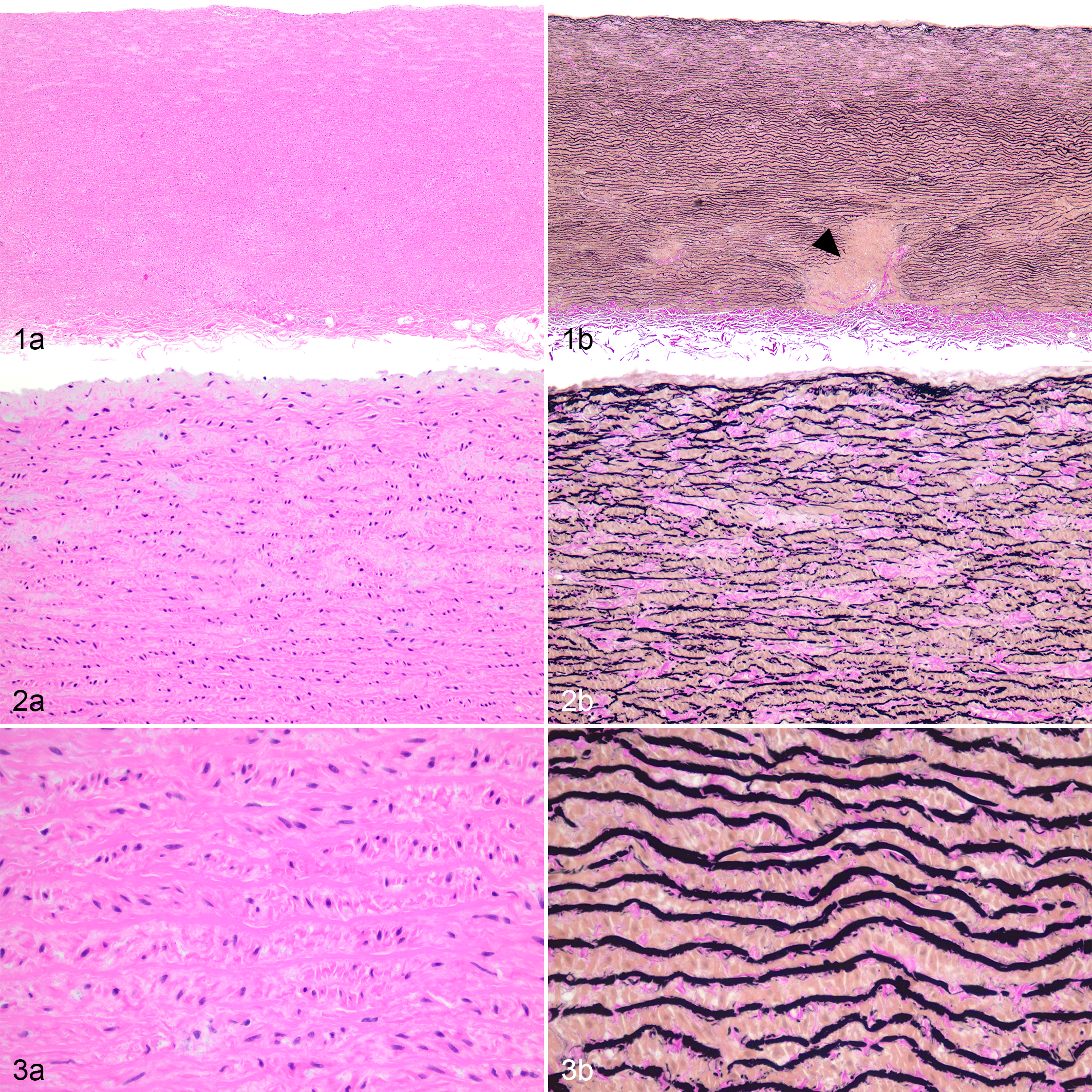
Aorta from a young dog, aorta, dog, case 7 (11 months old).
In the aorta of aged dogs, the overall (low magnification) tissue architecture of the aorta was not different from that of young dogs, but the intima of older dogs was variably irregular and thickened, and the media had subjectively less density of elastic fibers with wider intralamellar spaces (ie, the spaces between elastic laminae), patchy areas with loss of elastic fibers, and accumulation of fibrous connective tissue (Fig. 4a, b). In dogs of all ages, the lamellar units of the luminal third of the media had wider intralamellar spaces than those of the outer two thirds of the media (Figs. 1, 2). Additionally, disruptions of the medial lamellar units by larger vasa vasorum vessels in the outer third of the media were observed in dogs of all ages and were considered normal (Fig. 1b).
