Abstract
Despite the high prevalence of feline pancreatic disease, no detailed description on the histopathologic nature of this disease is currently available in the literature. In this study we characterize the distribution and histopathologic changes commonly found in feline pancreases, correlate the lesions with age and gastrointestinal GI and extra-gastrointestinal disease, and compare the pancreatic lesions in cats with those in humans. The entire pancreas was removed and examined from 115 cats presented for necropsy irrespective of the cause of death. Histologic sections from left limb, right limb, and body were scored for lesions of acute (AP) and chronic pancreatitis (CP) with a scoring system based on similar systems used in human and veterinary literature. The lesions of CP in cats resemble CP in humans, with fibrosis being more prominent than inflammatory changes. Cystic degeneration gradually increased as other lesions of CP were more prominent. A distinct nodular change of zymogen depletion and acinar cell dysplasia not associated with pancreatitis was prominent in 15.6% of the pancreases. Histologically, AP consisted of neutrophilic inflammation associated with interstitial edema and necrosis of mesenteric fat.
An overall prevalence of 67%, and 45% in clinically normal animals, was identified. CP was found in 69 (60.0%) pancreases, and 58 (50.4%) had CP only, with a significant correlation between age and occurrence of CP. There was a statistically significant higher prevalence of CP in the left limb in animals with gastrointestinal disease. AP was present in 18 animals (15.7%) of which 7 animals had AP only (6.1%).
Pancreatitis is an important and common disease of cats that may be associated with severe clinical disease and high mortality. 19, 26, 35, 38 Acute, recurrent acute, and chronic pancreatitis are all recognized as clinical entities. 26 Antemortem diagnosis can be challenging owing to the vague nonspecific clinical signs including anorexia, lethargy, and weight loss, and the lack of sensitive and specific conventional diagnostic tests such as radiography and measurement of serum lipase and amylase concentrations. Recent advances in diagnostic testing, including abdominal ultrasound and measurement of serum pancreatic lipase concentrations, have facilitated the antemortem diagnosis of feline pancreatitis, although pancreatic histology is still felt to represent the gold standard test for the diagnosis of feline pancreatitis. 19, 21, 30, 35, 37, 42
Histopathology is still considered the most reliable diagnostic test for pancreatitis in the cat. 10, 41, 42 However, despite the high prevalence of feline pancreatic disease, no detailed description on the histopathologic nature of this disease is currently available in the literature. Several articles that discuss the clinical aspects of pancreatitis occasionally provide minimal information on the histopathology of pancreatitis in the cat. 19, 26, 30, 39 The histopathology of experimentally induced chronic pancreatitis in the cat, either by intraductal or intraparenchymal injection of ethanol or by ligation of the pancreatic duct, has been described. 43, 44 Total obstruction, caused by intraductal damage by ethanol or ligation of the pancreatic duct, resulted initially in an acute necrotizing pancreatitis that progressed to a pronounced and progressive atrophy of both endocrine and exocrine tissues associated with prominent ductular dilation. Progressive chronic pancreatitis characterized by fibrosis, inflammatory cell infiltrate, and irregular stenosis and dilations of the pancreatic ductules was obtained in cats subjected to intraductal injections of alcohol and in all cats in which acinar and ductal damage was combined with partial obstruction. While diffuse acinar damage induced by multifocal ethanol injections resulted initially in severe acute hemorrhagic pancreatitis, it did not progress to chronic fibrosing lesions. 43, 44
Lesions associated with chronic pancreatitis in humans are progressive fibrosis associated with acinar atrophy and less prominent mononuclear inflammatory infiltrates, duct dysplasia with intraductal mucoprotein plugs, and cysts. 7, 18, 29 The major changes in the pancreas of humans associated with aging are lipomatosis, fine intra-acinar fibrosis, and duct alterations. 32, 36 Fang et al. (1997) and Longnecker et al. (1980) also describe the presence of atypical acinar nodules that were eosinophilic, basophilic, or vacuolated as common age-related findings. 14, 24, 25
The objectives of the current study were to 1) characterize the distribution and histopathologic changes commonly found in feline pancreases, 2) correlate the lesions with age and gastrointestinal and extra-gastrointestinal disease, and 3) compare the pancreatic lesions in cats with those in humans.
Material and Methods
Animals and tissues
The entire pancreas was removed and examined from 115 cats that were presented for necropsy between January and December 2002 at the Veterinary Medical Teaching Hospital, University of California, Davis. The animals were divided into 3 groups: group 1 included apparently healthy cats at the time of death. The animals with clinical signs at the time of death were further divided based on necropsy findings into 2 groups: group 2 included cats with gastrointestinal disease (GI-disease) and group 3, cats with extra-GI disease.
The entire pancreas was removed from each cat within 4 hours of death and immersion-fixed in 10% neutral-buffered formalin. After fixation for 48 hours, sections were collected from each pancreatic division (left and right limb and body), and from the mesentery with pancreatic-duodenal artery and gastroduodenal vein. All tissues were routinely processed for paraffin embedding and 4-μm-thick sections were cut and stained with hematoxylin and eosin (HE).
Histologic evaluation
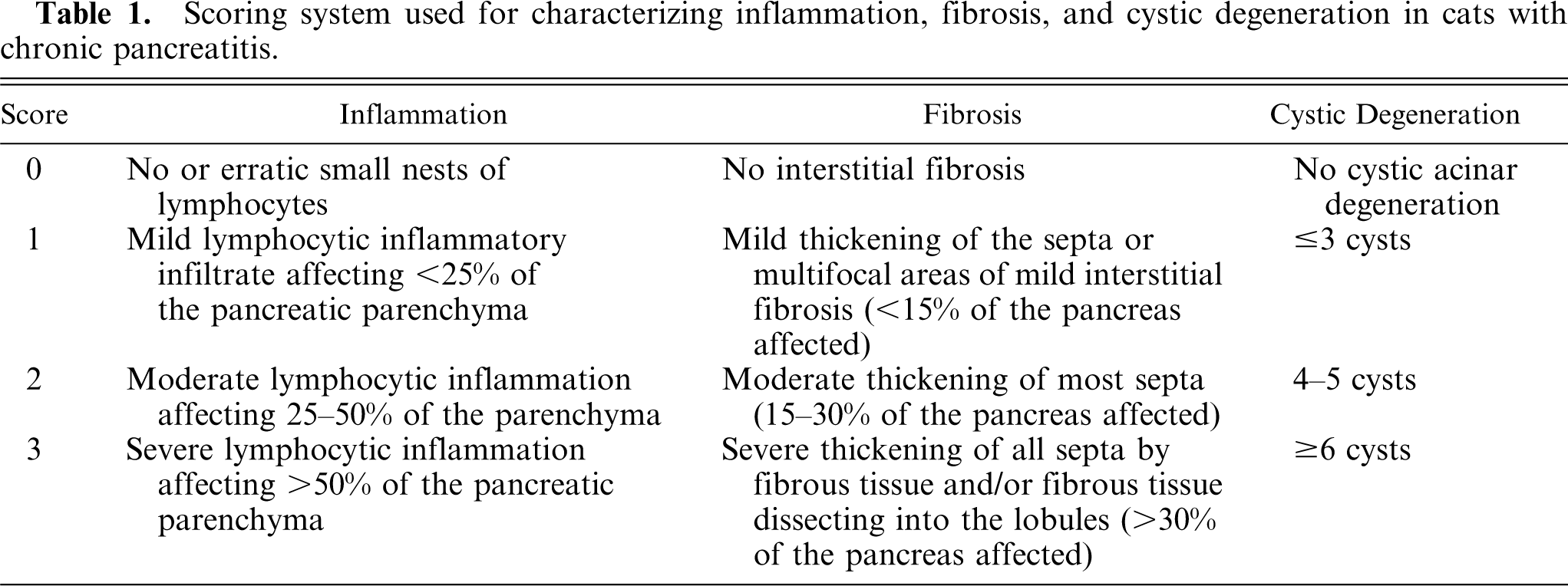
The histologic sections were reviewed and scored in a blinded fashion. A histopathologic scoring system, which reflects the severity of the lesions of acute and chronic pancreatitis as accurately as possible, was developed based on scoring systems used in the human and veterinary literature. 17– 19, 26, 30, 32 The grading system is based on a point system for 3 lesions that typically occur in chronic pancreatitis (CP): lymphocytic inflammation, interstitial fibrosis, and cystic acinar degeneration; and 2 lesions that typically occur in acute pancreatitis (AP): interstitial edema/necrosis of mesenteric fat and neutrophilic inflammation. Details of the points assigned to the different degrees of inflammation, fibrosis, and cystic degeneration in CP are explained in Table 1. Details of the points assigned to the different degrees of inflammation and edema/fat necrosis in AP are explained in Table 2. The sum of the points for each criterion in AP and CP was calculated with a maximal score of 9 for CP and 6 for AP. A total point score of 0 was considered normal pancreatic histopathology. A score of 1–3 total points was considered mild CP; 4–6, moderate; and 7–9, severe CP. A score of 1–2 total points was considered mild AP; 3–4, moderate; and 5–6, severe AP. All the sections of 1 division of the pancreas were present on a single slide, thus representing the entire division; therefore the score also reflected the entire pancreatic division.
