Abstract
Borna disease (BD) is a fatal disorder of horses, often characterized by blindness. Although degeneration of retinal neurons has been demonstrated in a rat model, there are controversial data concerning whether a similar degeneration occurs in the retina of infected horses. To investigate whether BD may cause degeneration of photoreceptors and possibly of other neuronal cells at least at later stages of the disease, we performed a detailed quantitative morphologic study of retinal tissue from Borna-diseased horses. BD was diagnosed by detection of pathognomonic Joest-Degen inclusion bodies in the postmortem brains. Paraffin sections of paraformaldehyde-fixed retinae were used for histologic and immunohistochemical stainings. Numbers of neurons and Müller glial cells were counted, and neuron-to-Müller cell ratios were calculated. Among tissues from 9 horses with BD, we found retinae with strongly altered histologic appearance as well as retinae with only minor changes. The neuron-to-Müller cell ratio for the whole retina was significantly smaller in diseased animals (8.5 ± 0.4; P < .01) as compared with controls (17.6 ± 0.8). It can be concluded that BD in horses causes alterations of the retinal histology of a variable degree. The study provides new data about the pathogenesis of BD concerning the retina and demonstrates that a loss of photoreceptors may explain the observed blindness in infected horses.
Borna disease (BD) was originally described as a fatal encephalitis primarily affecting horses and sheep, 8 caused by the infection with the neurotropic Borna disease virus (BDV). 5, 15 Natural BD has been well known as a behavioural and movement disorder for more than 100 years. A pathogenic role of BDV is suggested for particular human mental disorders. 4 BDV is a nonsegmented negative-strand RNA virus; it is the prototype of the family Bornaviridae within the order Mononegavirales (see reference 14 for a recent review). The virus infects neurons and glial cells in several neural tissues. 12 The disease has become a model for studying mechanisms of virus infection in the central nervous system including the retina. 12, 23 Intracerebral infection of laboratory animals results in the spread of BDV via the optic nerve into the eye. 18 Similarly, the virus may migrate centrifugally along the optic nerve into the retina in late stages of the disease in naturally infected horses. 15 The main outcome of the infection is an inflammatory reaction with mononuclear cells entering the neural tissue. Thus, BD is the result of a virus-induced immunopathologic reaction with virus-specific T cells (as well as macrophages/microglia) playing a significant role in the pathogenesis. 2, 26
Detailed studies referring to morphologic alterations 10, 16, 17, 23 and to the immune response 31 of the retina exist for Lewis rats infected with BDV. Massive neurodegeneration occurs, resulting in the reduction of the number of retinal neurons and of the retinal thickness to about one third after 6–8 months survival time. 16 Experimental BD retinitis and encephalitis are associated with behavioral abnormalities and blindness in rats. 23 Although blindness is regularly observed in later stages of natural BD in horses, there are less morphologic data from equine retinae. Whereas some studies are known from the first half of the 20th century 22, 30, 32– 34 (a comprehensive review of the older literature is given in reference 8), Richt et al. 26 reported about the lack of inflammation and degeneration in the retina of horses in a more recent review. Therefore, our study was aimed at a quantitative histologic investigation of retinal tissues from several naturally infected horses.
Materials and Methods
Nine horses (case Nos. 1–9) suffering from severe BD were euthanized for the reason of animal welfare at the Faculty of Veterinary Medicine, University of Leipzig. The age of the animals varied between 9 months and 15 years; the duration of disease (time from first clinical symptoms until death) was between 7 days and 6 months. During clinical diagnosis, we determined several parameters: number of cells within the cerebrospinal fluid (CSF), percentage of lymphocytes among the CSF cells, and BDV-specific antibodies in the CSF and in the serum. These data are summarized in Table 1. About 10 ml of CSF was taken from the cisterna magna by puncture between the occiput and first cervical vertebra immediately before euthanasia in deep anesthesia. The presence of BDV-specific antibodies was investigated using an indirect immunofluorescence assay. Four horses (case Nos. 10–13) that were euthanized because of other diseases served as controls (aged between 7 months and 7 years). The eyes were enucleated, and the retinae were removed and fixed in paraformaldehyde (4%). The tissue was embedded in paraffin, and 10-μm-thick radial retinal sections were cut and used for histologic and immunohistochemical stainings after deparaffination and rehydration.