Intima
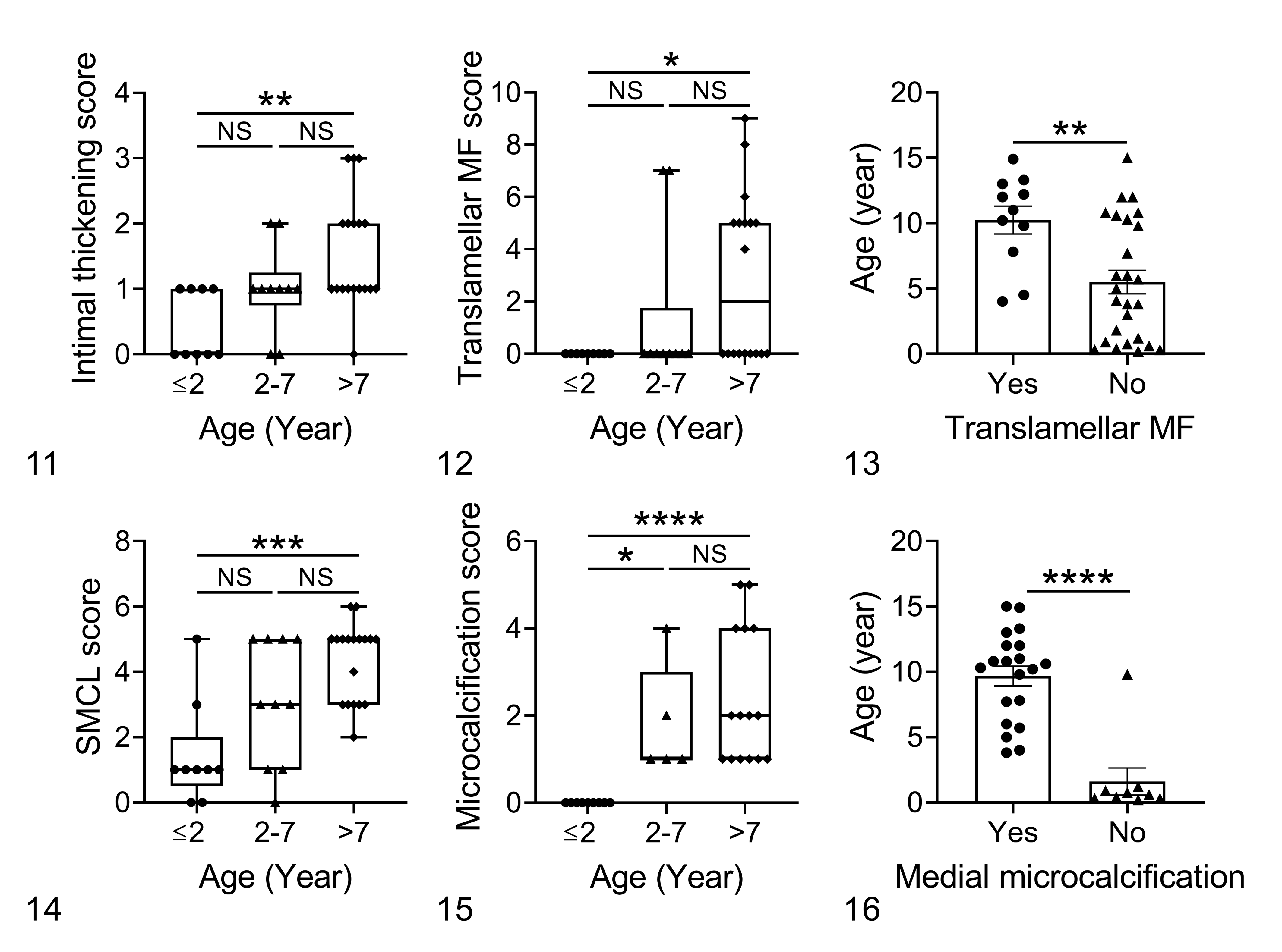
Intimal thickening was identified in 29 of 37 (78%) dogs and was characterized by an increased accumulation of layers of spindle cells and extracellular matrix (including collagen) deposited on a thickened and irregular first lamella highlighted by the VVG stain (Figs. 4b, 5). Of the 9 dogs that were ≤2 years old, 5 had no intimal thickening and 4 had mild segmental intimal thickening (2–5 layers of spindle cells), whereas moderate to severe intimal thickening (more than 6 layers of spindle cells) was only observed in dogs >2 years old. Dogs >7 years old had significantly higher severity scores for intimal thickening compared with dogs ≤2 years old (P = .003; Fig. 11). Intimal thickening was commonly seen in the proximal ascending aorta but was also present at the levels of the sinotubular junction, aortic sinus, and aortic valve annulus. Scattered inflammatory cells, usually histiocytes, were observed within the thickened intima in 8 of 37 (22%) dogs, but the ages of dogs with or without this lesion were not significantly different (P = .230, Student’s t test).
