Abstract
Immunohistochemical expression of immunocompetent cells bearing major histocompatibility complex (MHC-II) and interleukin 2-R (IL2-R) (CD25) molecules was performed on lymph nodes with spontaneous postweaning multisystemic wasting syndrome (PMWS). control lymph nodes displayed intense diffuse immunoreactivity to MHC-II in both follicles and interfollicular areas. A marked reduction of follicular MHC-II immunoreactivity and inconsistent staining of histiocytes in interfollicular areas was observed in PMWS cases with a slight lymphoid depletion; in those cases with moderate to severe lymphoid depletion, there was a progressive decrease in MHC-II expression. In controls and in slightly depleted nodes, IL2-R was equally expressed in interfollicular tissue and in follicles, whereas in moderate and severe cases, it was detected in interfollicular remnants only. Immunohistochemical staining was scored semiquantitatively. The mean MHC-II score was significantly reduced in PMWS cases compared with controls (Spearman test), whereas there was no difference in the IL2-R score. The evident reduction of MHC-II immunoreactivity suggests an impairment in MHC-II linked antigen presenting cell expression.
It is accepted that immune system dysfunction plays a central role in the pathogenesis of postweaning multisystemic wasting syndrome (PMWS); 14 what is unclear is why only a proportion of porcine circovirus 2 (PCV2) infected pigs develop the disease. 4 The pathogenic role of macrophages in triggering PCV2 replication has been clarified, but the mechanism responsible for the negative modulation (downregulation of markers) exerted on lymphocytic populations remains unknown. 6, 13
Immunohistochemical (IHC) studies on lymphoid tissues in PCV2-infected piglets that develop PMWS have documented changes in immunocompetent cells, consisting of a moderate to marked reduction or loss of B cells, decreased T lymphocytes, increased subcapsular and peritrabecular macrophages, and partial loss and redistribution of antigen presenting cells (APC) throughout lymphoid tissues. 2, 12 How lymphoid tissue depletion occurs in PMWS remains undetermined. 15 Apoptosis has recently been studied as the main mechanism underlying lymphoid depletion: PCV2-induced apoptosis led to selective B-lymphocyte depletion; 16 virus-induced apoptosis of histiocytic cells in lymphoid tissues was revealed in a mouse model of PCV2 infection. 8
The severe immune depression caused by PCV2 infection could also be explained by reduced immune-cell proliferation after reduced or absent production of growth factors for the immune system. 12 A further study on lymphoid depletion in PMWS lymph nodes indicated that the apoptotic index was lower in PMWS cases than controls both in lymphocytes and medulla-like tissue, which suggests that the reduced turnover appears more strongly influenced by decreased proliferation than by increased apoptosis. 9 Apoptosis was recently confirmed as an unremarkable feature of lymphoid depletion. 10
To find an explanation for the lymphoid depletion typical of PMWS, IHC techniques were used to evaluate the expression of immunocompetent cells bearing MHC-II and IL2-R in PMWS pigs. This study focused on the possible reduced expression of these molecules that play important roles in the immune response: MHC class II molecules are largely restricted to APCs (macrophages, dendritic cells, and B cells); 11 IL2-R, also known as CD25, drives T cells to enlarge, enter the cell cycle, and secrete cytokines that trigger the next stages of the immune response. 18
Ten superficial inguinal nodes were selected from 8- to 12-week-old moribund piglets or from those who died less than 2 hours before necropsy in an epidemic of PMWS. The presence of PCV2 genome was confirmed on blood, lung, and superficial inguinal node specimens from all pigs by polymerase chain reaction technique and on lymph node sections by in situ hybridization (ISH). 9 Three superficial inguinal nodes of 3-month-old SPF piglets negative for PCV2 served as controls.
