Abstract
Claw biopsy samples of 11 dogs with lupoid onychodystrophy were evaluated. They were stained with hematoxylin and eosin and with antibodies against CD 3 as a T-cell marker, BLA 36 and HM 57 (CD 79α) as B-cell markers, and lysozyme, Mac 387, and major histocompatibility complex (MHC) class II as a marker for histiocytes using an immunoperoxidase and avidin-biotin technique. Inflammatory cells were counted in five high-power fields. The inflammatory infiltrate comprised predominantly B cells and T cells. Macrophages were typically only present in small numbers. CD 3, BLA 36, lysozyme, and MHC class II preserved significant antigenicity during formalin fixation and short decalcification for 24–48 hours, whereas CD 79α and particularly Mac 387 seemed to be more susceptible to denaturation by the decalcification process.
Diseases limited to the canine claw have received little attention in the past, and most of the information available was based on anecdotal evidence. 12,23,30 In the last decade, a number of studies have been published evaluating claw disease in the dog. 3,5,15,25–27,33,34,36 These studies attempted to define claw disease and possible etiologies in more detail and gave useful information. One of these studies reported a number of dogs with exclusive claw disease, where the only histopathologic findings documented were a bandlike mononuclear infiltrate with interface dermatitis changes, 34 including basal cell hydropic degeneration, degeneration or necrosis of individual keratinocytes in the basal cell layer, and pigmentary incontinence. Interface dermatitis and a bandlike infiltrate of plasma cells and lymphocytes have been reported as suggestive of discoid (cutaneous) or systemic lupus erythematosus in the dog. 13,34,39 The proposed name for this claw syndrome was lupoid onychodystrophy. 34 No other signs of systemic lupus erythematosus, such as elevated antinuclear antibody titers, anemia, thrombocytopenia, or evidence of glomerulonephritis based on urinalysis and biochemistry panels, were present in dogs with lupoid onychodystrophy in a subsequent prospective study. 25 Furthermore, some dogs with these histopathologic features were diagnosed with bacterial infection or food adverse reaction, indicating that the presence of bandlike mononuclear infiltrates and interface dermatitis may be a “reaction pattern” of the canine claw. Vacuolization of claw matrix keratinocytes has been documented in normal canine claws, 27 complicating the interpretation of the histopathologic findings further.
Mononuclear cells commonly seen in dermal infiltrates include histiocytes/macrophages and lymphocytes. Histiocytes and activated lymphocytes may be difficult to distinguish using light microscopy with routine stains such as hematoxylin and eosin (HE). This study characterizes the nature and immunophenotype of the inflammatory infiltrate noted in dogs with clinical signs and the histopathology compatible with lupoid onychodystrophy using antibodies against CD 3, lysozyme, major histocompatibility complex (MHC) class II, BLA 36, and HM 57 (CD 79α) as markers for T cells, macrophages, and B cells.
Materials and Methods
Eleven samples from dogs with a histopathologic diagnosis of lupoid onychodystrophy and clinical disease restricted to the claws and claw beds were included in the study. The clinical signs included onychoschizia, onychomalacia, and onychomadesis. Complete blood counts, serum biochemistry panels, urinalyses, and total thyroxine (tT4) concentrations were within normal ranges. Antinuclear antibody titers and fungal cultures were negative. Routine histopathology of claw biopsy samples revealed interface dermatitis with basal cell hydropic degeneration and necrosis of individual basal keratinocytes, pigmentary incontinence, and a bandlike infiltrate of predominantly plasma cells and lymphocytes. Signalment, age of onset, duration of disease, response to treatment, and identified etiologic factors were recorded. The claw specimens were obtained by onychobiopsy without onychectomy, 26 initially fixed in formalin and then pretreated with formic acid for 12–48 hours to soften the tissue. Specimens were trimmed, processed, paraffin embedded, and stained with HE.
