Abstract
One 2-year-old, 7.5 months pregnant Aberdeen Angus out of a herd of 100 apparently healthy cows, died within 10 hours of hospitalization. At necropsy, multiple foci of mucosal hemorrhage and ulceration were observed in the spiral colon and cecum. Virus isolation from intestinal lesions yielded a cytopathic virus, which was revealed by electron microscopy to be an approximately 27 nm, nonenveloped virus. Further characterization by reverse transcription-polymerase chain reaction (RT-PCR), sequencing of the 5′UTR and partial VP1 coding region, and phylogenetic analysis classified the virus isolate as bovine enterovirus type 1 (BEV-1). No other significant pathogens were detected. This is the first report of BEV-1 isolated in the USA from an animal with fatal enteric disease in more than 20 years. Further investigation is required to determine the prevalence of BEV in North America and to establish the clinical relevance of this understudied virus.
Bovine enterovirus (BEV) belongs to the family Picornaviridae (picornaviruses), which consists of small (18–30 nm), nonenveloped viruses with an icosahedral capsid that encloses a single copy of positive-sense RNA genome. BEV is in the genus Enterovirus, along with poliovirus, human enterovirus, coxsackieviruses, swine vesicular disease virus, echovirus 11, and others. Originally classified into several serotypes, only 2 serotypes, BEV-1 and BEV-2, are now recognized. 10, 15, 18, 19 Because of the unavailability of type specific antisera or a commercially available diagnostic test, a genotypic classification, which supports previous recognized serological distinctions, has been proposed. 7 Despite the large volume of information available on other enteroviruses, very little documentation exists on the pathogenesis of BEV infections in cattle or on its prevalence in North America. Several case reports in the 1950s and 1970s document the isolation of BEV from feces and various tissues from apparently healthy animals or from animals with clinical signs that ranged from mild to moderate diarrhea to reproductive disease. 3, 4, 14, 20 However, these older reports are difficult to interpret as they relied solely on serological assays or had identified more than one infectious agent. This report describes the isolation and partial characterization of BEV-1 from a fatal enteric disease case in a heifer. A 2-year-old, 455 kg, 7.5 months pregnant Aberdeen Angus heifer was presented to the Boren Veterinary Medical Teaching Hospital of Oklahoma State University with a 5-hour history of progressive abdominal pain of sudden onset. According to the owner, the heifer was showing signs of colic by kicking at her abdomen and repeatedly getting up and down; she had been acting and eating normally the day prior to presentation. The animal had been raised on the farm and considered healthy until the episode of colic. Her vaccination (CattleMaster® 4 + VL5: IBR-BVD-PI3-BRSV-Campylobacter fetus-Leptospira canicola/ grippotyphosa/ hardjo/ icterohaemorrhagiae/ pomona, Pfizer, PA) and deworming (Ivermectin: Ivomec® 1% injection for cattle and swine, Merial, Iselin, NJ) status were current. She was kept on Bermuda grass pasture with 100 other cows of variable ages (all reportedly healthy) and supplemented with sweet feed. According to the owner, the heifer was not treated with any drugs prior to presentation. On initial physical examination, the heifer was in good body condition (Body Condition Score [BCS]: 7/9), with normal mentation status. The rectal temperature (38.8°C) and heart rate (79 beats/minute) were normal, but the respiratory rate was mildly elevated (49 breaths/minute), with a mild expiratory effort. The abdomen was moderately distended bilaterally, but more prominently in the right ventral quadrant. Right-sided gastrointestinal sounds were decreased, and rumen contractions were absent. Bilateral abdominal succussion and percussion revealed a right-sided fluid splash but no ping. A fetus was found on ballottement of the right abdomen. Rectal examination revealed the presence of a small amount of pasty black tarry stool and an enlarged rumen containing a moderate amount of gas in the dorsal sac. The right uterine horn contained a late-term fetus occupying the right quadrant of the abdomen and precluding palpation of the intestines. Orogastric intubation did not generate gas, frothy material, or excessive reflux of rumen fluid. Hematocrit of 36% and plasma total protein of 6.8 g/dl were considered to be within normal limits (reference range; 25–45% and 5.9–8 g/dl, respectively). Urinalysis (urine dipstick: Bili-Labstix, Bayer Corporation, Elkhart, IN) on free-catch sample revealed dark brown urine, with an elevated specific gravity (S.G. = 1.040) and moderately elevated ketones and bilirubin. Abdominal and renal ultrasonography was within normal limits; however, the motility of the intestines was reduced significantly. Abdominocentesis was negative, and fecal occult blood was positive. The animal was kept overnight for observation, and no treatments were administered. On the following morning, approximately 10 hours after admission, the animal developed severe abdominal distention and became recumbent with signs of cardiovascular collapse, including hypothermia (35.5°C), increased skin turgor and capillary refill time (>4 seconds), pale mucous membranes, bradycardia, and tachypnea (61 breaths/minute). The heifer died before any surgical or therapeutic intervention could be performed. The body of the animal was sent to necropsy, which commenced within 2 to 3 hours after death.