A histologic section of each lymph node from formalin-fixed paraffin-embedded material was tested. Each node was classified on the basis of the severity and the distribution of the lesions seen in each of the following anatomical areas of the nodes: follicular tissue, interfollicular tissue, and medulla-like tissue, which is composed of meshworks of reticulum and many fine collagen fibers that support a few lymphocytes and histiocytic cells. Follicle remnants, cell depletion in the lymphoid interfollicular tissue and prevalence of lymphoid with respect to medulla-like tissue are observed in PMWS cases with slight lymphoid depletion. 17 In moderate PMWS cases, the absence of follicles and more extensive cell depletion in the lymphoid interfollicular tissue is evident, but lymphoid tissue is still more prominent than the medulla-like tissue. Severe cases of PMWS are characterized by the absence of follicles, lymphoid cell depletion, and prominence of medulla-like tissue. 9, 12 Similar criteria have been adopted by other authors to classify lymphoid organs of affected pigs. 10
Four-micron-thick replicate sections were dewaxed in toluene and were rehydrated in a graded alcohol series. Endogenous peroxidase was blocked by 0.3% hydrogen peroxide for 30 minutes. Sections were rinsed in Tris buffer, immersed in citrate buffer (2.1 g citric acid monohydrate/liter distilled water), pH 6.0, and incubated for 4 periods of 5 minutes each in a microwave oven at 750 W. After microwave irradiation, sections were allowed to cool to room temperature (approximately 20 minutes). The sections were incubated overnight at 5°C with the 2 primary monoclonal antibodies. The first antibody, an anti MHC-II (clone 42A),a which recognizes the SLA-II-DQ fraction, was diluted 1°400 with Phosphate Buffered Saline (PBS). 5
The second antibody was an anti-IL2-R.1 (CD25/IL2-Rα),b specific to a 55 kD protein known as an IL2-R, which is expressed on the surface of activated T cells; 1 it was used diluted 1°100 in PBS within 1% bovine serum albumin. A replicate section from each node was treated with an irrelevant antibody in place of the primary antibodies and served as an internal control.
A highly sensitive streptavidin-biotin-peroxidase kitc was used to disclose the reaction. Diaminobenzidine (0.05% for 7 minutes at room temperature (RT)) was used as the chromogen; sections were counterstained with Papanicolaou hematoxylin (3 seconds), rinsed in tap water, dehydrated, and mounted with DPX mountant for histology (neutral solution of polystyrene and plasticizers in xylene).
IHC staining was scored by 2 investigators (L. M. and M. Z.) who did not know whether the slides were from affected or control animals. The following semiquantitative method was used to record observations: score 0 (completely negative staining), score 1 (up to 25% stained cells in the field), score 2 (25–75% stained cells), score 3 (more than 75% stained cells). Fields were selected from follicular, interfollicular, and medulla-like zones, and observed with a 40× lens. Three observations were made on each animal, and a mean score was recorded.
Data were tested with the Shapiro and Wilk's W test for normality. The Spearman test was used to compare groups. A conventional 5% level was used to define statistical significance.
According to the histopathologic criteria presented, 3 animals showed a slight lymphoid depletion; 4, a moderate depletion; and 3, a severe depletion. In controls, a strong dark-brown MHC-II immunoreactivity was detected in the cytoplasm and/or on the surfaces of cells located in the follicles and to a lesser extent in the interfollicular tissue (Fig. 1). At higher magnification, a few reactive cells (dendritic cells) were also detected in the medulla-like tissue, whereas in the capsule and the trabecular connective tissue, no staining was evident. In follicles the stained cells were almost all follicular dendritic cells (Fig. 1. Inset, upper right) and lymphocytes. In the interfollicular tissue, the dendritic cell population, whose cytoplasmic projections were well demarcated, was totally stained, (Fig. 1. Inset, lower right), whereas the lymphocytes did not stain consistently. In the medulla-like tissue, the antigen detection was limited to sparse cells with abundant cytoplasm, long cytoplasmic projections, and a large central nucleus, identified as macrophage/histiocytic type (dendritic) cells.