Scoring system used for characterizing inflammation, fibrosis, and cystic degeneration in cats with chronic pancreatitis.
Scoring system used for characterizing inflammation and edema in cats with acute pancreatitis.

In addition, the presence of other changes including atypical acinar cell nodules, acini with inspissated secretions, vacuolar degeneration, ductular alterations (e.g., epithelial hyperplasia/metaplasia, ductular dilation and inflammation), and islet cell amyloidosis were also noted. Vascular lesions were evaluated in the mesenteric section with the pancreatic-duodenal artery and gastroduodenal vein.
Statistical analysis
Statistical software SPSS for Windows (SPSS Inc., Chicago, IL) was used for the statistical analysis. The statistical analysis was based on the scores as described in the grading system in the Histologic evaluation section above. The analysis of variance for a repeated measures experimental design was used to evaluate the effect of study factors on AP and CP score. Clinical group, sex, and breed were incorporated in the analysis as categorical between subject factors, while location was used as a repeated measures factor. Age was a categorical covariate. Interaction effects were assessed in addition to main effects. The associations between AP and CP score and age were estimated parametrically using the Pearson correlation coefficient and nonparametrically using the Spearman rank correlation coefficient. A P value <.05 was considered statistically significant.
Results
General
Of the 115 cats included in the study, 82 were Domestic short hair; 13, Domestic long hair (DLH); 8, Domestic medium hair (DMH); 3, Siamese; 2, Abyssinian; 1, Burmese; 1, Bengal; 1, Maine Coon; 1, Himalayan; 1, Tonkinese; and 2, mixed breed. The age was known for 91 cats, with a mean of 102 months (SD ± 67 months; range 3–251 months). The other animals were all young adults. The sex was known for 93 animals: 38 were neutered males; 32, neutered females; 16, intact females; and 7, intact males. Forty-one animals were considered healthy (group 1) at the time of necropsy, including 28 cats from clinical studies that did not undergo any treatment (control animals) and 13 cats that had no history of disease and died of trauma (e.g., hit by car, dog bites). Lesions identified at necropsy involved the digestive system of 22 cats (group 2), including lymphoma (7) and other neoplastic processes (1 hemangiosarcoma in caudal abdomen, 2 intestinal carcinomas, 2 intestinal mast cell tumors, and 1 liver lymphoma), intestinal obstruction (1), megaesophagus (1), feline infectious peritonitis (FIP) (1), hepatic toxoplasmosis (1), intestinal mycobacteriosis (1), Parvovirus enteritis (1), and peritoneal actinomycosis (1). One cat had a pancreatic carcinoma. One cat had diffuse intravascular coagulation (DIC) and had thrombi throughout the GI-tract including the pancreas. Fifty-two cats had clinical conditions that did not involve the GI-tract (group 3): 10 cats had neoplasms (2 thoracic mesotheliomas, 1 transitional cell carcinoma, 1 adrenal carcinoma, 1 nasal carcinoma, 1 chondrosarcoma, 1 osteosarcoma, 1 leiomyosarcoma of the head, 1 skin lymphoma, 1 skin squamous cell carcinoma); 1 had a nasal polyp; 12 had lesions in the central nervous system; 10 had cardiovascular lesions; 5 had endocrine disease; 8 had end-stage kidney disease; and 1 had pneumonia. Two cats had myelodysplasia. Three animals were strays in poor body condition. Mean age for the animals for which the age was known was 86 months in the healthy (group 1), 104 months in the GI-disease (group 2), and 114 months in the extra-GI disease (group 3).
No lesions were identified in the left limb, right limb, or body in 38 of the 115 (33.0%) pancreases evaluated. AP was present in 18 cats (15.7%) of which 7 had AP only (6.1%). Both lobes and body were affected in 4 of these pancreases. In the other 3 cats there was 1 pancreas with the left limb and body affected, 1 with the right limb only, and 1 with the body only. CP was found in 69 (60.0%) pancreases, and 58 (50.4%) had CP only. The left limb had CP in 51 (44.3%) cats, the right limb in 49 (42.6%) cats, and the body in 57 (49.6%) cats. In 36 (31.3%) of the pancreases, the lesions were present in all divisions. In 4 cats, the left limb was affected only, in 4 the right limb only, and in 8 the body only; the left limb and body were affected in 7 cases, right limb and body in 6 cases, and left and right limbs in 4 cases. Eleven pancreases (9.6%) had evidence of both AP and CP; 3 of these had lesions of AP and CP in all divisions.
The mean score of AP in the left limb was 0.2832 (±0.9399; range 0–6), in the body 0.2124 (±0.7494; range 0–5), and in the right limb 0.2174 (±0.7106; range 0–4). The mean score of CP in the left limb was 1.3805 (±1.9925; range 0–9), in the body 1.2035 (±1.6211; range 0–7) and in the right limb 1.0870 (±1.6414; range 1–8). An overview of the mean scores for AP and CP in the different divisions of the pancreas for the different clinical groups is given in Table 3. Healthy animals always had the lowest score for AP and CP compared with animals from group 2 or group 3. Animals with GI-disease (group 2) had higher scores of AP compared with animals in group 3, whereas the reverse occurred with CP. The highest scores for CP occurred in animals with extra GI-disease (group 3). In animals of group 3, the CP scores were comparable in all divisions of the pancreas, while in animals with GI-disease the left limb had a much higher CP score compared with the right limb and body.