Clinical data of horses with Borna disease.
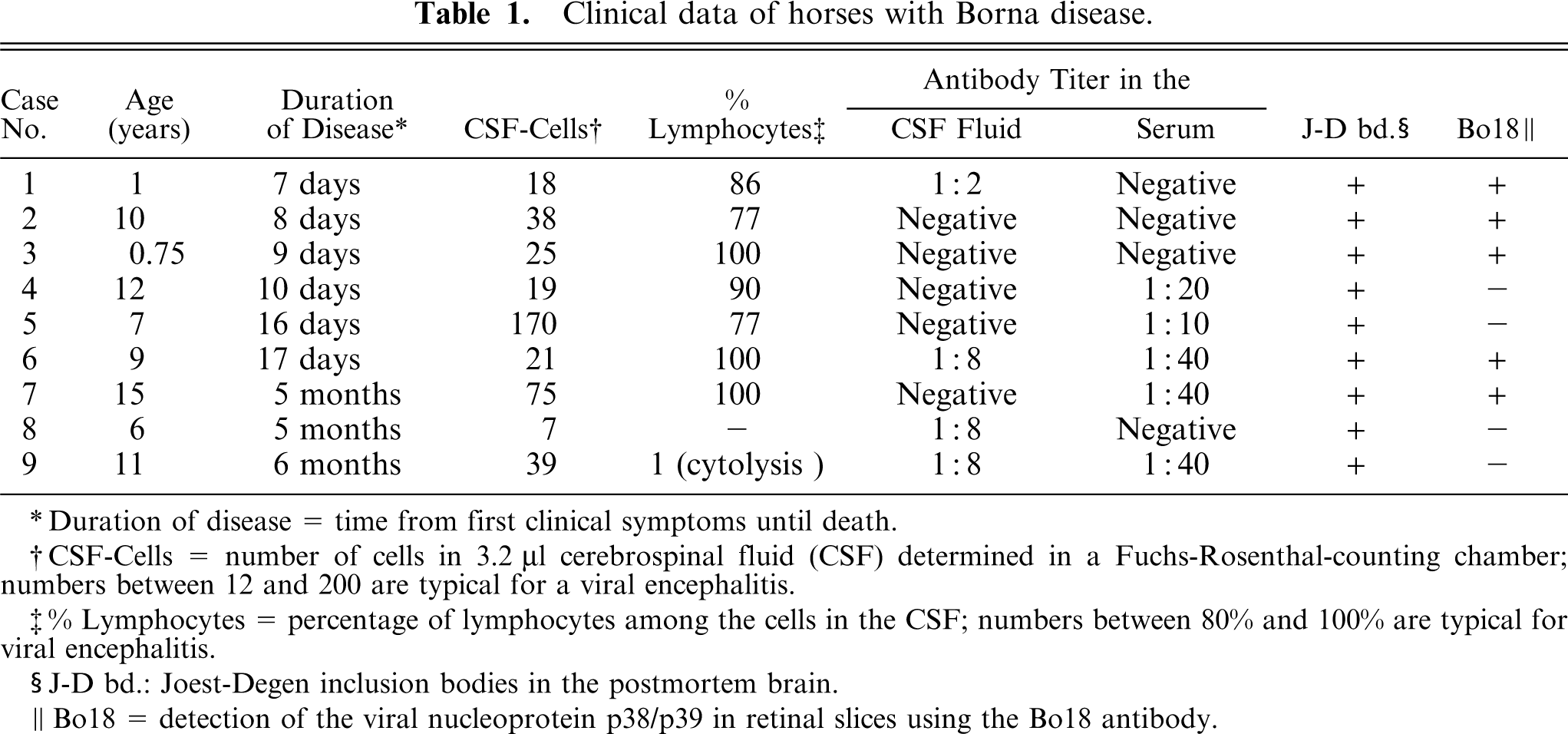
Duration of disease = time from first clinical symptoms until death.
CSF-Cells = number of cells in 3.2 4;µl cerebrospinal fluid (CSF) determined in a Fuchs-Rosenthal-counting chamber; numbers between 12 and 200 are typical for a viral encephalitis.
