Abstract
Borna disease is a severe viral-induced disorder of the central nervous system of horses, sheep, and a few other animal species, occurring in certain areas of central Europe. Pathogenesis and epidemiology of natural Borna disease virus (BDV) infections are still not fully understood; several unique epidemiologic features, however, point toward the existence of BDV reservoir populations other than the final hosts. In this study, 69 mice and 12 shrews were trapped and examined. The virus distribution was investigated in detail in 2 BDV-positive bicolored white-toothed shrews, Crocidura leucodon, by immunohistochemistry and TaqMan real-time reverse transcription polymerase chain reaction (RT-PCR). RT-PCR amplification products were sequenced, and the sequences were compared. These shrews had been collected in a BDV-endemic geographical region using live traps and did not show obvious clinical or pathological disease signs. BDV antigen and nucleic acid were identified in several organs, including the brain, mainly in nerve tissue and neurons, respectively, but also in parenchymal cells (eg, hepatocytes, Leydig cells) and epithelial cells, particularly of the respiratory and urogenital tract.
Keywords
Borna disease virus (BDV) is the causative agent of Borna disease (BD), a sporadic, mostly fatal meningoencephalitis of horses, sheep, and a number of other animal species, including cattle, rabbits, dogs, and some exotic ruminants kept in zoos. 22 It occurs sporadically in distinct endemic regions of Germany, Switzerland, the Principality of Liechtenstein, and Austria. BDV is a noncytolytic nonsegmented negative-stranded RNA virus belonging to the order Mononegavirales. A variety of warm-blooded vertebrates, ranging from rodents to nonhuman primates, are susceptible to experimental infection with BDV. In addition, for the past 20 years various publications indicated that humans could be a target for BDV infection, 2,15 implying a possible zoonotic risk. Recent findings, however, suggest that previous studies linking BDV to human neuropsychiatric diseases may have been compromised by sample contaminations. 3,18 Numerous studies with experimentally infected rats and mice have conclusively demonstrated that BD is caused by immunopathological mechanisms in which the antiviral CD8 T cell–mediated immune response results in neurological disease. 1,14,17,19 In adult rats, the infection causes severe neurological abnormalities and eventually death. 9 Neonatally infected rats may remain clinically healthy in spite of persistent infection. The same was observed after immunosuppression of adult rats by treatment with cyclophosphamide. 22 The natural way of transmission of BDV infection has been obscure so far. Free-living small rodents and inapparently infected sheep or horses have been discussed as vector candidates but never confirmed. 4,19,23
A recently published study by our group demonstrated for the first time that the bicolored white-toothed shrew, Crocidura leucodon, has to be considered a natural host of BDV. 10 These shrews had been trapped in Malix, a small village located approximately 4 km south of Chur, Switzerland, 760 m above sea level.
Bicolored white-toothed shrews are insectivores, 29 to 41 mm in length (head and body) and 7 to 13 g in weight. Their fur is clearly bicolored. In Switzerland, this shrew species is present in the Rhône Valley, the Basel region, Kanton Tessin, and in the Rhine Valley; 8 only in the latter area is BD endemic in horses, sheep, and other animal species.
The objectives of the present study were (1) to investigate further shrews, voles, and mice from a BD-endemic region close to but not identical to the region sampled before for the presence of BDV and (2) to investigate in detail the tissue distribution of BDV antigen and nucleic acid in several organs of this candidate species.
