Abstract
Normal human aging and diabetes are associated with a gradual decrease of cerebral flow in the brain with changes in vascular architecture. Thickening of the capillary basement membrane and microvascular fibrosis are evident in the central nervous system of elderly and diabetic patients. Current findings assign a primary role to endothelial dysfunction as a cause of basement membrane (BM) thickening, while retinal alterations are considered to be a secondary cause of either ischemia or exudation. The aim of this study was to reveal any initial retinal alterations and variations in the BM of retinal capillaries during diabetes and aging as compared to healthy controls. Moreover, we investigated the potential role of vascular endothelial growth factor (VEGF) and pro-inflammatory cytokines in diabetic retina.
Transmission electron microscopy (TEM) was performed on 46 enucleated human eyes with particular attention to alterations of the retinal capillary wall and Müller glial cells. Inflammatory cytokines expression in the retina was investigated by immunohistochemistry.
Our electron microscopy findings demonstrated that thickening of the BM begins primarily at the level of the glial side of the retina during aging and diabetes. The Müller cells showed numerous cytoplasmic endosomes and highly electron-dense lysosomes which surrounded the retinal capillaries. Our study is the first to present morphological evidence that Müller cells start to deposit excessive BM material in retinal capillaries during aging and diabetes. Our results confirm the induction of pro-inflammatory cytokines TNF-α and IL-1β within the retina as a result of diabetes.
These observations strongly suggest that inflammatory cytokines and changes in the metabolism of Müller glial cells rather than changes in of endothelial cells may play a primary role in the alteration of retinal capillaries BM during aging and diabetes.
Keywords
Introduction
An increased thickness of the capillary basement membrane (BM) has been found in a variety of tissues during aging and as a consequence of systemic diseases, such as hypertension and diabetes. 1 BM plays an important role in the regulation of capillary permeability and the thickening of these structures may alter the exchange of substances between the internal and external environment of the capillaries. Normal human aging is associated with a gradual decrease in cerebral blood flow due to modifications of vascular architecture, vessel walls, regulation of blood flow, or vascular plasticity and in the concentration of neurotransmitters due to a reduction of the metabolism in general. 2 These morphological abnormalities may reduce the blood supply to the brain parenchyma. The retina is an extension of the central nervous system (CNS) and extensive evidence suggests that the retinal vessels present age-related abnormalities similar to those found in the brain, such as thickening of the capillary BM and an increase in the incidence of microaneurysms, hemorrhages, and exudates. 3 Diabetic disease may also cause retinal damage that results in severe loss of vision in adults between the second and seventh decades of life in industrialized countries. 4 Despite the advances in medical care, therapeutic approaches are still inadequate, awing to the complex etiology of this disease. The risk of developing diabetic retinopathy increases with age and retinopathic changes are appreciable in 98% of patients with type 1 diabetes and 78% with type 2 diabetes. In addition, 33% of patients with type 1 diabetes and 17% of those with type 2 diabetes will also develop proliferative diabetic retinopathy (PDR): this occurs after neovascularization of the retina or iris, followed by fibrocontractive processes that cause retinal detachment and blindness. Some of the most visible ophthalmoscopic signs in the diabetic retina are vascular lesions (microaneurysms, capillary degeneration, vessel growth, increased vascular permeability, hemorrhages) and neuronal cell death, alterations in glial cell structure and function, and inflammation. 5 Thickening of capillary BM is well documented both in human diabetics as well as in animal models of experimental diabetes.6,7 Other authors have previously established that diabetes-induced peroxynitrite formation and inflammation are the major molecular mechanisms responsible for retinal endothelial dysfunction and vascular cell death.8,9 This diabetic retinopathy involves a chronic, low-grade inflammatory component. 10 Elevated cytokines activate microglia, thereby stimulating a cycle of inflammation that recruits leukocytes, causes vascular breakdown, and directly induces cell death through the release of cytotoxic substances. 11
Some studies suggest that diabetic retinopathy has many characteristics in common with low-grade inflammatory diseases. The levels of pro-inflammatory cytokines are increased in the diabetic retina and vitreous body, the retinal capillaries become non-perfused and ischemic with an increased number of platelet-fibrin thrombi. 12 Different molecular and cellular alterations typical of inflammatory processes have been observed in the retinas of diabetic animals and patients. Different evidence supports the idea according to which the DR is a manifestation of a chronic low-level inflammation in which different effectors, e.g. the cytokines and the leukocytes, are responsible for retinal damage. 13 Diabetic retinopathy presents many similarities to chronic inflammatory diseases: increase in vascular permeability, edema, infiltration of inflammatory cells, tissue destruction, neovascularization, and retinal expression of inflammatory cytokines. Recent publications present strong evidence of an important inflammatory involvement even in the early stages of DR. 14 It has been demonstrated that, both in animals and patients with diabetes, the leukocytes, including monocytes/macrophages (microglia), neutrophils, and a certain number of lymphocytes, show adhesion to the retinal vascular endothelium. This occurs simultaneously with impairment damage of the blood–retina barrier, capillary occlusion, and death of the endothelial cells. The expression of adhesion molecules, particularly of ICAM-1 (intracellular adhesion molecule-1) and VCAM-1 (vascular cell adhesion molecule-1), appears increased in the diabetic retina and is connected with an increased leukocyte adhesion and leukostasis. 15 Inflammation contributes to the development and the progression of retinopathy, as it is demonstrated by the fact that intravenous administration of glucocorticoids and anti-VEGF drugs may improve the clinical conditions. 16 Growth factors, neurotrophic factors, and hormones may alter the levels of certain molecules that have been found in diabetic subjects: insulin, IGF-1 (insulin-like growth factor-1), aldosterone, adrenomedullin, GH, endothelin, VEGF, PEDF, FGF (fibroblast growth factor), and TGF-β (transforming growth factor-β). These elements are all responsible for the structural changes of retinal vascularization (angiogenesis, ischemia, and increased vessel permeability) and for the progression of DR. 13 Neurotrophic factors such as insulin, PEDF, CNTF (ciliary neurotrophic factor), NGF (nerve growth factor), GDNF (glial cell line-derived neurotrophic factor), and BDNF (brain-derived growth factor), all of which appear altered in the diabetic subject, are also responsible for retinal neuronal damage. 13