Sections were then recut and stained with antibodies against CD 3 (Novocastra, Newcastle upon Tyne, UK) labeling T cells, BLA 36 (Biogenex, San Ramon, CA, USA) and CD 79α (HM 57, Dako, Carpenteria, CA, USA) labeling B cells, and lysozyme (Zymed, San Francisco, CA, USA), Mac 387 (Dako), and MHC class II (clone TAL.1B5, Dako) labeling macrophages, using an avidin–biotin immunoperoxidase technique, as described previously. 4,14,16 Heat-induced epitope unmasking, using sodium citrate buffer (BLA 36, CD 79α), ethylenediamine tetraacetic acid buffer (CD 3) or protease XIV antigen retrieval (Sigma, St Louis, MO, USA) (Mac 387, lysozyme) was applied to the tissues before the application of the primary antibody. Antigen retrieval was not done before staining for MHC class II. Diaminobenzidine was used as the chromogen substrate. Positive control slides of canine lymph node were included in each run and examined for appropriate staining. Details of antibody concentrations, incubation times, temperature, and concentration of secondary antibodies are outlined in Table 1. Increasing the heat-induced epitope retrieval step to 30 minutes from the usual 20 minutes was necessary in some cases to increase the intensity of the CD 3 stain.
Antibody concentrations, incubation times, temperature, and concentration of secondary antibodies used to stain canine claw tissue samples immunohistochemically.
Inflammatory cells were evaluated in sections stained with HE. Using the above markers, immunostained cells were identified in the claw matrix (which was defined as the epidermis giving rise to the claw horn) and in the underlying dermis. Total numbers of inflammatory cells and cells staining positively with each marker were counted in five high-power fields (× 400) measuring approximately 0.2 mm2 each. The average of the five counts was determined. Numbers were also expressed as a percentage of the cellular infiltrate present. The mean number of staining cells and their standard deviation as well as the mean percentage of the total inflammatory infiltrate of all sections stained with a particular marker were calculated. The percentage of stained cells of the total inflammatory infiltrate in the dermis and epidermis was compared for each marker with a Wilcoxon matched pairs test. Because the numbers were small and we wanted to decrease our chance of a type-II error and thus the chance of missing a real difference, P < 0.1 was considered significant.
Results
Clear immunostaining with antibodies against CD3, BLA 36, and lysozyme was observed in all specimens. Four of the 11 specimens did not show any significant staining with the CD 79α antibody but were positive for BLA 36. No significant staining was observed in any specimen using Mac 387, although intense staining was observed in the positive control slides, which were not subjected to tissue-softening pretreatments.
It was not always possible to evaluate exactly the same site for each of the stains because of processing artifacts. For this reason, it was not possible to count cells in more than three complete high-power fields (× 400) in specimen 7. In this case, the average number of cells counted in these fields was used for calculations. The percentage of cells stained with each marker of the total inflammatory infiltrate in each specimen is listed in Table 2. The mean number of staining cells and their standard deviation as well as the mean percentage of the total inflammatory infiltrate of all sections stained with a particular marker are listed in Table 3. Sections stained with HE were also evaluated; results are listed in Table 4. There was no significant difference between the percentage of cells stained with CD 3, MHC class II, or CD 79α in the epidermis versus the dermis. Cells stained with antibodies against BLA 36 comprised a significantly greater part of the epidermal infiltrate (P = 0.02). The cells staining for lysozyme formed a larger part of the infiltrate in the dermis (P = 0.02).