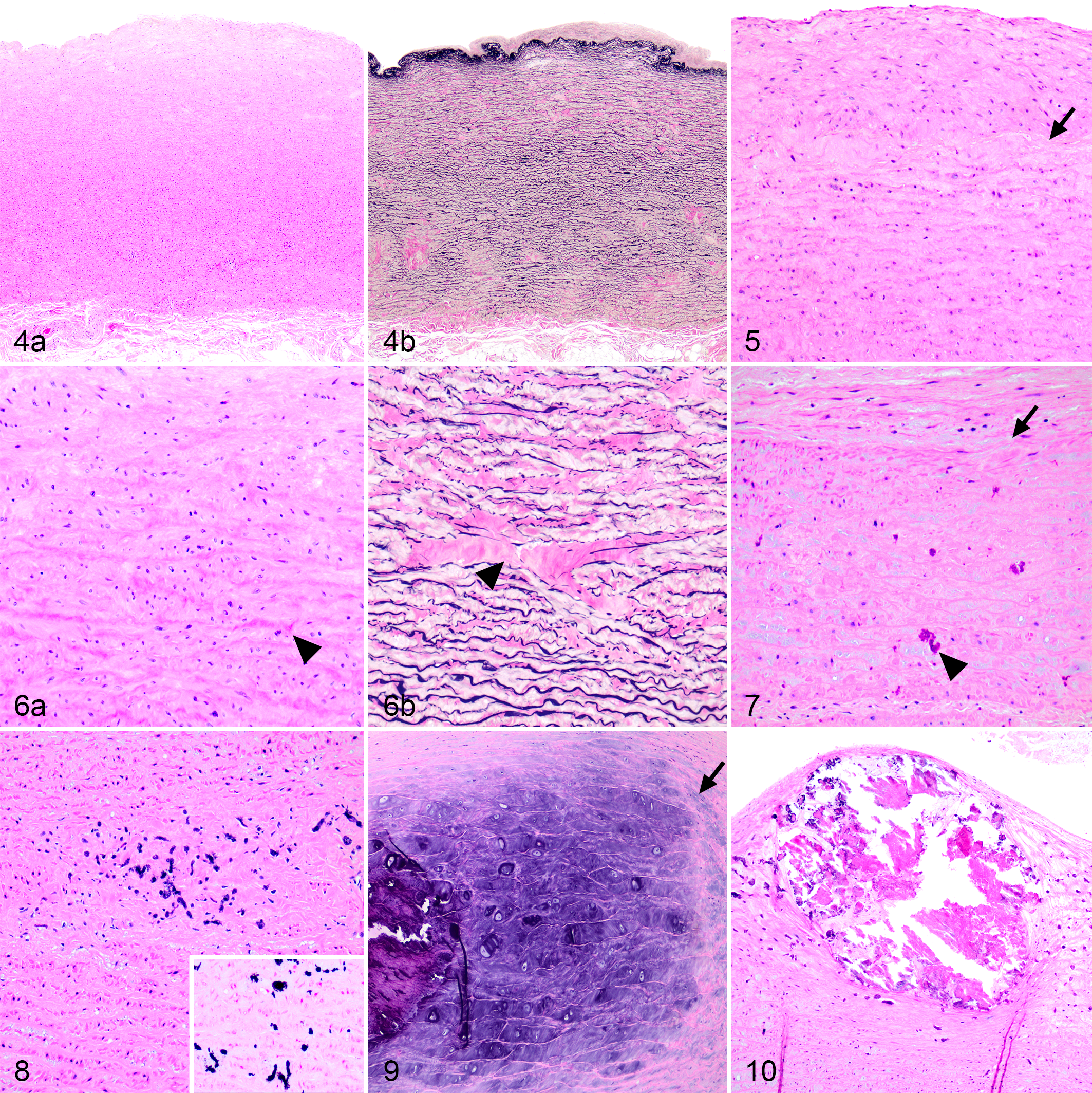
Age-related degeneration, aorta, dog, case 33 (12 years old).