Intraocular levels of inflammatory factors, which are expressed by alterations of migrating leukocytes, glial cells, and vascular endothelial cells, were significantly correlated with the severity of DR, suggesting they play an important role in the occurrence of macular edema. The structure of retinal vessels has been well described by many authors.17,18 The continuous lining of endothelial cells lies upon a homogeneous basal lamina, enclosing muscle cells or pericytes, which is particularly prominent where it separates the vessel from the nervous tissue. The outer basement membrane consists of two layers: the basal lamina of the outer vascular cells-mesodermal basement lamina (MBL) and that of the Müller cells-ectodermal basement lamina (EBL). Thickening of the perivascular basement membrane has been shown to develop with advancing age 19 and appears to be related to hydrostatic pressure, as well as to various pathological conditions, such as oxygen-poisoning, hypertension, myxoedema, polymyositis, and lupus erythematosus. 20 The various pathological manifestations of retinopathy, such as microaneurysms, zones of capillary non-perfusion, pericyte cell death, and thickening of vascular BM have been consistently reported.6,7 Of all these changes, BM thickening has received the most attention, being the major and earliest morphological characteristic of diabetic microangiopathy, not only in the retina, but also in the kidneys, 21 muscle, 22 and skin 23 of humans and diabetic animals. Several studies have sought to measure BM thickening of the retinal capillaries and have quantitatively confirmed BM thickening during diabetes. 24 These studies found significant differences in the BM thickness of capillaries from the different retinal layers in normal rats: capillaries in the nerve fiber layer (NFL) showed significantly thicker BM than capillaries located within either the inner plexiform layer (IPL) or the outer plexiform layer (OPL). Furthermore, a study on diabetic dogs showed that retinal capillaries located in an arterial environment had significantly thicker BM when compared to those of capillaries found in a venous environment. 25 Müller cells, the principal retinal glial cells, play an important causal role in the development of diabetic retinopathy. Müller cells may respond to retinal injury, insults, or changes by an altered expression of cytoskeletal proteins and enzymes, cellular hypertrophy, proliferation, and migration in the vitreous body. 26 Studies of Müller cells in animal models and tissue culture revealed the capacity of this cell type to generate tractional forces and cause retinal detachment. 27 Thickening of the capillary basement membrane, appearance of acellular capillary remnants, ultrastructural signs of pericyte degradation, and vascular tortuosity have also been observed in the aged rat retina. Pericytes, the mural cells on capillaries, are thought to play a role in the vessel stabilization (regulating endothelial cell proliferation and preventing capillary withdrawal) 28 and in the regulation of capillary blood flow. 29 Alterations in these cells with age and diabetes may contribute to the development of morphological and physiological abnormalities of the microvasculature. Close contact between pericytes and endothelial cells is necessary for maintaining vascular stability. 30 In the rat retina, during aging and diabetes, ultrastructural observations have shown that the decrease of pericytes and the alter pericyte-endothelial relationship may cause vessel instability. 31
The aim of this study was to identify changes of the retinal capillaries in humans during aging and diabetes. Immunohistochemical technique was employed to investigate the expression of growth factors and pro-inflammatory cytokines (ICAM-1, VEGF, tumor necrosis factor [TNF]-α, interleukin-1 [IL-1β], and interleukin-6 [IL-6]) involved in the pathogenesis of DR. In the present study, using TEM, retinal alterations were investigated, playing particular attention to alterations of the retinal capillary wall (BM and pericytes) and Müller glial cells in type 2 diabetes as compared to age-matched healthy controls.
Materials and methods
Forty-six enucleated human eyes (age range, 6–83 years) were selected for these electron microscopic studies. Samples of retina were harvested from the left eye during autopsies. Postmortem phenomena may produce early morphologic modifications of the eye structure, therefore our samples were harvested as early as possible after death in Hungarian hospitals (within 24 h). All experiments were conducted in accordance with the Declaration of Helsinki (1964) and in conformity with the ARVO Statement for use of human samples in Ophthalmic and Vision Research. The Ethical Committees of the hospitals involved gave their approval and the relatives of the dead humans gave their written informed consent. Four eyes, aged 6, 62, 81, and 83 years, were used only for qualitative analysis of age-related changes. Twenty-one of the eyes were harvested from patients suffering from type 2 diabetes (age range, 42–87 years; mean age, 70.9 years; 20 women, 11 men) and another 21 were used as age-matched controls for both qualitative and quantitative morphometric studies. These samples were shipped to our laboratory in 0.1 M Tris-buffered saline with protease inhibitors (0.025 M ethylenediaminetetraacetic acid [EDTA], 0.001 M benzamidine hydrochloride, 0.001 M phenylmethysulfonyl fluoride [PMSF], 0.01 M N-ethylmaleimide [NEM]) on wet ice. All tissues used in the current study were from patients with known type 2 diabetes or age-matched controls (individuals with no prior history of disease). Retinas were removed from whole eyes and posterior eye cups and were immersed overnight in 2–5% glutaraldehyde (in 100 mmol/L phosphate buffer pH 7.4). The retina was postfixed in 1% buffered osmium tetroxide and dehydrated using graded ethanol solutions. Ultrathin sections (0.5 µm) of selected areas were then prepared and stained with uranyl acetate and lead citrate for transmission electron microscope (H7650TEM, Hitachi, Tokyo, Japan). Capillaries were then photographed at magnifications in the range of 8.000–24.000, the images transferred to an image analysis system and calibrated according to magnification. The retinal layer in which each capillary was located, the NFL, the IPL, and the OPL were also recorded. The method used to measure the two-dimensional (2D) thickness of the retinal capillary BM has been previously described in detail by Stitt et al. 18 However, a brief description will also be given below. A 2D grid of lines 15 mm apart was superimposed over each capillary profile on the image analyzer and, at the points where a grid line intercepted the BM, the shortest distance across the BM was measured. This method resulted in approximately 25–30 measurements of BM thickness per capillary and these values were then used to determine the mean BM thickness for each individual capillary sampled. The results were then analyzed by a two-way analysis of variance (ANOVA) as well as by a two tailed Student’s t-test.