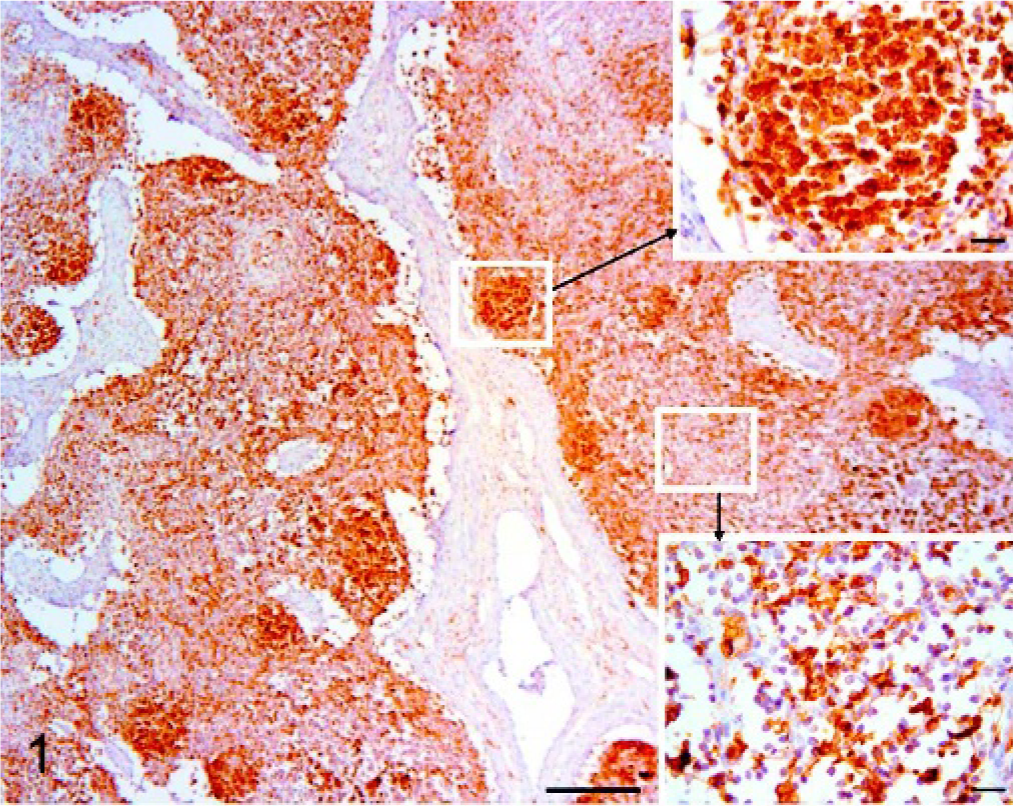
Superficial inguinal node, pig. Control. Immunohistochemistry for MHC-II. Bar = 200 μm. Dendritic cells both in follicles (Inset, upper right; Bar = 20 μm) and interdigitating cells in the interfollicular tissue (Inset, lower right; Bar = 20 μm). DAB staining, Papanicolaou hematoxylin counterstain.
In comparison with controls, a severe reduction of MHC-II both in the cytoplasm and on cell surfaces was observed in slightly depleted PMWS cases (Fig. 2). The reduction was more evident in follicles than in the interfollicular tissue. In the latter compartment, high endothelial venules (HEV) located in the paracortex of lymph nodes, and identifiable by rounded endothelial cells, 18 were also positive. The medulla-like tissue was negative for MHC-II.