Inflammatory cells of the claw matrix staining with various immunohistochemical markers expressed as a percentage of the total dermal (epidermal) inflammatory infiltrate.∗
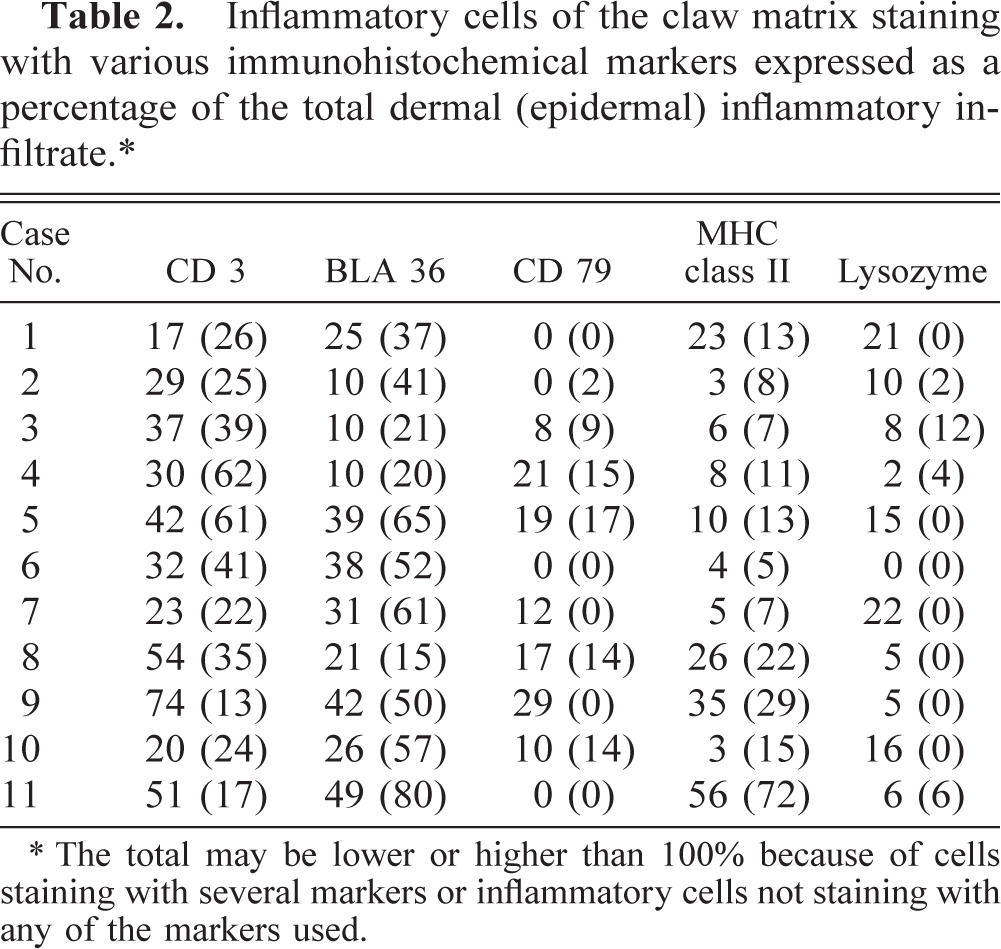
∗ The total may be lower or higher than 100% because of cells staining with several markers or inflammatory cells not staining with any of the markers used.
The mean number, standard deviation (SD), and percentage of the total inflammatory infiltrate of cells staining with a variety of immunohistochemical markers in the claw matrix of biopsy specimens of 11 dogs with lupoid onychodystrophy.∗
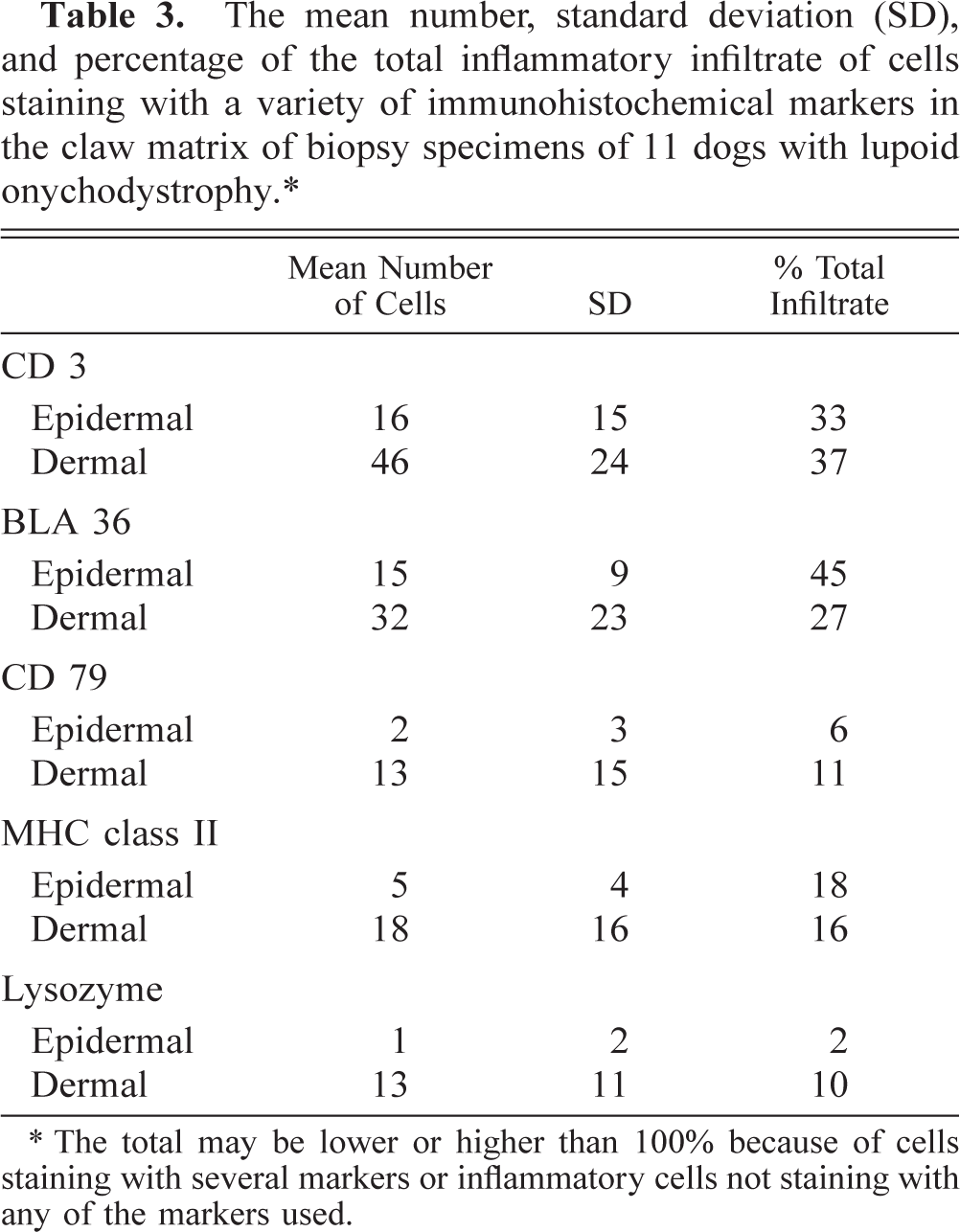
∗ The total may be lower or higher than 100% because of cells staining with several markers or inflammatory cells not staining with any of the markers used.
Summary of histopathologic findings in dogs with lupoid onychodystrophy on sections stained with hematoxylin and eosin.

Discussion
A number of publications in the recent past have evaluated claw disease in the dog and provided much-needed information. 3,5,25–27,33,34 A syndrome first described by Scott et al. 34 and named lupoid onychodystrophy by these authors showed histopathologic changes compatible with lupus erythematosus. However, in a subsequent prospective study of this syndrome, 25 no other systemic or cutaneous features of canine lupus erythematosus could be identified. Furthermore, in that study, dogs with claw disease due to pyoderma and food adverse reaction showed the same histopathologic features described by Scott et al. 34 These features included interface dermatitis, lymphocytic exocytosis, and hydropic degeneration of the basal cell layer, and the authors suggested that the observed interface dermatitis may be a reaction pattern of the canine claw matrix rather than being diagnostic for a particular etiology. As T and B lymphocytes cannot be differentiated readily on sections stained with HE and as activated lymphocytes and histiocytes may have a similar appearance, we attempted to further characterize the mononuclear infiltrate seen in canine lupoid onychodystrophy using a panel of monoclonal antibodies.
The sections stained with HE revealed the classic changes of interface dermatitis (Fig. 1). On the whole, the cell types observed in the dermis on HE correlated with the results of quantification of immunostained cells. Of the three dogs with a lack of necrotic basal keratinocytes, one dog had an underlying food adverse reaction and one had disease seasonal in summer and fall, thus raising the possibility of allergic claw disease. An underlying etiology could not be clearly identified in the seven cases, in which necrosis of basal keratinocytes was observed. An immune-mediated pathogenesis has been suggested for dogs with lupoid onychodystrophy and may have played a role in these dogs.