Immunohistochemical analysis
Control morphological sections were stained with hematoxylin-eosin stain (not shown). The following molecules were investigated: VEGF, IL-1β, IL-6, TNF-α, and ICAM-1. Small fragments of retina were washed in PBS, fixed in 10% formalin, and embedded in paraffin according to a standard procedure. The method employed for immunohistochemical tests was ABC/HRP technique (avidin complexed with biotinylated peroxidase). Serial 3-μm thick sections were cut using a rotative microtome, mounted on gelatin-coated slides, and processed for immunohistochemistry. These sections were deparaffinized in xylene and dehydrated. They were immersed in citrate buffer (pH 6) and subjected to microwave irradiation twice for 5 min (750 Watts). Subsequently, all sections were treated for 30 min with 0.3% hydrogen peroxide in methanol to quench endogenous peroxidase activity. To block non-specific binding, the slides were incubated in 6% normal goat serum in PBS for 30 min at room temperature. The slides were incubated overnight at 4°C with primary mouse monoclonal antibodies (dilution: 1/100) against human VEGF (Santa Cruz, sc-53462), against human TNF-α (Santa Cruz, sc-52746), against human ICAM-1 (Santa Cruz, sc-107), and with primary rabbit polyclonal antibody against human IL-1β (Santa Cruz, sc-7884) and against human IL-6 (Santa Cruz, sc-7920). Optimal antisera dilutions and incubation times were assessed in a series of preliminary experiments. After exposure to the primary antibodies, slides were rinsed twice in phosphate buffer and incubated for 1 h at room temperature with the appropriate secondary biotinylated goat anti-mouse or anti-rabbit IgG (Vector Laboratories, Burlingame, CA, USA; BA9200 and BA1000) and with peroxidase-conjugated avidin (Vector Laboratories, Burlingame, CA, USA; Vectastain Elite ABC Kit Standard* PK 6-100) for 30 min. After a further wash with phosphate buffer, slides were treated with 0.05% 3,3-diaminobenzidine (DAB) and 0.1% H2O2. Finally, sections were counterstained with Mayer’s hematoxylin and observed using a light microscope. Negative control experiments were done: (1) by omitting the primary antibody; (2) by substituting the primary antibody with an equivalent amount of non-specific immunoglobulins; and (3) by pre-incubating the primary antibody with the specific blocking peptide (antigen/antibody=5 according to supplier’s instructions). The staining assessment was made by two experienced observers in light microscopy. We assessed the immunoreactivity for VEGF, IL-6, IL-1β, TNF-α, and ICAM-1 in the retina. The intensity of the immune reaction was assessed microdensitometrically using an IAS 2000 image analyzer (Delta Sistemi, Rome, Italy) connected via a TV camera to the microscope. The system was calibrated taking the background obtained in sections exposed to non-immune serum as zero. Ten 100 µm 2 areas were delineated in each section by measuring the diaphragm.
Results
Transmission electron microscopy
Transmission electron microscopy (TEM) examination of the retinas from diabetic patients showed increased thickening of the capillary BM when compared with those of the control patients. The most relevant thickening of the BM was observed in diabetic capillaries, but it was similar to that due to aging. TEM examination on the cross-section through normal human retinal capillaries showed that they comprise inner continuous layers of endothelial cells surrounded by discontinuous processes of intramural pericytes. Retinal capillaries are formed by endothelial cells, pericytes, and BM. The BM consisted of: an inner subendothelial basement membrane (IBM) interposed between endothelial cells and pericytes; an external basement membrane (EBM) placed between pericytes and Müller glial cells (Figure 1a). In some areas the external basement membrane (EBM) showed double contours corresponding to endothelial-pericyte contact sites (Figure 1b). In retinal capillaries from control patients, numerous mitochondria and cytoplasmic organelles are detected both in endothelial cells and pericytes (Figure 1c), while in other areas in the EBM numerous vacuoles were observed (Figure 1d). We identified the presence of another contact site between an endothelial cell and pericyte together with a pinocytotic vesicle indicating a direct cytoplasmic communication between these cells (Figure 1c). In addition, the perivascular connective tissue space consisted of a basement membrane-like pericytic matrix (PMC) and numerous collagen fibrils. Normal adult human retinal capillaries (age range, 62–83 years) showed that the endothelial cell structure is well preserved while pericytes form an incomplete layer. A thin IBM between endothelial cells and pericytes may be seen all over. There was marked evidence of significant thickening of the EBM (Figure 2a). Endothelial cells contained numerous endosomes and cytoplasmic organelles while the thin cytoplasm of pericytes showed a poor content of organelles (Figure 2c). The endothelial cell-pericyte contact site may be observed (Figure 2b). Differently, the EBM presented very evident thickening and contained some rudimentary processes of pericytes (Figure 2a, b). In addition, the EBM showed some vacuoles or “empty” areas and electro-dense granules (Figure 2c). Furthermore, we detected some irregularities of the border between the EBM and Müller cells. TEM examination of retinas removed from type 2 diabetic patients showed increased thickening of the capillary BM when compared with those of the control patients. An extreme thickening of the BM was observed in diabetic capillaries located in the NFL close to the inner limiting membrane. In diabetic patients, the microvascular extracellular matrix (ECM) subcomponents, including the BM layer, are variously altered with loss of pericytes. The BM was vacuolated and contained osmophilic and cellular debris (probably resulting from degenerative pericytes) and occasionally reticulin fibers. The changes in the walls of capillaries were of a different nature and the greatly thickened laminated BM contained, in addition, blood elements such as lipid, platelets, and red cells. The pericytes within the wall were not usually involved in the thickening seen in the membrane around the vessel. The endothelial cells and pericytes had a normal appearance and there was a thin IBM, but the EBM containing some electro-dense granules and vacuoles was significantly thickened (Figure 3). The external membrane formed root-like extensions deeply inserted between Müller cells (Figure 4). An irregular border zone between the BM and Müller cells may also be observed. The data concerning the thickening of capillary basement membrane during aging and diabetes are provided in detail in Tables 1 and 2. Thickening of the BM of retinal capillaries during aging are shown in Table 1 with P value = 0.0004; thickening of the BM of retinal capillaries during diabetes are shown in Table 2 with P value = 0.0001.