% Lymphocytes = percentage of lymphocytes among the cells in the CSF; numbers between 80% and 100% are typical for viral encephalitis.
J-D bd.: Joest-Degen inclusion bodies in the postmortem brain.
Bo18 = detection of the viral nucleoprotein p38/p39 in retinal slices using the Bo18 antibody.
The presence of BDV was tested in retinal sections from all horses with BD (case Nos. 1–9) as well as in the retina of 2 control horses (case Nos. 10 and 12) using a mouse monoclonal antibody recognizing a BDV-specific antigen, the viral nucleoprotein p38/p39 (Bo18, 1°500, courtesy of J. A. Richt). Biotinylated goat anti-mouse antibodies followed by treatment with avidin-biotin-peroxidase complex and 3.3-diaminobenzidine tetrahydrochloride (DAB) as chromogen were used for visualization.
For the detection of the intermediate filament glial fibrillary acidic protein (GFAP), we applied the following protocol: Sections were treated with pronase (Boehringer, Mannheim, Germany; 1 mg/ml phosphate buffered saline, 15 minutes at 37°C). Endogenous peroxidase was blocked in 1% hydrogen peroxide for 30 minutes at room temperature. Next, the Vectastain-Elite-ABC-Kit (Vector Laboratories, Burlingame, CA) was employed according to the instructions of the manufacturer. Rabbit-anti-GFAP antibody (Sigma, Taufkirchen, Germany; diluted 1°500) was used as primary antibody; the sections were incubated overnight at 4°C. Peroxidase activity was then revealed with the DAB-peroxidase-substrate-kit (Vector Laboratories). Controls were obtained by omitting the primary antibody. Cell nuclei were counterstained with haemalum.
To evaluate the densities of neuronal cells and to estimate the neuron-to-Müller cell ratio, we counted the numbers of cell nuclei in the inner and outer nuclear layers (INL and ONL) as well as in the ganglion cell layer (GCL) within a frame provided by the image-analysis program, analySIS (Software Imaging System, Münster, Germany; based upon a microscope-coupled video camera). This frame corresponded to a 25-μm-wide stripe along the radial section (i.e., to a retinal surface area of 250 μm2 in 10-μm-thick sections). Thirty frames were counted from each retina. Raw data were corrected for cut nuclear segments. 9 Müller cells were counted as GFAP-immunoreactive trunks within the inner plexiform layer (IPL). 25 When the number of neuronal cells per Müller cell was evaluated, 1 nucleus per Müller cell trunk was subtracted from the total cell number in the INL (i.e., taken as a Müller cell nucleus). The thickness of Müller cell stem processes within the IPL was measured in sections stained for GFAP, employing the same computer program. Significance of differences was tested by the Mann-Whitney U-test. Data are given as mean values with standard error.
Results
Clinical data for the horses with BD are given in Table 1. The diagnosis of BD made primarily on the basis of clinical symptoms was verified histologically for each animal by the presence of pathognomonic Joest-Degen inclusion bodies in the postmortem brains. The presence of the viral nucleoprotein was tested in the retinae of all BD horses and was effectively detected in 5 of them (Table 1). The reason for the lack of a positive detection in 4 animals is not known. A similar finding was described by Gosztonyi and Ludwig, 11 who assumed technical reasons to be responsible for the negative immunohistochemical reaction. Because there were variations in the time between the death of the animal and the start of fixation of the retina as well as in the duration of fixation, technical problems may also apply in the present study. However, the animals with negative results displayed significant alterations of the retinal structure (Figs. 3A, B; 4A, B), and the presence of Joest-Degen inclusion bodies strongly suggested the virus infection of brain tissue. Although it cannot be excluded that retinal degeneration in the case Nos. 4, 5, 8, and 9 is caused by other reasons except BDV infection, we consider this unlikely. As expected, no viral nucleoprotein could be detected in retinal tissue from control animals.