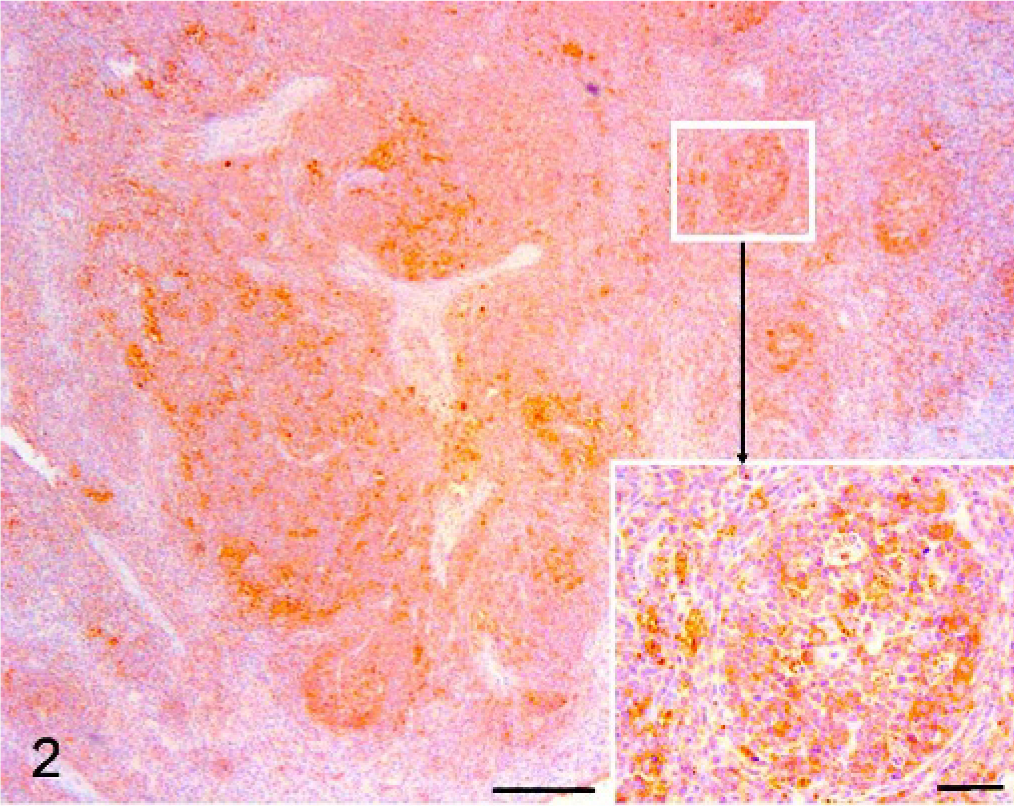
Superficial inguinal node, pig. PMWS-initial case. Immunohistochemistry. Diffused marked reduction of MHC-II expression (Bar = 200 μm) and marked reduction especially in follicles (Bar = 50 μm). (Inset: a follicle at higher magnification; Bar = 20 μm) DAB staining, Papanicolaou hematoxylin counterstain.
In PMWS cases with a moderate lymphoid depletion, the expression of MHC-II molecule in the follicles was not recognizable. In the remaining interfollicular tissue, interdigitating cells and cells arranged into nodular formations (Fig. 3) formed by 15–30 macrophages (granulomata) were focally labelled (Fig. 3. Inset). These cells, with abundant cytoplasm and round to oval nucleus, had a strong cytoplasmic positivity and contained the characteristic cytoplasmic circoviral inclusions in corresponding HE sections (Fig. 3. Inset, lower right). The medulla-like tissue was negative for MHC-II.