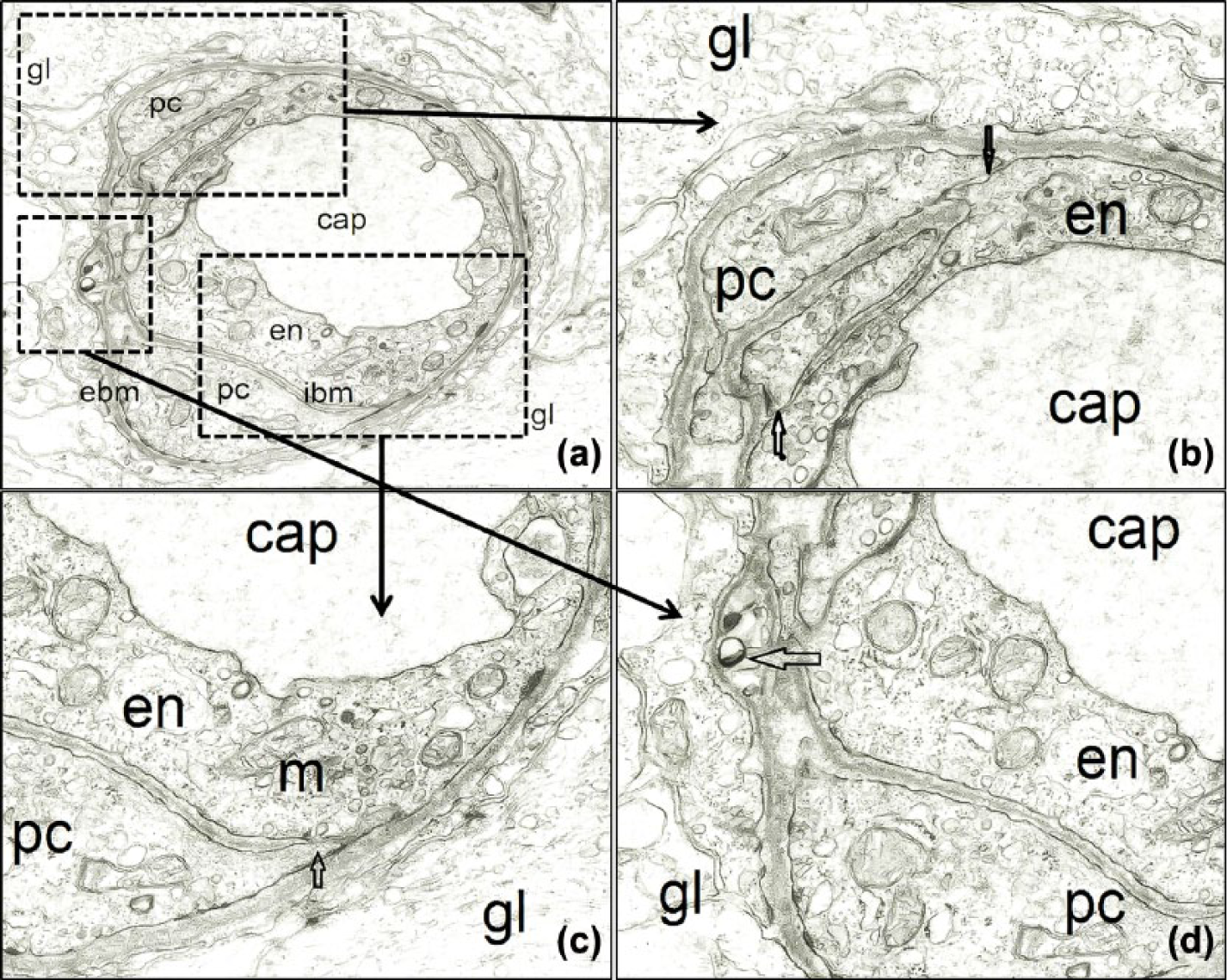
The retinal capillary wall of a 6-year-old boy. (a) Retinal capillaries (cap) are formed by two distinct layers: the inner layer (ibm) placed between the endothelial cells (en) and the pericytes (pc), and the external layer (ebm) interposed between the pericytes and the Müller glial cells (gl). Magnification 8000×. (b) Endothelial cells-pericyte contact sites (arrows). Magnification 24,000×. (c) Another contact site between pericyte (pc) and endothelial cell (en) with a pinocytotic vesicle indicating direct cytoplasmic communication between these cells. Both endothelial cells and pericytes have numerous mitochondria (m) and other cytoplasmic organelles. Magnification 24,000×. (d) In some places, we can see vacuoles in the external basement membrane (ebm). Magnification 24,000×.

The retinal capillary wall of an 81-year-old man. (a) The structure of endothelial cells (en) is well preserved. Pericytes (PC) form a discontinuous layer around the circumference of retinal capillary (cap) and a thin internal basement membrane can be seen between endothelial cells and pericytes. Significant thickening of the external basement membrane (ebm). Magnification 8000×. (b) Endothelial cell-pericyte contact site (arrow) can be identified in a photo at higher magnification. External basement membrane (ebm) contains some rudimentary processes of pericytes. Magnification 24,000×. (c) Endothelial cells (en) show numerous endosomes and cytoplasmatic organelles, while the cytoplasm of pericytes (pc) presents few organelles. The external basement membrane (ebm) contains some vacuoles (black arrowheads). The contact site between endothelial cell and pericyte can be observed (arrow). Magnification 24,000×. (d) The border between external basement membrane (ebm) and Müller cells (gl) is clearly visible. The external basement membrane (ebm) contains some vacuoles (black arrowheads). Magnification 24,000×.

Retinal capillary wall of a 62-year-old man affected by diabetes. Normal appearance of endothelial cells (en) and pericytes (pc). Internal basement membrane (ibm) is very thin and the external basement membrane (ebm) is significantly thickened with some electro-dense granules and vacuoles. Note that the border between basement membrane and Müller cells and adjacent Müller cells (star) is irregular. Magnification 20,000×.