Material and Methods
Between 2004 and 2006, 69 mice (2 Arvicola terrestris, 46 Apodemus sp., 9 Clethrionomys glareolus, 7 Mus domesticus, and 5 Microtus sp.) and 12 shrews (6 Sorex araneus and 6 C. leucodon), mostly adult animals, were collected using live traps (Longworth Mammal Trap With NestBox, Penlon Limited, Abingdon, UK) in Seewis, a small village in the Praettigau Valley, located approximately 15 km north-northeast of Chur, Switzerland, about 1000 m above sea level, 20 km from the previous trapping area. 10 This region is also considered endemic for BD in horses and sheep. The animals were humanely killed and dissected within 24 hours. Their brains were divided into 2 equal parts: One half was fixed in 4% formaldehyde, cut longitudinally into several equal parts, and embedded in paraffin wax for microscopic evaluation; the other half was stored in tubes at –20°C. Likewise, the heart, lung, liver, intestinal tract, kidney, reproductive tract, spleen, skin, salivary gland, adrenal gland, and a longitudinal section through the skull were fixed in 4% formaldehyde and embedded in paraffin wax for microscopic evaluation, while an equally sized part of the above-mentioned organs and tissues were stored frozen at –20°C.
BDV antigen staining was achieved employing 2 monoclonal antibodies individually directed against the p24 (Mab 21E7) and p38/40 (Mab B018) proteins of BDV. Formalin-fixed, paraffin-embedded sections were deparaffinized and rehydrated in xylene and alcohol. Sections were then counterstained with hemalum for 2 minutes and washed in H2O for 3 to 5 minutes. The following incubations were at ambient temperature, and, after each step, slides were thoroughly washed with phosphate-buffered saline, pH 8.0. After digestion with proteinase K (Dako REAL Proteinase K 40x, Dako, Copenhagen, Denmark) for 10 minutes, endogenous peroxidase was blocked with H2O2 (3% in H2O and supplemented with 0.2% NaN3) for 10 minutes. Separate sections were incubated for 12 hours in a 1:1000 dilution of monoclonal mouse anti-BDV antibody that recognizes the proteins P38/40 (gift from L. Stitz, FLI, Tuebingen, Germany) or a 1:500 dilution of a monoclonal antibody against protein P24 (L. Stitz).
For the identification of infected cells and structures, the following markers were used: GFAP (Dako, Zug, Switzerland) for astrocytes, CNPase (Chemicon International, Temecula, CA) for oligodendrocytes, NFP (Dako, Zug, Switzerland) for axons, MAP2 (Chemicon International, Temecula, CA) for myelin sheaths, and actin (Dako, Zug, Switzerland) for smooth muscle cells.
Subsequently, sections were washed and incubated with a secondary anti-mouse/anti-rabbit biotinylated antibody and then with streptavidin-peroxidase (both from the ChemMate kit, Dako, Zug, Switzerland) for 15 min each. Finally, the reaction was developed using AEC substrate (AEC-substrate chromogen kit, Zymed, San Francisco, CA).
A recently established TaqMan real-time reverse transcription polymerase chain reaction (RT-PCR) system (Applied Biosystems, Rotkreuz, Switzerland) was used to detect and quantify BDV nucleic acid. 17 For this purpose, the tissue samples (30 mg per 40 μl) were disrupted and homogenized with QIAshredder, and total RNA was subsequently isolated using the QIAamp Viral RNA Mini Kit (for skin and urine, the latter collected on blotting paper) and the RNeasy Mini Kit (for all other tissue samples; QIAGEN AG, Basel, Switzerland). Lysates were digested with 1 μl RNase-Free DNase (Promega, Madison, WI) for 1 hour at 37°C, followed by a deactivation step at 95°C for 5 minutes. Isolated and purified RNA was reverse transcribed into cDNA using the random primer Reverse Transcriptase System (Promega). Briefly, total RNA was incubated with random primer and 30 units of reverse transcriptase for 1 hour at 42°C. The enzyme was inactivated for 5 minutes at 95°C. The cDNA was stored at −20°C.
Quantification of the cDNA was carried out with specific double fluorescently labeled probes in a ABI Prism 7700 sequence detector (Applied Biosystems, Rotkreuz, Switzerland). TaqMan probes were labeled with FAM (5-carboxyfluorescein) as the 5′ fluorescent reporter and with TAMRA (6-carboxytetramethyl-rhodiamine) as the 3′ quencher. As an endogenous control and calibrator, eukaryotic 18S rRNA (Applied Biosystems, Foster City, CA) was amplified. The probe was labeled with VICTM dye -MGB (Applied Biosystems, Foster City, CA).