Histopathologic score for acute and chronic pancreatitis in the left and right limbs and body in apparently healthy cats, cats with gastrointestinal disease, and cats with extra-gastrointestinal disease.∗
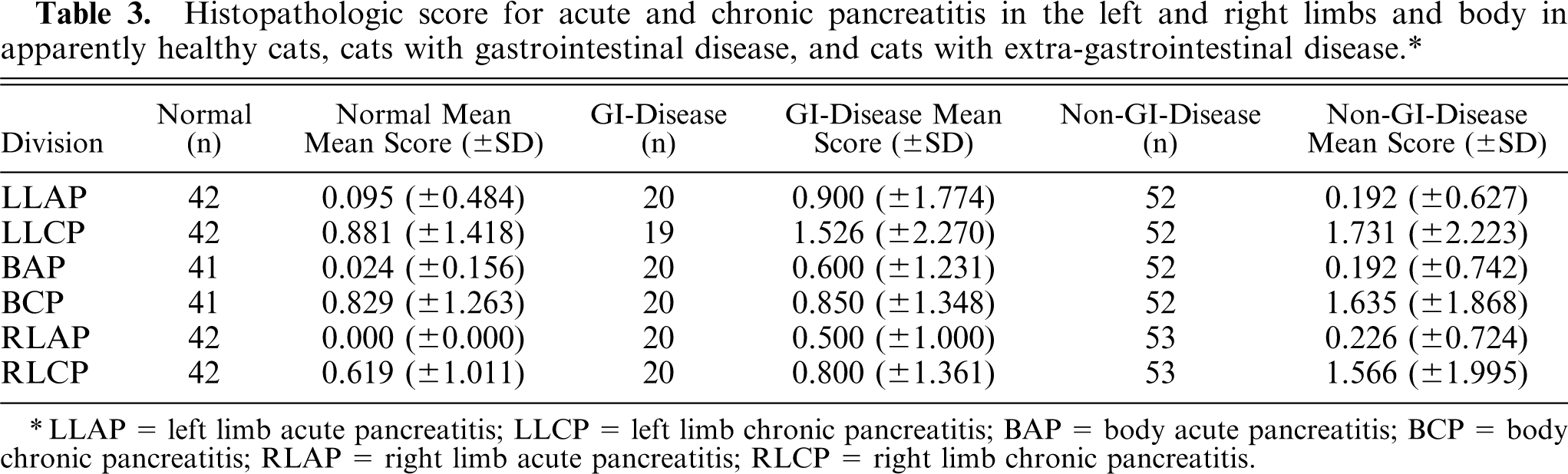
LLAP = left limb acute pancreatitis; LLCP = left limb chronic pancreatitis; BAP = body acute pancreatitis; BCP = body chronic pancreatitis; RLAP = right limb acute pancreatitis; RLCP = right limb chronic pancreatitis.
There was a statistically significant (P < .01) higher CP score in the left lobe compared with the right lobe in animals with GI-disease. There was no statistically significant difference in AP or CP score between the other clinical groups for the left limb, right limb, and body when adjusted for age. There was a statistically significant correlation (P < .01) between the presence of CP and location in the pancreas. There was also a statistically significant correlation (P < .01) between the presence of AP and location in the pancreas. A statistically significant correlation (P < .0005) was found between the age of the animal and the presence of CP in the left limb, right limb, and body unadjusted and when adjusted for other factors. There was no correlation between age and AP score in any location, both unadjusted and when adjusted for sex and breed. Sex and breed were shown to be nonsignificant factors in all analyses.
Histopathology
In general, the lesions of CP were multifocal. Only in the end-stage were the lesions confluent, although small nests of normal-appearing acini were always present. Lesions of AP were also randomly scattered throughout the pancreas and often extended into the surrounding mesenteric adipose tissue.
Fibrosis
Fibrosis was a very prominent feature of CP. In the mild stages of CP, fibrosis usually was minimal and composed of a thin layer of dense fibrous tissue that surrounded 1 or several pancreatic lobules (Fig. 1). In the more severe CP, the fibrous tissue expanded uniformly around 1 or more lobules and dissected into the lobule as thin strands that separated the individual acini. In the most severe cases, the fibrosis was marked in the interlobular and intralobular areas and often replaced large areas of pancreatic exocrine parenchyma (Fig. 2). In all cats with severe CP, small lobules with viable parenchyma were spared (Fig. 2). Occasionally fibrosis started within pancreatic lobules, individualizing the acinar structures without obvious interlobular fibrosis. The associated acini in these cases often were atrophic, and inflammation was frequently more prominent.
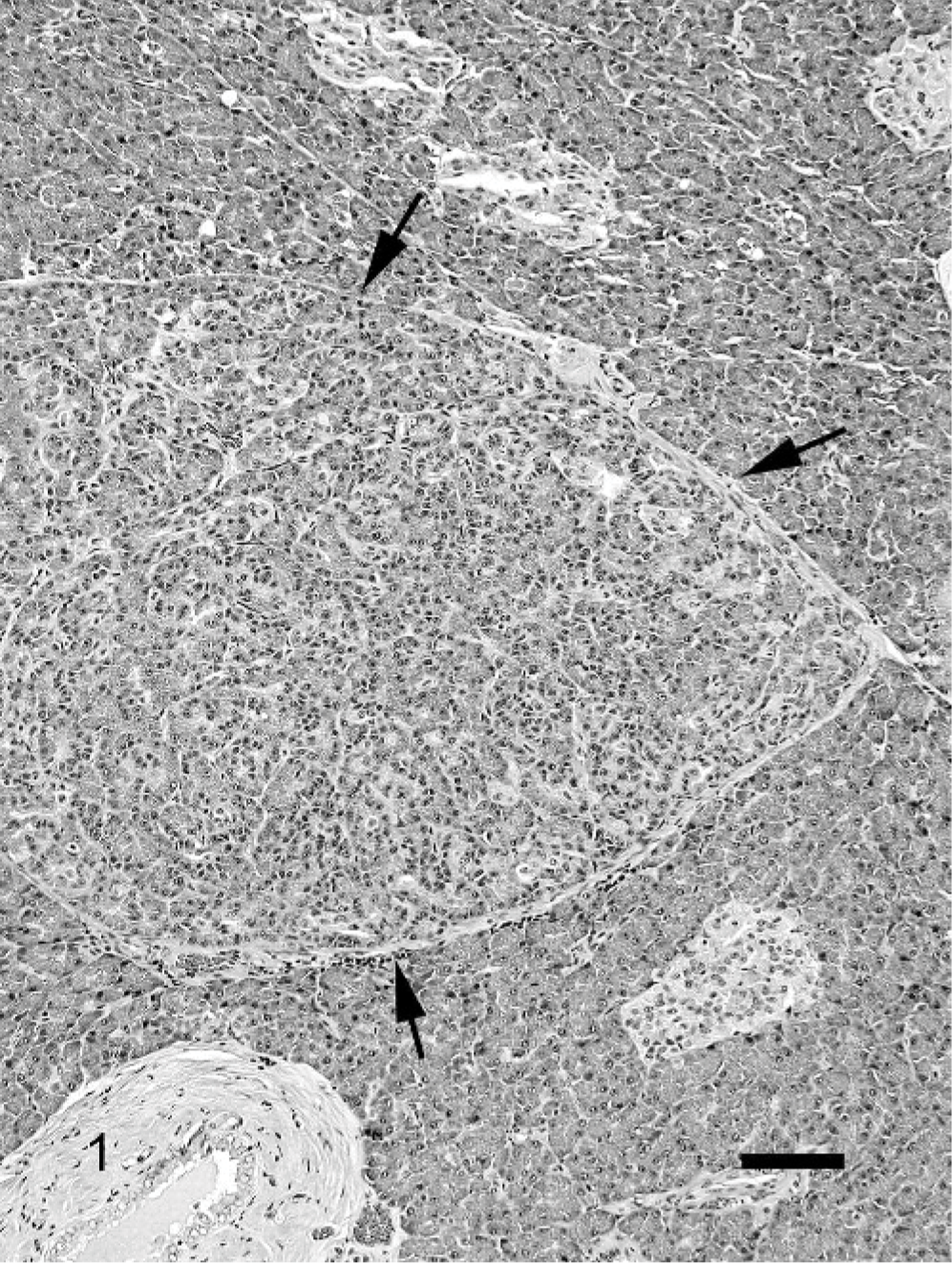
Pancreas; cat. Mild chronic pancreatitis; mild fibrosis (arrows) surrounding pancreatic lobules with occasional inflammatory cells. HE. Bar = 100 μm.