Because of the natural infection of horses, the actual duration of the infection was unknown. The appearance of retinal morphology in individual retinae suggests varying periods of time between infection and death of the animals. The retinal histology of a control animal (case No. 12) is shown in Fig. 1; that of 3 different animals with BD (case Nos. 3, 5, and 9), in Figs. 2–4. Clear differences between the retinae from control and diseased animals, but also among individual horses with BD, are visible. Obviously, the degeneration of neurons started in the ONL, where the photoreceptors are located (Fig. 2A, B). The thickness of this layer was reduced in all BD animals, but in some animals, there was also degeneration in the INL, although this was less pronounced in case Nos. 1, 3, and 6. In 2 cases (Nos. 8 and 9), the normal architecture of the retina was essentially effaced (Fig. 4A), in addition to the reduction in thickness of all layers. Death of BDV-infected neurons could also be a reason for the absence of immunohistochemical detection of virus in these cases. Interestingly, case Nos. 8 and 9, with the most extreme morphologic changes, had a duration of disease of several months. This suggests that the severity of neurodegeneration increases with the duration of infection, similar to the effects observed in experimentally infected rats. 16
To quantify the neurodegeneration, we counted the numbers of cell nuclei in the distinct layers and calculated the ratios between neurons and Müller cells. The 2 retinae that did not display a normal layering (case Nos. 8 and 9) were excluded from this quantification. Mean neuron-to-Müller cell ratios in each retinal layer and for the whole retina are given in Table 2 and Fig. 5, respectively. A significant decrease was found in each retinal layer, pointing to a specific cell death of neurons across the whole retina. Values of the neuron-to-Müller cell ratio ranged between 14 and 22 in the control animals and between 7 and 9 in all