PCR amplification was carried out in 25-μl volumes per well containing 12.5 μl of TaqMan Universal PCR Master Mix (2×) (Applied Biosystems, Foster City, CA), 600 nM of each primer, 80 nM (p40 probe) and 160 nM (p24 probe) of the probes, and 3.5 μl of cDNA. The 18S rRNA was amplified as an internal control to monitor the quality of the extracted RNA. The same 18S rRNA primers/probe were used for the amplification of cDNA from MDCK cells and tissues. The PCR conditions were as follows: 2 minutes at 50°C to activate the uracil-N-glycosilase, which detects and degrades the former PCR products, and then 10 minutes at 95°C to inactivate the Uracil-N-Glycosilase and activate the polymerase, followed by 40 cycles of denaturation at 95°C for 15 seconds and annealing and elongation at 60°C for 1 minute. Known negative samples, as well as water, were always carried through the experiments as controls. The data were analyzed using sequence detector software V1.7 (PE Systems, Fairfax, VA). Absorbance was measured in a microplate reader at a wavelength of 492 nm. Signals were regarded as positive if the fluorescence intensity (increase of fluorescence of the reporter dye represented by ΔRn) exceeded 10 times the standard deviation of the baseline fluorescence (threshold cycle, CT). CT values ≥ 40 were regarded as negative.
Selected frozen-stored parallel samples—including brain specimens from all animals that proved BDV-positive in the Zurich laboratory, as well as a brain specimen of a BDV-positive sheep from the same pasture where the small mammals were trapped—were sent to the Vienna laboratory for independent confirmation by conventional RT-PCR and subsequent sequencing of the amplification products. RT-PCR and sequencing were carried out as described by Kolodziejek et al. 12 The resulting amplicons were sequenced by employing the ABI PRISM BigDye Terminator Cycle Sequencing Ready Reaction Kit (PE Applied Biosystems, Foster City, CA) and an ABI Prism 310 genetic analyzer (PE Applied Biosystems, Foster City, CA).
Results
With the exception of C. leucodon, all other species of small mammals trapped and analyzed during this study proved negative for BDV antigen and nucleic acid—including 6 S. araneus shrews. Two of 6 C. leucodon shrews, however, (Nos. 67 and 68) were BDV positive (1 adult female and 1 adult male). These 2 shrews were trapped in August 2006—more than 3.5 years later and approximately 20 km from the place where 3 other BDV-positive C. leucodon shrews had been collected. 10
In these 2 shrews, BDV antigen was detected in many tissues and with high intensity by immunohistochemistry. Both antibodies produced a similar staining pattern, although the signal was stronger with the p38/40 antibody. A range of organs—including the brain, heart, lung, liver, intestinal tract, kidney, genital tract, skin, and a longitudinal section through the skull—showed labeling of small intranuclear inclusion bodies. In addition, some intracytoplasmatic staining was noted, mainly in nerve tissue as well as in epithelial and parenchymal cells within the various organs (Figs. 1–3).
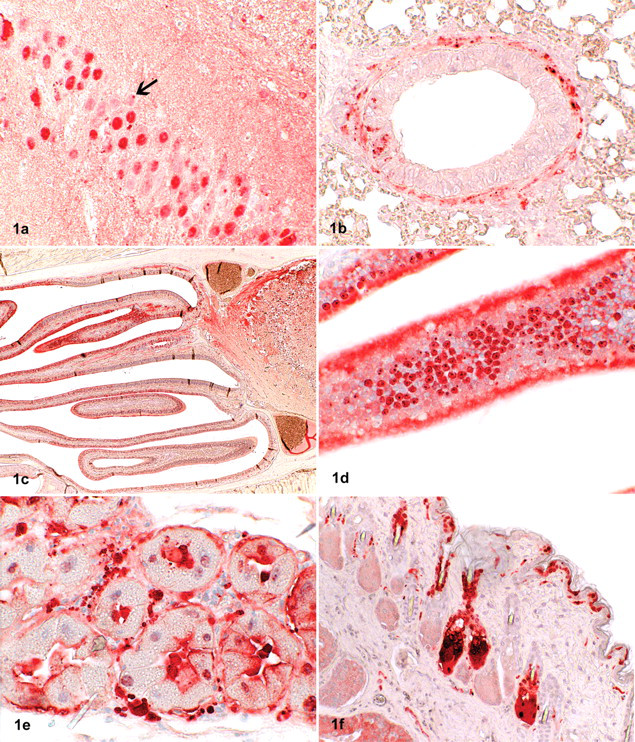
Demonstration of Borna disease virus (BDV) p24 antigen in tissues of bicolored white-toothed shrews (Crocidura leucodon) by immunohistochemistry (ChemMate method; Dako, Zug, Switzerland).