Medial Proteoglycans and Collagen
In dogs of all ages, there was an accumulation of extracellular matrix in the media, including Alcian blue–positive mucoid material (known as mucoid extracellular matrix accumulation or cystic medial degeneration) and collagen (medial fibrosis; Fig. 6a, b), which was associated with variable degrees of disruption of elastic fibers. Mild lesions were focal widening of intralamellar spaces with loss of the parallel arrangement of elastic fibers; severe lesions were complete loss of multiple layers of elastic fibers in a large area. Intralamellar and translamellar mucoid extracellular matrix accumulation was detected in all age groups with the youngest dog being 2 months old. Intralamellar and translamellar mucoid extracellular matrix accumulation were detected in 30 of 37 (81%) and 24 of 37 (65%) dogs, respectively. The severity scores for both lesions did not differ among age groups (Suppl. Figs. S19, S20). In contrast, medial fibrosis was only found in 12 of 37 (32%) dogs, and all of these were ≥4 years old. Of these 12 dogs, 11 had translamellar medial fibrosis and 1 dog had intralamellar medial fibrosis (Suppl. Figs. S15–S18). The severity score for translamellar medial fibrosis was significantly higher in dogs >7 years old compared with dogs ≤2 years old (P = .036; Fig. 12). Also, dogs with translamellar medial fibrosis were significantly older than dogs without this lesion (mean ± SEM = 10.3 ± 1.1 and 5.5 ± 0.9, respectively; P = .004; Fig. 13).
Relationship between age and histologic changes in the aorta in dogs without clinical cardiovascular disease.
Medial Smooth Muscle Cells
Smooth muscle cell nuclei loss, defined as the lack of identifiable smooth muscle cell nuclei within multiple lamellae of the media (Fig. 7) occurred in either a patchy pattern (Suppl. Fig. S13) or a band-like pattern (Suppl. Fig. S14), were identified. This lesion was found in 34 of 37 (92%) dogs. While mild focal loss of smooth muscle cell nuclei was identified in dogs as young as 2 months old, the severity scores were significantly higher in dogs >7 years of age compared with dogs ≤2 years of age (P = .001; Fig. 14).
Aortic Mineralization
Two patterns of aortic mineralization were identified: microcalcification and chondro-osseous metaplasia. Microcalcification was characterized by scattered aggregates of fine, amorphous, basophilic, von Kossa-positive stippling or cylindrical material (Figs. 7, 8), which was most frequently found in the inner third of the media but also occurred in other parts of the media. Microcalcification of the media was identified in 26 of 37 (70%) dogs, and all of these were ≥3 years of age. Of the 37 dogs, 8 had possible risk factors for metastatic calcification including renal disease (n = 2), hypercalcemia (n = 1), prior treatment with glucocorticoid (n = 4), or hyperadrenocorticism (n = 1). None were identified to have a history of exposure to vitamin D or its analogues. Therefore, statistical analysis was done on the remaining 29 dogs that had no indication of these risk factors. Microcalcification occurred in conjunction with other aortic lesions such as intralamellar and translamellar mucoid extracellular matrix accumulation, loss of smooth muscle cell nuclei (Fig. 7), and fibrosis (Fig. 8), but a significant association was only identified between microcalcification and the presence of smooth muscle cell nuclei loss (P = .029, χ2 test). The severity scores for medial microcalcification were significantly higher in dogs >7 years old and dogs between 2 and 7 years old compared with dogs ≤2 years of age (P < .0001 and P = .017, respectively; Fig. 15). Dogs with microcalcification were significantly older than dogs without this lesion (mean ± SEM = 9.7 ± 0.8 and 1.6 ± 1.0, respectively; P < .0001; Fig. 16). The evaluated section of the aorta from 22 of 29 dogs without predisposing factors for metastatic calcification (ie, without renal disease, hypercalcemia, glucocorticoid treatment, or hyperadrenocorticism) also contained aortic valves. Of these, 9 (41%) had valvular microcalcification with a similar appearance to that of the aortic media in both the fibrosa and spongiosa.