At necropsy, the most significant gross finding was marked serosal hyperemia of the distal jejunum, ileum, cecum, and spiral colon. The contents of the spiral colon and cecocolonic junction were bloody and the mucosal surfaces were dark red, with scattered, slightly depressed, bloody ulcers, 2 to 4 mm in diameter, and slightly raised, white/gray foci, 2 to 4 mm in diameter, corresponding to lymphoid tissue. The cecal mucosa had scattered, discrete, less than 2 to 4 mm diameter, sharply demarcated, flattened white foci, covered by a fibrinous exudate. There was no evidence of abdominal visceral displacement, intussusception, volvulus, or perforation. The uterus and fetus were grossly unremarkable and were not further examined.
Fresh, unfixed samples of colon, small intestine, liver, kidney, brain, and spleen were submitted for laboratory analysis. Fixed tissues were routinely processed for histopathology. Fixed intestinal and splenic tissues were tested for bovine viral diarrhea virus (BVDV) by immunohistochemistry as previously described. 1 Virus isolation was performed on specimens from brain, colon, small intestine, liver, spleen, and kidney using bovine turbinate (BT) and Madin Darby bovine kidney (MDBK) cells, following standard procedures. 8 When viral replication (as indicated by presence of cytopathic effects [CPE]) was observed in cell cultures, the cells were trypsinized, transferred onto spot slides, and stained by fluorescent antibody (FA) technique using fluorescein isothiocyanate-conjugated antibodies against bovine herpesvirus types 1 and 4, parainfluenza-3 virus, bovine respiratory syncytial virus, bovine coronavirus, bovine adenovirus types 3 and 5, orthoreovirus, and bovine parvovirus. When no CPE was observed, the cells were trypsinized, subcultured for an additional 6 days, and stained for noncytopathic BVDV by indirect FA using a BVDV-specific monoclonal antibody (Anti-BVDV IgG1 MAb 3.12F1: JT Saliki, OADDL, Stillwater, OK). The cultures showing CPE were amplified by subculture, concentrated by ultracentrifugation, and examined by negative staining electron microscopy.
The contents of the most severely affected section of the colon were used for aerobic and anaerobic bacterial culture and identification following standard procedures. 16 A portion of colon contents equivalent to about 50 μl was used to test for Clostridium difficile A/B toxins using a commercial ELISA kit (Clostridium difficile Tox A/B 11, TechLab, Blacksburg, VA).
Total RNA was extracted from colon samples, the virus isolate, and uninfected MDBK cell culture supernatant fluid. RNA was amplified by reverse transcriptase-polymerase chain reaction (RT-PCR) using the following primer pairs: BEV1-1000 forward: TTTAAAAgAgTcAgggggTTgTAccc; BEV1-1000 reverse: cgTcTAccAccggAcgTgTgAA, which cover the first 1000 bp of the BEV-1 genome (5'UTR and partial VP4); and AS2940U20: TcTAcAccAggTTTcccAAA/ AS4063L20: TTgTccAgcATAcTcTcAgT, which are based on astrovirus sequences but crossreact with bovine enteroviruses. Primers to β-actin were used in quality control to assure integrity of isolated RNA. RT-PCR products were visualized with ethidium bromide and agarose gel electrophoresis. Observed bands were excised and purified using High Pure PCR Product Purification kit (Roche, Indianapolis, IN). Purified products were cloned into pCR4-TOPO plasmid following manufacturer's protocols (Invitrogen, Carlsbad, CA). Plasmid DNA was purified with High Pure Plasmid Isolation Kit (Roche). Plasmid inserts were sequenced using Big Dye Terminator Cycle Sequencing Kit v3.1 (Applied Biosystems, Foster City, CA), and an ABI3100 Genetic Analyzer (Applied Biosystems). The resulting sequence was examined with Sequencher 4.41 software and subsequently compared to published sequences in Genbank by Blast software (GeneCodes, Ann Arbor, MI). 21 Further RT-PCR and sequencing were performed as above using alternative BEV primers to amplify 2 different regions of the genome. Those RT-PCR products were purified and sequenced directly. Edited sequences were aligned and phylogenetic analyses were performed using Clustal W with Lasergene software (DNASTAR, Madison, WI). The Genbank accession numbers for BEV sequences used in the phylogenetic analysis were D00214, AF123432, AF123433, AY508696, AY508697, and AJ250671.