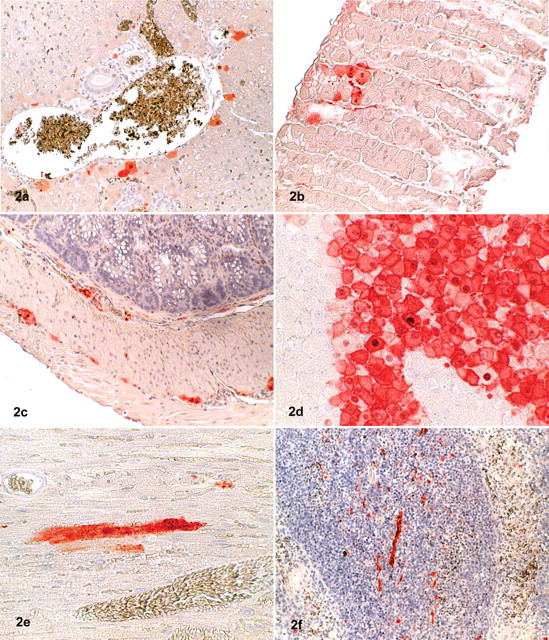
Demonstration of Borna disease virus (BDV) p24 antigen in tissues of shrews by immunohistochemistry (ChemMate method; Dako, Zug, Switzerland).
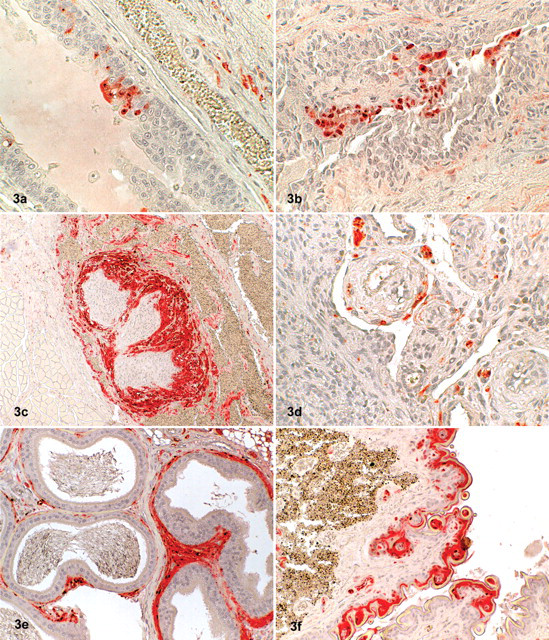
Demonstration of Borna disease virus (BDV) p24 antigen in tissues of shrews by immunohistochemistry (ChemMate method; Dako, Zug, Switzerland).
In the brain, high amounts of BDV antigen were detected in the cerebrum, hippocampus, cerebellum, and brain stem, mainly in neurons but also in oligodendrocytes, astrocytes, and neuropil and in Purkinje cells. Distinct labeling of the nuclei was observed, and some of the cells showed positive staining of Joest-Degen intranuclear inclusion bodies. Furthermore, a slight intracytoplasmatic staining was seen. Viral antigen was also present in the optic nerve but not within other ocular structures.