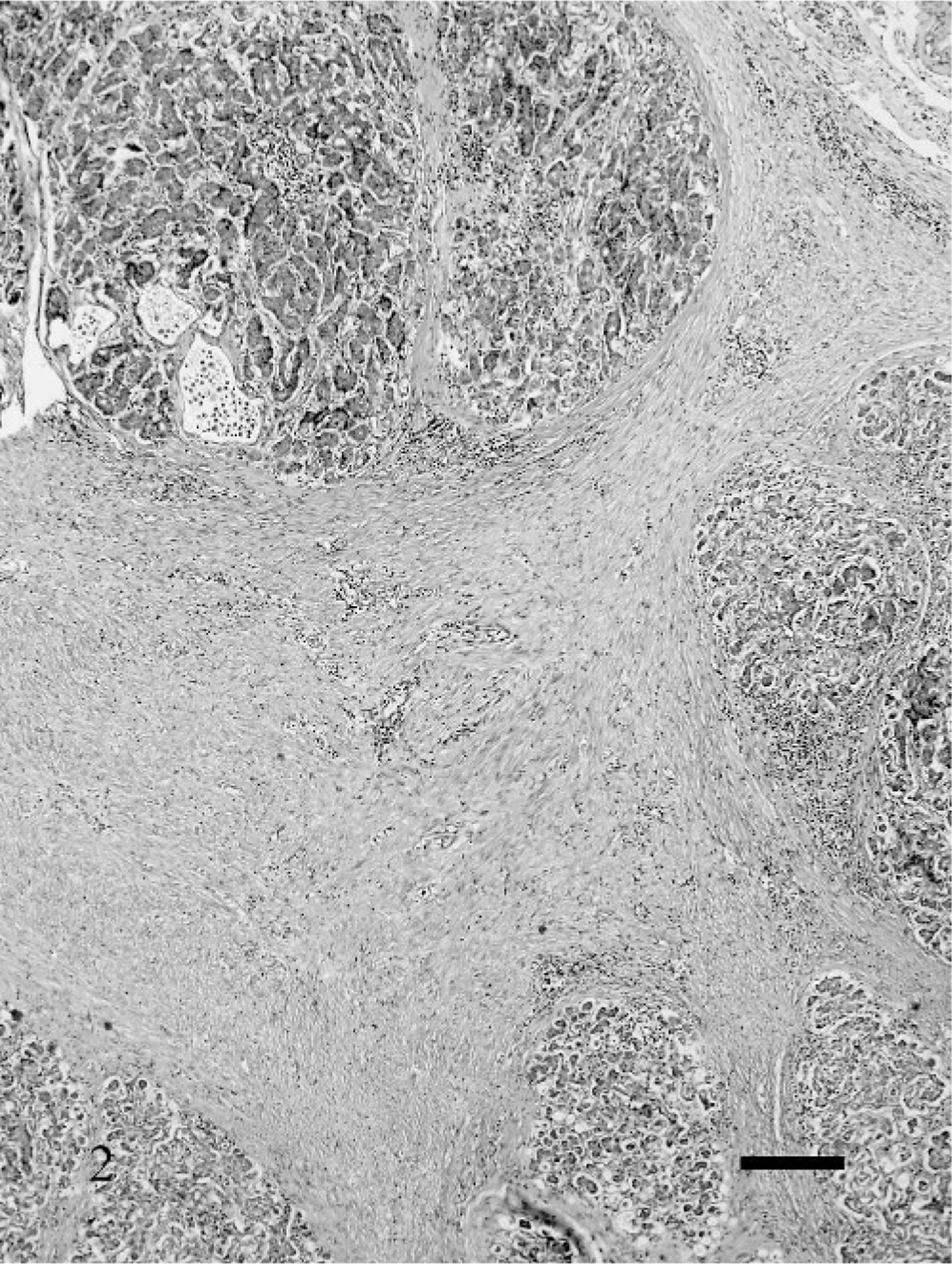
Pancreas; cat. Severe chronic pancreatitis; marked fibrosis in the interlobular and intralobular areas, replacing large areas of pancreatic exocrine parenchyma. Small lobules with viable parenchyma are spared. HE. Bar = 20 μm.
Fibrosis was not a feature of AP.
Inflammation
A few scattered lymphocytes within the pancreatic parenchyma was considered normal. Inflammation in CP was variable but often rather mild in comparison to the fibrous response. In the mild stages, only occasional inflammatory cells were scattered within the fibrous tissue (Fig. 1). As the amount of fibrous tissue increased, inflammation also became more prominent (Fig. 2) and more widely distributed within the lobules. Lymphocytes were always the major inflammatory component in CP, but occasionally eosinophils and macrophages were also present. Macrophages were especially prominent in the lumen of cystic acinar structures that developed in the more chronic stages of pancreatitis (see cysts) (Fig. 3).
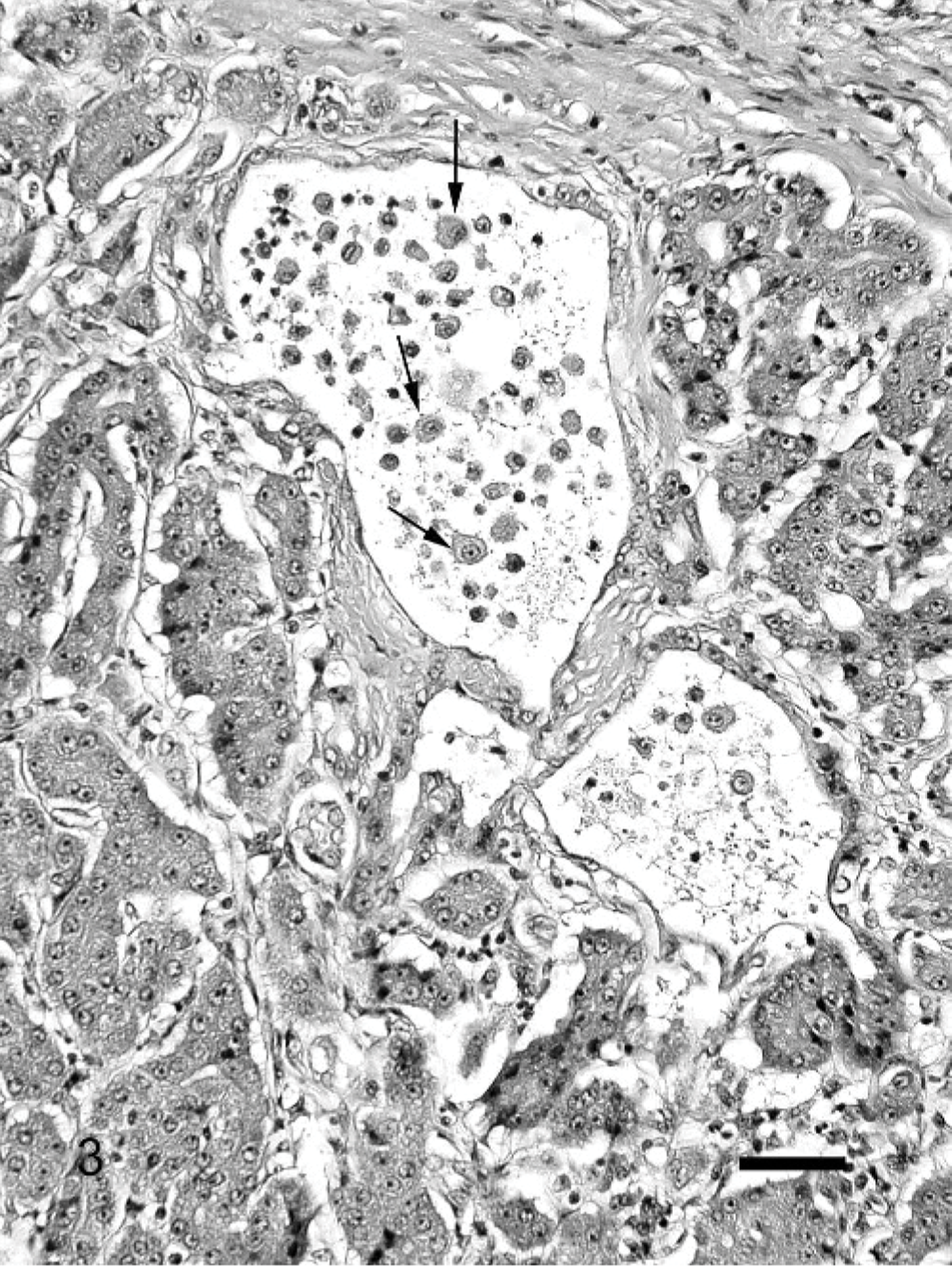
Pancreas; cat. Severe chronic pancreatitis; cystic acinar spaces with intraluminal macrophages (arrows). HE. Bar = 200 μm.