diseased horses. The mean value for each group is 17.6 ± 0.8 for controls (n = 4) and 8.5 ± 0.4 for animals with BD (n = 7), which is a significant difference (P < .01). Because immune cells were not specifically stained, it cannot be excluded that such cells were counted as neurons. In this case, the degree of neurodegeneration would have been underestimated. The density of Müller cells was not significantly affected in diseased retinae. The number of Müller cells per 25-μm-wide area varied between 2.5 and 3.2 in retinae from controls and from horses with BD. Thus, the neuron-to-Müller cell ratios mirror the proceeding neurodegeneration.
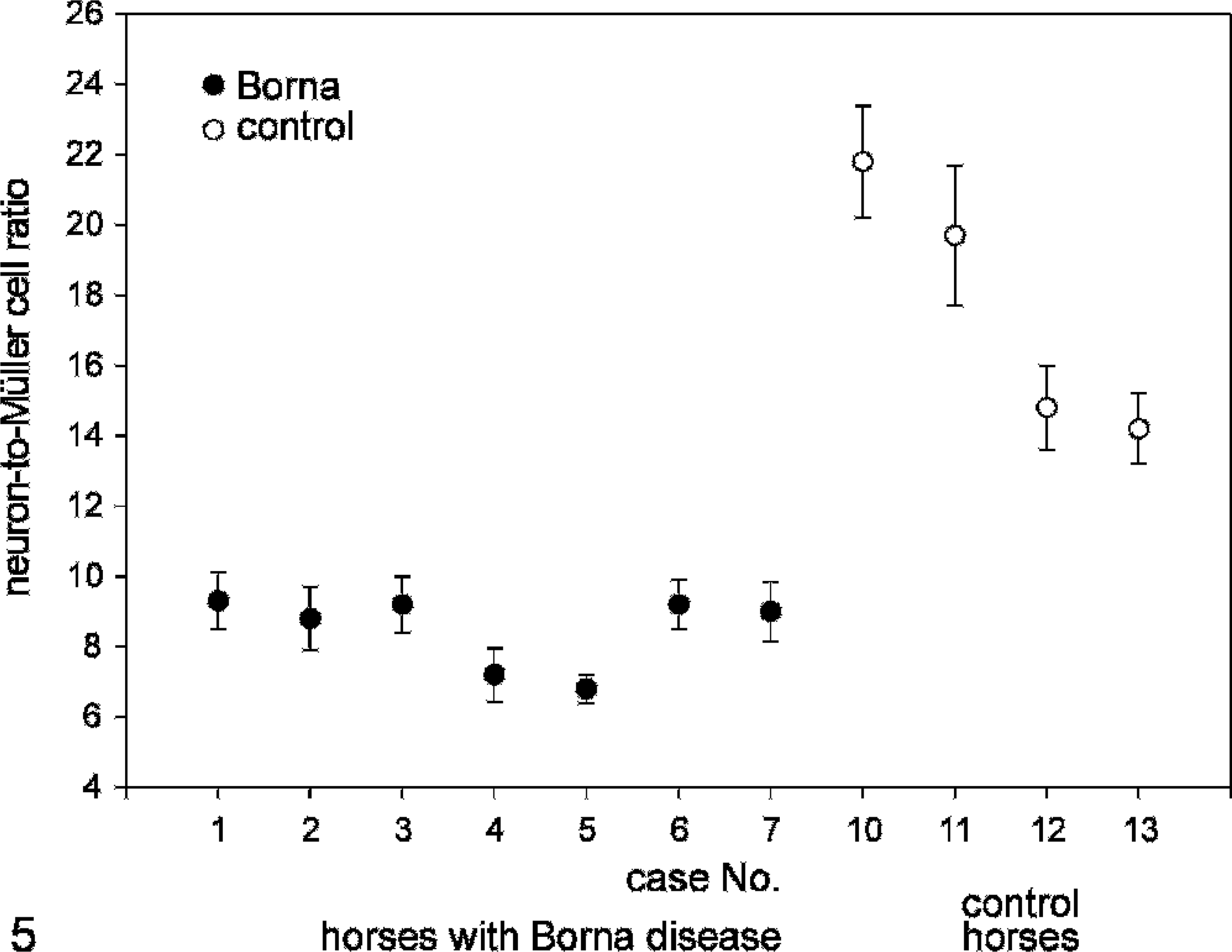
Neuron-to-Müller cell ratios for individual horses. Whereas the value for control animals is between 14 and 22 (case Nos. 10–13), it never reached more than 10 in any of 7 horses with BD (case Nos. 1–7). Mean values with standard errors are shown.
Neuron-to-Müller cell ratios in horse retinas.∗
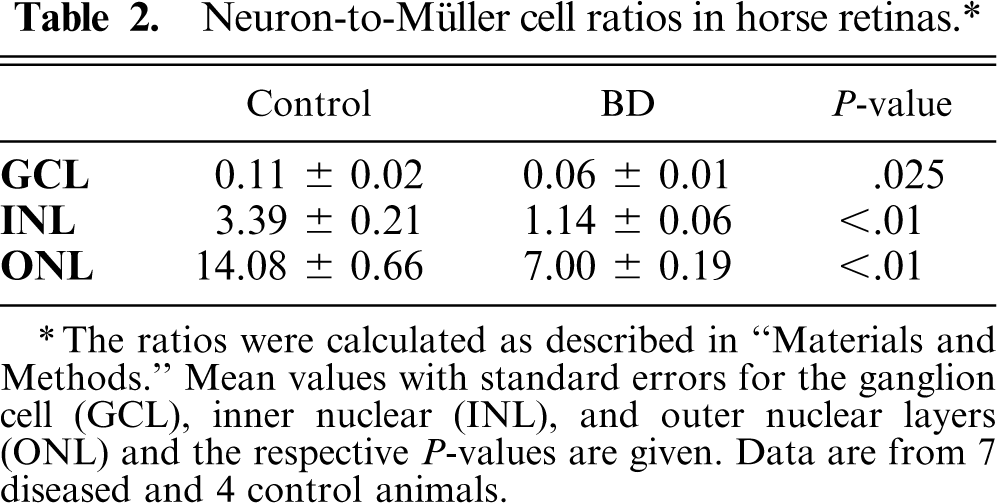
The ratios were calculated as described in “Materials and Methods.” Mean values with standard errors for the ganglion cell (GCL), inner nuclear (INL), and outer nuclear layers (ONL) and the respective P-values are given. Data are from 7 diseased and 4 control animals.
Although the number of Müller cells was virtually unchanged, certain alterations of cell morphology and protein expression could be observed. There are controversial data about expression of GFAP in normal Müller cells. In any case, it has been shown in different animal models that there is an upregulation of GFAP expression in cases of retinal injury or disease. 1, 20 We found a strong GFAP expression (Figs. 2B, 3B, and 4B) in Müller cells from animals with BD. Müller cells in control retinae were also immunoreactive for GFAP; however, their immunoreactivity was less prominent (compare Figs. 1B and 2B). This is in agreement with data from Chao et al., 7 who found GFAP-positive Müller cells in horse retina.