In many other organs—including the spleen, lung, liver, heart, kidney, salivary gland, skin, penis, testis, epididymis, stomach, intestine, urinary bladder, uterus, mammary gland, and adrenal—a distinct labeling of small nerves within the parenchyma and around blood vessels was observed. Virus antigen was also detected within parenchymal cells (tubular cells) of the kidneys, in hepatocytes close to the portal triad, in Leydig cells of the testicles, in myocardial cells, within epithelial cells of the epidermis and mucous membranes, in salivary glands (including saliva), in sebaceous glands, in cells of the transitional epithelium of the urethra, and in bronchiolar epithelial cells. The staining pattern of epithelial cells was either a distinct nuclear or a slight diffuse intracytoplasmatic labeling. Hepatocytes, myocardial fibers, peptic cells of the stomach, and Leydig cells exhibited intranuclear and/or diffuse intracytoplasmatic staining. Occasionally, intranuclear inclusion bodies could be identified in epithelial cells. Antigen was also detected in small nerves around blood vessels within the wall of the uterus and in the mammary gland. The adrenal gland exhibited a strong intracytoplasmatic and nuclear labeling of the medullary but not of the cortical cells (Table 1). Positive results were also obtained by TaqMan real-time RT-PCR; there was a good correlation of the intensity of staining seen in immunohistochemistry and the amount of viral RNA in the visceral organs and