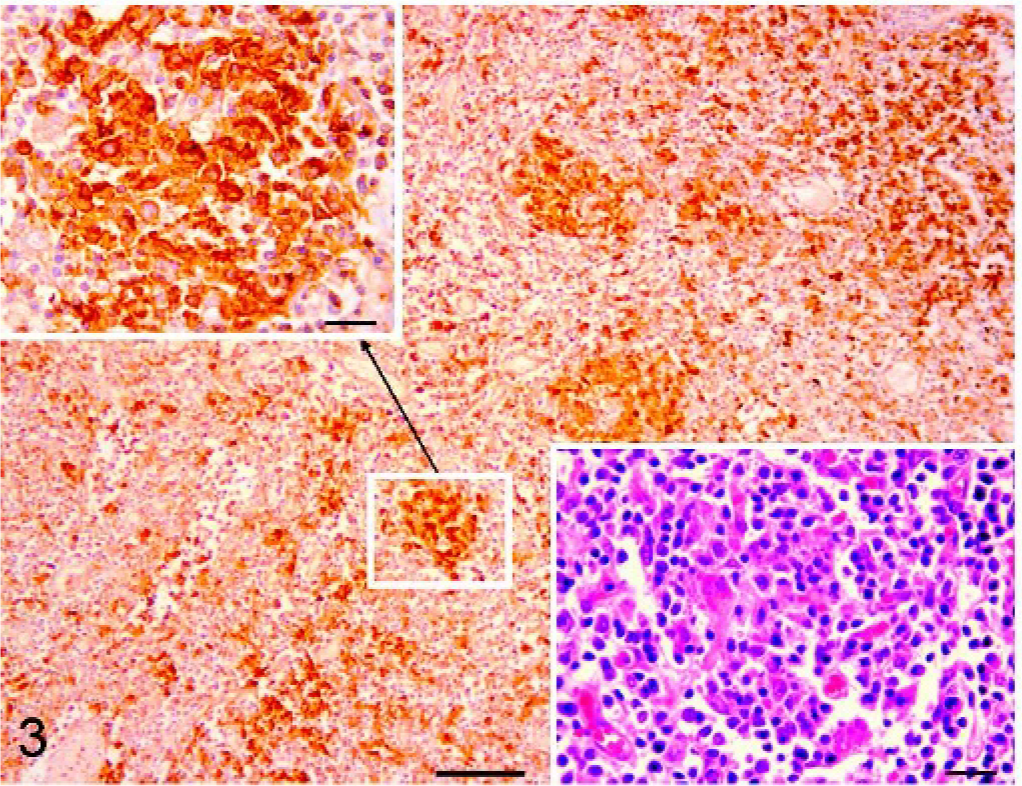
Superficial inguinal node, pig. PMWS case with moderate lymphoid depletion. Immunohistochemistry. In the remaining interfollicular tissue (Bar = 100 μm), interdigitating cells and cells arranged into nodular formations formed by 15–30 macrophages were labelled (Fig. 3. Inset; Bar = 20 μm). These granulomata contained the characteristic cytoplasmic circoviral inclusions in corresponding HE sections (Fig. 3. Inset, lower right; Bar = 20 μm). DAB staining, Papanicolaou hematoxylin counterstain.
In PMWS cases with a severe lymphoid depletion, the staining was scattered in the remaining interfollicular tissue; the stained cells consisted of histiocytes/macrophages and rare lymphocytes. In medulla-like tissue, which in this stage of disease was predominant over lymphoid tissue, no staining was observed.
The brown stain of the IHC reaction was observed on the cellular surface in the follicular (Fig. 4a) and interfollicular (Fig. 4b) lymphoid tissue of controls; within the medulla-like compartment immunoreactivity was not detected. The staining involved lymphocytes and lymphoblasts both in follicles and interfollicular tissue. The follicular dendritic cells and the interdigitating cells were negative for IL2-R.