Microscopically, the walls of the colon and cecum and their attached fatty mesentery were diffusely hyperemic and edematous, with markedly engorged blood vessels and lymphangiectasia. In the colonic and cecal mucosa, there were multiple foci of hemorrhage, necrosis, and ulceration (Figs. 1 and 2). The multifocally eroded and ulcerated mucosal surfaces were covered with blood and fibrin mixed with sloughed, necrotic enterocytes, bacteria, and birefringent plant material and debris. Multiple foci of hemorrhage expanded the lamina propria, separated the colonic glands, and elevated the overlying mucosal surfaces. Occasionally, the lamina propria collapsed over foci of moderately depleted submucosal lymphoid follicles. Occasionally, colonic and cecal glands were mildly dilated and contained sloughed, necrotic enterocytes. Mild, multifocal collections of neutrophils, lymphocytes, plasma cells, macrophages, and occasional eosinophils expanded the adjacent lamina propria. Rare enterocytes lining the colonic glands were infected with coccidial gamonts. In the colonic submucosa, dense collections of neutrophils expanded the tunica intima of some muscular arteries; the endothelium lining the engorged artery was diffusely hypertrophic (Fig. 3). Diffusely, a thin layer of fibrin strands, mixed with cellular debris, covered the serosal and mesenteric surfaces. In the submucosa of the small intestines, the lymphoid follicles were markedly depleted. In the spleen, the white pulp was moderately attenuated, characterized by lymphoid follicular depletion and lymphocyte necrosis. The liver had scattered, discrete foci of hepatocellular coagulative necrosis, with no associated inflammatory cellular infiltrates. The brain, kidney, heart, lung, and adrenal gland had no significant tissue alterations. Attempted immunohistochemical detection of BVD virus antigens on intestinal and splenic tissues was negative.
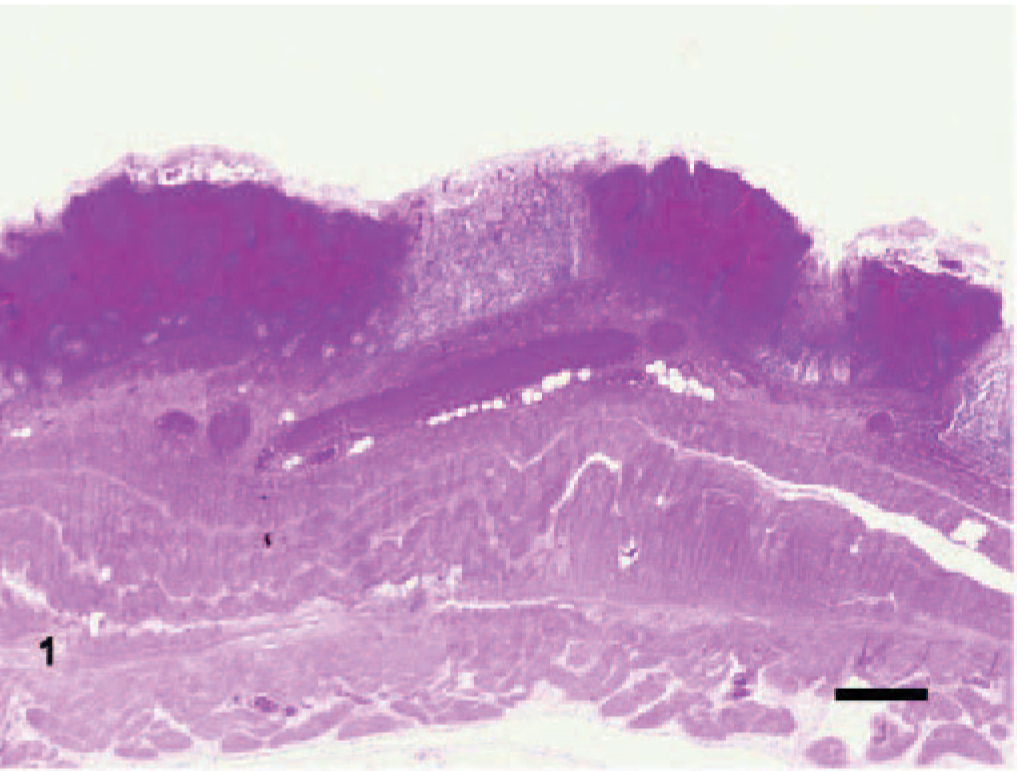
Large intestine, spiral colon; bovine. Colonic hemorrhage; the colonic mucosa has been obscured by multiple hemorrhagic foci, which expand the lamina propria and separates the colonic glands. In the submucosa, the blood vessels are engorged with blood. HE. Bar = 768 μm.