brain (Table 2) but not in the skin. Histological examination did not reveal any inflammatory or other pathological lesions, neither in the brain nor in the other organs investigated. Urine was collected from the shrews through a blotting paper placed on the bottom of the live traps, and it was subsequently examined by TaqMan real-time RT-PCR. The urine samples contained cells based on the 18s rRNA values, but no virus RNA could be detected.
Immunohistochemical Detection of Borna Disease Virus Antigen in Various Organs a
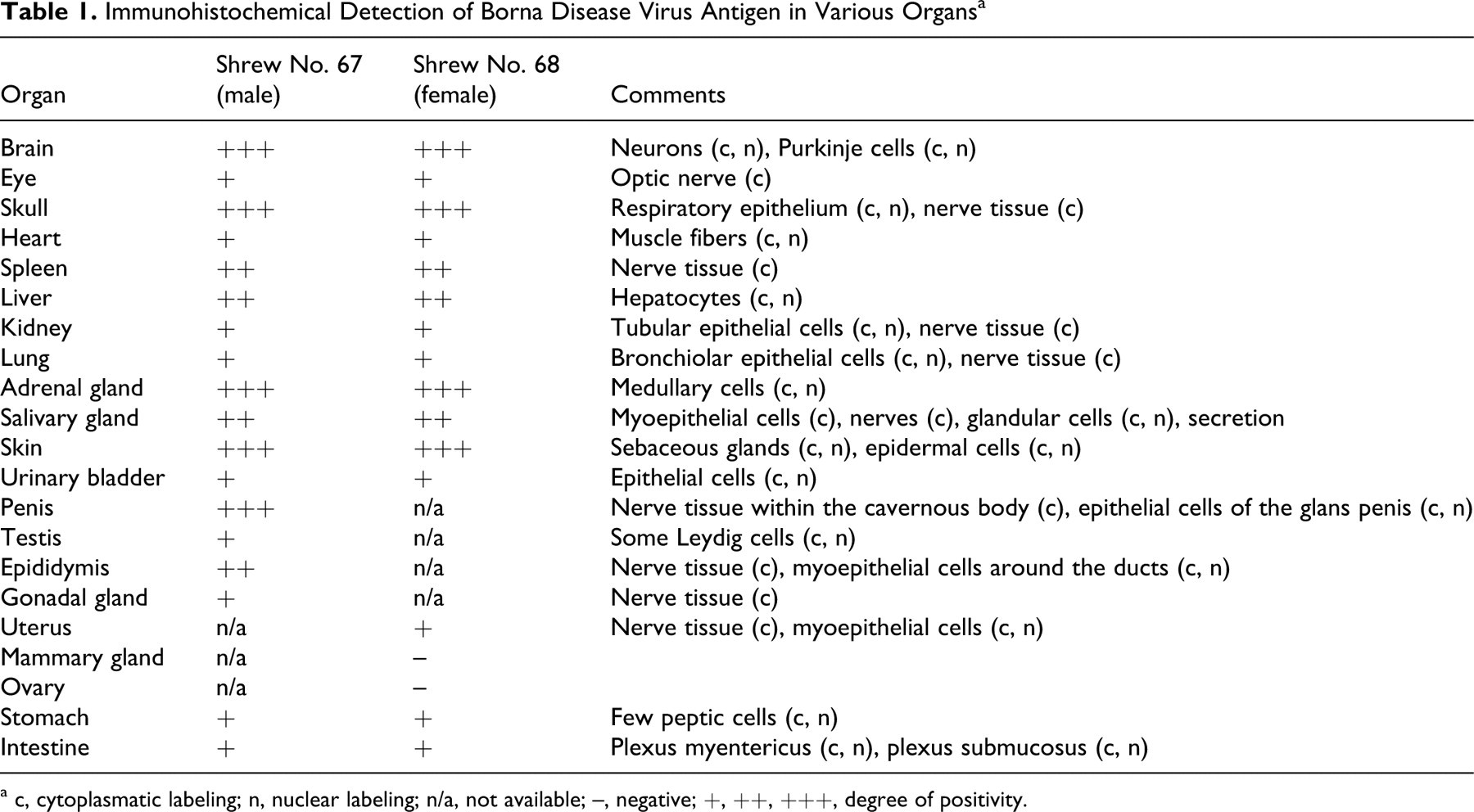
a c, cytoplasmatic labeling; n, nuclear labeling; n/a, not available; –, negative; +, ++, +++, degree of positivity.
Summary of Immunohistological and Real-Time Reverse Transcription Polymerase Chain Reaction Results a
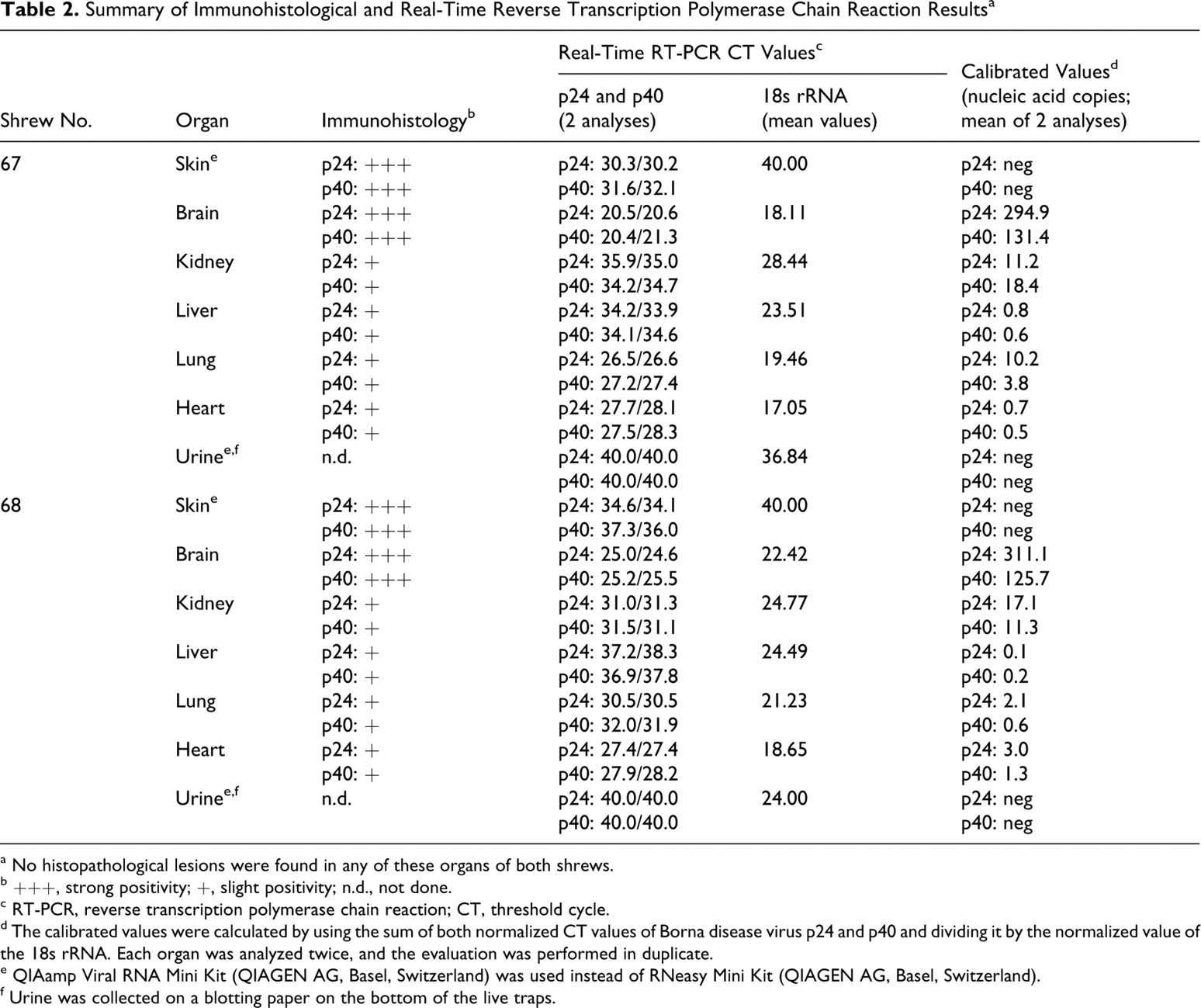
a No histopathological lesions were found in any of these organs of both shrews.
b +++, strong positivity; +, slight positivity; n.d., not done.
c RT-PCR, reverse transcription polymerase chain reaction; CT, threshold cycle.
d The calibrated values were calculated by using the sum of both normalized CT values of Borna disease virus p24 and p40 and dividing it by the normalized value of the 18s rRNA. Each organ was analyzed twice, and the evaluation was performed in duplicate.
e QIAamp Viral RNA Mini Kit (QIAGEN AG, Basel, Switzerland) was used instead of RNeasy Mini Kit (QIAGEN AG, Basel, Switzerland).
f Urine was collected on a blotting paper on the bottom of the live traps.