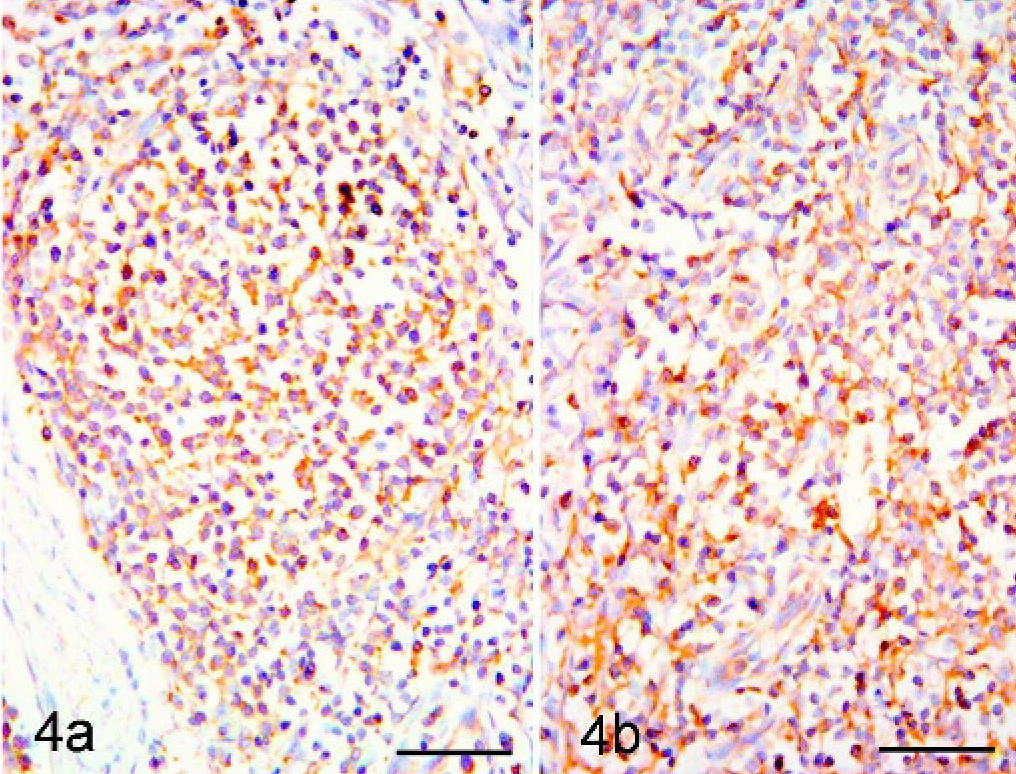
Superficial inguinal node, pig. Control. Immunohistochemistry for IL2-R. Immunoreactivity revealed in both follicular (Fig. 4a, left side. Bar = 50 μm) and interfollicular (Fig. 4b, right side. Bar = 50 μm) zone. DAB staining, Papanicolaou hematoxylin counterstain.
The type of expression (mainly membranous) and type of positive cells (lymphocytes) in PMWS cases with a slight lymphoid depletion did not differ from controls. IL2-R expression was distributed both in the remaining portions of follicles and in the interfollicular tissue.