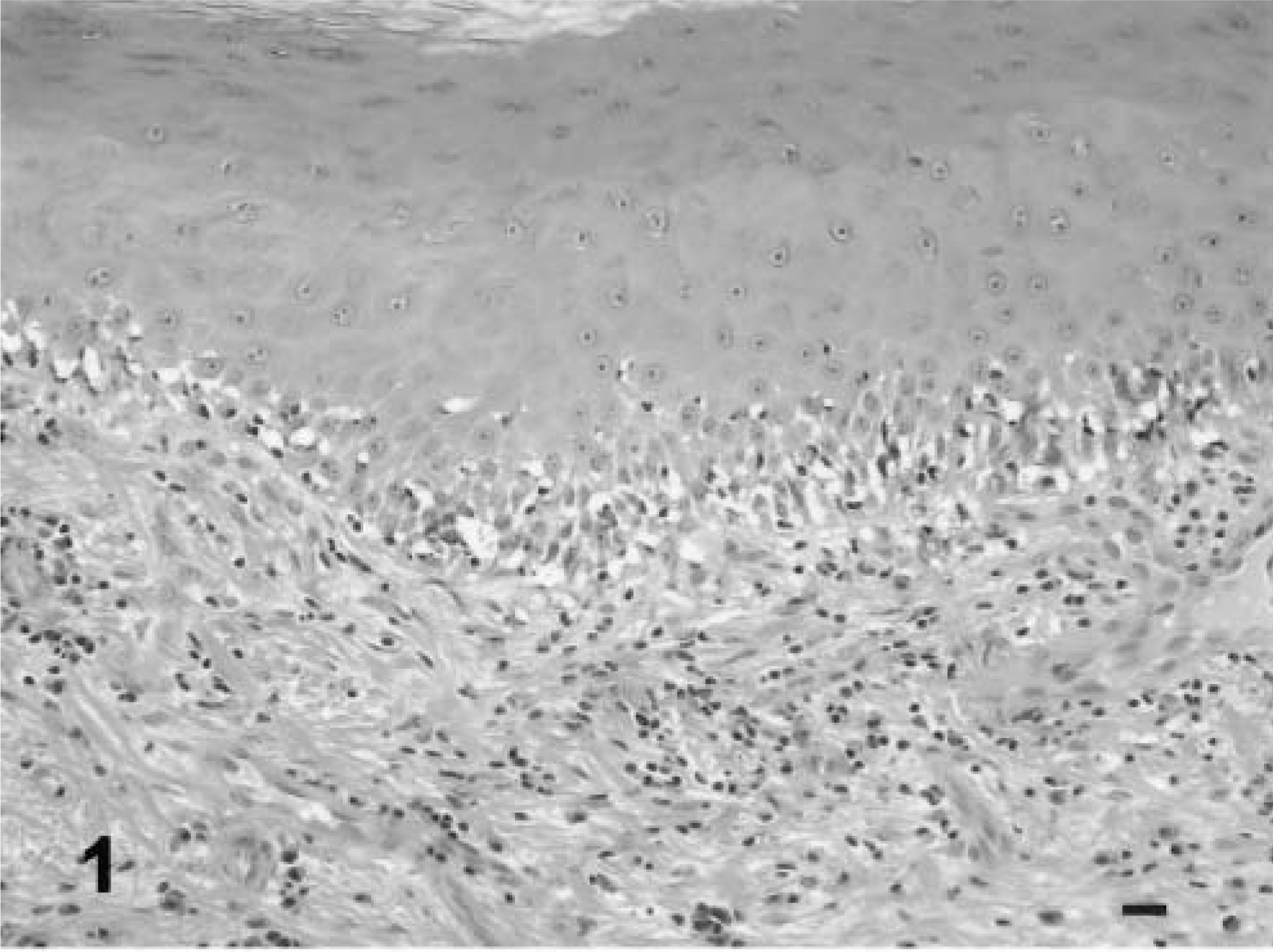
Claw matrix. Note the lymphocytic infiltrate, pigmentary incontinence, lymphocyte exocytosis, and satellitosis. HE. Bar = 40 µm.