The TaqMan real-time RT-PCR results were confirmed by conventional RT-PCR. The compiled BDV sequences derived from the 2 C. leucodon shrews as well as the sequence derived from a sheep originating from the same pasture as the shrews resulted in a stretch of 1,824 nucleotides composing the complete N-, X-, and P-protein-encoding regions as well as the 5′-untranslated region of the X/P transcript of the BDV genome. All sequences were unique, however closely related to each other. The BDV sequence derived from the sheep (GenBank accession No. EU095836) differed from the sequence derived from shrew No. 68 (GenBank accession No. EU095835) by 3 nucleotides and from the sequence derived from shrew No. 67 (GenBank accession No. EU095834) by 9 nucleotides. Shrews Nos. 67 and 68 showed an 8-nucleotide difference. The sequence of shrew No. 67 exhibited 99.8% identity to a BDV sequence (GenBank accession No. AY374550) derived from a horse that died of BD and that originated from Jenins (Graubuenden, Switzerland, close to Seewis, where the shrews were trapped) and 99.7% identity to a BDV sequence (GenBank accession No. AY374544) derived from a horse from Malans (Graubuenden, Switzerland) that died of BD near the location where the shrews were trapped. Finally, the sequences of shrew No. 68 and the sheep showed 99.9% identity to a BDV sequence (GenBank accession No. AY374549) derived from a sheep, which originated from a village called St Peter (again Graubuenden, Switzerland).