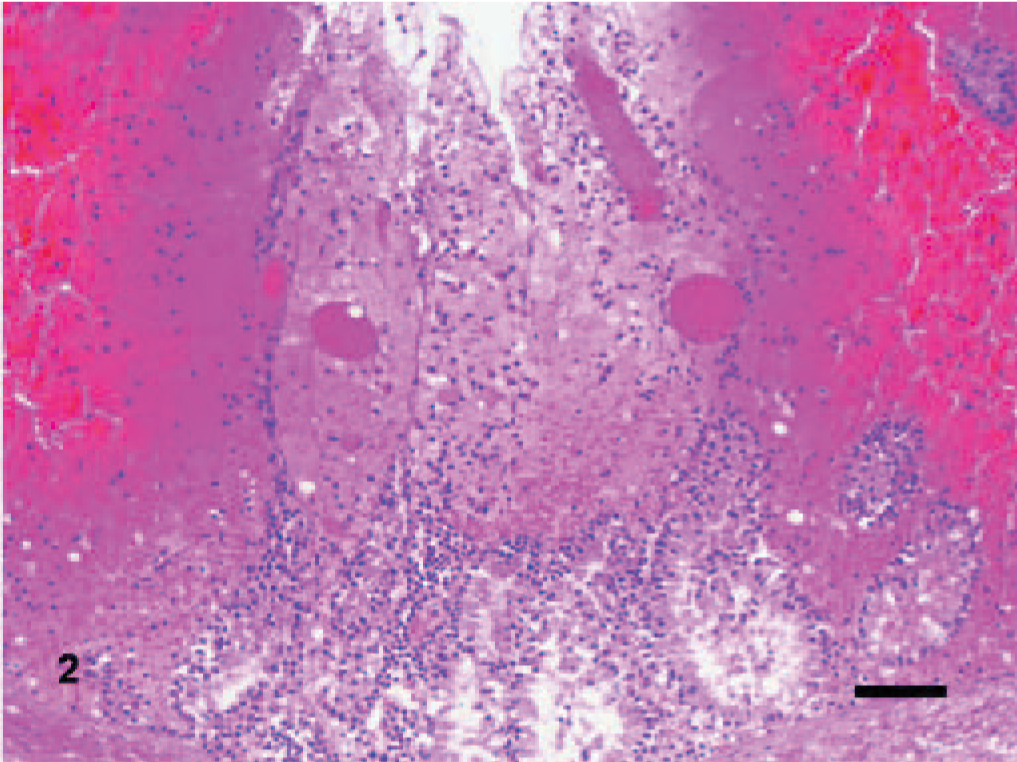
Large intestine, spiral colon; bovine. Higher magnification of Fig. 1. Overlying the mildly dilated colonic glands, scattered neutrophils, blood, fibrin, and engorged blood vessels expand the lamina propria. HE. Bar = 70 μm.