Retinal capillary wall of an 83-year-old woman affected by diabetes. External basement membrane is significantly thickened and root-like extensions formed deeply in between Müller cells. Magnification 20,000×.
Thickening of the BM of retinal capillaries during aging.

P value = 0.0004. The results were considered statistically significant when P <0.01.
Thickening of the BM of retinal capillaries during diabetes.

The results were considered statistically significant when P <0.01.
Immunohistochemistry
Sections of diabetic retina exposed to the primary/secondary antibodies, developed a dark-brown (intense), yellow-brown (slight), or no appreciable immunostaining. In diabetic retina, VEGF immunoreactivity was observed in several types of cells (Figure 5a, b), including retinal pigment epithelial cells, retinal microvascular endothelial cells, retinal pericytes, and Müller cells in cells infiltrating the subretinal space, particularly those in highly vascularized regions (Figure 5c). VEGF immunoreactivity was observed in the blood vessels situated in the diabetic and control retina but the numbers of blood vessels expressing VEGF were significantly higher in diabetic retina than in the control retina, owing to the fact that microvascular complications in diabetic patients are caused by prolonged exposure to high glucose levels. Weak evidence of cytoplasmic immunoreactivity for VEGF was noted in the inner nuclear layer of the control retina (Figure 5d).

Immunohistochemical staining of human retina for vascular endothelial growth factor (VEGF). In diabetic retina (a, b) VEGF immunoreactivity was observed in retinal pigment epithelial cells, retinal microvascular endothelial cells, retinal pericytes, and Müller cells. As it is visible in (c) VEGF immunostaining has been found in vascularized areas (boxed area). VEGF immunoreactivity was weakly noted in the inner nuclear layer in the normal control retina (d) (magnification 40×).
Intensive IL-1β staining was observed in the vascular endothelium as a direct consequence of chronic hyperglycemia and in the inner nuclear layer, where most of the cells expressed inflammatory cytokines. In addition, there were positive cells in the outer nuclear layer of the retina and in the ganglion cell layer cells and retinal pigment epithelium cells (Figure 6a). No IL-1β immunoreactivity was detected in the negative control sample (Figure 6b).
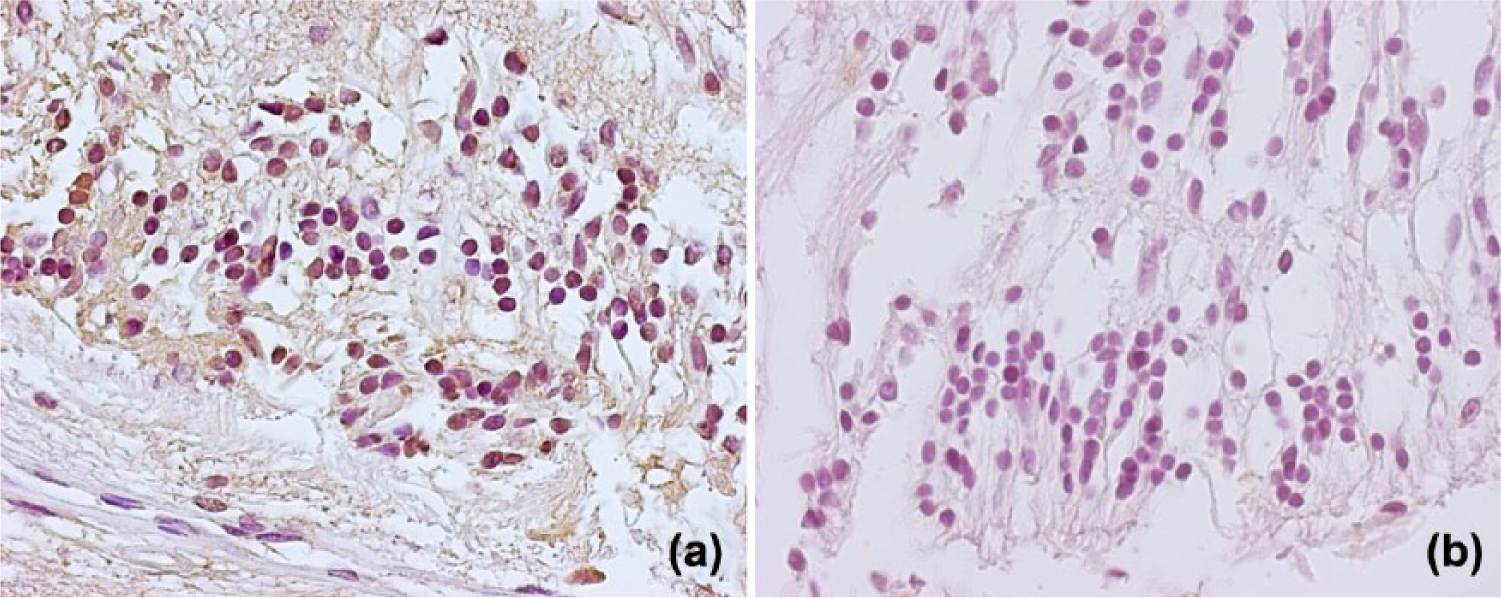
Immunohistochemical staining of human retina for interleukin-1beta (IL-1β). In diabetic retina (a) immunostaining for IL-1β was observed in the inner nuclear layer and in the retinal pigment epithelium. No immunoreactivity for IL-1β was detected in the negative control sample (b) (magnification 40×).
Subsequently, we performed immunohistochemistry for IL-6 and compared the expression level in the RPE layer between the diabetic and control retina. We found that IL-6 immunoreactivity was stronger in RPE cells from diabetic donor eyes (Figure 7a) compared with those from control eyes (Figure 7b).

Immunohistochemical staining of human retina for interleukin-6 (IL-6). Immunoreactivity for IL-6 was stronger in RPE cells from diabetic eyes (a). IL-6 immunoreactivity was not observed in the normal retina (b) (magnification 40×).