Discussion
In contrast to the distribution of BDV antigen and RNA in horses and sheep suffering from classical BD (in which the virus is restricted to the central nervous system), C. leucodon shrews harbor BDV in most of the tissues examined, without evidence of pathological lesions. These findings support the concept that shrews of the species C. leucodon can be persistently infected by BDV and may therefore act as reservoirs and vectors of BDV, as postulated in a previous publication. 10 Up to now, we did not find evidence of BDV infection in any of the other small mammals captured, including mice and other shrew species. The absence of inflammatory lesions points toward immune tolerance or immune suppression. Experimental investigations demonstrated that infection of neonatal rats (having an immature immune system) with BDV results in persistent infection. By intracerebral infection of 1-day-old Lewis rats, no clinical signs became evident, and these rats did not produce BDV-specific antibodies, whereas animals inoculated at 1 or 2 months of age showed inflammation in the brain; as such, the latter rats were able to produce anti-BDV antibodies. 11,20,22
From experimental BDV infections, it is known that there are considerable species-specific differences regarding pathogenesis of the disease. A major difference between the well-established BDV experimental rat model and the mouse model is that infection of adults induces neurological disease in rats but not in mice. 7,9,20 Whereas infection of newborn rats leads to immunological tolerance and almost disease-free persistent infection, neonatal infection of mice, dependent on the strain used, induces severe neurological disease. 7
Consistent with neonatal rats with BDV infection, C. leucodon shrews show a widespread infection without evidence of severe pathological lesions. 21 Mild degenerative lesions in the cerebellar cortex and the hippocampus have been reported in neonatally infected rats, which could not be seen in the shrews' brains. 5,24 The question of whether shrews are devoid of BDV-specific antibodies could not be answered so far, owing to the very limited amount of blood available postmortem.
Therefore, the following questions remain open: First, are shrews really immunotolerant toward BDV? Second, at which age do shrews become infected such that they develop a persistent infection that allows them to survive the infection without clinical disease and without inflammatory lesions in the central nervous system or in other organs? Therefore, an intrauterine infection appears probable, based on the presence of viral antigen in reproductive organs such as the uterus. The presence of BDV antigen in testis and epididymis are further arguments that point toward the possibility of a sexual and vertical transmission. Vertical transmission of BDV has been reported in a pregnant mare and its foal, as well as in experimentally infected mice. 6,13 Yet, infection in the perinatal age—for instance, by the oronasal route—is also conceivable in analogy to neonatally infected rats.
Another unsolved question is the way of transmission from shrews to other susceptible hosts, such as horses and sheep. Based on the high similarity of BDV sequences obtained from shrews and equine and ovine BD cases, a transmission appears very likely. A possible way could be via excretions and secretions, such as urine, saliva, or nasal secretions. In C. leucodon shrews, we could detect BDV antigen and nucleic acid in organs and tissues, such as urinary bladder, salivary glands, and respiratory epithelium, which leads to the conclusion that BDV is excreted and secreted by these shrews. BDV antigen, by immunohistochemistry, was also present in considerable amounts in skin structures—mainly, sebaceous glands and deep epidermal cells. The low recovery rate of BDV RNA by RT-PCR from skin tissue was possibly due to the fact that, based on the 18s rRNA RT-PCR, practically no cells were extracted from skin tissue. Possibly, the extraction kit used was not effective enough for skin tissue.
The port of entry of BDV in final hosts such as horses and sheep is generally thought to be the olfactory system, most likely through contact with contaminated feed. Although indirect experimental evidence suggests this route, proof of this hypothesis is lacking. 16
In summary, the findings presented here support the hypothesis that the shrew species C. leucodon plays an important role as reservoir and vector species of BDV. The exact way of acquisition and transmission of the infection remains to be investigated.
Footnotes
Acknowledgements
We thank Mr and Mrs Burger, Grüsch, Switzerland, for their help and cooperation in trapping mice and shrews.
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
This investigation was financially supported by the Swiss Federal Veterinary Office, Bern, Switzerland.