In 29 pancreases (25%) there were nests of lymphocytes without associated signs of pancreatitis. The lymphocytes were small and monomorphous, closely arranged and located in small aggregates scattered throughout the pancreatic parenchyma and surrounding mesenteric adipose tissue.
In AP, inflammation was very prominent surrounding areas of necrosis and usually infiltrated diffusely the rest of the pancreatic parenchyma tracing along the interlobular septa. Neutrophils dominated the infiltrate in AP with an occasional eosinophil. In AP secondary to FIP and actinomycosis, the inflammatory infiltrate also contained macrophages.
Cysts
Cystic dilation of the acini was a common finding in all stages of CP. Cysts developed centrolobularly, initially without much evidence of fibrosis or inflammation. The cysts contained small amounts of dark, eosinophilic-inspissated material interpreted to be mucoprotein. As the cystic lumen dilated and became more prominent, thin fibrous bands dissected between the surrounding acinar cells and macrophages appeared in the lumen (Fig. 3). In end-stage CP, cysts often were large and multiple.
Large cysts lined by acinar cells occurred in 8 pancreases without significant CP (Fig. 4). The lining cells usually had normal zymogen granules (Fig. 4). Four of the 8 cats with these types of cysts were long-haired breeds (DLH or DMH), which encompasses 40% of the long-haired breeds, whereas only 4% of the short-haired breeds had this change.
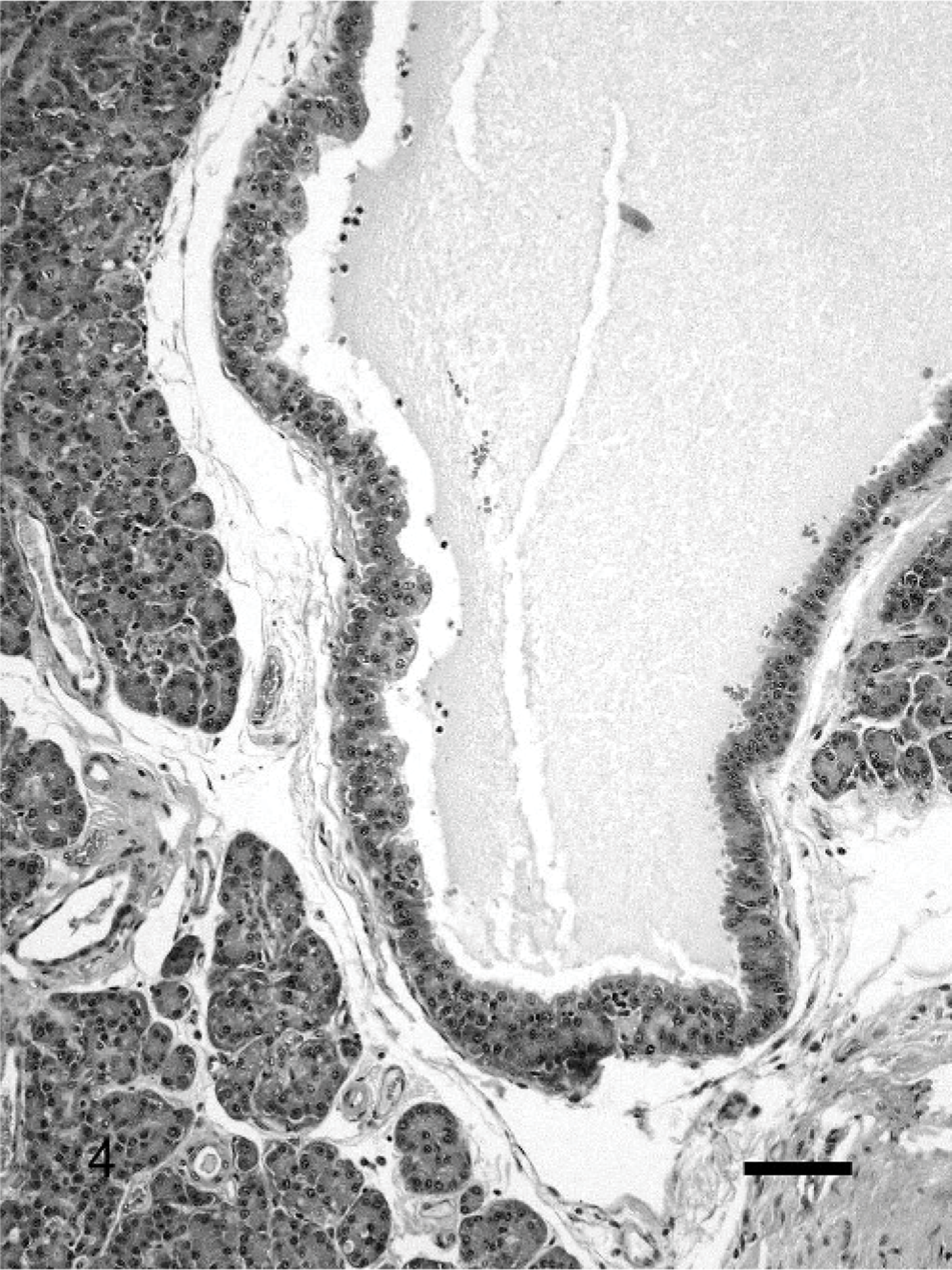
Pancreas; cat. Large cysts lined by acinar cells without significant chronic pancreatitis. The lining cells have normal zymogen granules. HE. Bar = 100 μm.
Necrosis
Necrosis exclusively occurred in association with AP, as randomly scattered foci within the pancreas and adjacent adipose tissue. The necrosis was present as small foci or involved entire lobules. Interlobular septa in these cases were separated by edema.
Atrophy
Fibrosis and inflammation was rarely associated with acinar cell atrophy or zymogen depletion especially in the mild stages of the disease. In the more severe stages, acini were replaced by fibrous tissue and acinar cells were atrophic, while adjacent acini remained active.