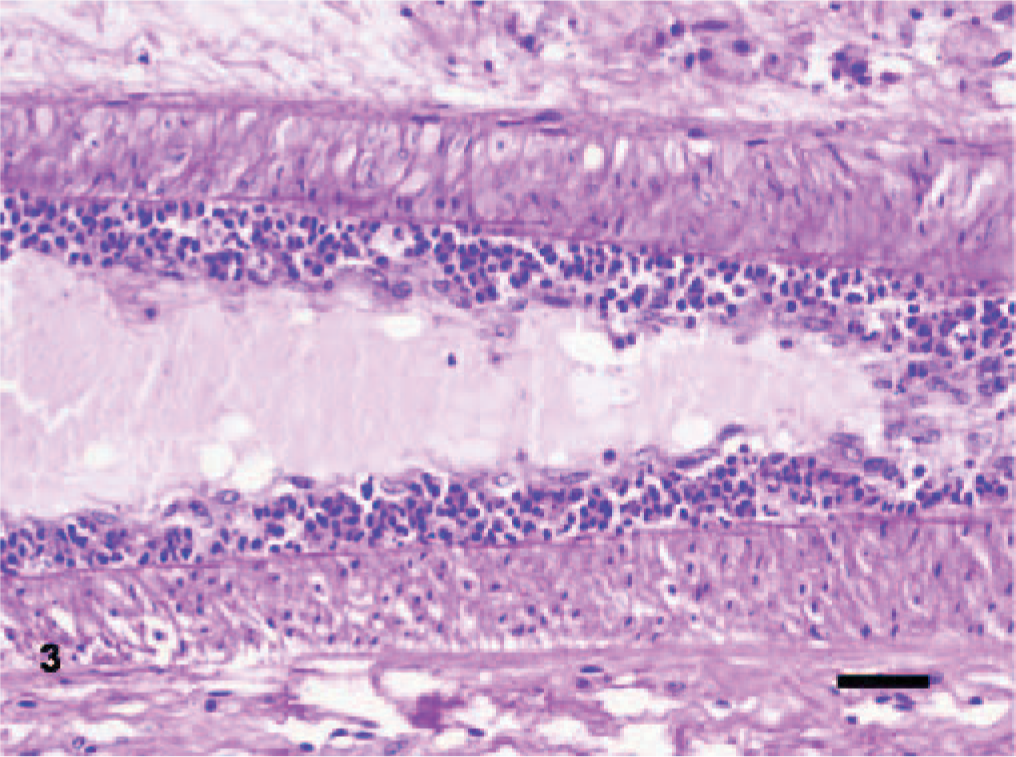
Large intestine, spiral colon; bovine. Endarteritis; a dense layer of neutrophils beneath the diffusely hypertrophic endothelium expands the intima of an artery within the submucosa of the spiral colon. Periodic Acid-Schiff (PAS) reaction. Bar = 42 μm.
The BT and MDBK cell monolayers inoculated with colonic and small intestinal tissue specimens exhibited CPE as early as 20 hours postinoculation and were completely infected by 42 hours postinoculation. Cell cultures inoculated with material from all other tissues exhibited no CPE through the second passage. Immunostaining for BVDV at the end of both passages was negative for all tissue specimens. The two CPE-positive samples did not stain for any of the viruses listed earlier. Negative-staining electron microscopic evaluation of the virus isolate revealed monomorphic small, nonenveloped viral particles of about 27 nm in diameter, consistent with the Picornaviridae, Astroviridae, or Hepeviridae (Fig. 4). 6 Based on the animal species and intestinal involvement, the virus isolate was tentatively classified as BEV.