Regarding immunostaining for TNF-α, faint immunostaining was barely detectable in the control retina, and was confined to a few glial cells and their processes and blood vessels (Figure 8d). However, the intensity of the immunostaining and the number of stained cells were noticeably greater in retina sections from diabetic eyes (Figure 8a–c). Based on the morphologic assessment, the immunostaining for TNF-α in diabetic eyes was mostly associated with glial cells located in the nerve fiber and retinal ganglion cell layers (Figure 8b, c). For example, at the level of light microscopy, Müller cells are characterized by their radial orientation, and astrocytes are characterized by their darker, smaller, and irregular nucleus relative to that of ganglion cells, and by their close localization to the blood vessels of the inner retina (Figure 8c). Immunohistochemical examination of retinal sections from control eyes revealed a clearly detectable immunostaining for TNF-α that was limited to a few glial cells and their processes (Figure 8d). In diabetic eyes, the intensity of the immunostaining and the number of stained cells for TNF-α were notably greater than that in normal eyes. Positive immunostaining for TNF-α receptor-1 in retinal sections from diabetic eyes was detectable in the cytoplasm as well as on the cell surface (Figure 5b). In addition to the faint immunostaining observed in all retinal layers, which was probably associated with glial cell processes or blood vessels, immunostaining for TNF-α was predominant in most large cell bodies in the retinal ganglion cell layer.

Immunohistochemical analysis of human retina for TNF-α. In diabetic retinas immunoreactivity for TNF-α was observed in the glial cells (a), in the nerve fiber and retinal ganglion cell layers (b, c). In control eyes immunostaining for TNF-α was observed in few glial cells (d). Magnification 40×.
Using the supply of diabetic and non-diabetic ocular tissue that was at our disposal, we sought to examine the localization of ICAM-1 in the diabetic retina compared to the non-diabetic retina (Figure 9). As shown in Figure 9a, diabetic retinas showed high levels of ICAM-1 expression, which was localized in the blood vessels of retina including the nerve fiber and outer-plexiform layers. Only faint immunoreactivity was seen in the retinas from control eyes (Figure 9b).

Immunohistochemical analysis of human retina for intracellular adhesion molecule-1 (ICAM-1). In diabetic retinas (a) immunoreactivity for ICAM-1 was localized in the inner layer and in blood vessels of the retina. Only faint immunoreactivity was seen in the retinas from control eyes (b). Magnification 40×.
The expression of ICAM-1 is increased early in diabetic retinopathy and has been associated with increased TNF-α, leukostasis, and subsequent endothelial cell death, characteristic of the disease. These findings suggest that cytokines and the resulting neuroinflammation cause several of the hallmark features of diabetic retinopathy.
Our results demonstrate a novel interaction which may occur between the production of a pro-inflammatory cytokine and VEGF involvement in diabetes. Immunohistochemical results for growth factors and pro-inflammatory cytokines are shown in Table 3; the results of immunopositivity were confirmed by statistical analysis (Figure 10). There were statistical differences between the histological of diabetic retina and control retina in terms of immunoreactivity for growth factors and pro-inflammatory cytokines.
Expression levels of VEGF, IL-1β, IL-6, TNF-α, and ICAM in DR and control specimens, and respective levels of statistical significance (t-test).
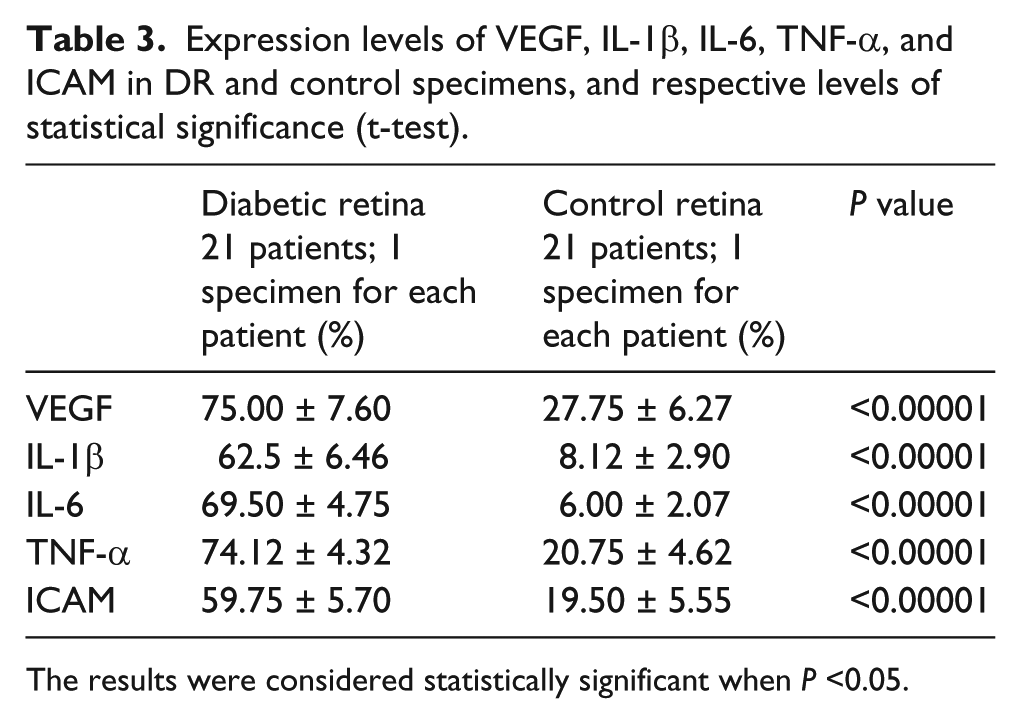
The results were considered statistically significant when P <0.05.