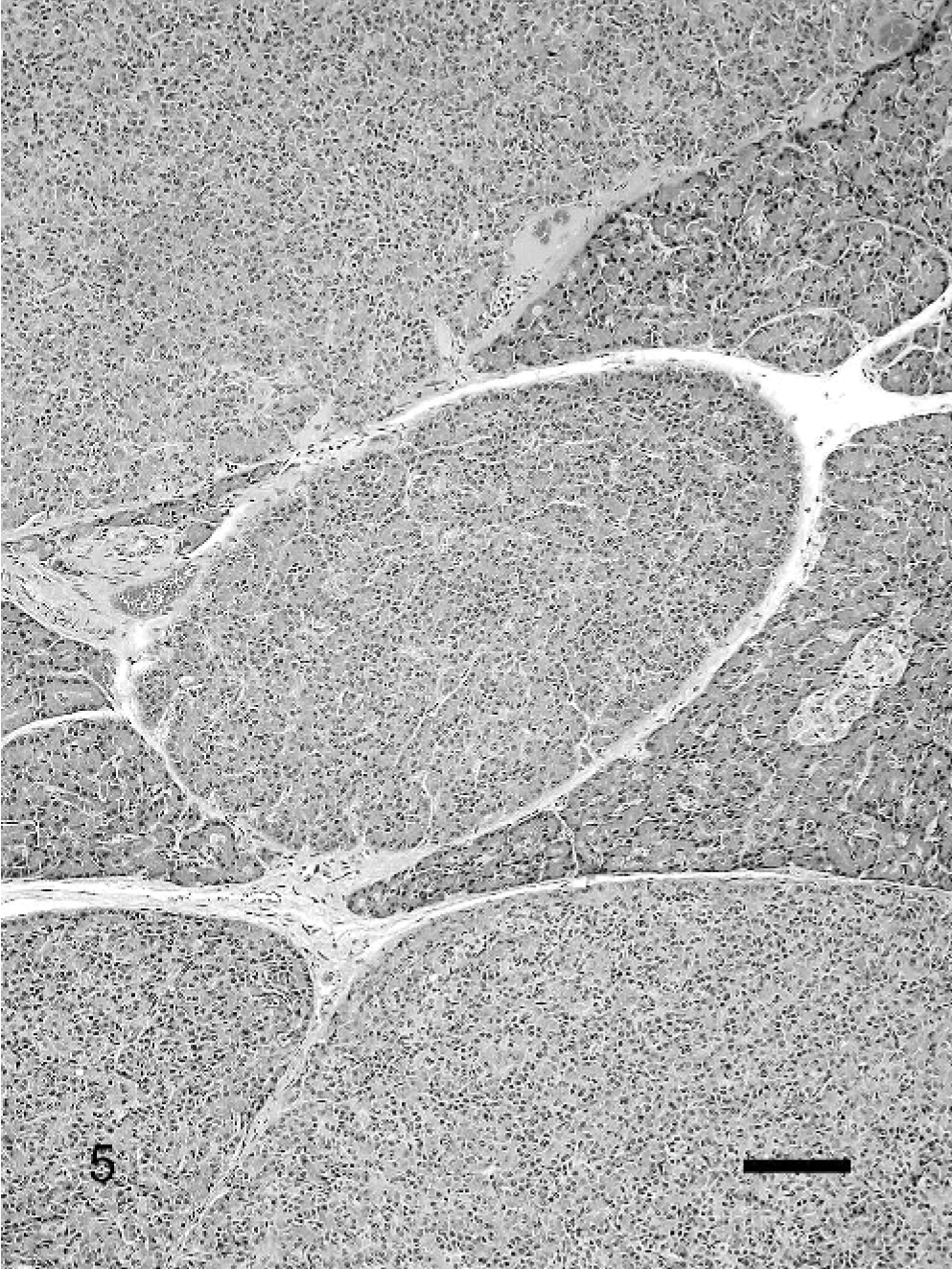
A striking feature found in 18 (15.7%) pancreases was prominent lobulation due to the intermingling of apparently normal looking lobules with prominent zymogen production and pale basophilic lobules with markedly less zymogen. (Fig. 5). The acinar cells often looked slightly atrophic with a small, round, pale basophilic nucleus and a small amount of dark basophilic cytoplasm that often contained microvacuoles. However, these lobules occasionally also consisted of mildly dysplastic acinar cells with large, oval nuclei, increased nuclear°cytoplasmic ratio, pale basophilic cytoplasm, and scattered mitotic figures. Interlobular ducts were not evident in these types of lobules. This feature appeared independently of other changes in the pancreas.
Pancreas; cat. Prominent lobulation owing to the intermingling of apparently normal looking lobules with prominent zymogen production and pale basophilic lobules with markedly less zymogen. No pancreatitis is apparent. HE. Bar = 100 μm.
In addition, atypical pancreatic nodules were found in 14 pancreases (12.2%). Two different types could be distinguished. In the basophilic/vacuolated type, the cells contained a prominent amount of cytoplasm that was pale basophilic or vacuolated (Fig. 6A). The vacuolation in these nodules was often more prominent in the antiluminal site of the acinar cell. In the eosinophilic type, the cytoplasm had a pale eosinophilic glassy-looking content (Fig. 6B). Vacuolation of the acinar cells was not always confined to atypical nodules. Scattered throughout the pancreas, small areas with cytoplasmic vacuolation, was not uncommon. This type of vacuolation was distinctively different from the vacuolation seen in atypical nodules, characterized by small well-demarcated vacuolar spaces randomly distributed within a basophilic cytoplasm.
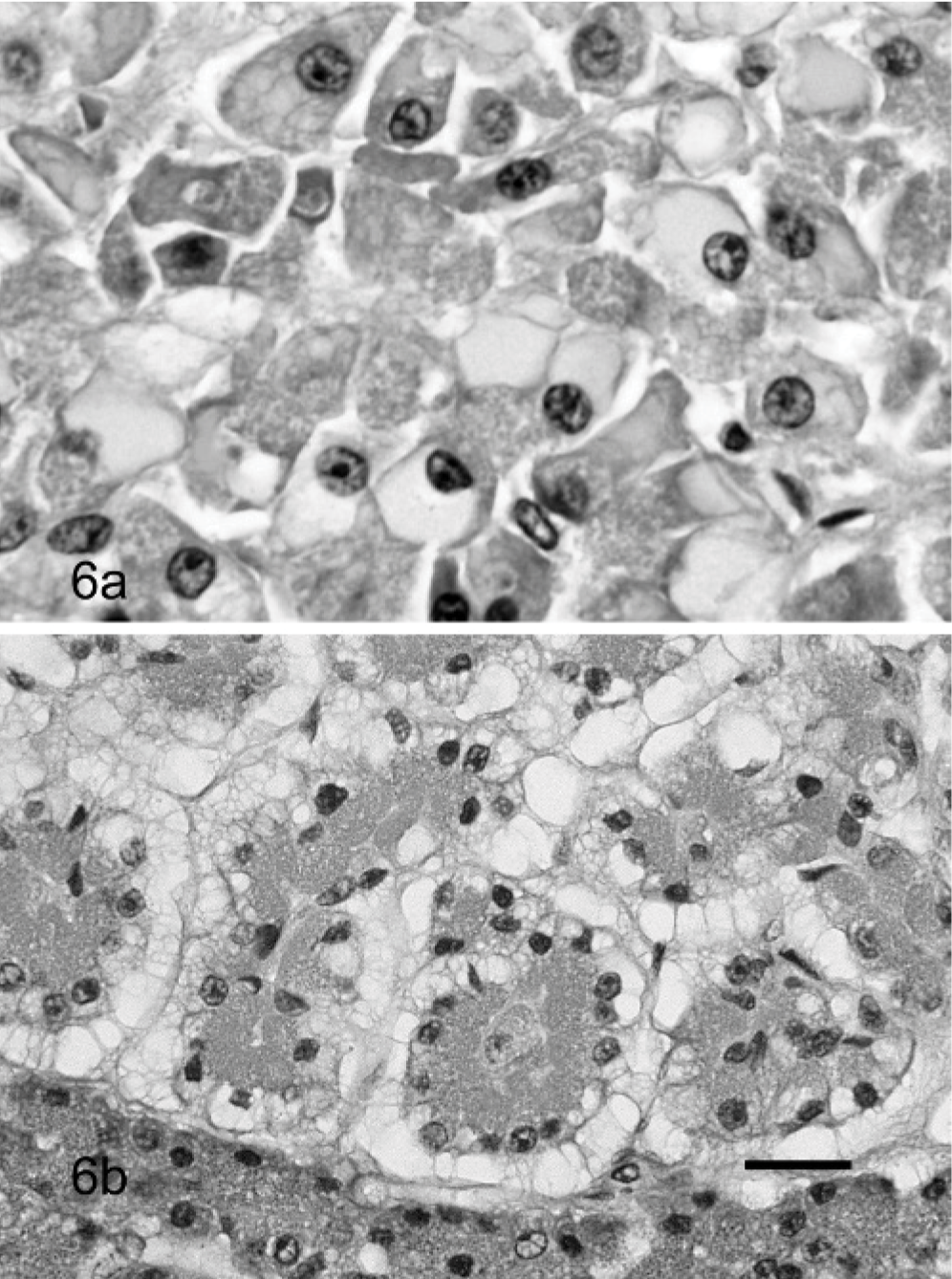
Pancreas; cat.