A moderately decreased expression of IL2-R with respect to controls was found in moderate PMWS (Fig. 5) and severe PMWS (Fig. 6) cases.
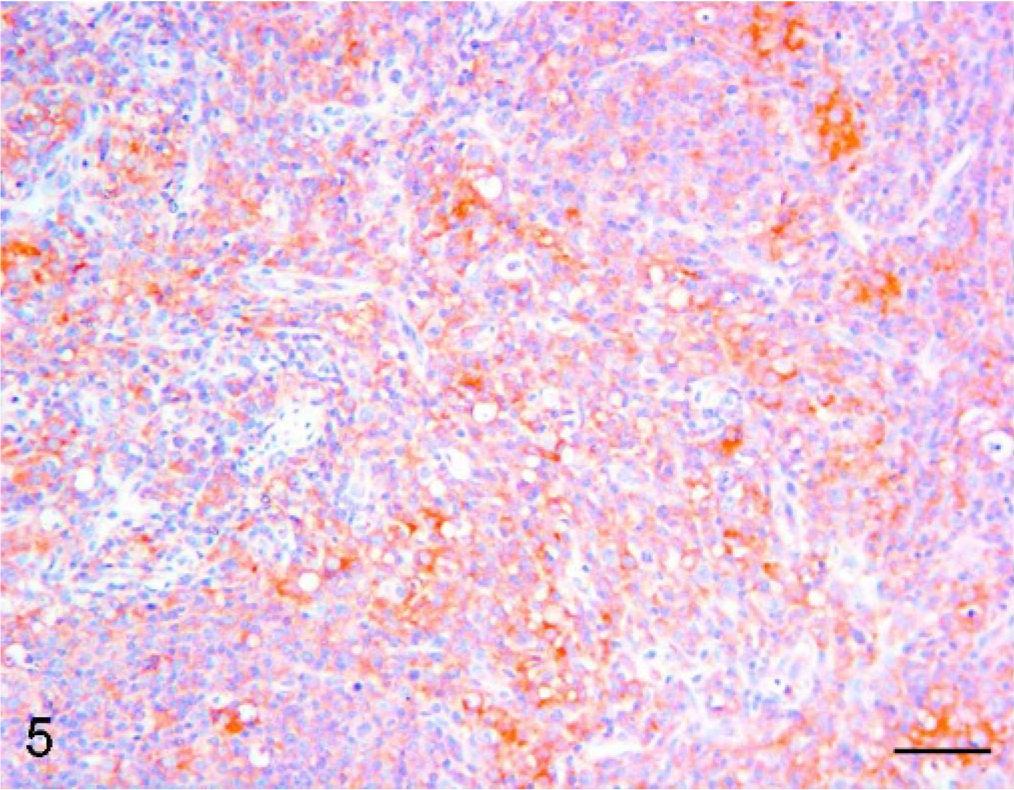
Superficial inguinal node, pig. PMWS case with moderate lymphoid depletion. Immunohistochemistry. IL2-R was displayed by lymphocytes (Bar = 50 μm). DAB staining, Papanicolaou hematoxylin counterstain.
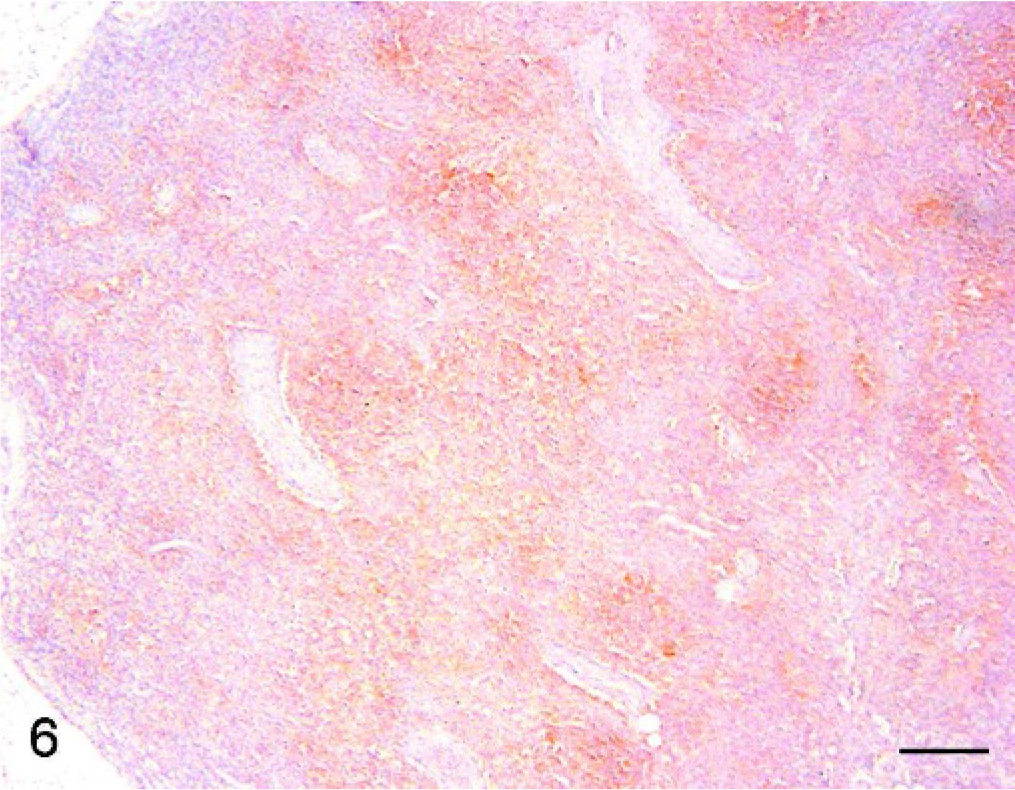
Superficial inguinal node, pig. PMWS case with severe lymphoid depletion. Immunohistochemistry. IL2-R immunoreactivity was displayed by lymphocytes (Bar = 200 μm). DAB staining, Papanicolaou hematoxylin counterstain.