The thickness of proximal stem processes of Müller cells in the IPL was assessed by measuring the GFAP-immunopositive structures. This value was significantly increased from 2.69 ± 0.07 μm in control retinae to 3.01 ± 0.06 μm in retinae from diseased horses (P < .01).
Discussion
BD is endemic in certain areas of Germany: There are a number of pathologic reports from the first half of the 20th century (mostly in the German language) about ocular manifestations of BD in horses. 8 However, we are not aware of a detailed quantitative histologic investigation as presented in our study. A short survey about observations on alterations in the horse retina caused by BD has been provided by Gosztonyi and Ludwig. 12 These authors quoted several reports about the development of amblyopia and amaurosis in horses, which was first described by Schmidt 30 and later confirmed by Zwick 34 and Müller and Fritsch. 22 Another early report of histologic alterations in the retina was given by Stofer. 32 Walther 33 described a nonpurulent, lymphocytic infiltration of the retina and a degeneration of the optic nerve. The study reported about impaired vision in 5 of 69 horses, but this could derive from degenerations in optic brain regions. In a more recent review, 26 it was pointed out that a loss of neurons occurs in the retina of rats and rabbits after BDV infection, whereas no similar observations exist for the retina of horses with BD. The main reason might be that most horses are euthanized before onset of retinal degeneration. 26 This is in agreement with reports from Bilzer et al., 2, 3 who investigated the retinae of horses and donkeys suffering from BD. Although virus RNA and protein could be found in the retina of some (but not all) animals, there were no signs of inflammation and only weak expression of MHC class I and class II molecules. Moreover, there are additional reports about the presence of BDV in the eyes (among other different tissues) from naturally infected horses. 13, 19
In the present study of tissue derived from naturally infected horses, we found retinae with an almost unaltered histologic appearance but also retinae with massive neurodegeneration. The quantification of cell numbers revealed that in all animals, the number of neurons (as demonstrated by the neuron-to-Müller cell ratio) is significantly decreased as compared with values from control animals without BD. The individual variability is assumed to be caused mainly by differences in the infection period. The 2 animals with severely destroyed retinae displayed a duration of disease of a few months, whereas this time interval was only several days for most other horses.
Our data demonstrate that similar to degeneration occurring in the rat retina after BDV-infection, 10, 23 neuronal cell death can be observed in the retina of the horse (a natural host of BD). Although the transmission route for viral infection of natural hosts has yet to be elucidated, virus entry through nasal secretions into the olfactory system seems to be very likely. 21, 27 Within the brain, the virus spreads by intra-axonal transport and possibly via the cerebrospinal fluid 27 and along the optic nerve into the retina. 18 Viral nucleoprotein can be demonstrated in the retina after intracranial experimental infection in rats. 16, 31 The time to spread to the retina via axonal transport may explain why some BDV-infected horses have no or minimal retinal neuronal effects. 2, 3 It is also possible that differences in BD-caused retinopathy in rats and horses may reflect species differences in retinal vascularization that might result in differences in the inflammatory process. A study similar to that performed by Stahl et al., 31 who demonstrated the time course of lymphocytic infiltration in BDV-infected rats, should be carried out on tissue from horses in order to assess such possible differences.
Whereas a more or less severe neurodegeneration in the horse retina is evident, more subtle alterations were found in Müller glial cells. Obviously, the Müller cells are less susceptible to the heavy inflammatory reaction observed after BDV infection. Müller cells were demonstrated to undergo reactive gliosis in experimentally infected Lewis rats. 24 Cells displayed increased membrane capacitances, which are a sign of cellular hypertrophy, and distinct changes in their membrane current pattern. Moreover, Kacza et al. 16 observed several immunocytochemical alterations in Müller cells from BDV-infected rats. Although we were not able to perform electrophysiologic recordings on Müller cells from Borna-diseased horses, Müller cell morphologic alterations similar to those seen in BDV-infected rats (thickened stem processes indicating hypertrophy and strong GFAP immunoreactivity) were found in horses. While astrocytes and other glial cells do not display massive degeneration, it is well documented that BDV replicates within these cells. 6 In addition to the gliotic response, glial cells in certain brain regions have been demonstrated to be involved in the process of inflammation by expression of cytokines 28 and chemokines. 29 This was also postulated for astrocytes and Müller cells in retinae of BDV-infected rats. 31 Whether glial cells play a similar role in the retinae of naturally infected horses remains to be elucidated in future studies.