Interlobular and intralobular ducts
In normal pancreases, the large intralobular ducts were often dilated and the lumen was often filled with a pale eosinophilic, watery, amorphous mucoproteinous material. The ducts were lined by a low columnar epithelium that often desquamated regionally. The latter was considered an artifact caused by manipulation of the tissues.
Changes in the ducts were generally minimal and not related to the extent of AP and CP in the surrounding exocrine pancreas. The most common change in the small and larger interlobular ducts was mild to moderate dilation. The mucoprotein varied in these ducts from pale eosinophilic to dark eosinophilic often with a slight granular, or occasionally more mucoid, pale basophilic appearance. Mineralization was rare and, if present, consisted of very small concrements. Pancreatic stones were not identified. The epithelium in markedly dilated interlobular ducts appeared low cuboidal to flat. In 8 cases, the epithelium was convoluted forming small invaginations and intraluminal papillary projections.
Ductal inflammation was found in 12 cases. In most cases, the inflammatory infiltrate was very mild, lymphocytic, and mainly periductular. In some cases, there was also a neutrophilic component and intraluminal infiltration, rarely with accompanying bacteria. Scattered epithelial cells with changes consistent with apoptosis were occasionally found in association with ductal inflammation. A prominent ductal inflammation characterized by a diffuse periductular lymphocytic inflammation with multifocal lymphoid follicle formation was found in 2 pancreases (Fig. 7). The amount of inflammation in these ducts was far more prominent than the inflammation of the surrounding parenchyma.
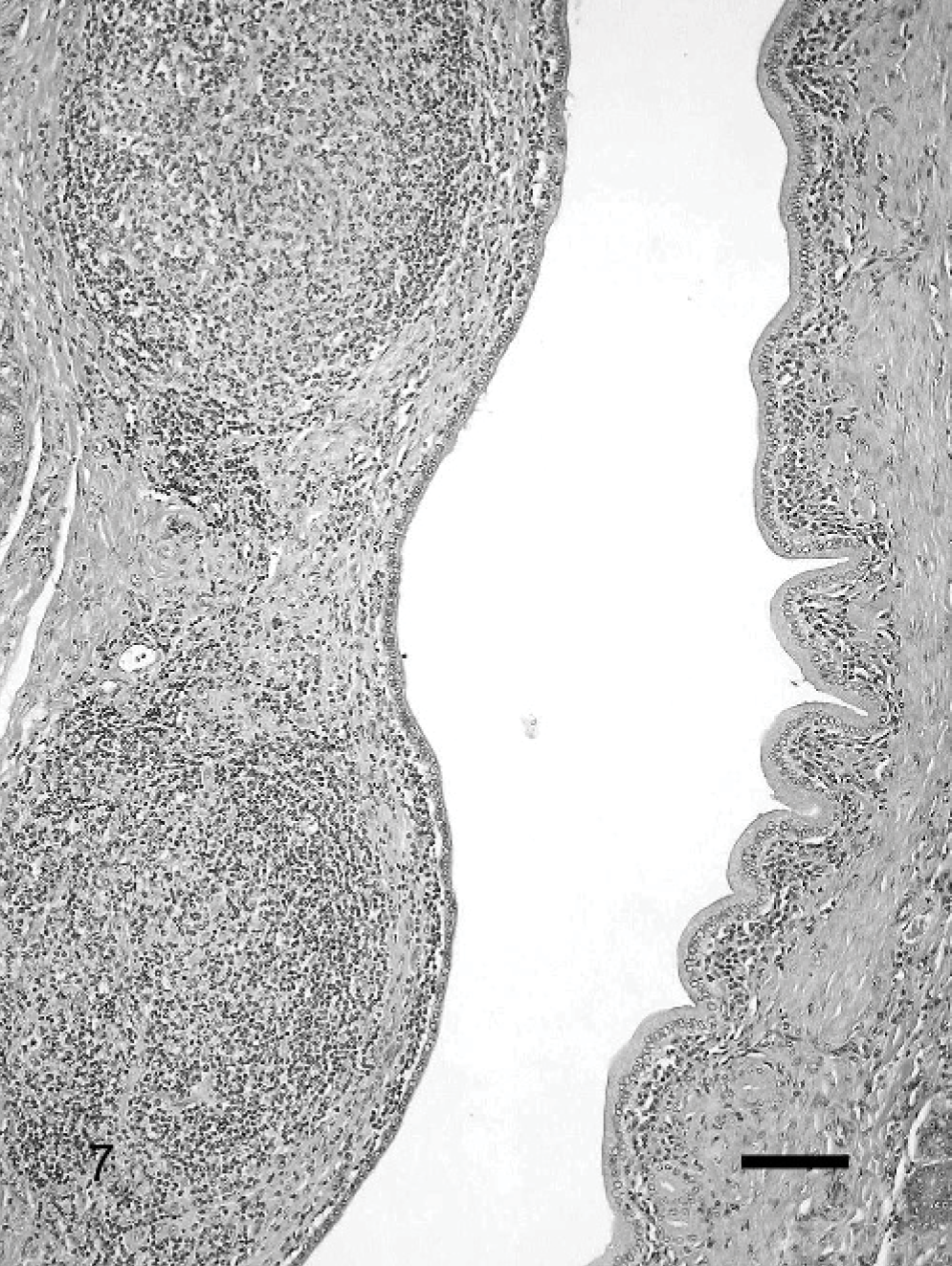
Pancreas; cat. Significant periductular lymphocytic inflammation forming lymphoid follicles. HE. Bar = 250 μm.
Islets
Amyloidosis was present in general in 34 (29.6%) of the pancreases, varying from mild (<50% of the islets affected) (10.4%), to moderate (50–75% of the islets affected) (10.4%), to severe (75–100% of the islets affected) (8.7%).
Vascular changes
Mild to severe arteriosclerosis, characterized by intimal fibrosis, was found only in 5 cats, either in the pancreatic-duodenal artery and gastroduodenal vein, or in the branches thereof within the pancreas.