A semiquantitative evaluation resulted in a significantly reduced mean MHC-II score in controls compared with PMWS (P < .05), whereas there was no difference in IL2-R score (Table 1). The tested data were not normally distributed.
Mean score of MHC-II and CD25 immunohistochemical evaluation in controls (CTR), in PMWS cases with slight, moderate, and severe lymphoid depletion. Spearman test was used to compare groups; a conventional 5% level was used to define statistical significance.

These IHC results yielded information on the distribution of cell types expressing MHC-II and IL2-R markers in nodal tissue from controls and PMWS cases. The tissue distribution of MHC class II molecules is restricted to antigen presenting cells (APC) 2 constitutively found on the B cells, the dendritic cells, and the macrophages and can be induced on T cells and keratinocytes. 18 In addition, endothelial cells that form HEVs are morphologically linked by discontinuous “spot-welded” junctions, facilitating the passage of recirculating lymphocytes, are induced to express MHC-II. 18
The antigen receptors of T lymphocytes are specialized to recognize a foreign antigenic peptide fragment bound to an MHC molecule. A T cell with a receptor specific for the complex formed between that particular foreign peptide and the MHC molecule can then recognize and respond to the APC. 7 An anti SLA-II-DQ antibody called BL2H5, confirming the macrophage origin of the labelled cells, demonstrated that both APC and PCV2 infected ISH and IHC positive cells have a very similar pattern of distribution, reinforcing the observation that all types of APC could be PCV2 infection target cells, not just macrophages. 2 These results are in line with the present observations that the MHC-II antibody was highly expressed by APC in controls, whereas its expression was decreased in PMWS cases.
Literature data on the IHC evaluation of the activation level of IL2-R are scant: a monoclonal IL2-R antibody anti-CD25 surface molecule that works on formalin-fixed paraffin-embedded material and on samples from peripheral blood identifying the activated lymphocytes has recently been used. 19
The reduced activation revealed by IL2-R expression in pig lymph nodes was ascribed to a reduction of cellularity rather than an effective downregulation. The modulation of IL2-R expression was not a significant finding in PMWS compared with control cases, appearing in contrast with the deficit of lymphocytic activation that follows a reduced expression of MHC-II molecules. This apparent discrepancy may be explained by the fact that the level of IL2-R expression is not exclusively MHC-II dependent but can also follow the MHC-I dependent activation of TCD8+ lymphocytes to that of B lymphocytes in response to the so-called T-independent antigens, other lymphocytic populations (i.e., natural killer cells), and lymphocytic activation by superantigens. 3, 18
The evaluation of the expression of the 2 markers of MHC-II and IL2-R disclosed that down-expression of MHC-II is the most important event in PMWS lymphadenopathy compared with the IL2-R expression, causing a significant impairment of APC function. According to the proposed pathogenic mechanism, the failure to trigger an immune response could be from a lack of interaction between APC and TCD4+ cells after the direct effect of PCV2 on APC. This could lead to impaired proliferation, with a loss of B and T cell renewal in a labile compartment, such as the lymphohemopoietic tissue, thereby culminating in cellular depletion characterized by a quantitative reduction of immunocompetent cells.
In conclusion, the negative modulation of MHC-II expression in PMWS lymphadenopathy seems to be a more important mechanism than the modulation of IL2-R expression of activated lymphocytes. Immunodeficiency may result from impaired antigen presenting function caused by decreasing MHC-II expression, rather than an intrinsic decrease in lymphocyte activation.
Sources and Manufacture
a. VMRD, Inc., Pullman, WA, USA
b. Labogen, Italy
c. BIO SPA, Milan, Italy
Footnotes
Acknowledgement
Anne Collins edited the English text.