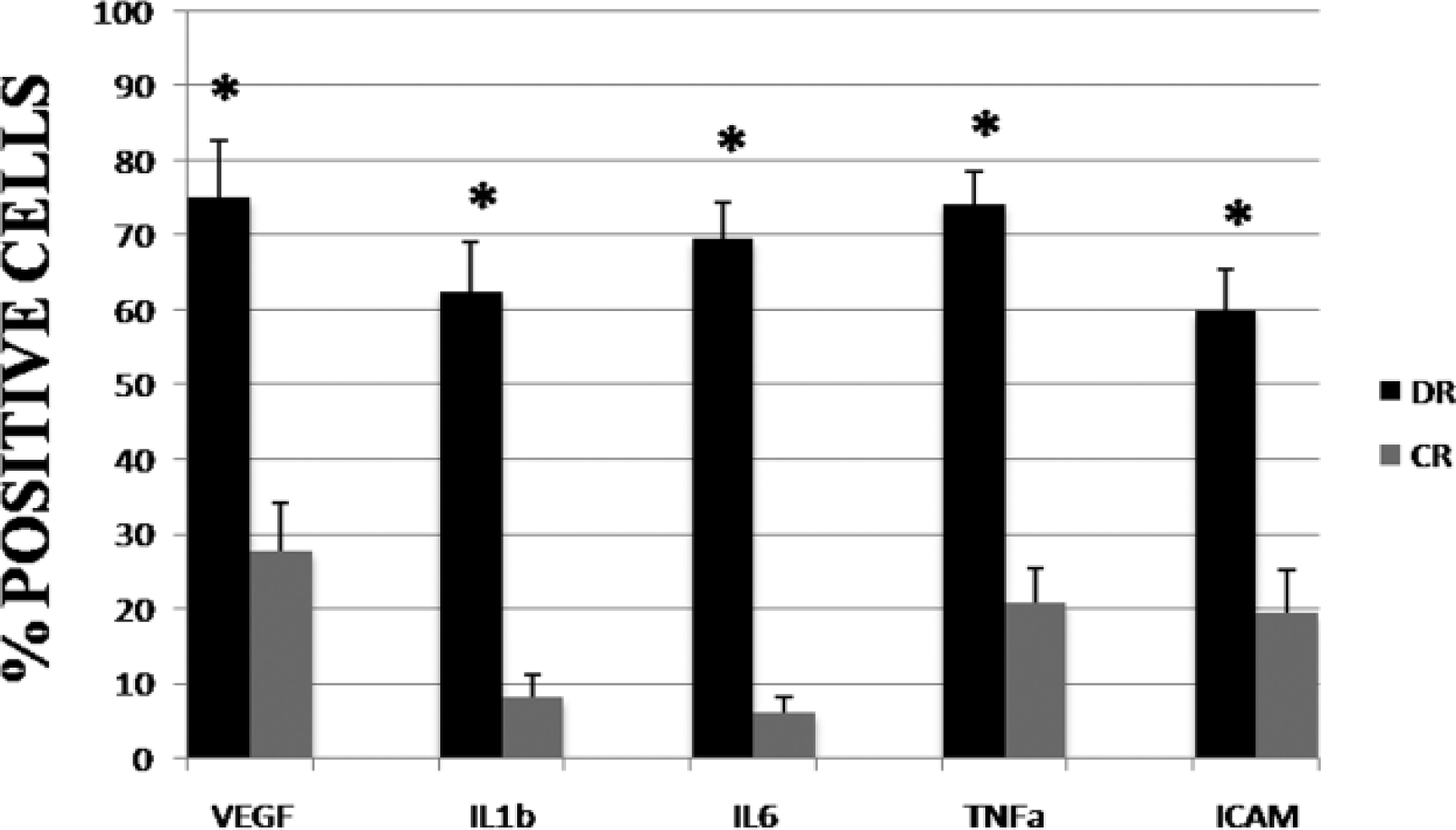
Pro-inflammatory cytokines and VEGF positive cell index in the diabetic and control retina. The bar graph indicates the mean % positive cells to growth factors and proinflammatory cytokines ± SD. Statistical analysis is performed using Student’s t-test. *P <0.05.
Control sections in which the primary antibodies were omitted or replaced with non-immune sera were all negative for specific immunostaining.
Discussion
BM thickening is one of the most widely studied morphological changes occurring in the microvascular system during diabetes and aging. It is not clear which of the many factors really contribute to the development of the vascular disease. A number of factors have been implied to contribute to an increase of BM thickening during aging and diabetes. It has been proposed that increased polyol pathway activity within the microvascular cells 32 may lead to damaging accumulations of intracellular polyalcohols in vascular cells and changes in the activities of enzymes involved in BM synthesis or breakdown. 33 There is also evidence of reduced activity of the enzymes involved in BM catabolism 33 and increased activity of enzymes involved in BM synthesis. Susceptibility to proteolytic resorption of BM may also be altered in diabetic patients through non-enzymatic glycosylation and associated oxidative modification of the basal membrane proteins. 34 The occurrence of capillary BM thickening in a wide variety of tissues suggests that this change in the capillary wall may be related to a deterioration of some tissue functions during senescence and disease. Capillary BM thickening, secondary to both diabetes and aging, is accompanied by pathological changes in the cellular components of the capillary wall. In diabetes, there is a selective loss of retinal capillary pericytes 35 while, during senescence, both pericytes and endothelial cells are lost, so that certain areas of the retina become avascular. This may underline the age-related loss of neural elements from the retina. 36
Our study has revealed that in adult humans, with increased age, vacuoles accumulated in the BM of retinal capillaries on the glial interface, demonstrating the existence of a statistically highly significant correlation between the amount of vacuolization and patient age. The fact that the vacuolization occurred in all of the samples we tested and that it was increased with age, strongly suggests that it was attributable to the normal aging process. Previous studies have shown that capillaries in mature animals of several species (monkeys, dogs, cats, and rats) do not present vacuoles in the BM as seen in adult humans. 37 It is therefore possible that the process of vacuolization concerning the BM might be a phenomenon specific in humans. It is not clear by what mechanism BM vacuoles are created and how the hypoxic stress might be a contributing factor. 38 It may be also possible that the vacuoles are remnants of degenerated pericytes. This would imply a turnover of degenerating and regenerating pericytes throughout the human lifespan, which could also explain the multiple BM sheaths. 39 We identified changes in the morphology of pericytes with a reduction in coverage of endothelium in the early stages of retinal vascular aging. Close contact between pericytes and the endothelium is thought to be necessary for vascular stability. 30 Our findings of reduced pericyte coverage and BM thickening may limit communication between endothelial cells and pericytes and thus contribute to the progressive age-related vessel instability. Pericytes also regulate endothelial cell proliferation. The vascular abnormalities such as spiraling and looping observed in the present study are suggestive of reduced contacts between pericytes and endothelial cells, combined with an increased level of VEGF expression. 31 The development of retinal vascular lesions is associated with the normal progression of aging, and this process has a striking similarity to the development of retinal vascular lesions observed in the pathogenesis of diabetic retinopathy. Interestingly, apoptosis was determined to be the underlying mechanism for the loss of endothelial cells and pericytes. Our studies indicate that the development of retinal vascular lesions shows histological and pathogenetic similarities both in the elderly and diabetics. Therefore, diabetes is considered a metabolic disease that accelerates the progression of aging through the development of vascular BM thickening in retinal and glomerular capillaries, 40 secretion of specific glycoproteins, 41 free radical formation resulting in oxidative stress, decline in cognitive function, 42 and BM thickening in muscle.