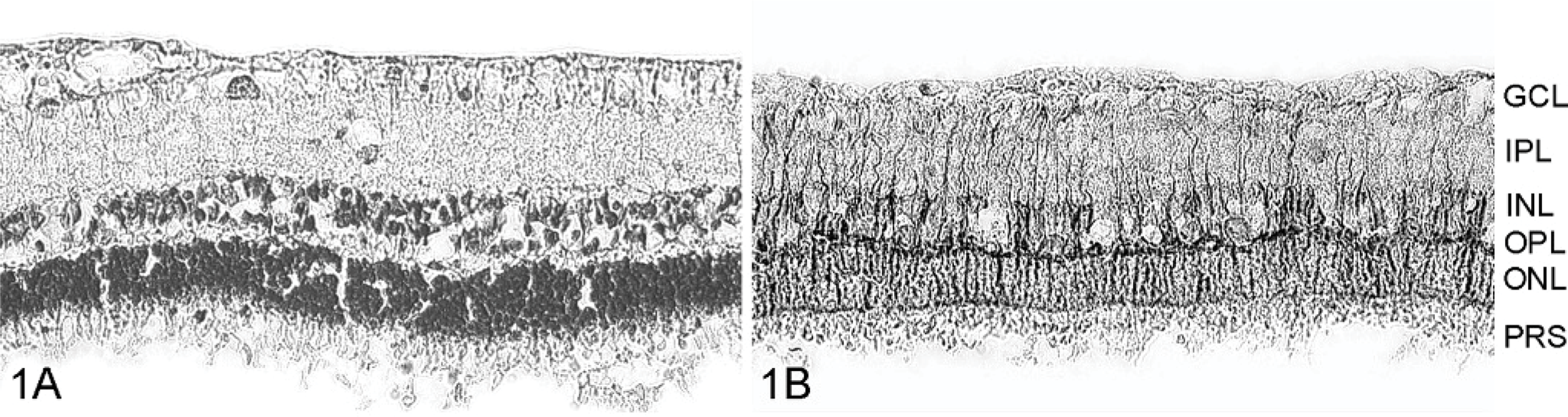
Retinal histology in slices from a control horse (case No. 12; Fig. 1A, B) and from 3 Borna-diseased horses (case No. 3: Fig. 2A, B; case No. 5: Fig. 3A, B; and case No. 9: Fig. 4A, B) displaying different degrees of degeneration. Slices were stained with hematoxylin (A) and with anti-GFAP antibodies (B). Only mild alterations were observed in case No. 3 with an apparent decrease of the ONL (compare with Fig. 1). The retina of case No. 5 is more heavily affected; the retinal structure is disturbed (therefore, the indication of retinal layers was omitted). The retina of case No. 9 totally lost the normal retinal layering and displayed a strong decrease in cell number. A weak immunoreactivity for GFAP was observed in Müller cells in retinae from control horses (Fig. 1B). BDV infection resulted in an increased GFAP immunoreactivity in all cases, demonstrating that Müller cells are surviving and display hypertrophy. Scale Bar in Fig. 4B represents 50 μm and is valid for all microphotographs in Figs. 1–4. GCL, = ganglion cell layer; IPL = inner plexiform layer; INL = inner nuclear layer; OPL = outer plexiform layer; ONL = outer nuclear layer; PRS = photoreceptor segments.
Footnotes
Acknowledgements
This work was supported by the Bundesministerium für Bildung, Forschung und Technologie, Interdisziplinäres Zentrum für Klinische Forschung (IZKF) at the Faculty of Medicine of the University of Leipzig (Project C5), and by Deutsche Forschungsgemeinschaft, Grant PA 615/1-1. The authors thank Prof. Dr. W. Drommer (Institut für Pathologie, Tierärztliche Hochschule Hannover) for kindly providing retinal tissues and Dr. J. A. Richt (United States Department of Agriculture, Ames) for the Bo18 antibody.