It is appreciated that the method of cell quantification in histologic sections must be considered arbitrary and that the “correct method of quantification” has not been established. In our study, there was a wide variation in cell numbers. This was predominantly due to two reasons. First, there was a variation in focal intensity of the infiltrate within any given section. To minimize the influence of chance, the cells in five high-power fields were counted manually, and the numbers were averaged. In most of the sections, the entire inflamed area could be examined because of the small size of the sampled claw matrix. In addition, we compared the cells stained with individual markers and expressed them as percentages of the total inflammatory cell population. Counting all the inflammatory cells present is problematic because fibroblasts with rounded nuclei may sometimes be mistaken for inflammatory cells and falsely elevate the total inflammatory cell count. Such fibroblasts are not very numerous and present in all specimens, limiting the influence of this possible error on the results. The second factor complicating the quantitative assessment is that at least seven sections were obtained from each specimen. At least one, but typically more, sections for each of the seven stains was required because by nature this staining frequently involves several dilutions. Thus, the focal variation in intensity of the inflammatory infiltrate also led to a variation in total cell numbers in different sections from the same specimen. The semiquantification was considered preferable to an evaluation based on estimates only.
The T-cell marker (CD 3) provided clear recognition of canine T lymphocytes in formalin-fixed specimens. CD 3 is a lineage marker exclusive to the T cell line. CD 3, a set of five polypeptides, is closely associated with the T-cell antigen receptor in the T-cell receptor complex. 1 The CD 3 complex is highly conserved among various mammalian species, and antibodies generated against peptide sequences from a conserved intracytoplasmic region of the CD 3 molecule have been shown to have reactivity to a very broad range of species, including the dog. 11,20 In our study, CD 3 antibody stained lymphocytes in all samples, indicating that it is a useful marker for T lymphocytes even with tissue-softening pretreatments between 24 and 48 hours (Fig. 2). T lymphocytes were a prominent cell type involved in claw matrix inflammation and comprised on average one third (between 17% and 74%) of the dermal inflammatory infiltrate.
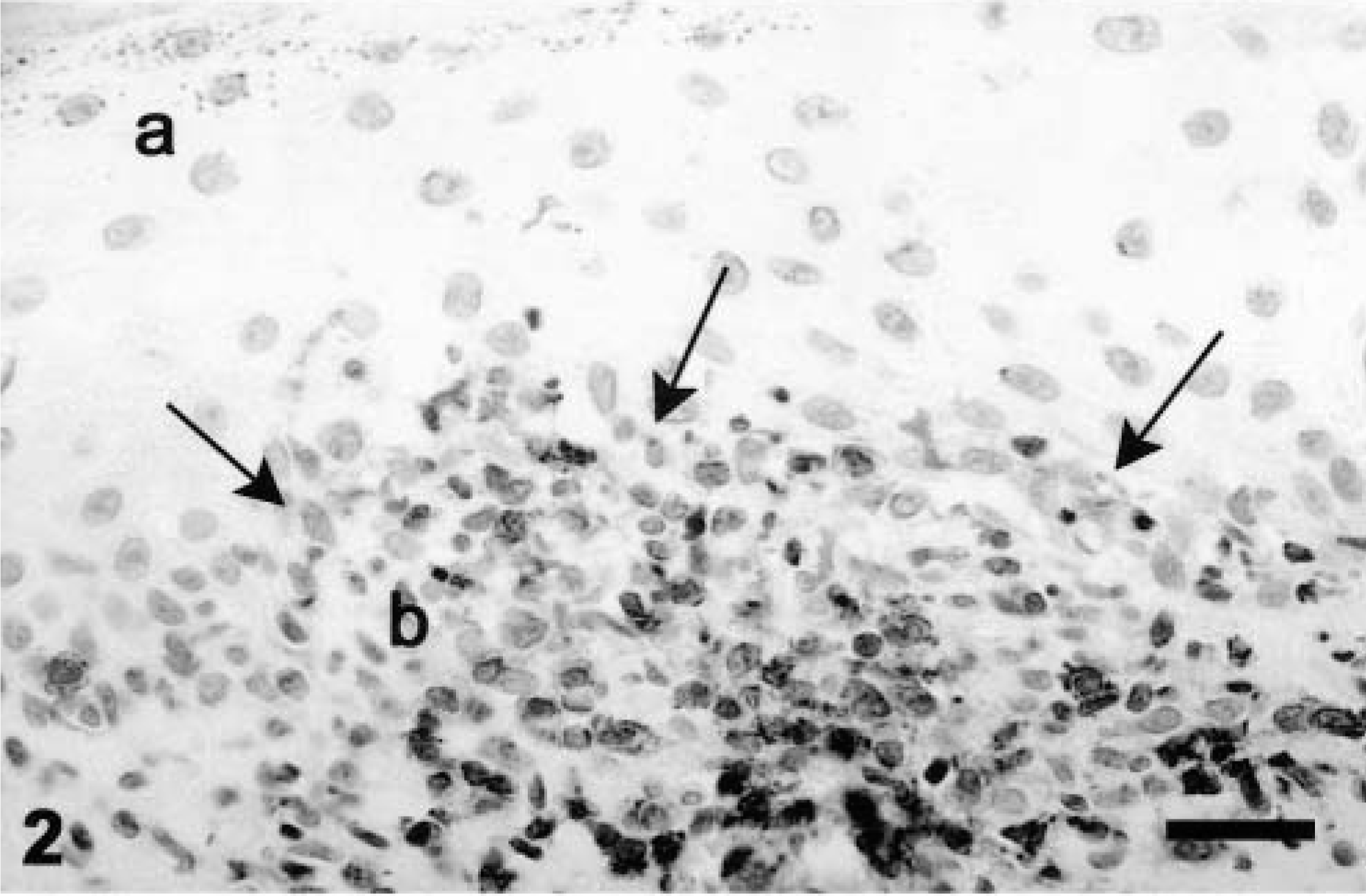
Claw matrix. Some cells are moving into the epidermis, obscuring the dermoepidermal junction (indicated by arrows) (a). Inflammatory cells are predominantly within the dermis. CD 3 antibody (b). Bar = 40 µm.