The second pattern of aortic mineralization, referred to as chondro-osseous metaplasia, was characterized by grossly visible, focal or multifocal, large, raised, hard nodules, which were linear, plaque-like, or ovoid (Suppl. Figs. S21, S22). Chondro-osseous metaplasia often occurred in the proximal ascending aorta, on the intima of the aorta at the sinotubular junction or within the aortic sinus. Microscopically, the nodules consisted of chondroid, osseous, or chondro-osseous material expanding the media and elevating the intima, bulging toward the luminal surface. The chondroid nodules predominantly consisted of chondrocytes with cartilaginous matrix that often expanded but did not disrupt the elastic fibers (Fig. 9). In comparison, the osseous nodules were predominantly composed of bone fragments without apparent osteoblasts, osteocytes, or osteoclasts. This osseous tissue often disrupted elastic fibers, and the adjacent aortic tissue frequently had microcalcification (Fig. 10). Chondro-osseous metaplasia was identified in 11 of 37 (30%) dogs, ranging from 7 months old to 12.2 years old. Two dogs had chondroid nodules and 8 dogs had osseous nodules, and 1 dog had both types of nodules. Dogs with the above-mentioned predisposing factors for metastatic calcification were excluded for statistical analysis. There were no associations between the presence of chondro-osseous metaplasia and other adjacent aortic lesions, including intralamellar and translamellar mucoid extracellular matrix accumulation, medial fibrosis, loss of smooth muscle cell nuclei, and microcalcification. The mean ages (± SEM) for dogs with or without chondro-osseous metaplasia were not significantly different (4.9 ± 1.3 and 7.9 ± 1.1 years old, respectively; P = .166; Suppl. Fig. S23).
Aortic Adventitia
Lesions of the vasa vasorum were only found in one dog, in which there was focal intimal thickening of the vasa vasorum.
Discussion
Using an adapted and modified histopathologic scoring system for canine aorta, we identified that intimal thickening, translamellar medial fibrosis, loss of smooth muscle cell nuclei, and aortic medial microcalcification increased in severity or incidence with age in dogs, whereas mucoid extracellular matrix accumulation and chondro-osseous metaplasia were not age-related changes.
Given the lack of a standardized description of canine aortic changes in the veterinary literature, we sought to evaluate the applicability of the grading scheme for human aortic medial degeneration to the canine aorta. The human grading scheme was modified to better fit the findings in dogs for the following reasons. First, not all lesions described in humans 9 were observed in our study. Similar aortic degenerative changes in both humans and dogs included mucoid extracellular matrix accumulation, loss of smooth muscle cell nuclei, medial fibrosis, and elastic fiber fragmentation and loss, whereas smooth muscle cell disorientation and laminar medial collapse were observed in humans but not in the dogs of our study. Second, medial microcalcification and intimal thickening, which was not part of the human grading scheme, occurred relatively frequently in this study and were therefore included in our scoring scheme. Last, while a set of lesions were evaluated to reach an overall grade for medial degeneration in humans, 9 it has not been determined whether the extrapolation of this overall grade to dogs is meaningful. Therefore, to better illustrate both the severity and distribution of each individual degenerative change in the canine aorta, we modified the human grading scheme into a “distribution-adjusted scoring scheme” with lesions that were identified in dogs in our study.
In this study, while 20 of 29 (69%) dogs from 2 to 15 years old without predisposing factors for metastatic calcification had aortic medial microcalcification, none of the dogs under 2 years old had this lesion, suggesting that it was an age-related change. Consistent with our findings, a study of young beagle dogs ranging in 9 to 20 months old found that aortic medial mineralization was rare, occurring in only 4 of 201 (2%) dogs. 10 In humans, microcalcification was frequently found in older individuals, and aortic calcium content increased with age. 18 The presence of microcalcification in our study was associated with loss of smooth muscle cell nuclei, and this could be attributed to several mechanisms as the pathogenesis of medial mineralization is complex. 11 Some studies suggested that microcalcification was a sequela of apoptosis of smooth muscle cells, 6 in which the apoptotic bodies acted as nucleating agents for the deposition of crystals. 17 Other studies suggested that aortic microcalcification resulted from loss of calcification inhibitors, such as in Marfan syndrome where fragmented elastic fibers led to hydrolysis of calcification inhibitors resulting in the generation of calcium phosphate crystals. 26 Together, as microcalcification occurs with similar morphology in both aging processes and pathological conditions, interpretation of its relevance is dependent on the clinical history.