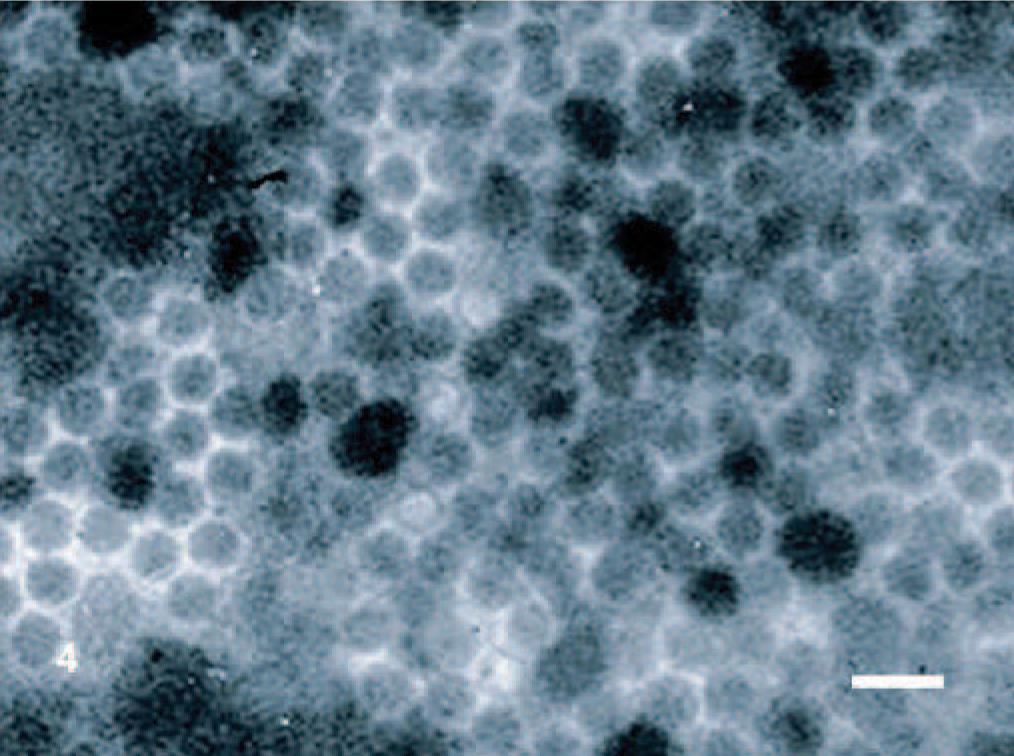
MDBK cell culture, spiral colon; bovine. Small, nonenveloped viral particles of about 27 nm in diameter were observed in the positive cell cultures. Negative staining electron microscopy. Bar = 54 nm.
Aerobic culture yielded moderate numbers of three types of Bacillus spp., small numbers of Escherichia coli and trace numbers of Klebsiella pneumoniae, which are usually considered normal microflora in mature cattle. Anaerobic culture from the colon content yielded large numbers of Clostridium sordellii, C. tertium, C. cadaveris, and Clostridum spp. No Salmonella spp. or C. difficile toxin A or B were detected.
Consistent with adequate RNA integrity, strong amplicons were obtained from all RNA extractions by RT-PCR for β-actin mRNA. To ascertain that the virus isolate was not a laboratory contaminant, RT-PCR was performed directly from RNA extracted from uninfected MDBK cell culture and from colon tissue samples; the uninfected cell culture was negative while the colon was positive for BEV, although the amplicon bands were faint and not at a concentration adequate for either direct sequencing or cloning. The RNA extracted from the virus isolate yielded strong bands that were used for cloning and sequencing. Blast analysis of the sequenced amplicon confirmed the virus isolate to be BEV (data not shown). Approximately 2230 nucleotides of the BEV isolate designated BEV-Oklahoma were sequenced, representing the 5'UTR (untranslated region) and partial VP4 coding region (a capsid protein) (∼1000 bases) and approximately 1200 bases of the VP1 coding region (immunodominant capsid protein).
Phylogenetic analysis of sequence from both regions grouped this virus isolate (BEV-Oklahoma) with BEV-1 and not with BEV-2 (Fig. 5). 5, 7, 12 Nucleotide sequence analysis of the 5'UTR and partial VP4 region (nucleotides 1–1000) revealed a 85.5% sequence identity with the UK BEV-1 VG527 strain and 77.8 to 80.7% identity with the Australian BEV-1 isolates, compared to 77.5 to 78% nucleotide identity with the US BEV-2 isolates. Analysis of a much smaller region of the 5'UTR including the 385 nucleotides of the US BEV1-261 isolate (originally isolated in Washington state in 1957) revealed an 87.1% nucleotide sequence identity (data not shown). With regards to the VP1 sequence, BEV-Oklahoma shared 79.6% nucleotide identity with the UK BEV-1 VG527 strain and 70.9 to 71.8% identity with the Australian BEV-1 isolates, compared to 61.2 to 61.5% nucleotide identity with the US BEV-2 isolates. The sequence data strongly support the identity of BEV-Oklahoma as a BEV-1.