Lymphocytic interface dermatitis was recently reported in claw beds of dogs with a positive diagnosis of leishmaniasis but clinically unaffected claws (A. F. Koutinas, personal communication). This suggests that an antigen elsewhere in the body may be able to drive an immunologic reaction at the claw bed interface. The same study also reported an identical interface reaction in those dogs with leishmaniasis, which clinically showed onychogryphosis. No immunohistochemistry studies had been performed on those samples. Other than complete blood counts, serum biochemistry, urinalysis, thyroid assays, and antinuclear antibody assays, the dogs in our study had not been evaluated for systemic disease, and a potential influence of chronic subclinical rickettsial disease cannot be ruled out.
Two B-cell markers were used in this study. The first B-cell marker is directed against CD 79α, which is one of two polypeptides forming the CD 79 heterodimer. CD 79α is noncovalently associated with membrane-bound IgM or IgD on the B-cell surface to form the B-cell antigen receptor complex. 6 These membrane immunoglobulins are exclusively expressed on the surface of B lymphocytes or myeloid precursors of B cells and are not secreted. Antibodies generated to a highly conserved intracytoplasmic peptide sequence 20 have been demonstrated to stain normal and neoplastic B cells and plasma cells of several species. 7,22,32,37 The second antibody was directed against BLA 36, a plasma membrane glycoprotein with growth-related functions expressed on B-cell progenitors and B cells and Hodgkin's and B cell lines in humans 17,18 and has been used to identify B-cell neoplasia in dogs. 2 This particular marker anecdotally and in the experience of one of the authors (KW) may also stain dendritic cells. Staining with the BLA 36 antibody was more prominent than with CD 79α in the majority of the samples (Fig. 3). In the remaining specimens, the numbers and distribution of cells stained with both BLA 36 and CD 79α were similar. One possible explanation for this discrepancy would be that CD 79α is more sensitive to tissue fixation or tissue-softening pretreatments, as has been reported previously. 8 Another possibility is the staining of dendritic cells in addition to B cells. The morphological appearance of particularly the stained cells within the epidermis frequently showed projections expected with dendritic cells such as Langerhans cells. However, because of the formalin fixation of the specimens, Langerhans cell markers could not be used to verify this explanation. Nevertheless, anti–BLA 36 antibody seems to be the preferred marker for B cells in tissue, which has undergone formalin fixation and tissue-softening pretreatments. On average, one fourth of the dermal inflammatory infiltrate (between 10% and 49%) seen in canine lupoid onychodystrophy comprised B lymphocytes, many of which were morphologically plasma cells. An increased number of plasma cells is likely associated with local antibody production.