Aortic medial chondroid or osseous metaplasia was not related to age in our study. Notably, dogs with chondro-osseous metaplasia had a younger mean age compared with dogs without this lesion, albeit not statistically significant. This may indicate the pathogenesis of chondro-osseous metaplasia differs from that of medial microcalcification. Aortic mineralization with osseous metaplasia was reported as an incidental finding in dogs without diseases related to metastatic mineralization in a previous study. 20 Although Rottweilers were reportedly overrepresented with aortic mineralization in the same study, 20 the current study included 2 Rottweilers among the 11 dogs that had chondro-osseous metaplasia (Supplemental Table S1) but the small number of each dog breed did not allow analysis of breed predilection. Chondro-osseous metaplasia can also be induced following traumatic events or injection of foreign proteins into the aorta. 7,15 Although the mechanism of chondrocyte development in the aorta is incompletely understood, smooth muscle cells in mice were shown to have substantial tissue plasticity in response to injury and were able to “trans-differentiate” to osteochondrogenic precursors and chondrocytes in arteries with medial calcification. 22 Since extensive vascular mineralization in humans can also lead to various clinical complications such as thromboembolism and aortic rupture, 4,11 future studies may be required to determine the pathogenesis of microcalcification and chondro-osseous metaplasia in dogs.
Mucoid extracellular matrix accumulation, more commonly referred to as “cystic medial necrosis” in the veterinary literature, was unexpectedly not related to age in our study. This finding contrasts with the human study where the accumulation of glycosaminoglycan, especially chondroitin-6-sulfate, increased with age in the aorta. 24 Based on the findings in our study, mucoid extracellular matrix may be normal in dogs. Therefore, pathologists should be cautious in considering mucoid extracellular matrix accumulation as a cause of aortic rupture or other subsequent aortic lesions. Interestingly, compared to the middle and outer layers, the inner third of the media in dogs of all ages demonstrated a wider lamellar units, as described previously where healthy dogs of various ages had separation of the intimal lamellae resulting in the formation of irregular cystic spaces. 13
Consistent with studies in humans and other animals, 14,21 intimal thickening and aortic medial fibrosis were both age-related changes in dogs. Whereas CD68-positive macrophages (histiocytes) were usually observed in aortas of aging humans, 9 we did not identify such an association with age, which may be due to the small number of cases with this lesion.
A few cases of aortic aneurysm and dissection in adult dogs (one middle-aged and one 10-year-old dog) have been reported. 1,25 Histologic lesions identified in the aortic media in those studies included pseudocystic spaces, extracellular matrix accumulation, elastic fiber fragmentation, and cartilaginous metaplasia, all of which were identified in aged dogs without cardiovascular diseases in this study. Without further studies that compare the severity and morphology of medial extracellular matrix accumulation in dogs with and without aortic rupture, aneurysm, or dissection, these aortic diseases in adult dogs should not be attributed solely to the presence of medial degeneration. In addition to genetic causes and aging, several risk factors that may increase aortic stiffness and lead to aortic aneurysm were described in adult humans, including hypertension, atherosclerosis, hypercholesterolemia, strenuous exercise, and pregnancy. 8,9 There were no reported underlying diseases in the 2 adult canine cases with aneurysm and dissection in the previous case reports. 1,25 Therefore, apart from inflammatory conditions such as Spirocerca infection and atherosclerosis, and as yet uncharacterized genetic conditions, risk factors for aortic diseases in adult dogs warrant future investigation.
This study has several limitations. First, the number of cases with available aortic tissue limited the scale of this study. Second, only the ascending aorta was evaluated due to the limitation of the standard postmortem sampling method. Last, medium- to large-breed dogs were overrepresented in our study population and breed predilection for specific aortic findings was not evaluated since mixed-breed dogs represented the largest group in our study.
Conclusions
Degenerative aortic changes including those reported in cases of aortic aneurysm are common in aged dogs without cardiovascular disease and should not be considered causative in the absence of additional data. Aortic changes such as mucoid extracellular matrix accumulation and wider separation of lamellae in the inner aspect of the media may be normal findings in dogs of any age without clinical significance. Histologic patterns of aortic mineralization should be further characterized as these may indicate different mechanistic processes. Evaluation of aortic histopathology using a comprehensive approach and standardized terminology along with the integration of signalment and clinical data can aid in distinguishing incidental age-related aortic findings from non-age-related diseases.
Supplemental Material
Supplemental Material, Combined_supplemental_materials-Yang_et_al - Age-Related Changes in the Canine Aorta
Supplemental Material, Combined_supplemental_materials-Yang_et_al for Age-Related Changes in the Canine Aorta by Ching Yang and Rebecca Kohnken in Veterinary Pathology
Footnotes
Acknowledgements
We thank the staff in the histology laboratory at The Ohio State University Department of Veterinary Biosciences for technical assistance. We thank Dr Samantha Kochie for the retrieval of clinical data. We also thank Tim Vojt for assistance with figure compilation.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