It has recently been suggested that the combination of glycation and oxidation may be very important in protein modification of the extracellular matrix occurring in diabetes and aging. Our work confirms pathological changes of the retinal capillary wall during aging and diabetes. In diabetes, there is a selective loss of pericytes in the retinal capillary, while during senescence, both pericytes and endothelial cells are lost, leaving certain areas of the retina as avascular. This confirms age- and diabetes-related loss of neural elements in the retina. Hence compensatory changes occur in the capillary walls, which have the potential to impair nutrient exchange and also to have a negative impact on neuronal function. A decrease in capillarity stability, culminating in vessel loss and localized angiogenesis in response to hypoxia, produces reduced ability of the microvasculature to meet the metabolic needs of the parenchyma.
Müller cells, astrocytes, and microglia are the cells of the local innate immune system. Under stress, they become active and produce pro-inflammatory cytokines and growth factors in order to reconstruct the tissue homeostasis, but in chronic pathologies such as diabetic retinopathy, the persistent inflammatory response leads to cellular death or damage. Liou et al. 43 have recently confirmed that the microglia are involved in activation even at the beginning of diabetic pathology, producing a wide range of pro-inflammatory cytokines, such as IL-1β, IL-3, IL-6, TNF-α, and other inflammation mediators like 51 ROS, glutamate, VEGF, lymphotoxins, metalloproteinase, and NO. These mediators induce the expression of adhesion molecules (I-CAM and V-CAM) in vascular cells for lymphocytes and monocyte-macrophages, cellular apoptosis, leukocyte infiltration, and the weakening of the hemato–retinal barrier. 43 The Langmann’s group has detected that the microglial cells are the ones responsible for an increased retinal production of iNOS, IL-1β, MIP-1α, IL-6, and M-CSF. 44 Shelton et al. 45 have observed an increase in IL-1β, IL-6, IL-8, IL-13, IP-10, ICAM-1, and NO in Müller and endothelial cells during the course of diabetes, confirming their participation in the inflammatory process. Owing to the fact that IL-1β is the main cytokine able to trigger the neuroinflammatory cascade, Liu et al. 5 have also considered its role in the amplification of the inflammation itself. This study has, in fact, highlighted that IL-1β secretion begins in the vascular endothelium as the direct consequence of chronic hyperglycemia, which stimulates the endothelial, macroglial, and microglial cells via the autocrine and the paracrine pathways, which do respond not only by activation signals, but also via the strengthening of IL-1β synthesis, thus exalting the inflammatory process. 46
The cytokine TNF-α is synthesized by stimulated monocytes and macrophages and functions in a cellular communication network. Additionally TNF-α stimulates the production of monocyte chemotactic protein by retinal pigment epithelial cells which makes the process self-stimulating. Cytokines such as IL-1β, TNF-α, and IL-6 have been shown to directly activate microglia. For example, TNF-α decreases cAMP levels within microglia, leading to a more destructive phenotype. Our results confirm the induction of pro-inflammatory cytokines TNF-α and IL-1β within the retina as a result of diabetes. The most likely source of these cytokines is retinal microglia. However, other retinal cell types (e.g. pericytes) release pro-inflammatory cytokines in response to diabetic stress. Upon their release, these cytokines may propagate the inflammatory response within the retina, leading to an increased leukocyte infiltration. Activation of leukocytes leads to enhanced synthesis of cyclooxygenase and lipoxygenase which convert arachidonic acid into peroxide metabolites that further stimulate cytokine secretion through a positive feedback mechanism. Given their functions, these signals represent potential targets for therapeutic procedures.
Besides inflammatory mediators mentioned above, it is well known that many other mediators are expressed inside the retinal tissue during the course of diabetes, since it is now clear that inflammation plays an essential role in diabetic retinopathy. 47 It is generally thought that the microglial cells also indirectly intervene in their production, due to their ability, via the production of mentioned molecules, to behave as chemoattractants towards the other leukocytes, especially the neutrophils and lymphocytes, which can strengthen the inflammatory process. Accumulating evidence suggests that leukocytes play an important role in the pathogenesis of diabetic retinopathy. Histopathologic studies have demonstrated many capillary occlusions caused by increased numbers of polymorphonuclear leukocytes in the retinas of diabetic patients and of leukocytes in the retinas of rats with chemically-induced diabetes. 48 Moreover, the activity of leukocytes trapped in the diabetic retina was found to be elevated, which may increase vascular permeability through endothelial dysfunction and damage to the blood–retina barrier: 48 these trapped leukocytes may produce more oxygen-derived free radicals than normal leukocytes. Retinal hypoxia and neovascularization were reported to be associated with the presence of leukocytes in the diabetic retina. 49
Additional clinical and experimental investigations appear to be necessary to better identify the causes and mechanisms of vascular changes during aging and diabetes, to determine whether treatment to prevent retinal capillary BM thickening is also effective in preserving the cellular elements of retinal capillaries and the surrounding tissue. Further research should focus on the inflammatory mechanisms of the development of diabetic retinopathy and should assess the possible effectiveness of anti-inflammatory therapy in the treatment and prevention of diabetic microangiopathy.
Footnotes
Acknowledgements
The authors are very grateful to Frances Corsetti, Stephanie Corsetti, and Sharon Hobby for the English language editing.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research received financial support from Fondazione ROMA and Italian Ministry of Health.