Inflammatory cells are seen predominantly within the epidermis; few cells are present in the dermis. BLA 36 antibody. Bar = 40 µm.
In human medicine, functional differences of macrophages are thought to be correlated to antigenic phenotypes in vitro, 21,31 and different antigenic phenotypes have been shown to exist in various reactive and inflammatory human conditions. 28 We chose three different markers for macrophages: Mac 387 and antibodies against lysozyme and MHC class II because there are no consistently definitive markers for all macrophages in fixed tissues from dogs. Mac 387 is a monoclonal antibody that was developed by Jones et al. 19 for identification of human phagocytes. Despite a strongly positive canine control, none of the specimens in this study showed significant staining with Mac 387 antibody. It is possible that the tissue-softening pre-treatment causes protein denaturation, which is responsible for the lack of staining.
Lysozyme is an enzyme that catalyzes the hydrolysis of specific glucosidic linkages and thus enhances the breakdown of some bacterial cell walls. It has been used as an immunohistochemical marker of histiocytic differentiation in human and canine histiocytic proliferative and granulomatous disorders. 24,29,35 Because neutrophils and macrophages are easily differentiated morphologically and other cells containing lysozyme are not usually present in the claw matrix, staining of round cells for lysozyme was considered highly specific for histiocytes/macrophages in our study. It is worth noting that neutrophils were observed in the upper layers of the claw matrix of case 11 and stained positive with the lysozyme stain. Higher counts of cells immunostained with antibody against lysozyme correlated well with the findings of the sections stained with HE, in which there was a moderate number of macrophages visible in the cellular infiltrate.
The MHC class II in humans and rodents is normally expressed by dendritic cells, macrophages, B cells, and vascular endothelial cells. 1 In dogs, it is constitutively expressed by all lymphocytes 9 and is thus the least specific of the markers used in our study. This particular molecule is essential for presentation of antigens to T cells. The human monoclonal antibody used in this study has been previously shown to react with canine MHC class II in paraffin-embedded, formalin-fixed specimens. 10,38 In one of these studies, 10 dogs with discoid lupus erythematosus often showed strong staining of the few present dermal macrophages with MHC class II antibody; in addition, lymphocytes occasionally were positively labeled. The staining technique used in this work rarely detects the lower level of MHC class II expression found on B cells, supported by the relative lack of correlation between B-cell markers and MHC class II in most cases in this study. In the few cases where there was a correlation, this may possibly be due to a higher number of MHC class II molecules expressed on highly activated B cells. Antibodies against lysozyme and MHC class II consistently stained some cells in almost all samples, indicating that they are useful markers for formalin-fixed tissues pretreated for softening. A possible explanation for the presence of macrophages could be the presence of a secondary infection. The significant increase in cells staining for lysozyme in the dermis versus the epidermis correlates well with the fact that the macrophages are rarely seen in the epidermis and have their predominant functions as part of the dermal infiltrate.
In summary, the mononuclear inflammatory infiltrate in the claw matrix of dogs with lupoid onychodystropy is composed predominantly of B cells and T cells; macrophages are typically only present in small numbers. Immunohistochemical staining correlated well with staining with HE in our study and provided no additional information. CD 3, BLA 36, lysozyme, and MHC class II preserve significant antigenicity during formalin fixation and short tissue–softening pre-treatment of 24–48 hours, whereas CD 79α and particularly Mac 387 seem to be more prone to denaturation by the softening process.
Footnotes
Acknowledgements
This study was financially supported by a grant from the American Academy of Veterinary Dermatology.