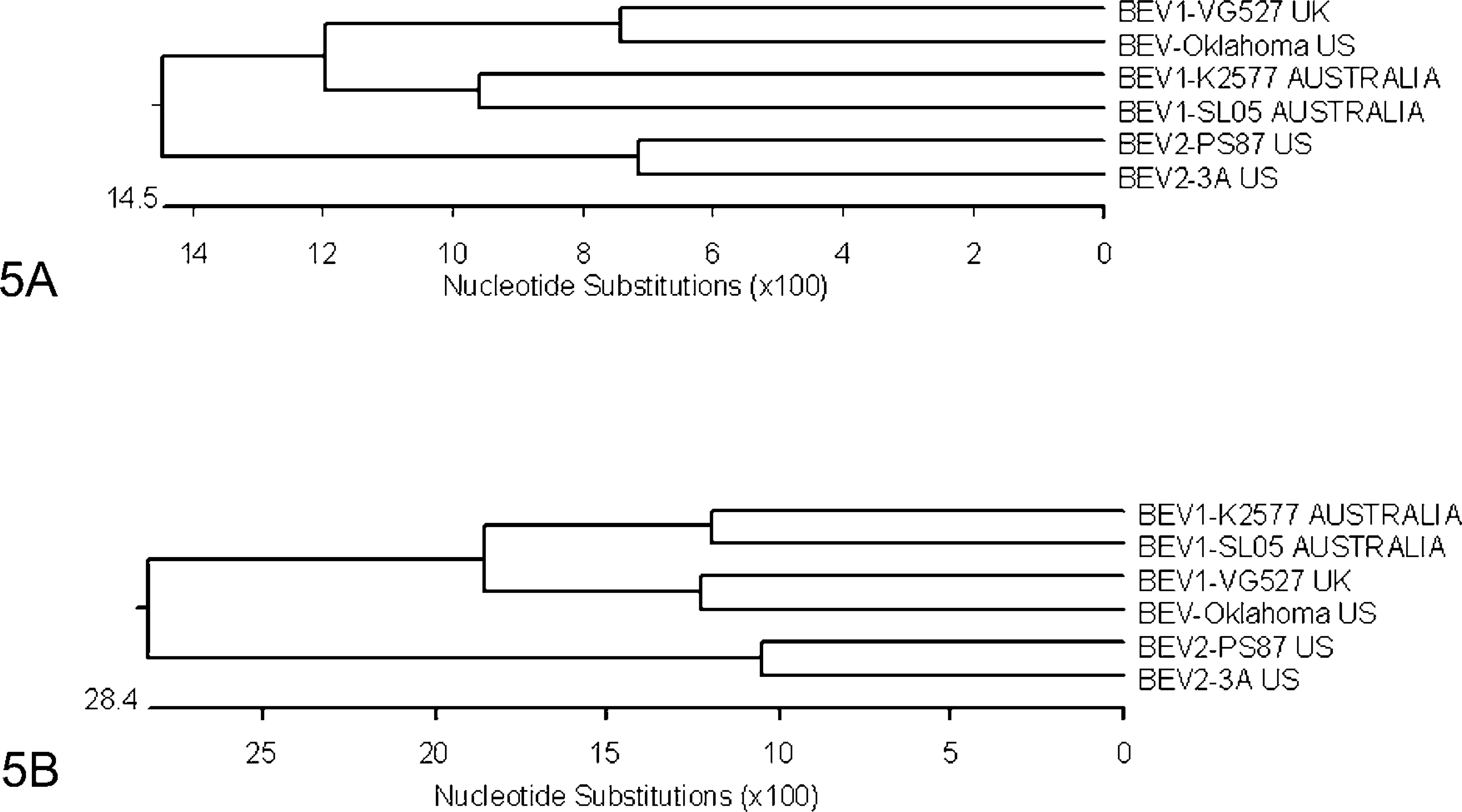
Nucleotide sequence phylogenetic analysis. A) 5' UTR and partial VP4 coding region; B) Partial VP1 coding region. Note that both regions clearly group BEV-Oklahoma with BEV-1 isolates from the UK and Australia and segregate them from the BEV-2 isolates from the USA.