Discussion
This study confirms that CP is common in cats, with an overall study prevalence of 67%, including 45% of apparently healthy cats. These figures are considerably higher than those previously reported, perhaps reflecting the high incidence of severe and complicated clinical conditions in the study population. 11, 26, 30, 38 In addition, the detailed examination of the entire pancreas detected even very mild lesions as reflected by the low mean CP scores found in the pancreases of the healthy cats. The finding is also supported by the observations of Steiner et al., who found a discrepancy between the prevalence of pancreatitis in clinical and pathologic studies from which they concluded that a significant number of cats have subclinical pancreatitis. 38
The high prevalence of CP in the cats with GI-disease is not surprising; however, cats with disorders not involving the abdomen had a similar occurrence of CP that was not attributable to age differences. This finding might suggests that the pancreas is very sensitive to drugs, stress, metabolic derangements, or ischemia associated with a wide variety of clinical conditions and potentially explains why mild pancreatic lesions are common even in clinically healthy animals. Similarly, Stamm found an unexpectedly high incidence of pancreatic lesions such as lipomatosis, fibrosis, alterations of ducts and ductal epithelium, inflammatory infiltrates, focal necrosis, acinar dilation, and vascular changes in his study of 112 human pancreases collected from unselected autopsies of adult patients with no known pancreatic disease. 36
A significant correlation was found between age and CP in the cat. Previous studies also have reported that older cats are more likely to present with pancreatitis. 11, 26 Mansfield and Jones noted that Siamese cats were more likely to develop pancreatitis compared with other breeds; however, we found no correlation between breed and pancreatitis in our study. 26 A plausible reason for this finding is that most cats in the Mansfield and Jones study were Domestic short hair (71%) and other breeds may have been under represented.
The lesions of CP in cats partially resemble the changes of CP in humans. As in humans, fibrosis is more prominent than inflammation. 18, 29 A major difference between feline CP in this study and human CP is involvement of the pancreatic ducts, which is a prominent feature in human CP, and is mild in cats. 27 Common lesions that occur in human pancreatic ducts are intraluminal mucoprotein accumulation and prominent mineralization/stone formation, ductular dilation, epithelial metaplasia, and ductal mucinous hyperplasia. 2, 3, 8, 22, 27, 29 Ductal mucinous hyperplasia, preneoplastic epithelial metaplasia, and intraductal stone formation were not identified in cat pancreases. Furthermore, ductular dilation, epithelial changes, mucoprotein accumulation, and mineralization were remarkably mild in affected cats. This finding is in contrast with the observations of Jubb et al., who describe CP as an extension from ductular inflammation. 20 Zhao et al. also report ductular epithelial flattening, epithelial proliferation, and stenosis in experimentally induced pancreatitis in cats. 44 A possible explanation is that the technique used to induce pancreatitis in the cats in this study, namely, ductular damage is not representative for the way the natural disease develops.
Acinar cell metaplasia is common in human CP. 9, 14, 33 The changes in the acinar cells as well as the ductular epithelial changes are considered preneoplastic lesions, and a gradual progression from mild to severe metaplasia and concurrent neoplasia has been well documented. 9, 14, 27, 33 While atypical nodules and nodular atrophy with occasional dysplasia (see below in discussion) were often present in the feline pancreases, transition to more distinct preneoplastic or neoplastic changes were not identified. The plausible reason for this difference is the different etiology of pancreatitis in humans and cats. Alcohol abuse is responsible for 70% of CP in humans, while the remaining 30% is autoimmune, dietary, genetic, idiopathic, or secondary to pancreatic duct obstruction. 8, 22, 27, 29 The cause of pancreatitis in cats is still highly speculative and, according to Steiner and Williams, 90% of the cases are idiopathic. 37 Correlations have been found between inflammatory bowel disease (IBD) and cholangiohepatitis and the presence of CP. 40
Differentiation between the different etiologies of CP based on histopathologic morphology was not possible in this study. This is similar to the findings in humans, indicating that pancreatic disease, independent of the underlying etiology, reaches a common immunologic stage beyond which it appears to progress as a single distinctive entity. 34 An exception is the recently documented autoimmune pancreatitis in humans characterized by prominent periductular lymphoplasmacytic inflammation associated with periductular sclerosis. 23, 28, 31 A comparable severe lymphocytic inflammation was observed in 2 feline pancreases (Fig. 7).
Some histopathologic features found in feline CP might be suggestive of a more specific etiology. Small lymphocytic infiltrates with no associated parenchymal lesions were common in the examined feline pancreases. It appeared as a subjective observation that these infiltrates were commonly associated with inflammatory bowel disease.
In addition, the multilobular atrophy (Fig. 5) sometimes associated with dysplasia, but not associated with inflammation found in 15.6% of the pancreases, is most likely associated with a specific insult to the pancreas. These lesions have been referred to in the literature as hyperplastic nodules. 20 However, it has never been investigated whether these lesions really are hyperplastic or not. In the pancreases examined for this project, the cells appeared rather atrophic based on the reduced amount of cytoplasm and lack of zymogen production. One possible reason for this is obstruction of smaller pancreatic ducts leading to atrophy of the associated lobule. Zhao et al. described extensive exocrine and endocrine pancreatic atrophy without fibrosis or inflammation in cases with complete obstruction of the pancreatic duct. 44 However, Zhao et al. also found dilated ducts in these particular cases, which was not present in the multilobular atrophy type of our study. 44 More research is warranted to determine the underlying cause for this specific lesion.
Some age-related changes described in the human pancreas were also frequently noticed in feline pancreases. This included acinar cell vacuolation and atypical eosinophilic and basophilic nodules. In addition, several pancreases had cystic structures not related to CP. It was remarkable that a high percentage of the cats with such cysts were of a long-haired breed. Since long-haired breeds have a predisposition for polycystic kidney disease syndrome, the pancreatic cysts might be related to this disease. 4– 6, 12 Similarly, in people with polycystic kidney disease, pancreatic cysts are not uncommon. 1
Pancreatic biopsy is still considered the gold standard for the diagnosis of CP in humans and cats. 10, 13, 17, 41, 42 Even acute necrotizing pancreatitis and CP in cats cannot be distinguished from each other solely on the basis of history, physical examination findings, results of clinicopathologic testing, radiographic abnormalities, or ultrasonographic abnormalities. 15 In humans, a highly significant correlation exists between direct function tests and histopathology of the exocrine pancreas. 17, 18 Surgical biopsy is facilitated by taking a biopsy of the most accessible part of the pancreas. An important observation in our study was the finding of higher mean CP scores in the left pancreatic limb in cats with GI-disease. The left limb is the most accessible portion of the pancreas, which should facilitate the diagnosis of CP in cats with GI-disease. In contrast, we did not find any differences in the severity of pancreatic scores in the left limb, right limb, or body in cats with extra-GI disease. The reason for the differences in CP scores in the different divisions of the pancreas in animals with abdominal disease is most likely related to anatomic characteristics, blood supply, or both in relation to the rest of the GI tract. The left limb, which had higher scores of CP, was also significantly bigger than the right limb. Therefore, it is assumed that differences in the size or morphology of the ducts might influence invasion of bacteria or regurgitation of bile from the GI tract. In contrast, in animals with generalized disease, the entire pancreas had comparable scores. It is thought that the lesions in these cases are related to blood-borne insults and therefore are more evenly distributed lesions.
Acute pancreatitis was relatively uncommon in this study as was the prevalence of AP and CP in the same pancreas. Forman et al. found a prevalence of 44% of AP and CP in the same pancreas in their study, which is significantly higher than the 9.6% found in this study. 16 The most likely reason for this is the difference in selected animals for both studies.
This study underscores the high prevalence of CP in cats. Histopathologic changes resemble the lesions found in human CP, although preneoplastic changes were not noticed in cats. While determination of the underlying cause might not be possible based on histopathologic examination of the pancreas in most cases, very typical lesions were noticed in feline pancreases that warrant further research to determine the underlying cause.
Footnotes
Acknowledgements
Sincere thanks are due to all the pathologists, clinicians, students, and technicians of University of California, Davis, who helped collect cases for this project. Specifically, we would like to thank Monica Kratochvil for her excellent assistance. The authors appreciate the critical review of this manuscript by Dr. James N. MacLachlan, Department of Pathology, Microbiology, and Immunology, University of California, Davis, School of Veterinary Medicine.