Before considering BEV-1 as a potential contributor to the disease syndrome in this case, several rule outs were considered. In the USA, differential diagnoses in cattle older than 2 years of age with acute hemorrhagic typhlocolitis may include salmonellosis, mucosal disease, coccidiosis, malignant catarrhal fever, adenovirus infection, winter dysentery (coronavirus), arsenic, other heavy metals, oak or acorn poisoning, and trichuriasis. 2 The criteria used to rule out these disease syndromes was based on history, gross and microscopic findings, and laboratory test results. In salmonellosis, both the small and large intestines are usually affected; in our case, the sudden history of abdominal pain, without fever or diarrhea along with lesions limited to the cecum and colon and negative cultures of Salmonella spp. from the intestines, makes salmonellosis a less likely etiologic diagnosis. While the gross and microscopic lesions in mucosal disease and malignant catarrhal fever can be limited to the large intestine as in this heifer, these syndromes usually are accompanied by other lesions elsewhere in the gastrointestinal tract; we found no evidence of BVD virus infection in this animal and, although we did not test for MCF virus, we found no evidence of lymphadenopathy or of lesions in the brain, eye, trachea, or other organs. While microscopic evidence of coccidiosis was detected in this case, the burden of colonic gland infection was very low; furthermore, other evidence such the age of the heifer, the absence of other sick animals in the herd, and the acute course of the disease suggest that coccidiosis was unlikely to be the primary cause of the hemorrhagic typhlocolitis in this heifer. While bovine coronavirus infection potentially causes similar gross and microscopic lesions to the ones recognized on this heifer, we were not able to isolate the virus from the lesion, or other viruses such as bovine adenoviruses 3 and 5; moreover, there was no evidence of dysentery in this or in any of the other animals on the farm. Based on history, gross, and microscopic findings, the likelihood of intoxication with heavy metal or severe enteric parasitism was low as well. The isolation of bovine enterovirus from tissues with lesions of this animal in the absence other significant enteric pathogens suggests that this virus had a potentially significant role in the development of the lesions observed in this case.
The earliest reports concerning the virulence of BEV suggested that there might be an association between BEV infections and a range of diseases in cattle, as viruses were isolated from cattle with clinical signs that varied from respiratory to enteric to reproductive disease and infertility. 2, 19 Bovine enterovirus has since been isolated from apparently healthy cattle and wildlife. 6, 10, 12 Interpretation and comparisons between these reports are difficult because in many cases other infectious agents were identified or were not investigated.
As reviewed by Jiménez-Clavero and co-workers, enteroviruses are highly stable in the digestive tract, are shed in large amounts, and persist in the environment for prolonged times, and thus serve as indicators of fecal pollution. 9 In general, most enterovirus infections, which follow the fecal-oral transmission route through contamination of water and feed, occur at a young age, are usually transient, and produce lifelong immunity. In susceptible individuals, enteroviruses cause subclinical infection or mild disease, although occasional infections may cause serious disease, such as that caused by poliovirus in humans. 17 The apparent low morbidity and high mortality observed in this case may or may not be consistent with the epidemiology of enteroviruses in general, as nowhere have results been published reporting the status of either individual animals or groups of animals experimentally infected with virulent BEV-1, the lesions associated with infection and disease, its pathogenesis, or its epidemiology in cattle. Host factors, such as stage of gestation of this heifer, who was in her last trimester, and disease susceptibility may have had an effect on the apparent low morbidity, but such association remains unproven. Likewise, specific BEV-1 factors may have had an effect as well. Having knowledge about the susceptibility of young calves to challenge, about of the pathology associated with infection, if any, and about the prevalence of BEV-1 infection in herds will be essential for the understanding of the epidemiology and characterization of this virus.
A specific cause for the association between the apparent generalized lymphoid necrosis and depletion to the fatal disease in the heifer was not determined. While BEV-1 was apparently gut-specific, a significant lesion in the animal was necrosis in and depletion of lymphoid organs. Such lesions, which may have been caused by concurrent infection with undetected pathogens, endogenous release or administration of cortisol, environmental toxins, and others, may have played a potentially significant role in the susceptibility of this animal to BEV through immune suppression
Although a causal relationship between BEV-Oklahoma and the observed enteric lesions was not established in this report, an association between BEV infection and disease seems to exist in this case. The isolation of BEV-1 from the affected intestinal tissues as well as the absence of other significant enteric pathogens of similar clinical and pathological presentation supports a possible role for the BEV-1 isolate. The occurrence of this virus in an animal during its last trimester of gestation might be significant, as pregnancy could have potentially modulated its susceptibility to infectious disease. This is the first report of a partial genomic sequence of a BEV-1 isolated in the USA from an animal with fatal enteric disease in the absence of other known pathogens. The absence of studies on the pathogenesis of BEV and of its prevalence in cattle warrants further studies on the possible pathogenic significance of BEV.
Footnotes
Acknowledgement
The authors wish to acknowledge Dr. B. J. Johnson, Director of the Oklahoma Animal Disease Diagnostic Laboratory, for his support in the accomplishment of this work.
